
Electrochemical techniques for monitoring stress corrosion cracking of
Type 40Cr steel in acidified chloride solution
AN Li-juan(安丽娟), LI Qing-fen(李庆芬)
College of Material Science and Chemical Engineering, Harbin Engineering University, Harbin 150001, China
Received 15 July 2007; accepted 10 September 2007
Abstract: Electrochemical impedance spectroscopy (EIS) and electrochemical noise (EN) techniques were used to detect stress corrosion cracking (SCC) on 40Cr steel specimens exposed to the acidified chloride solution at ambient. To test these two techniques, slow strain rate tensile (SSRT) tests were performed with 40Cr specimen in the identical corrosive solution at room temperature. In impedance measurements, phase shifts in frequency range from 1 to 1 000 Hz show a clear difference between the stressed and non-stressed specimens, suggesting that stress corrosion cracks are detected by the impedance measurements. EN signals in the process of SCC were recorded and then analyzed by standard deviation (STD). On the other hand, the mechanical properties, such as maximum tensile strength (MTS) and fracture strain (FS) measured by the SSRT, decrease significantly when the specimens are exposed to the corrosive solution relative to that in an inert medium. The SSRT results are consistent with fractography of the tested specimens by scanning electron microscopy (SEM). Analysis of the fracture surface clearly shows intergranular attack, suggesting that stress corrosion cracks are formed.
Key words: stress corrosion cracking; electrochemical impedance spectroscopy; slow strain rate tensile test; electrochemical noise;
40Cr steel
1 Introduction
Stress corrosion cracking (SCC) is well-known corrosion process, which can cause failures of structures and their components[1]. Since the SCC can lead to sudden failures, it is believed to be the most dangerous form of corrosion-assisted failure of materials.
There are many test methods that can be used to determine the basic properties of metals regarding stress corrosion cracking. One of the most common is slow strain rate tensile test[2-3]. Recently people have used electrochemical techniques such as electrochemical impedance spectroscopy (EIS) and electrochemical noise (EN) to research the process of SCC.
EIS has been used only occasionally to investigate SCC phenomenon. In the past time researchers have used EIS on non-stressed samples to investigate SCC. BOSCH et al[4-6] have used EIS during slow strain rate tensile (SSRT) tests of SS 304 specimen in a 5 mol/L H2SO4+0.1 mol/L NaCl solution at room temperature. They have measured the phase shift in a particular frequency range during the tests and correlated crack initiation to changes in this phase shift.
EN may be caused by the spontaneous fluctuation of corrosion potential and current generated by the corrosion reaction. Transient electrochemical measurements (e.g. measurements of electrochemical current and/or voltage generated by exposure of bare metallic surface) are often used to study the factors determining the initiation, propagation, and repassivation of localized corrosion damage[7-13]. EN is now recognized as a powerful tool in the study of corrosion processes, since it produces information that can be used to define corrosion mechanisms without the need of perturbing the system[14].
In this study, EIS and EN measurements were performed continuously during the SSRT tests, respectively. In the EIS experiments, phase shifts at particular frequencies show a clear difference between the stressed and non-stressed specimens. This suggests that it is possible to use EIS for monitoring stress corrosion cracking. EN technique was used to measure the stochastic fluctuations of the corrosion potential and the corrosion current during the SCC process. The characteristic noise signals were analyzed by standard deviation (STD).
2 Experimental
Tensile specimens with the dimensions shown in Fig.1 were used for SSRT tests. The chemical composition(mass fraction, %) of the 40Cr steel is: C 0.42, Mn 0.65, Si 0.21, Cr 0.9, Fe balance. Specimens were firstly annealed at 850 ℃ for 15 min, oil quenched, then tempered at 580 ℃ for 2 h and water cooled. The working surface of the specimen was polished to 1 000 grit using standard grinding paper degreased with acetone and rinsed with distilled water before the SSRT tests. All the tests were performed using the non-deaerated acidified chloride solution (pH=1) at room temperature. The SSRT tests were conducted using a tensile test machine operated at a crosshead speed of 0.002 mm/min, which corresponded to an initial strain rate of 2.35×10-6 s-1 of the tested samples. Details about the experimental installation were described in Ref.[15].
EIS experiments were performed using a potentiostat/galvanostat EG & G PAR model 283 and a frequency response detector model 1025 in the amplitude of the sinusoidal perturbation of 5 mV. The impedance measurements were performed over a frequency range from 10 mHz to 100 kHz. The cell consisted of the sample, used as the working electrode (WE), a Hg/Hg2Cl2 (saturated calomel electrode, SCE) reference electrode (RE) and a platinum counter electrode (CE).
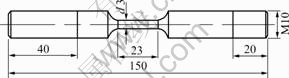
Fig.1 SSRT test specimen configuration (dimension in mm)
EN measurements were conducted using two nominally identical electrodes with the same surface area which were positioned in parallel to each other to be used as the working and the counter electrodes. SCE was used as the reference electrode for the measurement of potential noise. EN was measured in a freely corroding system (without externally applied current or voltage). Simultaneous potential and current data were recorded with a sampling frequency of 2 Hz. The area of the working electrode immersed in the test solution was 1.22 cm2.
The morphology of the fractured specimens was examined through a scanning electron microscope (SEM, Philips XL-30).
3 Results and discussion
3.1 EIS with Type 40Cr steel in acidified chloride solution
During the SSRT test, the electrochemical impedance measurements were performed every 30 min. The phases of four different frequencies, 1 000, 100, 10 and 1 Hz, were analyzed and plotted versus time. Fig.2 shows the results of the stressed and non-stressed test at different frequencies. The phases of the stressed specimens are smaller than those of the non-stressed specimens.
At 1 000 Hz a clear difference between the phases of the stressed and non-stressed tests can be seen from approximately 3.5 h onwards. At 1 and 100 Hz the difference is even clearer, also starting from about 3.5 h. The phenomenon is most likely to be related to the beginning of the SCC process shown in Fig.3. For application of EIS to the monitoring of stress corrosion cracking under these circumstances a frequency between 1 and 1 000 Hz seems the most appropriate choice. The correlation with SCC at 10 Hz is not so evident and is not plotted in Fig.3. When the SSRT test starts, the phase shift (=phasestressed-phasenon-stressed) starts to increase, suggesting the formation of stress corrosion cracks especially at 1 000 Hz.
3.2 EN analysis of Type 40Cr steel in acidified chloride solution
The numerous transients of the potential and current for stressed Type 40Cr steel in the acidified chloride solution are shown in Fig.4, which is attributed to stress corrosion cracking process. There are enormous numbers of noise peak, and the amplitude of the potential and current fluctuations is considerably high. It is seen from Fig.5 that the current transients with a value as high as 7-8 μA/cm2, which shows the typical SCC pattern of rapid rise and a slow fall in a stepwise fashion[16].
Standard deviation (STD) is often used to analyze EN. The deviation value depends on the number of sample points used in the calculation[17]. Figs.6 (a) and (b) show the STD of data in Fig.5 with different sample points of 10 and 100, respectively. There are four main current density peaks as shown in Fig.5. Corresponding to that, the same phenomenon is seen in Fig.6 at about the same time during this period. But the STD of current density and the total number of peaks remarkably decrease with an increased number of sample points. In Fig.6(a) the variation of STD peaks is well consistent with the appearance of EN amplitudes, while the value of main STD peaks is almost equal, as shown in Fig.6(b). Comparing Fig.6(a) with (b), the smaller number of sample points gives a more accurate result.
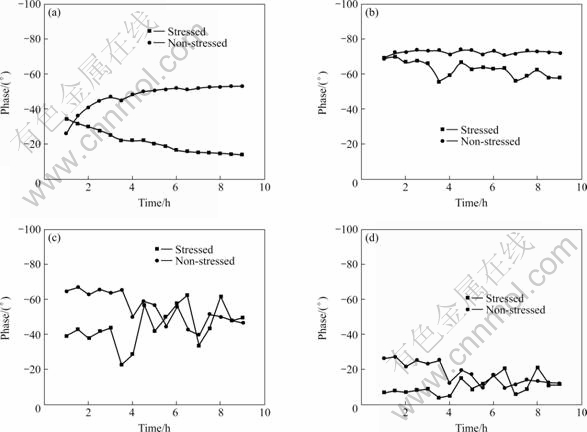
Fig.2 Phases of stressed and non-stressed tests vs time at various frequencies: (a) 1 kHz; (b) 100 Hz; (c) 10 Hz; (d) 1Hz
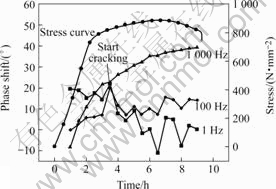
Fig.3 Phase shift evolution during SSRT test with Type 40Cr steel in acidified chloride solution
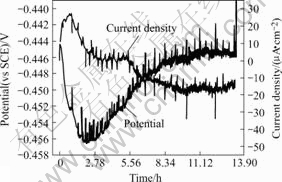
Fig.4 Visual records of potential and current density signals during SCC in acidified chloride solution
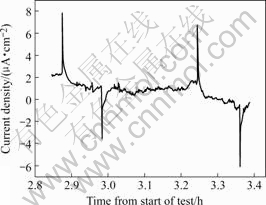
Fig.5 Characteristic noise of Type 40Cr steel in acidified chloride solution
3.3 SSRT results and fractography
The slow strain rate tensile tests were also performed with Type 40Cr steel specimens, which were subjected to an inert environment such as glycerin. Fig.7 shows the stress/strain curves of two tests, with and without the corrosive environment. The difference in fracture strain clearly reflects the reduction in elongation due to the stress corrosion cracking process. The total strain of the specimen without SCC is about 12%, while that of the specimen with SCC is approximately 8%. The maximum tensile strength of the former is 975.71 MPa,while the latter is 888.72 MPa, resulted from stress corrosion cracking process.
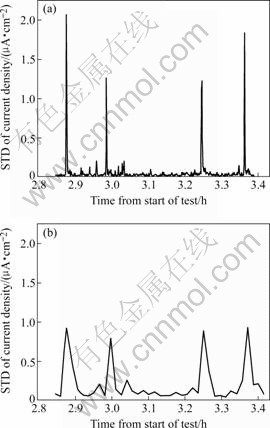
Fig.6 STD of current densities in Fig.5 processed with different sample points: (a) 10; (b) 100
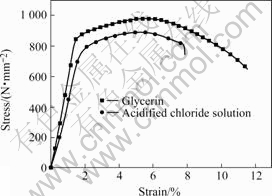
Fig.7 Stress―strain curves for Type 40Cr steel with and without corrosive environment
To prove that stress corrosion cracking certainly took place, the fracture surface was examined by using scanning electron microscopy (SEM). Fig.8 shows a picture of the fracture surface at two different magnifications. The photographs show clearly the intergranular fracture, related to the SCC process. Due to the severe conditions of the test solution, localized attack along the grain surfaces occurs.

Fig.8 SEM fractographs after SSRT tests: (a) Fracture surface; (b) Intergranular SCC
4 Conclusions
1) Intergranular SCC is observed when 40Cr steel is strained at a strain rate of 2.35×10-6 s-1 in the acidified chloride solution at room temperature.
2) The mechanical properties, such as maximum tensile strength and fracture strain, decrease significantly owing to the SCC occurrence when the specimens are exposed to the acidified chloride solution.
3) Impedance measurements show that a change in phase shift can be related to the stress corrosion cracking process. This implies that EIS can be used to detect stress corrosion cracks.
4) The sharp fluctuations of the potential and the current are recorded. Furthermore, some typical current transient signals are noticed and analyzed by STD during the SCC process.
References
[1] Leban M, Legat A, Dole?ek V. Detection and differentiation of SCC processes[C]//NACE 57th Annual Conference and Exposition. (CORROSION NACEXPO/2002), Houston, TX: NACE International, 2002: 1-10.
[2] YI Y, KIM H, PARK Y, KIM J. Effect of an inhibitor on the stress corrosion cracking behavior of Alloy 600 in a high-temperature caustic solution[J]. Corrosion, 2005, 61(5): 403-410.
[3] Saxena A, Singh Raman R K, Muddle B C. Slow strain rate testing for monitoring cracking of mild steels for vessels and pipes for processing using caustic solutions[J]. Int J Pressure Vessels Piping, 2006, 83(5): 399-404.
[4] Bosch r w, Moons F, ZHENG J H, BOGAERTS W F. Application of electrochemical impedance spectroscopy for monitoring stress corrosion cracking[J]. Corrosion, 2001, 57(6): 532-539.
[5] Bosch r w. Electrochemical impedance spectroscopy for the detection of stress corrosion cracks in aqueous corrosion systems at ambient and high temperature[J]. Corros Sci, 2005, 47(1): 125-143.
[6] Bosch R W, Moons F, Zheng J H, Bogaerts W F. Application of electrochemical impedance spectroscopy (EIS) for monitoring of SCC cracks[C]//NACE 54th Annual Conference and Exposition(CORROSIONNACEXPO 1999). Houston, TX: NACE International, 1999: 1-11.
[7] LEBAN M, Dole?ek V, Legat A. Comparative analysis of electrochemical noise generated during stress corrosion cracking of AISI 304 stainless steel[J]. Corrosion, 2000, 56(9): 921-927.
[8] Stewart J, Wells D B, Scott P M, Williams D E. Electrochemical noise measurements of stress corrosion cracking of sensitised austenitic stainless steel in high-purity oxygenated water at 288 ℃[J]. Corros Sci, 1992, 33(1): 73-88.
[9] Kolman D G, Gaudett M A, Scully J R. Modeling of anodic current transients resulting from oxide rupture of plastically strained β+α titanium[J]. J Electrochem Soc, 1998, 145(6): 1829-1840.
[10] Legat A, Dole?ek V. Corrosion monitoring system based on measurement and analysis of electrochemical noise[J]. Corrosion, 1995, 51(3): 295-300.
[11] Kolman D G, Scully J R. Limitation of potentiostatic repassivation techniques and their relationship to the applicability of the high field approximation to the repassivation of titanium[J]. J Electrochem Soc, 1995, 142(7): 2179-2188.
[12] Kolman D G, Scully J R. On the repassivation behavior of high-purity titanium and selected α, β and β+α titanium alloys in aqueous chloride solutions[J]. J Electrochem Soc, 1996, 143(6): 1847-1860.
[13] Kova? J, Leban M, Legat A. Detection of SCC on prestressing steel wire by the simultaneous use of electrochemical noise and acoustic emission measurements[J]. Electrochimica Acta, 2007, 52(27): 7607-7616.
[14] Duran M G, Macdonald D D. Stress corrosion cracking of sensitized type 304 stainless steel in thiosulphate solution. Ⅱ. Dynamics of fracture[J]. Corros Sci, 2006, 48(7): 1608-1622.
[15] Zheng J H, Bogaerts W F, BRABERS M J. Stress corrosion cracking and anodic dissolution of 316L stainless steel in hot lithium hydroxide[J]. Corrosion, 1992, 48(4): 320-331.
[16] Anita T, Pujar M G, Shaikh H, Dayal R K, Khatak H S. Assessment of stress corrosion crack initiation and propagation in AISI type 316 stainless steel by electrochemical noise technique[J]. Corros Sci, 2006, 48(9): 2689-2710.
[17] LUO J L, QIAO L J. Application and evaluation of processing methods of electrochemical noise generated during stress corrosion cracking[J]. Corrosion, 1999, 55(9): 870-876.
Corresponding author: AN Li-juan; Tel: +86-532-85843210; E-mail: anlijuan1977@yahoo.com.cn
(Edited by CHEN Wei-ping)