J. Cent. South Univ. Technol. (2010) 17: 56-61
DOI: 10.1007/s11771-010-0011-9 
Mechanism of bioleaching chalcopyrite by Acidithiobacillus ferrooxidans in agar-simulated extracellular polymeric substances media
YU Run-lan(余润兰)1, 2, TAN Jian-xi(谭建锡)1, 2,
GU Gou-hua(顾帼华)1, 2, HU Yue-hua(胡岳华)1, 2, QIU Guan-zhou(邱冠周)1, 2
1. Key Laboratory of Biometallurgy of Education Ministry, Central South University,
Changsha 410083, China;
2. School of Minerals Processing and Bioengineering, Central South University, Changsha 410083, China
? Central South University Press and Springer-Verlag Berlin Heidelberg 2010
Abstract: The mechanism of leaching chalcopyrite by Acidithiobacillus ferrooxidans (A. ferrooxidans) in agar-simulated extracellular polymeric substances (EPS) media was investigated. The results indicate that bacterial EPS can release H+ and concentrate Fe3+; Fe2+ is movable between agar-simulated EPS phase and bulk solution phase, but it is difficult for Fe3+ to move due to its hydroxylation and EPS complex action; A. ferrooxidans first prefer Fe2+ as energy to metabolize compared with chalcopyrite, and a suitable simulated EPS environment for bacterial living is at about pH 1.8; the iron precipitates and jarosites formed by a lot of biologically oxidized Fe3+ cover the simulated EPS easily and form an impermeable deposit acting as a limited barrier of ion transport that attenuates the aggressiveness of the bioleaching attack. The EPS layer blocked by iron precipitates or jarosites is responsible for the chalcopyrite passivation.
Key words: Acidithiobacillus ferrooxidans; chalcopyrite; agar-simulated EPS media; bioleaching; mechanism
1 Introduction
Acidithiobacillus ferrooxidans (A. ferrooxidans) is one of the most important mesophiles for the extraction of metals from sulfidic ores by bioleaching [1]. The extracellular polymeric substances (EPS) of A. ferrooxidans consisting of neutral sugars and lipids mediate the contact between the mineral surface and the cell, and play an active role in the dissolution of sulfide minerals via a “contact leaching” mechanism [2-7]. The EPS containing complex Fe(Ⅲ) ions comprise a reaction space, in which the dissolution process takes place. The behavior of attached bacteria is greatly dependent on the molar ratio of Fe3+ to Fe2+ in the EPS layer, which is, in turn, greatly dependent upon the redox potential in solution and the concentration of soluble irons [8-9]. Bacterial cells change the chemical composition of their exopolymers according to the substrate in use [10]. The EPS might facilitate Fe3+ concentration by complexation through uronic acids or other residues on the mineral surface, resulting in an enhanced oxidative attack on the sulphide [4, 10]. FU et al [11] observed that EPS biofilm with some porous passages formed on the surface of chalcopyrite after bioleaching for 14-21 d. The chemical reactions occurred outside of the cells, outside of the outer membrane, but still in the EPS-generated microenvironment [12]. Up to now, the conditions within this reaction space, e.g., pH, redox potential, are not clear [1-2]. Jarosite precipitation is a very important phenomenon for chalcopyrite passivation in the bioleaching process [4, 10].
Agar (C12H18O9)n is a compound consisting of agarose agaropectin. Agarose is a non-ionizing, straight chain saccharan arranged alternately by D-galactosyl and 3,6-dehydration-L-galactosyl. Agaropectin is a complex saccharan containing certain amount of sulfate, glucuronic acid and malonadlhydic acid. Agar is a sol with thick and out-of-order in hot water, its saccharan chain twists and assembles cavum with some water. It is a good biological polymer to simulate and fasten bacterium EPS due to its high stability, favorable affinity for biological materials and good passageway for ions and water. EPS formed gradually in the course of bioleaching by A. ferrooxidans will pile up in the agar phase. At the same time, ions and water can also pass through it freely. Consequently, the changes of potential and pH values in the agar phase measured by pH meter can basically reflect the information on the interface of bacterial EPS/ sulfide mineral.
Nevertheless, the influence of EPS may be related partly to the extracting techniques used, and the relationship between the properties of the natural material and the extractions of EPS is still uncertain [13-17]. Ideally, the characteristics of the simulated EPS by agar should also reflect the properties of the stabilizing EPS in the natural environment.
In this work, leaching behavior of chalcopyrite by A. ferrooxidans in an agar-simulated EPS media was investigated to provide useful information to understand the EPS behavior, action with jarosite and their effect on bioleaching chalcopyrite.
2 Experimental
2.1 Microorganisms and culture
A. ferrooxidans strains obtained from our laboratory were cultured in ferrous-free 9K but containing CuFeS2 (1.0 g/L) as energy source, shaken at 28 ℃ and 180 r/min. The compositions of ferrous-free 9K medium were as follows: 3.00 g/L (NH4)2SO4, 0.10 g/L KCl, 0.50 g/L K2HPO4, 5.00 g/L MgSO4?7H2O and 0.01 g/L Ca(NO3)2. The pH value of the medium was adjusted to 2.0 by adding dilute H2SO4. All cultures were sterilized at 121 ℃ for 20 min before being inoculated. After being cultured for 14 d, cell numbers were up to 2×107 mL-1 counted microscopically using a counting chamber and this culture was used for the experimental inoculation.
2.2 Chalcopyrite preparation
A flotation chalcopyrite concentrate obtained from Dexing Mine (Jiangxi Province, China) partly containing pyrite (about 5%) was washed by 1mol/L HCl, 2 mol/L H2SO4 in turn three times, and then washed with acetone to remove the flotation reagents. The chalcopyrite concentrate was put in air for 4 h, then dried at 50 ℃ for 24 h. Finally, the mineral was sterilized by UV for 24 h in asepsis room.
2.3 Experimental method
Experimental equipment is shown in Fig.1.

Fig.1 Schematic diagram of experimental equipment
100 mL Fe2+-free 9K medium containing 3% (ratio of mass to volume) agar and 3% (ratio of mass to volume) NaCl was sterilized by autoclaving at 121 ℃ for 20 min to form a sol. When the temperature of the sol reduced to 45-50 ℃, 1.0 g chalcopyrite sample mentioned above was mixed with the sol rapidly to form a gel. Then a pH/ potential electrode was put into the gel whose outside was wrapped by gauze, which was called as gar-chalcopyrite phase.
2.3.1 Experiment 1
The agar-chalcopyrite phase without A. ferrooxidans was put at room temperature for 24 h, and then immersed in 200 mL iron-free 9K medium (pH=2.2) in a 250 mL beaker. Another pH/potential electrode was put in the solution. Then the solution of the beaker system was cultured at 30 ℃ and shaken at 100 r/min. The pH and potential values of the agar-chalcopyrite phase and the solution phase were measured by pH-3C meter simultaneously every day. The aim of this experiment is to investigate the behavior of pure acidic leaching in agar phase so as to compare with the situation with A. ferrooxidans.
2.3.2 Experiment 2
All experimental conditions were the same as those in experiment 1 besides that 10 mL A. ferrooxidans culture mentioned above was inoculated into the agar-chalcopyrite phase.
2.3.3 Experiment 3
Experimental conditions were the same as those in experiment 2, but the solution contained 0.5 g/L Fe2+ initially.
In order to keep the same pH and redox measurement conditions, the pH value was measured by pH electrode directly. Although the redox potential was measured by Pt electrode and reference electrode, there was certain definite relationship between two kinds of measured results by pH electrode and Pt electrode. So the results of redox potential measured by pH meter should be discussed as important as the results measured by Pt electrode.
3 Results and discussion
3.1 Simple acidic leaching behavior of chalcopyrite in agar-simulated system
In the no presence of A. ferrooxidans (experiment 1), chalcopyrite is basically and non-oxidatively leached by H2SO4 as follows [18]:
CuFeS2+4H+ Fe2++2H2S+Cu2+ (in agar phase) (1)
Fe2++2H2S+Cu2+ (in agar phase) (1)
This reaction is an acid-consumed reaction. But in fact, the pH values of both agar phase and H2SO4 solution phase decrease and their redox potentials increase gradually, and the change in agar phase is more obvious than that in H2SO4 solution phase from beginning to the third day (see Fig.2). So the facts mentioned above cannot be explained clearly by the non-oxidative leaching of chalcopyrite by H2SO4. The electrochemical galvanic action by FeS2 in chalcopyrite concentrate and complexing catalysis by glucuronic acid and HPO42- may be mainly responsible for these results. Relative reactions are as follows:
CuFeS2-4e=Fe2++Cu2++2S (2)
S+4H2O-6e=SO42-+8H+ (3)
4Fe2++O2+4H+=4Fe3++2H2O (4)
O2+4H++4e=2H2O (5)
Fe2+-e+nH2O=Fe(OH)n(3-n)++nH+ (6)
Mn++pHxY=(MYp)n-px+pxH+ (7)
where Mn+ represents metal ions and HxY represents organic acid in agar phase.

Fig.2 Change of pH value (a) and potential (b) with time in agar-simulated system without bacteria
At the same time, H+ in agar phase moves into the solution phase. Consequently, pH values of the two phases reduce gradually with the increase of time. But as can be seen from Fig.2(a), from the third day on, pH values of agar phase increase suddenly and then decrease again. It should be noted that the system is open to air, which potentially allows some chemical reactions of oxygen with the leaching solution [19]. Reactions (4) and (5) are shown above, which increase the pH value of solution. The hydroxylation action of Fe3+ is stronger than its complexing action at high pH value. When acidity reduces to lower pH value, Fe(OH)n(3-n)+ will liberate OH- that reacts with H+ to produce H2O as organic acids in agar complex Fe3+. At the same time, chalcopyrite with outside shell of agar phase can also be continuously leached by acid in solution due to the effect of the electrochemical galvanic action, so the pH values of H2SO4 solution phase only decrease continuously. Finally, the reactions in this system are up to equilibrium, and equilibrium pH value of agar phase at later stage is slightly higher and its redox potential is lower than that of solution phase.
Therefore, the hydroxylation of Fe3+ results in the acidity decrease and agar plays a role in reducing the redox potential by complexing Fe3+. However, agar cannot concentrate H+.
3.2 Bioleaching behavior of chalcopyrite in agar- simulated system
In the presence of A. ferrooxidans (experiment 2), chalcopyrite in agar phase is chiefly oxidized by bacteria [19], producing acid and Fe3+ according to reactions (8)-(11). Consequently, the pH values of both agar phase and solution phase decrease gradually with the increase of time, while their potentials increase gradually with the increase of time. The changes are more rapid compared with those of simple acidic leaching in experiment 1. Finally, equilibrium pH value of agar phase maintains at about 1.8, and its potential is about 300 mV at later stage (see Fig.3).
CuFeS2+4O2 FeSO4+CuSO4 (8)
FeSO4+CuSO4 (8)
4Fe2++O2+4H+ 4Fe3++2H2O (9)
4Fe3++2H2O (9)
Fe3++nH2O=Fe(OH)n(3-n)++nH+ (10)
3Fe3++M++2HSO4-+6H2O→MFe3(SO4)2(OH)6+8H+ (11)
The change trends of pH values in both solution and agar phases are similar before the 13th day. However, the former is always slightly lower than the latter,and the potential is on the contrary. It is just the period that EPS forms gradually according to FU et al [11]. After the 13th day, intact EPS film is formed, and the equilibrium pH value in EPS phase is slightly lower than that in solution phase. As the EPS mainly consist of proteins, carbohydrates and phenolic compounds that contain functional groups such as carboxyl, hydroxyl and amine, responsible for the binding of metal ions [20-21], it may be suggested that EPS can release H+.
Fe3++3EPS-H→Fe(EPS)3+3H+ (12)
Nevertheless, the pH values in solution phase suddenly rise from the 14th day to the 17th day. In the meantime, the presence of A. ferrooxidans can be observed microscopically in solution phase. Obviously, A. ferroxidans move into H2SO4 solution from agar phase. The metabolism of A. ferrooxidans consuming acid results in the rise of pH value in solution according to the following reactions:
Fe2+-e→Fe3+ (Iron enzyme center) (13)
4H++O2+4e→2H2O (Cytoplasm) (14)
2H2O+CO2→Hydrocarbon+Energy (Cytoplasm) (15)
After a few nutrition Fe2+ ions in the solution are consumed rapidly by bacteria, H+ from agar phase makes the pH value of solution phase decrease again. Finally, bioleaching in agar phase, H+ migration and consuming acid of A. ferrooxidans metabolism maintain dynamically equilibrium in solution phase, which makes a slight increase in pH values of solution phase from the 14th day to the 17th day.
Generally, the mixed potential is mainly dependent on the molar ratio of Fe3+ to Fe2+ and pH values. Fe3+ from Fe2+ oxidized by A. ferrooxidans in solution phase will make solution phase maintain much higher potential. But in fact, the potential of agar-EPS phase is always higher than that in solution phase. This indicates that A. ferrooxidans in solution only have a few EPS; a small amount of Fe3+ in solution phase is bound up by this part of EPS of A. ferrooxidans; and Fe3+ migration from agar-EPS phase into solution phase is very difficult.
Thereby, it can be inferred that: (1) EPS can concentrate bioleached Fe3+; (2) Fe2+ is somehow movable from agar-EPS phase into solution phase, but it is difficult for Fe3+ to move from agar-EPS phase into solution phase due to its hydroxylation and complex action; (3) bacteria create a suitable EPS environment for their living, that is, pH at about 1.8.
Experiment 3 (solution contains initial 0.5 g/L Fe2+) was carried out in order to understand further behaviors of Fe3+ and bacteria between solution phase and agar-EPS phase. The results are shown in Fig.4. It can be found that there are several differences compared with Fig.3.
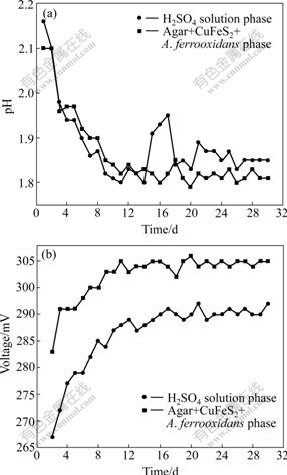
Fig.3 Change of pH value (a) and potential (b) with time in agar-simulated system with bacteria
Firstly, the pH values in the solution phase rise suddenly from the 6th day to the 9th day, and then decrease again. In the meantime, A. ferrooxidans are observed microscopically in solution phase in the 6th day. Obviously, A. ferrooxidans enter into solution phase from agar phase much early compared with experiment 2. This indicates that A. ferrooxidans first prefer Fe2+ as energy to metabolize. The solution phase maintains higher pH values of about 1.88 at the later stage of bioleaching according to reactions (13)-(15).
Secondly, although solution phases of experiments 2 and 3 maintain nearly the same redox potential of about 290 mV at the later stage, the equilibrium potential of the agar-EPS phase of experiment 3 (about 276 mV) is much lower than the corresponding values of both experiment 2 (about 300 mV) and experiment 1 (about 283 mV) without A. ferrooxidans. At the same time, the equilibrium pH value of the agar-EPS phase of experiment 3 at later stage (about 1.67) is much lower than the corresponding value of experiment 2 (about 1.88). HANSFORD and VARGAS [22] thought that the behavior of attached bacteria is strongly dependent on the molar ratio of Fe3+ to Fe2+ in the EPS layer, which is, in turn, greatly dependent upon the redox potential in solution and the concentration of soluble iron. Based on the experimental results mentioned above, it can be inferred reasonably that: (1) in the natural case without Fe2+ added, EPS formed by A. ferrooxidans create a suitable environment for bacterial living and for oxidizing sulfide, maintaining higher potential and suitable pH value at about 1.8; (2) in the presence of Fe2+, the iron precipitates or jarosites formed by a lot of biologically oxidized Fe3+ cover the outer shell of agar phase gradually in the solution phase and hinder bioleaching, and matter migration such as H+ and Fe2+, resulting in lower pH value and potential of agar phase at the later stage of bioleaching, so the appearance of large amount of iron precipitate or jarosite blocking EPS layer is responsible for the chalcopyrite passivation; (3) seen from the results that the pH values of solution phases of experiments 2 and 3 maintain higher value, and once entering into the solution phase, A. ferrooxidans will not go back to agar phase again.
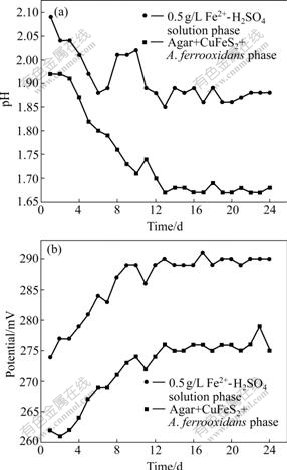
Fig.4 Change of pH value (a) and potential (b) with time in agar-simulated system with bacteria and 0.5 g/L Fe2+ added
4 Conclusions
(1) Fe2+ is movable between EPS phase and solution phase, but it is difficult for Fe3+ to move due to its hydroxylation and EPS complex action.
(2) A. ferrooxidans first prefer Fe2+ as energy to metabolize. Bacterial EPS can release H+ and concentrate Fe3+.
(3) Bacteria create a suitable EPS environment for their living, that is, at pH value of about 1.8; the iron precipitate or jarosite formed by a lot of biologically oxidized Fe3+ covers EPS easily and hinders bioleaching, and matter migration such as H+ and Fe2+ ions. Maybe the iron precipitate or jarosite blocking EPS layer is responsible for the chalcopyrite passivation.
References
[1] BARRETT J, HUGHES M N, KARAVAIKO G I, SPENCER P A. Extraction by bacterial oxidation of minerals [M]. Chichester: Metal Ellis Horwood, 1993.
[2] WATLING H R. The bioleaching of sulfide minerals with emphasis on copper sulphides: A review [J]. Hydrometallurgy, 2006, 84(1/2): 81-108.
[3] SAND W, GEHRKE H. Extracellular polymeric substances mediate bioleaching/biocorrosion via interfacical processes involving iron(Ⅲ) ions and acidophilic bacteria [J]. Research in Microbiology, 2006, 157(1): 49-56.
[4] KINZLER K, GEHRKE T, TELEGDI J, SAND W. Bioleaching: A result of interfacial processes caused by extracellular polymeric substances (EPS) [J]. Hydrometallurgy, 2003, 71(1/2): 83-88.
[5] GEHRKE T, TELEGDI J, THIERRY D, SAND W. Importance of extracellular polymeric substances from Thiobacillus ferrooxidans for bioleaching [J]. Appl Environ Microbiol, 1998, 64(17): 2743-2747.
[6] POGLIANI C, DONATI E. The role of exopolymers in the bioleaching of a non-ferrous metal sulphide [J]. J Ind Microbiol Biotech, 1999, 22(2): 88-92.
[7] BOON M. The mechanism of ‘direct’ and ‘indirect’ bacterial oxidation of sulphide minerals [J]. Hydrometallurgy, 2001, 62(1): 67-70.
[8] C?RDOBA E M, MU?OZ J A, BL?ZQUEZ M L, GONZ?LEZ F, BALLESTER A. Leaching of chalcopyrite with ferric ion. Part IV: The role of redox potential in the presence of mesophilic and thermophilic bacteria [J]. Hydrometallurgy, 2008, 93(3/4): 106-115.
[9] STOTT M B, WATLING H R, FRANZMANN P D, SUTTON D. The role of iron-hydroxy precipitates in the passivation of chalcopyrite during bioleaching [J]. Miner Eng, 2000, 13(10/11): 1117-1127.
[10] HARNEIT K, G?KSEL A, KOCK D, KLOCK J H, GEHRKE T, SAND W. Adhesion to metal sulfide surfaces by cells of Acidithiobacillus ferrooxidans, Acidithiobacillus thiooxidans and Leptospirillum ferrooxidans [J]. Hydrometallurgy, 2006, 83(1/4): 245-254.
[11] FU Jian-hua, QIU Guan-zhou, HU Yue-hua, XU Jing. The role of EPS of Thiobacillus ferrooxidans during bioleaching [J]. Acta Laser Biology Sinica, 2004, 13(1): 62-66.
[12] GEHRKE T, HALLMANN R, KINZLER K, SAND W. The EPS of Acidithiobacillus ferrooxidans: A model for structure-function relationship of attached bacteria and their physiology [J]. Water Sci Technol, 2001, 43(1): 159-167.
[13] DADE B W, DAVIS J D, NICHOLS P D, NOWELL A R M, THISTLE D, TREXLER M B, WHITE D C. Effects of bacterial exopolymer adhesion on the entrainment of sand [J]. Journal of Geomicrobiology, 1990, 8(1): 1-16.
[14] BLACK K S, SUN H, CRAIG G, PATERSON D M, WATSON J, TOLHURST T. Incipient Erosion of Biostabilized sediments examined using particle-field optical holography [J]. Environmental Science and Technology, 2001, 35(11): 2275-2281.
[15] TOLHURST T J, BLACK K S, SHAYLER S A, MATHER S, BLACK I, BAKER K, PATERSON D M. Measuring the in situ erosion shear strength of intertidal sediments with the cohesive strength meter (CSM) [J]. Estuarine, Coastal and Shelf Science, 1999, 49(2): 281-294.
[16] de BROUWER J F C, WOLFSTEIN K, STAL J L. Physical characterisation and diel dynamics of different fractions of extracellular polysaccharides in an axenic culture of a benthic diatom [J]. European Journal of Phycology, 2002, 37(1): 37-44.
[17] de BROUWER J F C, RUDDY G K, JONES T E R, STAL L J. Sorption of EPS to sediment particles and the effect on the rheology of sediment slurries [J]. Biogeochemistry, 2002, 61(1): 57-71.
[18] NICOL M J, LAZARO I. The role of non-oxidative processes in the leaching of chalcopyrite [M]. Montreal: Canadian Institute of Mining, Metallurgy and Petroleum, 2003: 367-381.
[19] THIRD K A, CORD-RUWISCH R, WATLING H R. The role of iron-oxidizing bacteria in stimulation or inhibition of chalcopyrite bioleaching [J]. Hydrometallurgy, 2000, 57(3): 225-233.
[20] GUIBAUD G, TIXIER N, BOUJU A, BAUDU M. Relation between extracellular polymers composition and its ability to complex Cd, Cu and Pb [J]. Chemosphere, 2003, 52(10): 1701-1710.
[21] AL-ASHEH S, DUVNJAK Z. Sorption of cadmium and other heavy metals by pine bark [J]. J Hazard Mater, 1997, 56(1/2): 35-51.
[22] HANSFORD G S, VARGAS T. Chemical and electrochemical basis of bioleaching processes [J]. Hydrometallurgy, 2001, 59(2/3): 135-145.
Foundation item: Project(2010CB630900) supported by the National Basic Research Program of China; Project(50621063) supported by the National Nature Science Foundation of China
Received date: 2009-02-16; Accepted date: 2009-05-31
Corresponding author: YU Run-lan, PhD, Professor; Tel: +86-731-88877216; E-mail: YRL715@sina.com.cn
(Edited by CHEN Wei-ping)