
Effect of sodium hexametaphosphate on separation of serpentine from pyrite
LU Yi-ping1, ZHANG Ming-qiang2, FENG Qi-ming1, LONG Tao1, OU Le-ming1, ZHANG Guo-fan1
1. School of Mineral Processing and Bioengineering, Central South University, Changsha 410083, China;
2. Changsha Design and Research Institute of Chemical Industry Ministry, Changsha 410016, China
Received 1 February 2010; accepted 6 September 2010
Abstract: The effect of sodium hexametaphosphate (SHMP) on the separation of serpentine from pyrite and its mechanism were studied systematically through flotation tests, sedimentation tests, surface dissolution, �� potential tests, adsorption measurements, and infrared spectroscopic analyses. The results show that the SHMP could significantly reduce the adverse effect of serpentine on the flotation of pyrite and make the mixed sample of pyrite and serpentine more disperse in the alkaline condition, thus improve the adsorption of xanthate on pyrite. The action mechanism of the SHMP is that it lowers the pH value at the isoelectric point of serpentine and enhances the negative charge through the dissolution of magnesium from the surface of serpentine and adsorbing on the surface of serpentine. It changes the total interaction energy between serpentine and pyrite from gravitational potential energy to repulse potential energy, according to the calculation of the EDLVO theory.
Key words: serpentine; sodium hexametaphosphate; dissolution; pyrite; flotation
1 Introduction
There is still no good solution for the efficient separation of serpentine from sulfide ore in the flotation of the serpentine-bearing sulfide ore, especially in the flotation of the copper-nickel sulfide. The main gangue mineral has a much higher content of serpentine, and it is prone to being clayed, which seriously interferes with the flotation of nickel-containing minerals[1-8]. In order for improving the flotation of the copper-nickel sulfide, sodium hexametaphosphate, sodium silicate, guar gum, carboxy methyl cellulose (CMC), and other agents are used as the inhibitors of serpentine[9-13].
Sodium hexametaphosphate disperses serpentine slime very well. It is widely used in mineral processing as a flotation regulator and has been applied in the separation of serpentine sulfide ore. Furthermore, there are a number of studies reporting the mechanism of sodium hexametaphosphate. GUO et al[14] explored the effects of sodium hexametaphosphate on the flotation of nickel-bearing pyrrhotite and its mechanism. They pointed out that the complexation of metal ions, from useful minerals, with sodium hexametaphosphate has a great effect on the flotation of nickel-bearing pyrrhotite. Based on the literature review[15], sodium hexametaphosphate has a good inhibitory effect on antigorite. According to another document, sodium hexametaphosphate adsorbed on the surface of the antigorite, lowering the adsorption of the anionic collector on the antigorite. XIA et al[16] studied the dispersion mechanism of serpentine. They found that sodium hexametaphosphate could change the surface potential of the serpentine and disperse the minerals.
However, the relationship between the dispersion of serpentine and pyrite and the flotation behavior has not been systematically studied. The action mechanism of sodium hexametaphosphate has not been systematically explained.
In the present work, the influence of sodium hexametaphosphate on the flotation and dispersion of the serpentine and pyrite is systematically studied. Moreover, the mechanism of sodium hexametaphosphate is also discussed providing a reference for further study.
2 Experimental
2.1 Samples and reagents
The serpentine (Mg6[Si4O10](OH)8) used for all experiments was obtained from Donghai, Jiangsu Province, China. A batch of 20 kg of serpentine rocks was ground and sieved. The grain size distribution is shown in Table 1. And the mineral composition of the serpentine selected for experiments and tested by XRD was as follows: serpentine 90%, chlorite 5%, and illite + amphibole 5%. Pyrite was obtained from Yunfu, Guangdong Province, China. The result of chemical analysis showed that the composition included Fe 43.96% and S 49.98%, and the grain size distribution of the sample is shown in Table 2.
Table 1 Grain size distribution of serpentine sample

Table 2 Grain size distribution of pyrite sample

Before each test, the pyrite was cleaned for 3 min in the SB3200 ultrasonic cleaner.
HCl, NaOH, and sodium hexametaphosphate used in all experiments were analytical reagents. Methyl isobutyl carbinol (MIBC) was a commercially pure reagent. The purity of potassium amyl xanthate (PAX) was 97.5%. The once-distilled water was the test media.
2.2 Equipments
The main test equipments were WGZ-3 scattered light turbidimeter, pHS-3C precision pH meter, Coulter delsa 440sx potentiometer, XFG hanging trough flotation machine, TU1810 UV-Vis spectrophotometer, FTIR spectrometer NEXUS670, PS-6 Vacuum-type Radial Plasma Spectrometer, and the SB3200 ultrasonic cleaner.
2.3 Methods
Coagulation and dispersion between serpentine and pyrite were studied using the sedimentation test. After the settlement of the slurry with 1 g/L concentration at a fixed time (3 min), the dispersion of the supernatant liquor was characterized by its turbidity.
The flotation test was performed in a 40 mL cell, using an XFG laboratory flotation machine.
The ore sample was ground to less than 2 ?m. �� potential and pH value were measured after mixing according to the test procedures and test conditions.
The adsorption of PAX was measured using the residual concentration method, with a TU1810 UV-Vis spectrophotometer. The characteristic absorption peak of PAX was at the wavelength of 301 nm and its concentration range was 0-9��10-5 mol/L; the Beer��s law was obeyed.
The infrared spectroscopy measurement of the serpentine sample was carried out by using a FTIR spectrometer NEXUS670.
The supernatants can be obtained by centrifuged pulp that has been stirred for 5 min in a solid content of 0.5 g/L and pH=9. The content of Mg and P of the supernatant was analyzed using a PS-6 Vacuum-type Radial Plasma spectrometer.
3 Results and discussion
3.1 Effect of sodium hexametaphosphate on flotation separation of serpentine from pyrite
The effect of sodium hexametaphosphate on the flotation separation of serpentine from pyrite was studied through flotation tests and sedimentation tests. According to Fig.1 and 2, serpentine has a great impact on the flotation of pyrite, and the more the amount of serpentine, the greater the impact is. When there is only 5% of additive serpentine in the mixed mineral of serpentine and pyrite, the productivity of the mixed mineral decreases from 86% to 32%. However, the addition of sodium hexametaphosphate (60 mg/L) could significantly weaken the suppression effect of serpentine as a result of the sedimentation tests as shown in Fig.2. It can be inferred that the heterogeneous coacervation between serpentine and pyrite is intense. Sodium hexametaphosphate could effectively disperse serpentine and pyrite.
In order to further investigate the effect of sodium hexametaphosphate on the flotation separation of serpentine from pyrite, the adsorption quantity of PAX on artificial mixed ore (pyrite 15 g/L, serpentine 5 g/L) was investigated and determined by the TU1810 UV-Vis spectrophotometer. From Fig.3, it can be seen that PAX has basically not absorbed on the surface of serpentine; the absorption amount of PAX on the surface of the serpentine-bearing pyrite pulp increases with the increasing amount of sodium hexametaphosphate. The

Fig.1 Effect of SHMP on flotation separation of serpentine from pyrite with flotation condition of pH=9.0, c(PAX)=1��10-4 mol/L, c(MIBC)=18.5 mg/L and ��(pyrite)=50 g/L

Fig.2 Relationship between turbidity of serpentine and pyrite and pH value in flotation test with different conditions

Fig.3 Relationship between adsorbed xanthate on mineral with dosage of SHMP in flotation separation test under condition of pH=9.0, c(PAX)=1��10-4 mol/L
results correspond to Figs.1 and 2. The fact further demonstrates that sodium hexametaphosphate reduces the coverage of serpentine on pyrite.
3.2 Mechanism of sodium hexametaphosphate separating serpentine from pyrite
3.2.1 The influence of sodium hexametaphosphate on electrical property of serpentine
The relationship between �� potential and pH value of pyrite and serpentine particle surface is presented in Fig.4. It can be seen that the pH value at the isoelectric point of serpentine is 10.20. The pH value at the isoelectric point of pyrite is less than 2. The surface of the pyrite particles shows negative electricity in the pH range of 2-14. In the pH value range of 2-14, the absolute value of �� potential of the surface of serpentine particle is lower and aggregation occurs easily among the particles. Meanwhile, the potential of the surface of pyrite is negatively high in the pH range of 2-14, and the electrical properties of serpentine and pyrite are opposite.
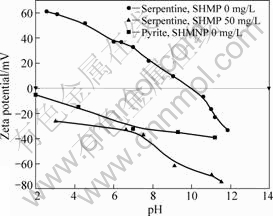
Fig.4 Relationship between Zeta potential and pH value
Sodium hexametaphosphate lowers the pH value at the isoelectric point of serpentine, and the negative charges on the surface increase. The surface potential of serpentine changes from positive to negative in the weak alkaline condition. And the sodium hexametaphosphate weakens the heterogeneous coacervation between the serpentine and pyrite.
3.2.2 Interaction between sodium hexametaphosphate and the surface of serpentine
A number of studies reported that sodium hexametaphosphate could lower the surface �� potential of serpentine. There are two explanations for the action mechanism of sodium hexametaphosphate. First, a stable hydrophilic complex is able to form due to the complexing of sodium hexametaphosphate with metal ions from the surface of serpentine. Second, sodium hexametaphosphate can adsorb on the surface of serpentine, thus changing the electrical properties of serpentine. We investigated the interaction between sodium hexametaphosphate and the surface of serpentine in two aspects. One is the dissolution of the surface of serpentine and the other is the adsorption of sodium hexametaphosphate on the surface of serpentine.
Sodium hexametaphosphate (NaPO3)6 is a kind of long-chain inorganic salt, a spiral chain polymer derived by polymerization from numerous basic structural units, and it can be expressed as (NaPO3)n, where n=20-100.
Sodium hexametaphosphate has a good capacity of complexation with metal ions, including calcium ions, magnesium ions, and other metal ions, generating soluble complexes.
Some magnesium ions may be dissolved from the surface of serpentine by sodium hexametaphosphate. Reactions may occur as follows:
(NaPO3)6+Mg2+=[MgNa2P6O18]2-+4Na+ (1)
[MgOH]++[Na4P6O18]2-=MgNa4P6O18+OH- (2)
The result of the dissolution test is shown in Table 3. The magnesium on the surface of the serpentine dissolves significantly in the sodium hexametaphosphate solution. Based on the reports, the brucite layer of serpentine has damaged in the fracture and the surface-exposed magnesium ions of serpentine are emplaced ions. Negative charges increase with the decrease in the amount of magnesium ions on the surface of serpentine and then the electrical property of serpentine is changed.
Table 3 Dissolution test results of serpentine in centrifugal liquid
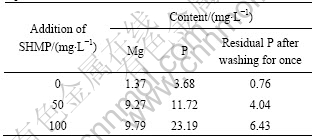
It could be seen that sodium hexametaphosphate remains mainly in the solution by the dissolved P measured in the supernatant liquid. However, when the surface of the serpentine is washed once, there is still residual sodium hexametaphosphate on the surface of the serpentine, which shows that a certain amount of sodium hexametaphosphates have adsorbed on the surface of serpentine. This also leads to a change in the electrical property of serpentine.
In order to verify the adsorption of sodium hexametaphosphate on the surface of serpentine, the FTIR spectrum of serpentine under the action of sodium hexametaphosphate was measured, and the results are shown in Fig.5.
The infrared spectrum of the sample of sodium hexametaphosphate is shown as line 1 (Fig.5), where
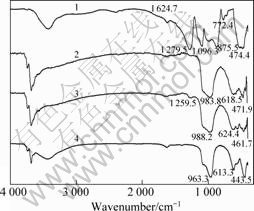
Fig.5 FTIR spectra of serpentine with or without SHMP: 1 SHMP; 2 SHMP+serpentine, pH=9.0; 3 SHMP+serpentine, pH=6.0; 4 Serpentine
the peaks at 1 279.54, 1 096.33 and 875.52 cm-1 correspond to the characteristic peak of P=O, P��O, and P��O��P, respectively. The infrared spectrum of pure serpentine is shown as line 4. The infrared spectrum of serpentine after adding sodium hexametaphosphate in different pH conditions is shown as lines 2 and 3. It is able to obtain the stretching frequency of hydroxyl changed after the action of sodium hexametaphosphate. It is indicated that sodium hexametaphosphate changes the surface hydroxyl of serpentine. Compared with line 4, the peak at 1 096.33 cm-1 on lines 2 and 3 are strengthened after the action of sodium hexametaphosphate. It demonstrates that the sodium hexametaphosphate has adsorbed on the surface of serpentine. There is a peak at 1 259.5 cm-1 on line 3, which also demonstrates that the sodium hexametaphosphate has adsorbed on the surface of serpentine. Na in sodium hexametaphosphate could be replaced by H, and hence cause the change of stretching frequency.
Therefore, there are two kinds of explanations for the action mechanism of sodium hexametaphosphate. First, a certain amount of magnesium ions are dissolved; second, sodium hexametaphosphate adsorbs on the surface of serpentine, which not only results in the change of the �� potential of serpentine, but also changes the space steric hindrance.
3.2.3 Interaction energy between serpentine and pyrite particles
The extended DLVO theory model is as follows. Taking the energies of electrostatic interaction, van der Waals interactions, and steric hindrance effects into consideration [17-18],
VT=VW+VE+VSR (3)
where VT is the interaction energy; VW is van der Waals interaction energy; VE is electrostatic interaction energy; VSR is the steric hindrance interaction energy. And
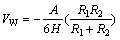 (4)
(4)
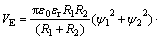
 (5)
(5)
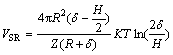 (6)
(6)
The radius of serpentine particle R1 is 3 ��m, the radius of pyrite particle R2 is 11 ��m; �� is the thickness of the electric double-layer, ��=0.180 nm-1; ��0, ��r represents the vacuum dielectric constant and the relative dielectric constant of the continuous phase, with given value of 6.95��10-10 C2/(J��m), and H represents the distance between particles; A represents the effective Hamaker constant for the serpentine and pyrite in aqueous solution, and A=1.97��10-20 J; �� is the thickness of the adsorbed layer after the absorption of sodium hexametaphosphate on the surface, and ��=5 nm[17]; K is the Boltzmann constant, and K=1.381��10-23 J/K; Z is the absorbing area of macromolecules on the surface of the particles, the given value is 1.9��10-16 m2[18]; ��1 and ��2 are the surface potential, mV. A dynamic potential can be used.
Replacing the relative data into Eqs.(2) and (3), the result is shown in Fig.6.

Fig.6 Relationship between interaction energy of pyrite�C serpentine particles and particles distance
The total interaction energy between serpentine and pyrite of the samples without SHMP is negative, as shown in Curve 1 of Fig.6. They attract each other and the aggregation of each other occurs easily. The total interaction energy between serpentine and pyrite is positive after adding SHMP, as shown in Curve 2 of Fig.6. Mutual exclusion will appear among the particles. This is consistent with the experimental results.
4 Conclusions
1) The heterogeneous coacervation between serpentine and pyrite is intense when pH>7.5 in the alkaline condition. Sodium hexametaphosphate can effectively disperse the serpentine and pyrite, reduce the coverage of serpentine on pyrite and then increase the adsorption of PAX on the surface of the pyrite, improving the flotation recovery of the pyrite.
2) The main reason for the heterogeneous coacervation between serpentine and pyrite is the higher pH value at the iso-electric point of serpentine. Sodium hexametaphosphate lowers the pH value at the electric point of the serpentine significantly, and then the space hindrance comes into being. The total interaction energy changes from the gravitational potential energy to the repulsion potential energy, effectively weakening the heterogeneous coacervation.
3) The change in the electrical properties of serpentine is due to the dissolution of magnesium from the surface of serpentine by sodium hexametaphosphate, and its adsorption on the surface of serpentine. The former is the main reason.
References
[1] HU Xian-zhi, ZHANG Wen-bin. Research progresses on removal of MgO in the flotation concentrate of copper-nickel sulfides [J]. Non-Ferrous Mining and Metallurgy, 2003, 19(1): 21-25. ( in Chinese)
[2] GENCE N, OZDAG H. Surface properties of magnesite and surfactant adsorption mechanism [J]. International Journal of Mineral Processing, 1995, 43(1/2): 37-47.
[3] FORNASIERO D, RALSTON J. Cu(II) and Ni(II) activation in the flotation of quartz, serpentine and chlorite [J]. International Journal of Mineral Processing, 2005, 76(1/2):75-81.
[4] GALLIOS G P, DELIYANNI E A, PELEKA E N, MATIS K A. Flotation of chromite and serpentine [J]. Separation and Purification Technology, 2007, 55(2): 232-237.
[5] BREMMELL K E, FORNASIERO D, RALSTON J. Pentlandite-lizardite interactions and implications for their separation by flotation [J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 2005, 252(2/3): 207-212.
[6] PRIETROBON M C, GRANO S R, SOBIERAJ S, RALSTON J. Recovery mechanisms for pentlandite and MgO-bearing gangue minerals in nickel ores from Western Australia [J]. Minerals Engineering, 1997, 10(8): 775-786.
[7] CHEN Jin, The research on the influence of sulfite on violarite flotation [J]. Metallic Ore Dressing Abroad, 1980, 3: 1-10. (in Chinese)
[8] LU Yi-ping, LONG Tao, FENG Qi-ming,OU Le-ming, ZHANG Guo-fan. Flotation and its mechanism of fine serpentine [J]. The Chinese Journal of Nonferrous Metals, 2009, 19(8): 1493-1497. ( in Chinese)
[9] HIRASAWA R, HORITA H. Dissolution of nickel and magnesium from garnierite ore in an acid solution [J]. International Journal of Mineral Processing, 1987, 19(1/4): 273-284.
[10] LI Zhi-hua. The effect of gangue minerals containing magnesium on pentlandite flotation [J]. Journal of Central-South Institute of Mining and Metallurgy, 1993, 24(1): 36-44. ( in Chinese)
[11] RAIJA J. Surface chemistry of froth flotation [M]. Beijing: Metallurgical Industry Press, 1987: 601-603
[12] KIRJAVAINEN V, HEISKANEN K. Some factors that affect beneficiation of sulphide nickel-copper ores [J]. Minerals Engineering, 2007, 20(7): 629-633.
[13] EDWARDS G R, KIPKIE W B, Agar G E. The effect of slime coatings of the serpentine minerals, chrysotile and lizardite on pentlandite flotation [J]. International Journal of Mineral Processing, 1980, 7(1): 33-42.
[14] GUO Chang-huai, Deng Zhu-sheng, HU Xi-geng. The effect of sodium hexametaphosphate on the flotation of nickel bearing pyrrhotite [J]. Nonferrous Metals (Mineral Processing), 1985(6): 16-19. ( in Chinese)
[15] DONG Nai-liang, CUI Bao-bin. Study of the flotation characteristics of antigorite and restrain Mechanism [J]. Journal of BGRIMM, 1984, 3: 31-36. ( in Chinese)
[16] XIA Qi-bin, LI Zhong, QIU Xian-yang, DAI Zi-lin. Investigation of action mechanism between sodium hexametaphosphate and serpentine [J]. Mining and Metallurgical Engineering, 2002, 22(2): 51-54. ( in Chinese)
[17] QIU Guan-zhou, HU Yue-hua, WANG Dian-zuo. Mechanism of carrier flotation of ultrafine hematite [J]. Nonferrous Metals, 1994, 46(4): 23-28. ( in Chinese)
[18] WANG Yu-hua, CHEN Xing-hua, HU Yue-ming, LAN Ye. Influences of phosphates on dispersion of fine alumin-silicate minerals [J]. Journal of Central South University of Technology (Science and Technology), 2007, 38(2): 238-244. (in Chinese)
��ƫ������������ʯ�����������е�����
¬����1������ǿ2��������1������1��ŷ����1���Ź���1
1. ���ϴ�ѧ ��Դ�ӹ������﹤��ѧԺ����ɳ 410083��
2. ��������ɳ����о�Ժ����ɳ 410116
ժ Ҫ��ͨ����ѡ���顢�������顢�����ܽ⡢�Ƶ�λ���ԡ����������Ժͺ�����ײ����о���ƫ������������ʯ�����������е�����������������������ƫ�����ƿ���������������ʯ�Ի�����ѡ�IJ���Ӱ�죬�����ڼ���������ʹ����ʯ��������ɢ���Ӷ����ҩ�ڻ�������������������ƫ�����Ƶ�������ͨ���ܳ�����ʯ�����þ�������������������������ʯ����ȵ��pH����ǿ�����ԡ�DLVO���ۼ����������ƫ��������ʹ����ʯ������������������������λ��ת��Ϊ����λ�ܡ�
�ؼ��ʣ�����ʯ����ƫ�����ƣ��ܽ⣻������ѡ
(Edited by LAI Hai-hui)
Foundation item: Project(2007CB613602) supported by the National Basic Research Program of China
Corresponding author: ZHANG Ming-qiang; Tel/Fax: +86-731-88830913; E-mail: luyp309@sohu.com
DOI: 10.1016/S1003-6326(11)60701-2