改性生物质材料对水溶液中Cu2+的吸附
来源期刊:中国有色金属学报(英文版)2015年第3期
论文作者:董颖博 林 海
文章页码:991 - 996
关键词:生物质材料;香菇培养基废料;改性;吸附;重金属;Cu2+
Key words:biomass material; spent shiitake substrate; modification; adsorption; heavy metal; Cu2+
摘 要:研究改性生物质材料――香菇培养基废料对水溶液中重金属铜离子的吸附性能。结果表明:改性可以大大提高香菇培养基废料的吸附性能,其铜离子去除率高达95%以上;与天然香菇培养基废料相比,改性后吸附材料的比表面积和电负性增大,表面官能团数目及种类增多。在吸附0.75 h后,改性香菇培养基废料对铜离子的吸附达到平衡,该吸附过程是一个放热反应。改性香菇培养基废料吸附铜离子前后的扫描电镜和能谱分析表明,吸附铜离子的材料表面可明显看到孔隙被占据,且铜含量明显增加,而钠含量减少。
Abstract: Cu2+ adsorption from simulated aqueous solution was investigated using a modified spent shiitake substrate (MSSS). The results showed that the MSSS has a high adsorption efficiency and removal performance. The Cu2+ removal rate of the MSSS reached above 95%. Compared with spent shiitake substrate (SSS), the specific surface area, electronegativity and surface functional groups of the MSSS were all improved, resulting in a high adsorption capacity. The Cu2+ adsorption of MSSS reached equilibrium after 0.75 h and was an exothermic reaction. The SEM and EDS analyses of the MSSS before and after Cu2+ adsorption showed that the pores on the surface of the MSSS were occupied after adsorption and the Cu content increased but the Na content decreased.
Trans. Nonferrous Met. Soc. China 25(2015) 991-996
Ying-bo DONG1,2, Hai LIN1,2
1. School of Civil and Environment Engineering, University of Science and Technology Beijing, Beijing 100083, China;
2. Key Laboratory for High-efficient Mining and Safety of Metal Mines, Ministry of Education, University of Science and Technology Beijing, Beijing 100083, China
Received 23 April 2014; accepted 13 July 2014
Abstract: Cu2+ adsorption from simulated aqueous solution was investigated using a modified spent shiitake substrate (MSSS). The results showed that the MSSS has a high adsorption efficiency and removal performance. The Cu2+ removal rate of the MSSS reached above 95%. Compared with spent shiitake substrate (SSS), the specific surface area, electronegativity and surface functional groups of the MSSS were all improved, resulting in a high adsorption capacity. The Cu2+ adsorption of MSSS reached equilibrium after 0.75 h and was an exothermic reaction. The SEM and EDS analyses of the MSSS before and after Cu2+ adsorption showed that the pores on the surface of the MSSS were occupied after adsorption and the Cu content increased but the Na content decreased.
Key words: biomass material; spent shiitake substrate; modification; adsorption; heavy metal; Cu2+
1 Introduction
Heavy metals as raw materials or catalysts are used widely in many industrial production, such as mining, metallurgy, electrolysis, electroplating and leather [1]. They can be subsequently released into the environment through wastewater, which has become a threat to human and local environment [2]. Because the heavy metals in water are difficult to be biodegraded and tend to accumulate in organisms through the food chain [3], the wastewater containing heavy metals should be treated before discharge. Beside heavy metal ions, the pH of mine water is between 3 and 4, due to a series of reactions of sulfide minerals in the presence of air, water and microorganisms.
Wastewater containing heavy metals is usually treated through neutralized sedimentation, precipitation with sulfides, redox, membrane separation, ion exchange, constructed wetlands and adsorption [4-7]. Among them, adsorption is a simple, inexpensive, effective and universal method [8]. Development and application of low-cost adsorption material have become the hotspot of current research [9-10]. Agricultural wastes have recently attracted increasing interest from researchers because they are readily available and can be used as adsorbent for effectively removing heavy metals from wastewater [11-14].
Spent shiitake substrate (SSS) is the discarded bacteria stick from shiitake cultivation that is rich in dead fungal hyphae and cultivation materials, such as sawdust and bran [15]. Studies have shown that materials such as bran and sawdust adsorb heavy metals, thus SSS may be a suitable adsorbent [16-17]. TU et al [18] studied the performance of SSS in adsorbing lead ions and found that lead ions can be effectively adsorbed when the pH of wastewater ranged from 4.09 to 6.00 [18]. Nevertheless, our preliminary study indicated that the organic matter in SSS dissolves when it is directly used as an adsorbent, thereby increasing the chemical oxygen demand (COD) and chroma. Therefore, SSS needs to be modified before it can be used as an adsorbent.
Few studies have been conducted on SSS modification. To improve the performance of SSS and decrease the amount of organic matter dissolved during adsorption, which causes secondary pollution, SSS was modified via phosphate esterification supplemented with alkali and microwave treatment. The adsorption performance of SSS and the modified SSS (MSSS) was compared. MSSS was also used to treat simulated AMD at low pH and the underlying Cu2+ adsorption mechanism was studied.
2 Experimental
2.1 Materials
The SSS, provided by a company in Beijing, was dried at 80 °C to a constant mass, with a moisture content of 10%. The dried SSS was pulverized and filtered with a 850 μm screen. The SSS was placed in polyethylene bags and stored in a dryer.
2.2 Water sample
Simulated wastewater samples containing different concentrations of Cu2+ were prepared according to the condition of the AMD. The reagent used in the experiment was cupric sulfate. The pH of water samples was adjusted with H2SO4 (0.1 mol/L). All of the reagents used are analytically pure.
2.3 Modification of SSS
50 g of SSS was mixed with 750 mL of NaOH solution (mass fraction, 10%), and shaken for 2 h at 140 r/min and 30 °C, filtered and washed with deionized water until the pH of the washing fluid became neutral. The SSS was dried at 80 °C to a constant mass and placed in phosphoric acid solution (0.5 mol/L) with a solid-to-liquid ratio of 1:15. The SSS was heated in the microwave for 2.5 min, and then washed to remove the excess phosphoric acid solution. Finally, the resulting MSSS was dried at 80 °C to a constant mass, filtered with a 850 μm screen, placed in polyethylene bags and stored in a dryer.
2.4 Adsorption experiment
Water samples containing 150 mg/L of Cu2+ were prepared. The water samples in 500 mL flasks were mixed with 5 g of MSSS and rotated in a thermostated rotator (30 °C, 140 r/min) to achieve adsorption equilibrium. After adsorption, the materials were filtered. The Cu2+ concentration of the water sample was then measured. All the experiments were carried out in triplicate and some were repeated several times to establish the reproducibility of the results.
2.5 Analytical test
The Cu2+ concentration in the water sample was analyzed using atomic absorption spectrometry. The removal effectiveness was evaluated based on the adsorption quantity (qt, mg/g) and removal rate (r, %), which were calculated by the following equations:
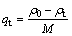 (1)
(1)
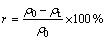 (2)
(2)
where ρ0 represents the initial Cu2+ concentration; ρt represents the remaining Cu2+ concentration after adsorption (in mg/L); M represents the amount of adsorbent added (in g/L).
3 Results and discussion
3.1 SEM analysis of SSS and MSSS
The SEM micrographs of the SSS and MSSS are shown in Figs. 1(a) and (b), respectively. In Fig. 1(a), the surface of SSS is loose, with large pores and burrs on the surface. In Fig. 1(b), the cytoderm structure of the MSSS is damaged, with more small pores, greatly increasing its specific surface area. Furthermore, the pores are aligned. Greater specific surface areas are generally correlated with better adsorption performance. Therefore, the structure of MSSS is better for adsorption.

Fig. 1 SEM micrographs of SSS (a) and MSSS (b)
3.2 Zeta potentials of SSS and MSSS
The zeta potentials of the SSS and MSSS are shown in Fig. 2. Both the MSSS and SSS were negatively charged in the solution; thus, they can absorb Cu2+ through electrostatic attraction. The electronegativity of MSSS and SSS increased when pH increased, leading to the increase of adsorption capacity [19]. The electronegativity of MSSS was higher than that of SSS. This result indicates that after modification, the negative charge on the surface increases, which is beneficial for Cu2+ adsorption.
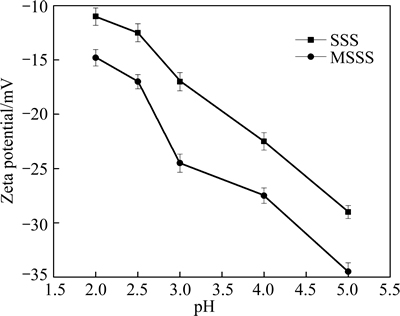
Fig. 2 Zeta potentials of SSS and MSSS
3.3 FTIR analyses of SSS and MSSS
The FTIR spectrograms of the MSSS and SSS were compared, as shown in Fig. 3. In the FTIR analysis of SSS, the strong peak near 3422.34 cm-1 is attributed to the stretching vibration of O―H, the peak near 2925.69 cm-1 is the antisymmetrical stretching vibration of ―CH2, the peak near 2362.81 cm-1 is probably the peak of CO2, the strong peak near 1633.71 cm-1 is the absorption band of amide II, the strong peak near 1321.70 cm-1 is the absorption band of amide III, the peak near 1051.29 cm-1 is the stretching vibration of C―OH and the peak near 786.83 cm-1 is the symmetrical stretching vibration of Si―O―Si [20].

Fig. 3 FTIR spectrograms of MSSS and SSS
Comparison of the FTIR spectrograms of the MSSS and SSS revealed that the stretching vibration peak of O―H significantly changed. The peak was stronger and shifted from a wave number 3422.34 to 3450.64 cm-1. This result strongly indicated that the modification of the SSS increased the number of O―H. The absorption bands of amide II and III significantly changed. The peaks were thinner and shifted to 1641.15 and 1357.66 cm-1, respectively. This result is attributed to phosphate esterification, a part of the ―NH2 in the amide molecules reacts with ―POOH, and the phosphoryl group is formed. Finally, the stretching vibration peak of C―OH changed from 1051.29 cm-1 to 1113.98 cm-1. The peak strength decreased because of the esterification reaction. According to the above analysis, phosphoric acid groups were successfully introduced through phosphate esterification supplemented with alkali and microwave treatment. Moreover, the number of hydroxide radicals increased and new functional groups, such as phosphoryl and ester groups were formed, which enhanced the adsorption capacity of MSSS.
3.4 Cu2+ adsorption experiment of MSSS under different pH values
The Cu2+ removal rate and the adsorption quantity of the MSSS at different pH values are shown in Fig. 4. The results showed that the Cu2+ removal rate and adsorption quantity of MSSS rapidly increased at pH values ranging from 0.7 to 3.0. The Cu2+ removal rate at pH 3.0 reached 86.99% and the adsorption quantity reached 13.05 mg/g. When pH increased to 4.8, the Cu2+ removal rate and the adsorption quantity both reached the highest values, 94.93% and 14.24 mg/g, respectively. These results indicated that MSSS reached adsorption saturation. The Cu2+ removal rate remained stable from pH 3 to 5.
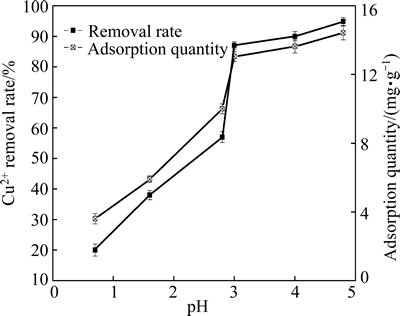
Fig. 4 Cu2+ removal rate and adsorption quantity of MSSS under different pH values
At pH 0.7, the Cu2+ removal rate of MSSS reached 19.66% and its adsorption quantity reached 2.95 mg/g. These results are significantly better than those of peanut shells, rice husks and so on [21,22]. Hence, MSSS can be applied in the treatment of wastewater at the low pH.
3.5 Adsorption time
The Cu2+ removal rate and adsorption quantity of the MSSS with different adsorption time are shown in Fig. 5. In the first 0.5 h of adsorption, the Cu2+ removal rate and adsorption quantity of the MSSS rapidly increased and reached 88.63% and 13.29 mg/g, respectively. When the reaction time exceeded 0.75 h, the Cu2+ removal rate and the adsorption capacity of the MSSS slowly increased and reached adsorption equilibrium, which is convenient for industrial application. If the copper ion concentration of the effluent is not lower than the standard value, the multistage series treatment can be used. Moreover, the MSSS which have been at sorption saturation can be combusted to recover the copper metal.

Fig. 5 Effect of adsorption time on Cu2+ removal
3.6 Adsorption thermodynamics
The Cu2+ adsorption quantity of the MSSS under different temperatures is shown in Fig. 6. The results showed that the Cu2+ adsorption quantity decreased with increasing temperature. However, when the temperature increased from 20 °C to 40 °C, the adsorption quantity of MSSS decreased by 1.98 mg/g, which indicates that temperature has minimal effect on the Cu2+ adsorption of the MSSS.
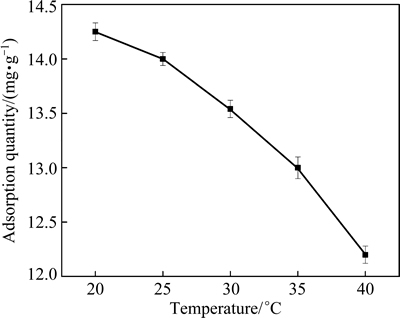
Fig. 6 Cu2+ adsorption quantity of MSSS under different temperatures
Studies on the adsorption thermodynamics achieved the regular trend, intensity, and driving force in the adsorption process. Therefore, calculating the adsorption experiment results by the variations of standard thermodynamic functions is a vital part in adsorption thermodynamics. The relationships among thermodynamic functions △GΘ, △HΘ, and △SΘ, and equilibrium adsorption distribution coefficient Kd are shown as [23]:
 (3)
(3)
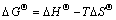 (4)
(4)
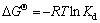 (5)
(5)
The equilibrium adsorption distribution coefficient Kd could be calculated using
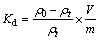 (6)
(6)
where Kd is the equilibrium adsorption distribution coefficient, △GΘ is the standard free energy of adsorption, △HΘ is the standard adsorption enthalpy, △SΘ is the standard adsorption entropy change, R is the ideal gas constant, T is the temperature, V is the solution volume, and m is adsorbent dosage, ρ0 is the initial Cu2+ concentration, ρt is the remaining Cu2+ concentration after adsorption.
Table 1 Thermodynamics parameters of MSSS for Cu2+adsorption
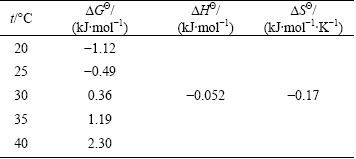

Fig. 7 SEM-EDS results of MSSS before and after Cu2+ adsorption
Kd, △GΘ, △HΘ and △SΘ were calculated based on MSSS adsorption thermodynamics experiment results of Cu2+ at different temperatures. The thermodynamics parameters for Cu2+ adsorption of the MSSS are shown in Table 1. The △GΘ was negative at temperatures ranging from 20°C to 25 °C, which indicated that the adsorption was spontaneous. The △GΘ was positive at temperatures ranging from 30 °C to 40 °C, which indicates that the adsorption was not spontaneous beyond a certain temperature and external energy is needed to reach the activation energy. The △HΘ and △SΘ remained negative across the whole temperature scope, which indicated that the adsorption was exothermic and the entropy of the adsorption process greatly reduced.
3.7 SEM-EDS analyses of MSSS before and after Cu2+ adsorption
The results of SEM-EDS analyses of MSSS before and after Cu2+ adsorption is shown in Fig. 7. After adsorption, the pores on the surface of MSSS were occupied. The MSSS contained C and O, as well as Na, Mg, Al, Si, P, Ca and Cu, before and after adsorption.
The results of the surface spectral analysis of MSSS before and after Cu2+ adsorption are shown in Table 2.
Table 2 Surface spectral analysis results of MSSS before and after Cu2+ adsorption
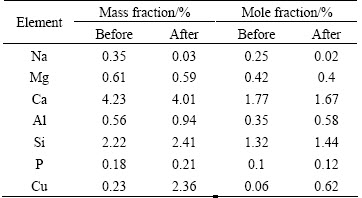
The contents of Na and Mg changed after adsorption, especially Na, which greatly decreased. Therefore, the Cu2+ adsorption by MSSS includes ion-exchange adsorption.
4 Conclusions
1) The MSSS obtained through the phosphate esterification with alkali treatment of the SSS has efficient adsorption performance and high removal efficiency of Cu2+ from wastewater containing heavy metal, even at a low pH. Cu2+ adsorption of MSSS reached equilibrium after 0.75 h.
2) The specific surface area and electronegativity of the MSSS were greater than those of the SSS. Furthermore, the number of hydroxide radicals increased and new functional groups, such as phosphoryl and ester groups formed on the surface of the MSSS. These results indicated that the modification improved adsorption capability of the MSSS.
3) The SEM and EDS analyses of the MSSS before and after Cu2+ adsorption showed that the pores on the surface of MSSS were occupied after adsorption, and the Cu content increased but the Na content decreased. The Cu2+ adsorption thermodynamics experiments showed that the Cu2+ removal by MSSS was exothermic and the entropy of the adsorption reduced greatly.
References
[1]  D, GORGIEVSKI M,
D, GORGIEVSKI M,  N,
N,  N. Adsorption of heavy metal ions by beech sawdust―Kinetics, mechanism and equilibrium of the process [J]. Ecological Engineering, 2013, 58: 202-206.
N. Adsorption of heavy metal ions by beech sawdust―Kinetics, mechanism and equilibrium of the process [J]. Ecological Engineering, 2013, 58: 202-206.
[2] XU M Y, YIN P, LIU X G, TANG Q H, QU R J, XU Q. Utilization of rice husks modified by organo multi phosphonic acids as low-cost absorbents for enhanced adsorption of heavy metal ions [J]. Bioresource Technology, 2013, 149: 420-424.
[3] SAYGIDEGER S, GULNAZ O, ISTIFLI E S, YUCEL N. Adsorption of Cd(II), Cu(II) and Ni(II) ions by Lemna minor L: Effect of physicochemical environment [J]. Journal of Hazardous Material, 2005, 126(1-3): 96-104.
[4] KALIN M, FYSON A, WHEELER W N. The chemistry of conventional and alternative treatment systems for the neutralization of acid mine drainage [J]. Science of the Total Environment, 2006, 366(2-3): 395-408.
[5] YANAGISAWA H, MATSUMOTO Y, MACHIDA M. Adsorption of Zn(II) and Cd(II) ions onto magnesium and activated carbon composite in aqueous solution [J]. Applied Surface Science, 2010, 256(6): 1619-1623.
[6] ZHANG M L. Adsorption study of Pb(II), Cu(II) and Zn(II) from simulated acid mine drainage using dairy manure compost [J]. Chemical Engineering Journal, 2011, 172(1): 361-368.
[7] LUPTAKOVA A, UBALDINI S, MACINGOVA E, FORNARI P, GIULIANO V. Application of physical-chemical and biological- chemical methods for heavy metals removal from acid mine drainage [J]. Process Biochemistry, 2012, 47(11): 1633-1639.
[8]  C A, WILLIAMS C D, ROBERTS C L. Removal of heavy metals from acid mine drainage (AMD) using coal fly ash, natural clinker and synthetic zeolites [J]. Journal of Hazardous Material, 2008, 156(1-3): 23-35.
C A, WILLIAMS C D, ROBERTS C L. Removal of heavy metals from acid mine drainage (AMD) using coal fly ash, natural clinker and synthetic zeolites [J]. Journal of Hazardous Material, 2008, 156(1-3): 23-35.
[9] GUO Xue-yi, LIANG Sha, XIAO Cai-mei, TIAN Qing-hua. Adsorption of heavy metal ions from aqueous solutions by MgCl2 modified orange peel [J]. The Chinese Journal of Nonferrous Metals, 2011, 21(9): 2270-2277. (in Chinese)
[10] LU S G, GIBB S W. Copper removal from wastewater using spent-grain as biosorbent [J]. Bioresource Technology, 2008, 99(6): 1509-1517.
[11]  Adsorption of heavy metals from electroplating wastewater by wood sawdust [J]. Bioresource Technology, 2007, 98(2): 402-409.
Adsorption of heavy metals from electroplating wastewater by wood sawdust [J]. Bioresource Technology, 2007, 98(2): 402-409.
[12] SARIN V, PANT K K. Removal of chromium from industrial waste by using eucalyptus bark [J]. Bioresource Technology, 2006, 97(1): 15-20.
[13] BANSAL M, GARG U, SINGH D, GARG V K. 2009. Removal of Cr(VI) from aqueous solutions using pre-consumer processing agricultural waste: A case study of rice husk [J]. Journal of Hazardous Material, 2009, 162(1): 312-320.
[14] LI X S,LIU S L,NA Z Y, LU D N, LIU Z. Adsorption concentration, and recovery of aqueous heavy metal ions with the root powder ofEichhornia crassipes [J]. Ecological Engineering, 2013, 60: 202-206.
[15] WANG D H, XIANG Q B, CHEN G Y. Research progress of ecological higher value application of spent mushroom compost [J]. Nonferrous Metals Engineering & Research, 2007, 28(23): 262-266.
[16] FAROOP U, KOZINSKI J A, KHAN M A, ATHAR M. Biosorption of heavy metal ions using wheat based biosorbents―A review of the recent literature [J]. Bioresource Technology, 2010, 101(14): 5043-5053.
[17] ZHANG Y S, LIU W G, ZHANG L, WANG M, ZHAO M J. Application of bifunctional Saccharomyces cerevisiae to remove lead(II) and cadmium(II) in aqueous solution [J]. Applied Surface Science, 2011, 257(23): 9809-9816.
[18] TU X, ZENG G M, CHEN G Q. Sorption of Pb2+ in waters by culture medium waste of mushroom [J]. China Environmental Science, 2006, 26(S): 45-47.
[19] TONG X J, LI J Y, YUAN J H, XU R K, ZHOU L X. Adsorption of Cu(II) on rice straw from acidic aqueous solutions [J]. Environmental Chemistry, 2012, 31(1): 64-68.
[20] YUAN H J, QUAN X J, RAN X Z, XIANG J X, JIANG L. Adsorption performance of Camellia cakes as adsorbent for removal of Ni2+ [J]. Chemical Engineering Journal, 2011, 62: 986-993.
[21] WITEK-KROWIAK A, SZAFRAN R G, MODELSKI S. Biosorption of heavy metals from aqueous solutions onto peanut shell as a low-cost biosorbents [J]. Desalination, 2011, 265(1-3): 126-134.
[22] JEON C. Removal of copper ion using rice hulls [J]. Journal of Industrial and Engineering Chemistry, 2011, 17(3): 517-520.
[23] HUO H X, LIN H, DONG Y B, CHENG H, WANG H, CAO L X. Ammonia-nitrogen and phosphates sorption from simulated reclaimed waters by modified clinoptilolite [J]. Journal of Hazardous Material, 2012, 229-230(8): 292-297.
董颖博1,2,林 海1,2
1. 北京科技大学 土木与环境工程学院,北京 100083;
2. 北京科技大学 金属矿山高效开采与安全教育部重点实验室,北京 100083
摘 要:研究改性生物质材料――香菇培养基废料对水溶液中重金属铜离子的吸附性能。结果表明:改性可以大大提高香菇培养基废料的吸附性能,其铜离子去除率高达95%以上;与天然香菇培养基废料相比,改性后吸附材料的比表面积和电负性增大,表面官能团数目及种类增多。在吸附0.75 h后,改性香菇培养基废料对铜离子的吸附达到平衡,该吸附过程是一个放热反应。改性香菇培养基废料吸附铜离子前后的扫描电镜和能谱分析表明,吸附铜离子的材料表面可明显看到孔隙被占据,且铜含量明显增加,而钠含量减少。
关键词:生物质材料;香菇培养基废料;改性;吸附;重金属;Cu2+
(Edited by Yun-bin HE)
Foundation item: Project (51204011) supported by the National Natural Science Foundation of China; Project (20121000803) supported by the Foundation of the Advisor of Beijing Excellent Doctoral Dissertation of China; Project (2013T60063) supported by the China Postdoctoral Science Foundation
Corresponding author: Hai LIN; Tel: +86-10-62332526; E-mail: linhai@ces.ustb.edu.cn
DOI: 10.1016/S1003-6326(15)63689-5

