�����ȴ�����-�����Ʊ�����Ti-Alϵ�����仯������
��Դ�ڿ����й���ɫ����ѧ����Ӣ�İ棩2011���8��
�������ߣ����岨 ��ҵ�� �ŵ� ������ ����� ������
����ҳ�룺1722 - 1727
�ؼ��ʣ����㸴�ϲ��ϣ�Ti-Alϵ�����仯������������ӷ�Ӧ�������������˻�
Key words��multilayered composites; Ti-Al based intermetallics; self-propagating high-temperature synthesis; voids; cold rolling; annealing
ժ Ҫ��ͨ���Ѳ������������ս��Ʊ�������Ti-Alϵ�����仯����Ͻ��ġ��Բ�ͬ�ս������»�õİ����֯������ɽ��з�����������������ս��¶ȵ��ﵽAl���۵�����ʱ�����������ӷ�Ӧ(SHS)��Ti����Al��֮�䷢����������-Ti��Ti3Al��TiAl��TiAl2��TiAl3���ࣻ�����ս�ʱ����ӳ�����-Ti��TiAl2��TiAl3����ʧ�����ջ�ð���Ti3Al��TiAl�ĵ���ṹ��ġ����������ۻ����¿ϴ��ЧӦ�ͷ�Ӧǰ����Ħ������ı仯���ս�����в��������Ŀ���������ѹ��������������������ܵĺϽ��ġ�
Abstract:
Multilayered Ti-Al based intermetallic sheets were fabricated by sintering alternately layered titanium and aluminum foils. The microstructure and phase formation of the obtained sheets under different sintering conditions were evaluated by various techniques. The results reveal that when the sintering temperature is above the melting point of aluminum, the self-propagating high-temperature synthesis reaction occurs between Ti and Al, and forms various phases of Ti-based solid solutions including ��-Ti Ti3Al, TiAl, TiAl2 and ��-Ti including TiAl3, etc. When the sintering time increased, Ti-based solid solution, TiAl2 and TiAl3 disappeared gradually, and the sheet containing Ti3Al and TiAl phases in a multilayered structure formed finally. A lot of voids were also observed in the sintered structures, which were caused by the melting Al, Kirkendall effect and the difference of molar volumes between reactants and products. The voids were eliminated and a dense sample was obtained by the following hot press.

SUN Yan-bo, ZHAO Ye-qing, ZHANG Di, LIU Cui-yun, DIAO Hao-yan, MA Chao-li
Key Laboratory of Aerospace Advanced Materials and Performance of Ministry of Education,
School of Materials Science and Engineering, Beihang University, Beijing 100191, China
Received 18 October 2010; accepted 10 December 2010
Abstract: Multilayered Ti-Al based intermetallic sheets were fabricated by sintering alternately layered titanium and aluminum foils. The microstructure and phase formation of the obtained sheets under different sintering conditions were evaluated by various techniques. The results reveal that when the sintering temperature is above the melting point of aluminum, the self-propagating high-temperature synthesis reaction occurs between Ti and Al, and forms various phases of Ti-based solid solutions including ��-Ti Ti3Al, TiAl, TiAl2 and ��-Ti including TiAl3, etc. When the sintering time increased, Ti-based solid solution, TiAl2 and TiAl3 disappeared gradually, and the sheet containing Ti3Al and TiAl phases in a multilayered structure formed finally. A lot of voids were also observed in the sintered structures, which were caused by the melting Al, Kirkendall effect and the difference of molar volumes between reactants and products. The voids were eliminated and a dense sample was obtained by the following hot press.
Key words: multilayered composites; Ti-Al based intermetallics; self-propagating high-temperature synthesis; voids; cold rolling; annealing
1 Introduction
Ti-Al intermetallic compounds have great potential as high temperature structure materials in the applications of automobile and aerospace industries due to their low density, high specific strength, high melting temperature and environmental resistance at elevated temperatures [1-3]. The application of Ti-aluminide sheets is considered to be one of the breakthroughs of practicability of Ti-Al intermetallic compounds [4-5]. However, Ti-aluminide sheets are challenging to manufacture and have not been commercialized for its poor room temperature ductility and fracture toughness [6].
To explore an alternative approach to the conventional sheets production, studies have been recently undertaken to fabricate near-net-shaped sheets by self-propagating high-temperature synthesis (SHS) reaction using elemental Ti and Al foils [7-10]. This technique of forming intermetallic materials has the advantage that the starting materials are pure metals and therefore it is easy to fabricate near-net-shaped products. Otherwise, the reactions are sustained with the reaction front propagating through the unreacted materials by the exothermic heat of SHS, so a simplified fabrication and energy conserving process could be expected. Moreover, this method could form multilayered Ti-Al intermetallic composites [7]. The multilayered structures are designed based on the small spacing of layers and multi-interfacial effects to improve the impact and fracture resistance of materials, as a result, the materials would be insensitive to the original cracks [11-12]. Another advantage of using foils is that it could reduce the oxygen content of Ti-Al intermetallics made by powder processing, which might limit the usefulness of Ti-Al intermetallics [13-15].
For such a technological need, further study was carried out in this work on the fabrication of multilayered Ti-Al based intermetallic sheets by SHS after stacking thin Ti and Al foils, alternately. In order to investigate the phase formation and growth behaviors, several kinds of multilayered sheets were prepared by varying the heat treatment condition. The mechanism of voids formation was detailedly discussed. Hot press was applied at last for minimizing voids formed during SHS and diffusive reactions. Thus, dense Ti3Al/TiAl multilayered composites were obtained finally.
2 Experimental
Elemental Ti foils (purity 99.8%, thickness 0.045 mm) and elemental Al foils (purity 99.99%, thickness 0.027 mm) were employed. The foils were cleaned ultra-sonically in methanol and cut into 11.5 cm in width before preparing Ti/Al multilayers. Ti/Al multilayer was firstly prepared by alternately stacking one elemental Ti foil and one elemental Al foil, and then convolved it into scroll shape. The scroll was inserted into a Ti tube (purity 99.8%; d15.9 mm (outer diameter) �� 150 mm, wall thickness 0.6 mm), which was used to keep the scroll shape and prevent the sample from oxidation and leakage of Al when it was heated above the melting point of Al (660 ��C). Next, the tube filled with scroll would be rolled several passes at room temperature, which could enhance the Ti/Al interfacial bonding and break the oxide films of the foils. Three samples prepared as above were then respectively sealed into vacuum quartz tubes to prevent oxidation during heat treatments. The samples were subjected to heat treatment at 950 ��C for 30 min, 2 h and 24 h in the tube furnace, respectively. Hot press was subjected to the sample annealed at 950 ��C for 24 h. Hot press process was conducted under the compression stress of 35 MPa at 1 000 ��C for 4 h in a hot-press furnace. The furnace was first evacuated and then backfilled with argon to provide an inert atmosphere.
After heat treatments and hot press, the samples were sectioned and inlaid with epoxy resin. After the resin was solidified, the samples were prepared as a metallographic specimen through grinding with metallographic abrasive papers and then polishing. The microstructures and compositions were characterized by JXA-8100 electron probe microanalyzer (EPMA) equipped with an OXFord INCApentaFET��3 energy dispersive spectroscope (EDS). Phases formed inside multilayers were further confirmed by a D/max-IIIA X-ray diffraction instrument (Cu Target, 40 kV, 40 mA). Micro-hardness was measured on the hot pressed sample using a Leco M-400-Hl hardness testing machine with a Vickers indenter.
3 Results and discussion
3.1 Microstructure and phase formation after heat treatment
Figure 1(a) shows the microstructure of the as-cold rolling Ti/Al multilayered samples without heat treatment. As shown in Fig. 1(a), white regions and dark regions represent Ti layers and Al layers, respectively. After cold rolling, the thickness of Ti foils is about 30 ��m, but the decrement of Al foils is not homogeneous with the thickness range of 17-25 ��m. Besides, Ti/Al interfacial bonding is good and smooth, and no cracks and interfacial phases are observed.
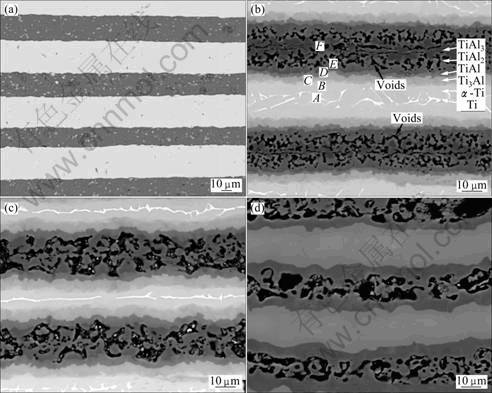
Fig. 1 EPMA back scattered electron images of as-processed sheets: (a) As-cold rolling without heat treatment; (b) Heat treatment at 950 ��C for 30 min; (c) Heat treatment at 950 ��C for 2 h; (d) Heat treatment at 950 ��C for 24 h
The microstructure of the samples held at 950 ��C for 30 min, 2 and 24 h after cold rolling are shown in Figs. 1(b)-(d), respectively. In Fig. 1(b), it is seen that the microstructure consists of six regions. Figure 2(a) presents the XRD pattern of the sample annealed at 950 ��C for 30 min. Peaks of Ti, Ti3Al, TiAl, as well as TiAl2 and TiAl3 are observed. The six points denoted on the sample in Fig. 1(b) were analyzed by EDS to compare the compositions, and the result of EDS analysis is shown in Table 1. In Ti-Al binary system[16], six kinds of phases appear depending on the compositions and temperatures, as shown in Fig. 3, namely Ti based �� solid solution, Ti3Al, TiAl, TiAl2, TiAl3 and Al based solid solution. According to the Ti-Al phase diagram, the results of XRD and EDS indicate that Ti, Ti based �� solid solution, Ti3Al, TiAl, TiAl2 and TiAl3 are aligned in order from white regions to dark regions, as marked in Fig. 1(b). No aluminum is detected. Comparison of the positions of these six regions in Fig. 1(b) with those of the Ti and Al foils in Fig. 1(a) suggests that: 1) aluminum foils are replaced with voids and dark gray regions, 2) all the voids are shown in the starting aluminum layers and 3) the starting titanium foils become thinner and only a few pure Ti are left.

Fig. 2 XRD patterns of multilayered sheets: (a) Heat treatment at 950 ��C for 30 min; (b) Heat treatment at 950 ��C for 2 h; (c) Heat treatment at 950 ��C for 24 h
Table 1 EDS analysis of positions in Fig. 1(b)


Fig. 3 Phase diagram of Ti-Al system
Figures 1(c) and (d) are BEI images of the samples held at 950 ��C for 2 and 24 h, respectively. The XRD patterns of the two specimens are shown in Figs. 2(b) and (c), respectively. Figure 2(b) shows the peaks of Ti, Ti3Al, TiAl as well as TiAl2. Figure 2(c) shows just the peaks of Ti3Al and TiAl. Compared to Fig. 1(b), the phases in Fig. 1(c) should be Ti, Ti based �� solid solution, Ti3Al, TiAl, and TiAl2 aligned in order from white region to gray region. The phases in Fig. 1(d) should be Ti3Al and TiAl, represented with light gray region and dark gray region, respectively. The voids observed in Fig. 1(b) coarsen accordingly to the heat treatment condition, as shown in Figs. 1(c) and (d).
3.2 Reactive process according to soaking time
As observed in Fig. 1(b), when the multi-stacked Ti/Al sheets are held at 950 ��C for 30 min, the phases of Ti/��-Ti/Ti3Al/TiAl/TiAl2/TiAl3 form. With the heat treatment time extends to 2 h (Fig. 1(c)), TiAl3 disappears, and Ti as well as Ti based �� solid solution becomes thinner. At the same time, Ti3Al, TiAl and TiAl2 phases all become thicker. While the soaking time increases to 24 h, just Ti3Al and TiAl are left, as shown in Fig. 1(d).
In the experiment, the initial temperature of 950 ��C is enough for the self-propagating high-temperature synthesis (SHS) reaction occurring between solid Ti and melting Al, according to CHE and FAN [17]. TiAl3 would form firstly, and then the solid-state reaction is dominated by the diffusive process in the Ti-TiAl3 couple, so Ti3Al, TiAl and TiAl2 are formed between Ti and TiAl3 (Fig 1(b)). After all the liquid Al is consumed, competing diffusion happens among Ti/��-Ti/Ti3Al/TiAl/ TiAl2/TiAl3, and the thickness of Ti3Al, TiAl and TiAl2 increases accompanying with the absence of TiAl3 (Fig. 1(c)). When TiAl3 is exhausted, Ti, ��-Ti and TiAl2 start to decrease, while Ti3Al and TiAl keep increasing. Finally, Ti3Al/TiAl multilayer is fabricated after 24 h heat treatment, as shown in Fig. 1(d).
The process of the solid-solid diffusion reaction can be described as follows: diffusion of Al atoms in TiAl3 into the Ti layer leads to the converting of Ti layer into Ti based �� solid solution layer and Ti3Al layer. Meanwhile, diffusion of Ti atoms into the TiAl3 layer leads to converting of part of the TiAl3 phase near the original Ti layer into TiAl layer. Accordingly with the present of TiAl, TiAl2 forms between TiAl layer and TiAl3 layer. With further interdiffusion between Al and Ti atoms, the TiAl3 layer and Ti layer become thinner and thinner. Meanwhile, the Ti3Al layer and TiAl layer become thicker and thicker. However, the amount of TiAl2 layer becomes thicker at first, and then become thinner when entire TiAl3 layer disappears[18]. With a longer heat treatment time, TiAl will grow accompanying with decreasing TiAl2, and eventually a multilayered sheet of Ti3Al and TiAl is achieved.
3.3 Void formation
As shown in Figs. 1(b)-(d), many voids are observed in original Al layers and no voids are seen in original Ti layers. With prolonging soaking time, voids grow up and coarsen. In the present study, it is concluded that the voids are mainly caused by the melting Al, Kirkendall effect and the difference of molar volumes between reactants and products.
In the study, the annealing temperature is 950 ��C which is much higher than the melting point of aluminum (660 ��C). Otherwise, the emission of the reactive heat by melting Al and solid Ti could make the maximum temperature reach 1 350-1 400 ��C [19]. So Al would be totally melted at the beginning of the reaction. With the appearance of melting Al, WANG et al [14] recognized that swelling takes place during the heating of Ti-Al systems caused by flowing away of the melting Al and increases with an increasing amount of Al. In addition, there is a sintering effect at temperature higher than 1 000 ��C in the system; and the shrinkage of the sample takes place. With the swelling and shrinkage, many voids would be generated in the procedure. So, the appearance of the melting Al is one of the causes of voids formation.
Secondly, void formation as an accompanying phenomenon also takes place due to the great diffusion discrepancy between Ti and Al elements in intermediate phases such as TiAl3. This is the so-called Kirkendall effect which has been found in several intermetallic compounds[20]. JIANG et al [21] investigated the voids formation mechanism using Ti/Al diffusion couple. The results show that there is a directional net mass flow along the diffusion direction of Al element since Al atoms diffuse faster than Ti atoms. Moreover, the realization of atom diffusion is through the exchange between atoms and vacancies instead of the direct exchange between atoms. Otherwise, the situation of Ti-Al system is different from other cases. As shown in Fig. 3, Ti forms a solid solution with Al in a wide composition range, while almost no solid solution area exists in pure Al or TiAl3 sides. This means that Al can diffuse into Ti phase, while Ti can hardly diffuse into pure Al or TiAl3 regions. So the vacancies contributing to the diffusion of Al into Ti region from Al and TiAl3 regions are not available for the diffusion of Ti into Al and TiAl3 regions. Consequently, there must be a directionally net vacancy flow balanced with the above-mentioned mass flow. Thus, the vacancies are expected to be accumulated throughout the Al and TiAl3 regions. With the continuation of the diffusion reaction of Ti and Al elements, the vacancy concentration is saturated. To reduce the system energy, these excessive vacancies collapse into voids preferentially at the sites of defects such as grain boundaries and dislocations. As a result, a large number of Kirkendall voids generated in the original Al layer, as shown in Figs. 1(b)-(d).
Finally, the difference of molar volume between reactants and products is another cause of void formation. It was supposed that there was no mass change after reaction, YI et al [13] calculated that the ratio of the molar volume of the reactants and products was 0.911 for TiAl, 0.932 for TiAl3 and 0.969 for Ti3Al. This means that, for example, if there are no dimensional changes after reaction, the relative product volume would be 0.911Vr (Vr is the volume of reactants) for TiAl, which means there is 8.9 % porosity attributable to the molar volume difference between Ti + Al and TiAl. As a result, there are many voids caused by the difference of molar volume between reactants and products at the end of the reaction. Therefore, the technique of hot press is necessary to be applied during the reaction synthesis in order to eliminate the voids.
3.4 Microstructural changes after hot pressing
Figure 4(a) shows the microstructure of sample hot pressed under compressive stress of 35 MPa and 1 000 ��C for 4 h. As observed in Fig. 4(a), only two layers are achieved finally, and the voids decrease significantly. The X-ray diffraction pattern of this specimen is presented in Fig. 4(b), and Ti3Al and TiAl peaks are observed. The energy dispersive spectroscopy (EDS) line profile in Fig. 4(a) indicates more detection of Al in dark gray layers and whereas more Ti in light gray layers, so the two phases in Fig. 4(a) are identified to be Ti3Al (light gray layers) and TiAl (gray layers).
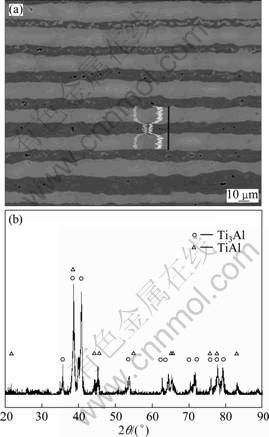
Fig. 4 EPMA back scattered electron image (a) and X-ray diffraction pattern (b) of sample after hot press under 35 MPa and 1 000 ��C for 4 h
Hardness tests were carried out on the hot-pressed sample, where load of 0.49 N was applied for 15 s at least 5 locations for each layer. The micro-hardness values are HV (285��5) on the Ti3Al layers and HV (350��5) on the TiAl layers, respectively. Figure 5 shows the indentation under a load of 9.8 N applied. No cracks are observed around the indentation and some plastic deformation could be seen. This suggests the multilayered materials possess a certain plasticity and toughness.

Fig. 5 Indentation with load of 9.8 N
4 Conclusions
1) The SHS reaction occurs between the solid Ti and the melting Al when the annealing temperature is above the melting point of Al. TiAl3 forms firstly, then the solid-state reaction is dominated by the diffusive process among Ti/��-Ti/Ti3Al/TiAl/TiAl2/TiAl3, until Ti3Al/TiAl multilayer is fabricated finally.
2) Many voids are fabricated in original Al layers. The voids are mainly caused by melting Al, Kirkendall effect and difference of molar volumes between reactants and products.
3) The voids are significantly decreased by hot press, and the dense multilayered Ti3Al/TiAl sheet is finally obtained. Hardness tests show that the multilayered materials possess a certain plasticity and toughness.
Acknowledgment
The authors would like to thank Prof. ZHOU Wan-cheng of Northwestern Polytechnical University of China for parts of the experiments carried out in his laboratory.
References
[1] KIM Y W. Intermetallic alloys based on gamma titanium aluminide [J]. JOM, 1989, 41(7): 24-30.
[2] DIMIDUK D M. Gamma titanium aluminide alloys-an assessment within the competition of aerospace structural materials[J]. Mater Sci Eng A, 1999, 263(2): 281-288.
[3] WU Xin-hua. Review of alloy and process development of TiAl alloys[J]. Intermetallics, 2006, 14(10-11): 1114-1122.
[4] CLEMENS H. Intermetallic ��-TiAl based alloy sheet materials- processing and mechanical properties[J]. Zeitschrift Fur Metallkunde, 1995, 86(12): 814-822.
[5] DRAPER S L, KRAUSE D, LERCH B, LOCCI I E, DOEHNERT B, NIGAM R, DAS G, SICKLES P, TABERNIG B, REGER N, RISSBACHER K. Development and evaluation of TiAl sheet structures for hypersonic applications[J]. Mater Sci Eng A, 2007, 464(1-2): 330-342.
[6] HANAMURA T, HASHIMOTO K. Ductility improvement of direct-cast gamma TiAl-based alloy sheet[J]. Materials Transactions, JIM, 1997, 38(7): 599-606.
[7] RAWERS J C, MAUPIN H E. Metal-intermetallic composites formed by reaction-sintering metal foils[J]. Journal of Materials Science Letters, 1993, 12(9): 637-639.
[8] FUKUTOMI H, MASAYASU U, NAKAMURA M, SUZUKI T, KIKUCHI S. Production of TiAl sheet with oriented lamellar microstructure by diffusional reaction of aluminum and textured titanium foils[J]. Materials Transactions, JIM, 1999, 40(7): 654-658.
[9] OH J, LEE W C, PYO S G, PARK W, LEE S, KIM N J. Microstructural analysis of multilayered titanium aluminide sheets fabricated by hot rolling and heat treatment[J]. Metallurgical and Materials Transactions A, 2002, 33(12): 3649-3659.
[10] LUO Jian-guo, ACOFF V L. Using cold roll bonding and annealing to process Ti/Al multi-layered composites from elemental foils[J]. Mater Sci Eng A, 2004, 379(1-2): 164-172.
[11] WAS G S, FOECKE T. Deformation and fracture in microlaminates[J]. Thin Solid Films, 1996, 286(1-2): 1-31.
[12] SURESH S. Modeling and design of multi-layered and graded materials[J]. Progress in Materials Science, 1997, 42(1-4): 243-251.
[13] YI H C, PETRIC A, MOORE J J. Effect of heating rate on the combustion synthesis of Ti-Al intermetallic compounds[J]. Journal of Materials Science, 1992, 27(24): 6797-6806.
[14] WANG G X, DAHMS M, LEITNER G, SCHULTRICH S. Titanium aluminides from cold-extruded elemental powders with Al-contents of 25�C75 at% Al[J]. Journal of Materials Science, 1994, 29(7): 1847-1853.
[15] JAKOB A, SPEIDEL M O. Microstructure and tensile properties of TiAl compounds formed by reactive foil metallurgy[J]. Mater Sci Eng A, 1994, 189(1-2): 129-136.
[16] SCHUSTER J C, PALM M. Reassessment of the binary aluminum-titanium phase diagram[J]. Journal of Phase Equilibria and Diffusion, 2006, 27(3): 255-277.
[17] CHE H Q, FAN Q C. Microstructural evolution during the ignition/quenching of pre-heated Ti/3Al powders[J]. Journal of Alloys and Compounds, 2009, 475(1-2): 184-190.
[18] MA Yan, FAN Qun-cheng, ZHANG Jing-jue, SHI Jian, XIAO Guo-qing, GU Mei-zhuan. Microstructural evolution during self-propagating high-temperature synthesis of Ti-Al system[J]. Journal of Wuhan University of Technology: Materials Science Edition, 2008, 23(3): 381-385.
[19] YANG W Y, WEATHERLY G C. A study of combustion synthesis of Ti-Al intermetallic compounds[J]. Journal of Materials Science, 1996, 31(14): 3707-3713.
[20] FUKUTOMI H, NAKAMURA M, SUZUKI T, TAKGI S, KIKUCHI S. Void formation by the reactive diffusion of titanium and aluminum foils[J]. Materials Transactions, JIM, 2000, 41(9): 1244-1246.
[21] JIANG Yao, DENG Chu-ping, HE Yue-hui, ZHAO Yao, XU Nan-ping, ZOU Jin, HUANG Bai-yun, LIU C T. Reactive synthesis of microporous titanium-aluminide membranes[J]. Materials Letters, 2009, 63(1): 22-24.
���岨����ҵ�࣬�� �ϣ������ƣ�����ң�������
�������պ����ѧ ���Ͽ�ѧ�빤��ѧԺ �����Ƚ���������۽������ص�ʵ���ң����� 100191
ժ Ҫ��ͨ���Ѳ������������ս��Ʊ�������Ti-Alϵ�����仯����Ͻ��ġ��Բ�ͬ�ս������»�õİ����֯������ɽ��з�����������������ս��¶ȵ��ﵽAl���۵�����ʱ�����������ӷ�Ӧ(SHS)��Ti����Al��֮�䷢�������ɦ�-Ti��Ti3Al��TiAl��TiAl2��TiAl3���ࣻ�����ս�ʱ����ӳ�����-Ti��TiAl2��TiAl3����ʧ�����ջ�ð���Ti3Al��TiAl�ĵ���ṹ��ġ����������ۻ����¿ϴ��ЧӦ�ͷ�Ӧǰ����Ħ������ı仯���ս�����в��������Ŀ���������ѹ��������������������ܵĺϽ��ġ�
�ؼ��ʣ����㸴�ϲ��ϣ�Ti-Alϵ�����仯������������ӷ�Ӧ�������������˻�
(Edited by FANG Jing-hua)
Foundation item: Project (2010DFA51650) supported by the Ministry of Science and Technology of China
Corresponding author: MA Chao-li; Tel/Fax: +81-10-82339772; E-mail: machaoli@buaa.edu.cn
DOI: 10.1016/S1003-6326(11)60921-7

