烟气抗硫脱汞材料的研究进展
来源期刊:中南大学学报(自然科学版)2021年第1期
论文作者:王卉 沈畅 沈昊天 吴建飞
文章页码:133 - 144
关键词:脱汞;烟道气;抗硫;活性位;催化剂;吸附剂
Key words:mercury removal; flue gas; SO2 resistance; active site; catalyst; adsorbent
摘 要:综述了近年来烟气抗硫脱汞材料的研究进展,提出抗硫脱汞材料设计的4种方法,分别为活性位保护法、活性位增加法、氧活性位规避法和贵金属催化法。活性位的保护可以利用材料的亲硫性或抗硫性来实现,活性位的增加可以通过表面氧缺陷构造或者气氛调控来实现,氧活性位的规避可以通过抗硫活性位的设计来实现,而贵金属催化剂的抗硫脱汞机理则复杂多变。这4种方法的提出将为抗硫脱汞材料的进一步研发提供参考。
Abstract: The recent research progress on mercury removal materials with SO2 resistance was reviewed. Four methods for the design of mercury removal materials with SO2 resistance were proposed, which were active site protection method, active site increase method, oxygen-containing active site evasion method and noble metal catalysts method. The protection of active sites can be achieved by doping the materials with sulfur affinity or sulfur resistance. The increase of active sites can be achieved by the enrichment of surface oxygen defects or the adjustment of reaction atmosphere. The evasion of oxygen-containing active sites can be achieved through the design of SO2-resistant active sites. The mechanisms of mercury removal with SO2 resistance of noble metal catalysts are complicated which can vary with certain materials. The proposed four methods will provide the theoretical reference for further development of mercury removal materials with SO2 resistance.

DOI: 10.11817/j.issn.1672-7207.2021.01.013
沈畅1, 2,王卉2,沈昊天2,吴建飞2
(1. 南京理工大学 钱学森学院,江苏 南京,210094;
2. 南京理工大学 能源与动力工程学院,江苏 南京,210094)
摘要:综述了近年来烟气抗硫脱汞材料的研究进展,提出抗硫脱汞材料设计的4种方法,分别为活性位保护法、活性位增加法、氧活性位规避法和贵金属催化法。活性位的保护可以利用材料的亲硫性或抗硫性来实现,活性位的增加可以通过表面氧缺陷构造或者气氛调控来实现,氧活性位的规避可以通过抗硫活性位的设计来实现,而贵金属催化剂的抗硫脱汞机理则复杂多变。这4种方法的提出将为抗硫脱汞材料的进一步研发提供参考。
关键词:脱汞;烟道气;抗硫;活性位;催化剂;吸附剂
中图分类号:X511 文献标志码:A 开放科学(资源服务)标识码(OSID)
文章编号:1672-7207(2021)01-0133-11
SHEN Chang1, 2, WANG Hui2, SHEN Haotian2, WU Jianfei2
(1. Tsien Hsue-shen College, Nanjing University of Science and Technology, Nanjing 210094, China;
2. School of Energy and Power Engineering, Nanjing University of Science and Technology,Nanjing 210094, China)
Abstract: The recent research progress on mercury removal materials with SO2 resistance was reviewed. Four methods for the design of mercury removal materials with SO2 resistance were proposed, which were active site protection method, active site increase method, oxygen-containing active site evasion method and noble metal catalysts method. The protection of active sites can be achieved by doping the materials with sulfur affinity or sulfur resistance. The increase of active sites can be achieved by the enrichment of surface oxygen defects or the adjustment of reaction atmosphere. The evasion of oxygen-containing active sites can be achieved through the design of SO2-resistant active sites. The mechanisms of mercury removal with SO2 resistance of noble metal catalysts are complicated which can vary with certain materials. The proposed four methods will provide the theoretical reference for further development of mercury removal materials with SO2 resistance.
Key words: mercury removal; flue gas; SO2 resistance; active site; catalyst; adsorbent
汞是一种具有高挥发性、强神经毒性、持续生物累积性等特性的元素[1-3]。煤燃烧仍是全球最大的人为汞排放源[4]。根据预测,全球汞排放量目前仍呈现增长趋势[5]。为此,世界各国都出台了日趋严格的汞排放标准[6]。然而,目前较为成熟的活性炭喷射脱汞技术仍然存在许多技术性和经济性问题[7-8],因此,烟气汞控制技术的进一步研发具有显著意义[9-11]。对一般的脱汞材料来说,SO2的存在往往会造成其脱汞性能的大幅下降[12-14],降幅最大可达80%[15]。其抑制机理可初步归纳为:SO2的表面吸附(抢占活性位点)和表面氧化(消耗活性位点)[16-19],因此,现有的脱汞材料将无法满足高硫环境下的汞控制要求。而随着技术的发展和环保政策的不断出台,高硫环境下的汞控制问题已得到广泛关注。高SO2环境的非传统烟气的抗硫脱汞技术的研发,已成为汞控制技术发展的共性问题,在碳捕集减排领域[20],富氧燃烧技术[21-22]被认为具有广阔的应用前景和经济优势[23]。然而,循环富氧烟气中的汞控制问题是其无法商业化应用的重要原因[24-26]。由于富氧烟气的循环特性,其中的SO2和Hg0的含量都普遍高于普通空气燃烧烟气的含量[27-28],因此,抗硫脱汞技术的研发对于富氧燃烧技术的商业化应用具有重要意义。在有色金属冶炼烟气处理领域,由于冶炼烟气中含有比普通燃煤烟气更高浓度的SO2和Hg0[29],故应用于该工况下的汞控制技术有着更高的抗硫要求[30-31]。在固体废弃物焚烧烟气处理领域,高SO2浓度也已成为烟气汞控制的一大难题[32]。由于汞控制技术抗硫能力的限制,固体废弃物焚烧烟气的汞排放贡献已达全球人为汞排放的7.27%[33]。综上可得,高硫环境下的汞控制问题,已成为进一步限制汞排放的共性问题,亟须在理论和工程上加以解决。然而,该技术的研制还处在萌芽阶段,相关的理论指导还比较匮乏。本文综述了近期国内外抗硫脱汞材料的研究进展,提出脱汞材料抗硫性能设计的4种方法,分别为活性位保护法、活性位增加法、氧活性位规避法和贵金属催化法,旨在为抗硫脱汞材料的进一步研发提供参考。
1 活性位保护法
脱汞材料的活性位点在高硫环境下会受到SO2的毒化[34-35]。过渡金属氧化物催化材料(如MnOx)能够先行与SO2结合,使其中毒失活[36-39]。为了保护脱汞活性位点,可在体系中设置一种与SO2亲和力更强的保护金属,Cu[40]和Mo[41]是目前研究较多的亲硫保护金属元素。
目前,许多学者利用Cu的亲硫特性成功设计出抗硫脱汞材料。CuO掺杂的催化剂是研究较为广泛的抗硫脱汞材料。CHEN等[42]研究了CuO掺杂的商用SCR催化剂V2O5-WO3/TiO2的抗硫脱汞性能,结果发现500×10-6(体积分数,下同)SO2的加入几乎不影响其脱汞性能,其脱汞效率在200~350 ℃范围内可达90%。LI等[43]研究了CuO改性的CeO2(5)-WO3(9)/TiO2催化剂的抗硫脱汞性能,结果发现CuO可以先行和SO2结合,进而保护CeO2活性位点。YANG等[44]研究了CuO改性MnOx/TiO2催化剂的抗硫脱汞性能,结果发现在1 000×10-6 SO2的作用下,该体系仍可保持90%左右的脱汞效率。YUE等[45]制备了CuaCebZrcO3/γ-Al2O3抗硫脱汞催化剂,结果发现在500×10-6 SO2的作用下,该体系仍可保持80%左右的脱汞效率。当Cu以其他形式存在时也能提高体系的抗硫脱汞性能。ZHANG等[46]制备了CuCl2改性活性焦吸附剂,发现在没有O2参与作用的情况下,800×10-6 SO2可以显著提高该种吸附剂的汞控制能力;加入6% O2后,其汞控制能力进一步增强。其抗硫脱汞机理为:一方面,SO2借助表面活性氧生成了利于汞氧化脱除的SO3;另一方面,由于Cu元素对S元素的亲和力更强,故可以保护吸附剂表面的活性Cl不被SO2破坏。
另有学者研究了掺杂Mo元素后材料的抗硫脱汞性能。LIU等[41]发现,MoO3-CeO2改性圆柱型活性焦可以显著抵抗SO2对脱汞的抑制作用。其机理如图1所示[41]:一方面,MoO3可以先行与SO2反应,从而保护CeO2活性位点;另一方面,SO2也可以促进弱HgO和HgSO4的形成。同时,该种吸附剂也具有很好的再生作用。赵波[47]探究了Mo改性载Mn碳纳米管的抗硫脱汞性能,结果发现在6% O2的环境下,1 000×10-6 SO2能够使汞氧化效率从80%提升到90%,而1 500×10-6 SO2的加入则会使汞氧化效率降至70%。其抗硫机理为:一方面,MoOx能够加快SO2向SO3的转化过程,而SO3在毒化MnOx的同时,也能使Hg0与SO3反应生成稳定的HgSO4;另一方面,与Mn相比,Mo对SO2的亲和力更强,故Mo的存在可以保护Mn的催化活性。
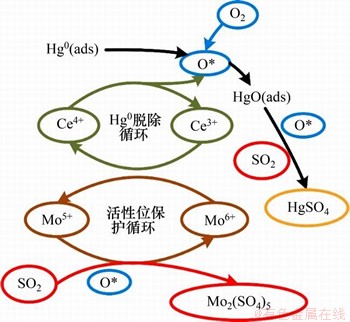
图1 MoO3-CeO2的抗硫脱汞机理[41]
Fig. 1 Mercury removal mechanism of MoO3-CeO2 with SO2 resistance[41]
少数学者采用Fe和Co作为保护元素,设计了抗硫脱汞材料。ZHANG等[48]探究了溶胶-凝胶法制备的Mn-Fe改性的沸石脱汞吸附剂,结果发现在5% O2和600×10-6 SO2气氛作用下,该体系仍可保持80%左右的脱汞效率,而仅用Mn改性的沸石只有50%左右的脱汞效率,这表明Fe的加入能够提高体系的抗硫性能,其原因可能是Fe可以防止SO2侵入Mn2O3内,从而保护了Mn2O3。LI等[49]研究了共沉淀法制备的Co改性CeO2-TiO2脱汞催化剂,结果发现在1 000×10-6 SO2的作用下,该体系仍能保持80%以上的脱汞效率,而当没有加入Co时,CeO2-TiO2体系的脱汞效率仅有40%。这说明Co改性对于该体系的抗硫脱汞性能有着显著的增强,其机理主要为Co可以先行与SO2反应生成CoSO4,从而保护CeO2活性成分。
上述方法主要是利用亲硫金属与SO2的优先反应特性来保护其他脱汞活性位点,但该法在提高体系抗硫脱汞性能的同时,也会降低亲硫金属的催化氧化性能。为了减少催化材料中的活性物质的降低程度,一些学者也尝试采用涂层技术或者表面掺杂技术,降低SO2的表面吸附能,进而提高体系的抗硫脱汞能力。ZHANG等[50]运用SiO2涂层技术降低了催化剂的表面碱性,从而减少了表面SO2的累积。研究发现,500×10-6 SO2的加入仍可使该体系的汞脱除效率保持在80%以上,比同等条件下不加涂层的脱汞效率高15 %左右,这表明SiO2涂层能够增强材料的抗硫脱汞性能。ZHANG等[51]通过DFT研究发现,对MnOx进行Cr掺杂可以有效地降低体系表面的SO2吸附能,进而提高体系的抗硫脱汞性能。实验发现,加入1 500×10-6 SO2后,Cr-Mn脱汞体系的脱汞效率仍然达到94%以上。
综上所述,采用活性位保护法能够显著提高材料的抗硫脱汞性能。目前,活性位的保护主要有2种方法,其一是利用亲硫金属与SO2优先反应的特性来实现(如掺杂Cu、Mo、Fe、Co等亲硫金属元素),其二是通过降低体系表面的SO2的吸附能来实现(如采用SiO2涂层技术或表面Cr掺杂技术等)。
2 活性位增加法
脱汞材料无法在高硫环境下工作,主要是因为其中的活性位点受到SO2的毒化。但若脱汞活性位点数量足够多,则该毒化效应便能在一定条件下转为促进效应。以表面活性氧为例,LI等[52]研究发现:一方面,SO2首先借助吸附剂表面的Lewis酸性位点与表面的化学吸附氧反应,生成SO3;另一方面,Hg0也可以与表面活性氧反应生成HgO,而HgO能够与SO3反应生成稳定的HgSO4,进而促进汞的化学吸附。相关反应可由下式描述[52]:

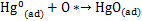



由此可推知:虽然部分研究指出SO2能够消耗表面活性氧[16, 18, 37],进而造成脱汞吸附剂的部分失活,但上述分析表明,如果表面活性氧足够多,那么SO2便可参与并促进汞的化学吸附,进而提升材料的抗硫脱汞性能。
Ce元素能够提高脱汞体系的氧储备能力,进而提高其抗硫脱汞性能[37]。LI等[37]研究了Mn-Ce基材料的抗硫脱汞性能,结果发现当存在6% O2时,该材料对800×10-6 SO2仍有较强的抵抗作用,汞脱除效率保持在90%左右。其抗硫机理可能为,CeO2能够借助氧气补充活性位点。XIE等[53]研究了Mn与Ce物质的量比为1:1的情况,也得到了类似的结论。上述学者的研究表明,添加Ce元素有助于脱汞体系在O2气氛下获得表面活性氧的补充,进而提升材料的抗硫脱汞性能。CHI等[54]研究了Ce-Cu改性的商用V2O5/TiO2基SCR催化剂的抗硫脱汞性能,结果发现7% Ce配合1% Cu可以大幅提高材料的抗硫脱汞性能,5% O2和1 000×10-6 SO2的加入仍可使之保持80%左右的脱汞效率。他们没有给出具体的抗硫机理,但综合以上研究来看,Ce元素的活性氧获取能力可能是其优良抗硫脱汞性能的主导因素。WU等[55]研究了Ce-Mn/TiO2的抗硫脱汞性能,结果发现加入2000×10-6 SO2后,该体系仍然具有80%的脱汞效率。LI等[56]通过DFT方法系统研究了SO2对CeO2的影响,结果发现,一方面SO2生成的SO3可以导致CeO2的表面电子分布重组,进而促进Hg0的氧化;另一方面,由于SO2和Hg0在CeO2表面的吸附位点相同,且SO2和CeO2的结合能大于Hg0和CeO2的结合能,故SO2会首先抢占Hg0的吸附位点。
除Ce元素外,Nb,Cr和Co等元素也能使体系中发生电荷重组和产生氧空位,进而借助O2生成更多的氧活性位点。LIU等[36]研究了Nb改性的MnTiOx SCR催化剂的汞控制性能,研究发现:虽然Nb元素的脱汞能力比Mn元素的弱,但是加入少量2% Nb可以显著提高催化剂脱汞的抗硫性能(其机理如图2所示[36])。在4% O2和100×10-6 SO2作用下,其汞氧化效率仍然达到90%以上。XPS分析结果表明,该体系中的Nb元素主要以Nb5+的形式存在,这将造成体系表面电荷不均匀和产生氧空位,进而借助O2生成更多的氧活性位点,最终提高汞的吸附效率和材料的抗硫特性。CHEN等[57]采用溶胶-凝胶法制备了CrOx改性的MnOx/TiO2脱汞催化剂,结果发现6% O2和600×10-6 SO2的加入几乎不影响其脱汞效率,仍可保持在95%左右。其机理可能是,Cr的加入能够使MnOx/TiO2体系中的氧储备位点增多。GAO等[58]采用超声波浸渍改性法制备了CoOx改性的MnOx生物质活性炭,当有6% O2和900×10-6 SO2的混合模拟气氛存在时,该体系仍然保持近90%的脱汞效率。其机理可能为:当CoOx加入时,CoOx与MnOx发生了相互作用,降低了表面氧空位的生成能,形成了更多的氧缺陷,进而能够借助O2来补充表面活性氧。
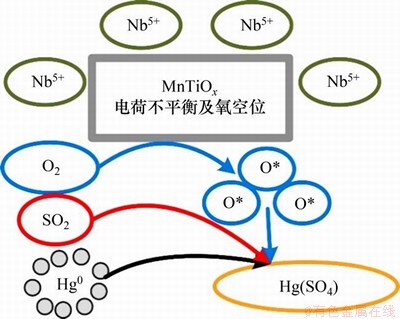
图2 Nb改性MnTiOx的抗硫脱汞机理[36]
Fig. 2 Mercury removal mechanism of Nb modified MnTiOx with SO2 resistance[36]
上述方法主要是通过重组脱汞体系的表面电荷、制造氧缺陷来实现活性位的增加的(如掺杂Ce,Nb,Cr和Co等金属元素,进而借由O2补充表面活性氧)。另一些学者从反应气氛调控的角度,也发现了相应的抗硫脱汞方法。所谓反应气氛调控,即通过调节脱汞体系的工作气氛(如增加活性气体O2,NO和HCl的体积分数),以期在脱汞体系表面制造更多的活性位点,提高其抗硫脱汞能力。YANG等[59]研究了CuAlO2催化剂脱汞体系,结果发现当3000×10-6 SO2单独作用时,汞氧化效率由不加入SO2时的96.1%降至33.8%,其原因可能是SO2消耗了表面的活性成分。而当500×10-6 NO加入时,在(500~3 000)×10-6 SO2范围内,其汞氧化效率均在95%以上。其抗硫脱汞机理为:SO2可能首先在催化剂表面生成Bronsted酸性位点,该酸性位点可以结合NO转变为活性氮氧位点,进而氧化Hg0为Hg(NO3)2。LI等[60]研究了CeO2-WO3/TiO2催化剂的脱汞性能,结果发现在6% O2环境下,添加1×10-7 HCl或者1×10-8 NO后,该体系能够抵抗(500~3 000)×10-6 SO2的毒化,并保持汞氧化效率在90%以上,其机理可能为NO和HCl能够生成可以掩盖SO2不利作用的脱汞氮活性位和氯活性位(图3)。LIU等[61]研究发现,SO2可以抑制CuBr2改性硅藻土吸附剂的汞控制能力,但在4% O2,1 200×10-6 SO2和300×10-6 NO联合作用下,该体系的脱汞效率重新升至93%。其抗硫脱汞机理可能为,当NO加入时,其表面可以生成新的活性位点(如NO+,NO2,
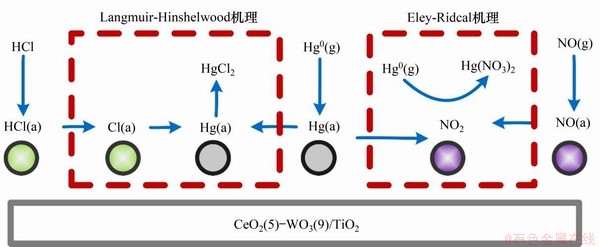
图3 HCl和NO对表面活性位的影响[59]
Fig. 3 Effect of HCl and NO on surface active sites[59]
综上所述,利用活性位增加法增强材料的抗硫脱汞性能主要有2种途径:一种是借助Ce,Nb,Cr和Co等金属元素的掺杂,对脱汞体系表面电荷进行重组、制造氧缺陷,并借助O2来补充表面活性氧;另一种则是通过反应气氛调控,增加活性烟气组分(如O2、NO、HCl)的体积分数,以期在脱汞体系表面创造更多的活性位点,抑制SO2对脱汞的不利影响。
3 氧活性位规避法
含氧官能团对脱汞材料的汞控制能力有重要影响。研究表明羰基、羧基和酯基等含氧官能团能够促进汞的化学吸附[62],而SO2能够消耗脱汞材料表面的含氧官能团[37],造成失活。因此,要使脱汞材料具有抗硫性能,可以规避设计氧活性位,取而代之以其他的抗硫活性位。目前,运用较多的抗硫活性位是硫活性位。硫活性位不会被SO2毒化,与氧活性位氧化Hg0的产物HgO相比,硫活性位能直接氧化Hg0至不与SO2反应的HgS[63],从而提升脱汞材料的抗硫性能。其反应途径[31]为:


HCl和NO对表面活性位的影响[59]见图3。许多学者探究了金属硫化物改性材料的抗硫脱汞性能。LIU等[31-32]研究了用Cu(NO3)和Na2S溶液浸渍改性的CuS活性焦的抗硫脱汞性能,结果发现在5 000×10-6 SO2作用下,该体系的汞吸附能力达到16 mg/g以上。可能的原因是,该体系表面的

金属硫化物改性结合掺杂Co元素工艺,能够进一步提高材料的抗硫脱汞性能。LIU等[68]研究了Co掺杂的ZnS脱汞体系,结果显示,当加入3 500×10-6 SO2时,该体系的汞控制容量仍能达9.53 mg/g。其抗硫脱汞机理可能为:该体系中的活性位点为Co3+和
另一些学者也尝试了使用含硫活性气体H2S或SO2对材料进行硫活化,从而增加材料表面的硫活性位,提升材料的抗硫脱汞能力。KONG等[69]研究发现,H2S改性的Cu/TiO2催化剂具有良好的抗硫脱汞性能。在1% SO2的作用下,仍能保持近100%的脱汞效率,其抗硫脱汞机理如图4所示。ZOU等[63]研究发现,H2S改性的天然铁钛矿石拥有良好的抗硫脱汞性能。HUO等[70]研究发现ZnS可以催化分解H2S,生成活性S*和活性H*,有利于Hg0氧化为HgS,进而可能提升材料的抗硫脱汞性能。SHE等[71]研究了SO2浸渍改性多孔碳的脱汞性能,结果表明,SO2浸渍改性能够为多孔碳表面增加不同的含硫活性位,这可能有利于材料的抗硫脱汞性能。CHEN等[72]研究发现,SO2改性后的多孔碳具有更多含硫活性位,其汞吸附形态主要为HgS,这可能有利于提高材料的抗硫脱汞性能。
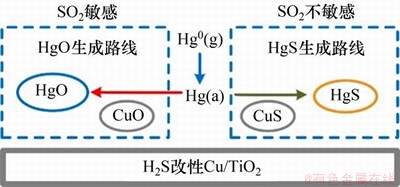
图4 H2S改性Cu/TiO2的抗硫脱汞机理[68]
Fig. 4 Mercury removal mechanism of H2S modified Cu/TiO2 with SO2 resistance[68]
除硫活性位外,Se0活性位也具有抗硫脱汞性能。LIU等[29]研究了Zn-Se-S纳米颗粒体系的抗硫脱汞性能,当加入2 000×10-6 SO2时,其脱汞效率仍可达到80%~90%。研究发现该体系中的活性位点主要为活性Se0和活性S,其抗硫脱汞机理如图5所示。由图5可见:该体系中的活性Se0和活性S不与SO2发生反应,从而避免了活性位点的失活。此外,SO2能够还原Se2+生成Se0,进而与Hg0结合生成稳定的HgSe。
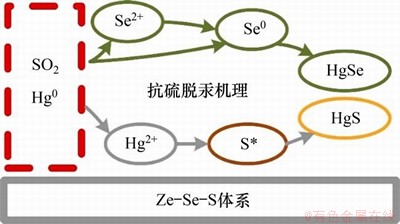
图5 Zn-Se-S体系的抗硫脱汞机理图[29]
Fig. 5 Mercury removal mechanism of Zn-Se-S composite with SO2 resistance[29]
综上可得,氧活性位规避法是目前应用较为广泛的抗硫脱汞材料设计方法,其中硫活性位的设计是目前运用最多的方法。硫活性位的设计方法主要有2大类,其一是通过金属硫化物(如CuS,MoS3和Mn-SnS2等)对材料进行改性,其二是利用含硫活性气体(如H2S和SO2等)对材料进行硫活化。此外,Se0活性位也是一种新型的抗硫脱汞活性位。
4 贵金属催化法
贵金属材料是一类具有良好汞催化氧化性能的材料[73]。这类材料往往具有成本高、活性强、可循环利用等特性;此外,贵金属材料通常在静电除尘器或布袋除尘器的下游工作,因此,其工作温度一般比普通的脱汞材料的工作温度低[74]。在抗硫脱汞材料研究领域,一些贵金属材料(如Au)能够催化分解SO2生成活性硫[75],进而促进Hg0的化学吸附。目前,Au,Ag,Pd和Ru等贵金属已被证明具有抗硫脱汞的性能,但其抗硫脱汞机理相对复杂。
Au元素能够催化分解SO2生成活性硫,进而促进Hg0的化学吸附。GOMEZ-GIMENEZ等[75-76]研究了纳米Au/C可再生吸附剂的脱汞性能,结果发现SO2的加入会使该种吸附剂的汞氧化能力增强。其机理可能为(图6):纳米Au颗粒可能作为催化分解SO2的催化剂,生成活性硫沉积在吸附剂表面,从而促进Hg0的脱除。
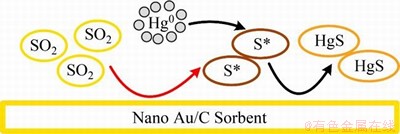
图6 纳米Au/C吸附剂的抗硫脱汞机理[75]
Fig. 6 Mercury removal mechanism of nano Au/C sorbents with SO2 resistance[75]
Ag元素能够在特定环境下,提高材料的抗硫脱汞性能。ZHAO等[77]研究了Ag-Mo改性的SCR催化剂的脱汞性能,结果发现Ag的加入可以使得该体系在500×10-6 SO2作用下的脱汞效率提高10%~20%。该现象可能归结于Ag的加入提高了材料的氧化性。然而,Ag的抗硫脱汞特性与体系的相组成有关,李成伟等[78]发现,SO2能显著抑制Ag/BiOI光催化剂溶液的脱汞效果。这表明在湿法脱汞方法下,Ag的添加并不能显著提高其抗硫脱汞性能。因此,在进行抗硫脱汞技术开发时,体系的相组成也是需要考虑的因素。
Pd作为催化材料,其抗硫性能存在两重特性。YANG等[16]发现浸渍改性制备的Pd/CuCl2/γ-Al2O3催化剂拥有良好的抗硫脱汞性能。Pd和CuCl2是其中的脱汞活性位点。其抗硫机理为:一方面,当加入SO2时,有更为稳定的含硫物质(如SO―OH和SO2―OH)在该体系的表面生成,从而减少了CuSO4的生成,减轻了Hg0和SO2在CuCl2活性位点上的竞争吸附;另一方面,Pd和CuCl2的两相组合,可能促进了表面活性Cl的生成和迁移,克服了SO2的抑制效应。HUO等[79]使用浸渍法制备了Pd/Ce/γ-Al2O3脱汞材料,虽然该种材料的抗SO2特性的试验并未进行,但是他们研究了(150~200)×10-6 H2S气氛下体系的汞吸附形态。通过XPS分析,发现表面存在HgS和PdS。综合上述的研究结果,可以推测:Pd作为催化材料,一方面能够促进体系表面活性位的生成和迁移,另一方面Pd也面临着被毒化成PdS的风险。
Ru元素的抗硫脱汞性能需要配合O2气氛方可实现。张志会等[80]研究发现,RuO2的添加可以提高MnOx/TiO2体系的抗硫脱汞性能,原因可能是RuO2可先行和SO2反应生成RuSO4进而保护活性MnOx。ZHANG等[81]进一步研究发现,RuSO4能够进一步与O2发生反应,生成新的RuO2活性位点。这表明Ru元素的添加可以提高体系的抗硫脱汞性能,但其抗硫能力必须由O2辅助。LIU等[82]也得出了类似的结论。
综上所述,不同的贵金属材料其抗硫脱汞机理不同。Au能够催化分解SO2生成活性S,Ag能够提高体系的氧化性,Pd能够促进体系表面活性位的生成和迁移,Ru则需有O2的配合才能实现抗硫脱汞性能。值得一提的是,虽然贵金属材料有着良好的催化性能,但由于其成本较高,制备难度较大,故目前该种材料尚未在工业上得到应用[16]。但是,贵金属催化材料拥有良好的再生性能,能够实现汞捕集后的循环利用[76]。通过纳米技术,可以在减少贵金属材料用量的同时,实现催化材料的大比表面积特性[75]。故贵金属催化法在烟气抗硫脱汞材料制备方面仍然具有广阔的发展空间。
5 结论
1) 提出了烟气抗硫脱汞材料设计的4种方法,即活性位保护法、活性位增加法、氧活性位规避法和贵金属催化法。
2) 活性位保护法原理如下:一方面可以利用亲硫金属与SO2优先反应的特性来实现(如掺杂Cu,Mo,Fe和Co等亲硫金属元素),另一方面也可以通过降低体系表面的SO2吸附能来实现(如采用SiO2涂层技术或表面Cr掺杂技术等)。
3) 活性位增加法原理如下:一方面可以通过重组脱汞体系的表面电荷、制造氧缺陷来实现(如掺杂Ce,Nb,Cr和Co等金属元素,进而借由O2补充表面活性氧);另一方面也可以通过反应气氛调控来实现(如增加O2,NO和HCl等活性气体的体积分数,进而创造更多的活性位点)。
4) 氧活性位规避法原理如下:一方面可以通过硫活性位的设计来实现(如:利用金属硫化物CuS,MoS3和Mn-SnS2等对材料进行改性,或通过含硫活性气体H2S、SO2等对材料进行硫活化),另一方面也可以通过Se0等抗硫活性位的设计来实现。
5) 贵金属催化法原理如下:Au能够催化分解SO2生成活性S,Ag能够提高体系的氧化性,Pd能够促进体系表面活性位的生成和迁移,Ru则需有O2的配合才能实现其抗硫脱汞性能。
参考文献:
[1] O'CONNOR D, HOU D, OK Y S, et al. Mercury speciation, transformation, and transportation in soils, atmospheric flux, and implications for risk management: a critical review[J]. Environment International, 2019, 126: 747-761.
[2] PLETZ J, SáNCHEZ-BAYO F, TENNEKES H A. Dose-response analysis indicating time-dependent neurotoxicity caused by organic and inorganic mercury: implications for toxic effects in the developing brain[J]. Toxicology, 2016, 347(10): 1-5.
[3] MCKINNEY M A, PEDRO S, DIETZ R, et al. A review of ecological impacts of global climate change on persistent organic pollutant and mercury pathways and exposures in arctic marine ecosystems[J]. Current Zoology, 2015, 61(4): 617-628.
[4] WICHLINSKI M, WIELGOSZ G, KOBYLECKI R. The effect of circulating fluidized bed boiler load on the emission of mercury[J]. Journal of the Energy Institute, 2019, 92(6): 1800-1806.
[5] STREETS D G, HOROWITZ H M, LU Z, et al. Global and regional trends in mercury emissions and concentrations, 2010-2015[J]. Atmospheric Environment, 2019, 201: 417-427.
[6] 吴清茹, 赵子鹰, 杨帆, 等. 中国燃煤电厂履行《关于汞的水俣公约》的差距与展望[J]. 中国人口资源与环境, 2019, 29(10): 52-60.
WU Qingru, ZHAO Ziying, YANG Fan, et al. Gaps and prospects for the implementation of minamata convention on mercury by China′scoal-fired power plants[J]. China Population, Resources and Environment, 2019, 29(10): 52-60.
[7] ZHOU Qiang, TAO Xin, LEI Yibo, et al. Effect of molybdenum incorporation on the activity of a magnetic Fe-Mn sorbent for the capture of elemental mercury[J]. Energy & Fuels, 2019, 33(3): 2390-2398.
[8] ZHOU Qiang, LEI Yu, LIU Yibo, et al. Gaseous elemental mercury removal by magnetic Fe-Mn-Ce sorbent in simulated flue gas[J]. Energy & Fuels, 2018, 32(12): 12780-12786.
[9] YANG Jianping, LI Qin, LI Min, et al. In situ decoration of selenide on copper foam for the efficient immobilization of gaseous elemental mercury[J]. Environmental Science & Technology, 2020, 54(3): 2022-2030.
[10] YANG Jianping, ZHU Wenbing, QU Wenqi, et al. Selenium functionalized metal-organic framework MIL-101 for efficient and permanent sequestration of mercury[J]. Environmental Science & Technology, 2019, 53(4): 2260-2268.
[11] ZHAO S L, PUDASAINEE D, DUAN Y F, et al. A review on mercury in coal combustion process: content and occurrence forms in coal, transformation, sampling methods, emission and control technologies[J]. Progress in Energy and Combustion Science, 2019, 73: 26-64.
[12] WANG Hui, SHEN Chang, DUAN Yufeng, et al. Synergistic effect between H2O and SO2 on mercury removal by activated carbon in O2/CO2 conditions[J]. Journal of Chemical Technology and Biotechnology, 2019, 94(4): 1195-1201.
[13] TANG Hongjian, YOU Wenqi, WANG Zewei, et al. Detrimental effects of SO2 on gaseous mercury(II) adsorption and retention by CaO-based sorbent traps: competition and heterogeneous reduction[J]. Journal of Hazardous Materials, 2020, 387: 121679.
[14] ZHOU Qiang, DUAN Yufeng, CHEN Mingming, et al. Effect of flue gas component and ash composition on elemental mercury oxidation/adsorption by NH4Br modified fly ash[J]. Chemical Engineering Journal, 2018, 345: 578-585.
[15] LI Yaning, DUAN Yufeng, WANG Hui, et al. Effects of acidic gases on mercury adsorption by activated carbon in simulated oxy-fuel combustion flue gas[J]. Energy & Fuels, 2017, 31(9): 9745-9751.
[16] YANG Shu, LIU Cao, LIU Zhilou, et al. High catalytic activity and SO2-poisoning resistance of Pd/CuCl2/γ-Al2O3 catalyst for elemental mercury oxidation[J]. Catalysis Communications, 2018, 105: 1-5.
[17] 周强, 段钰锋, 冒咏秋, 等. NH4Cl改性活性炭脱除气态Hg0的特性分析[J]. 化工进展, 2018, 37(10): 4068-4073.
ZHOU Qiang, DUAN Yufeng, MAO Yongqiu, et al. Gaseous Hg0 removal by NH4Cl modified activated carbon [J]. Chemical Industry and Engineering Progress, 2018, 37(10): 4068-4073.
[18] WANG Yan, LIU Yangxian, SHI Shou. Removal of nitric oxide from flue gas using novel microwave-activated double oxidants system[J]. Chemical Engineering Journal, 2020, 393: 124754.
[19] YANG Yang, XU Wenqing, WANG Jian, et al. New insight into simultaneous removal of NO and Hg0 on CeO2-modified V2O5/TiO2 catalyst: a new modification strategy[J]. Fuel, 2019, 249: 178-187.
[20] LI Hailong, QU Mingyu, HU Yingchao. Preparation of spherical Li4SiO4 pellets by novel agar method for high-temperature CO2 capture[J]. Chemical Engineering Journal, 2020, 380: 122538.
[21] WU Ying, LIU Daoyin, DUAN Lunbo, et al. Three-dimensional CFD simulation of oxy-fuel combustion in a circulating fluidized bed with warm flue gas recycle[J]. Fuel, 2018, 216: 596-611.
[22] WU Ying, LIU Daoyin, ZHENG Dong, et al. Numerical simulation of circulating fluidized bed oxy-fuel combustion with dense discrete phase model[J]. Fuel Processing Technology, 2019, 195: 106129.
[23] ANDERSSON V, FRANCK P-y, BERNTSSON T. Techno-economic analysis of excess heat driven post-combustion CCS at an oil refinery[J]. International Journal of Greenhouse Gas Control, 2016, 45: 130-138.
[24] WANG Fumei, SHEN Boxiong, YANG Jiancheng, et al. Review of mercury formation and capture from CO2-enriched oxy-fuel combustion flue gas[J]. Energy & Fuels, 2017, 31(2): 1053-1064.
[25] WANG Hui, WANG Shen, DUAN Yufeng, et al. Activated carbon for capturing Hg in flue gas under O2/CO2 combustion conditions. Part 1: experimental and kinetic study[J]. Energy & Fuels, 2018, 32(2): 1900-1906.
[26] WANG Hui, WANG Shen, DUAN Yufeng, et al. Activated carbon for capturing Hg in flue gas under O2/CO2 combustion conditions. Part 2: modeling study and adsorption mechanism[J]. Energy & Fuels, 2018, 32(2): 1907-1913.
[27] SARBASSOV Y, DUAN L, JEREMIAS M, et al. SO3 formation and the effect of fly ash in a bubbling fluidised bed under oxy-fuel combustion conditions[J]. Fuel Processing Technology, 2017, 167: 314-321.
[28] CHAMBERLAIN S, REEDER T, STIMPSON C K, et al. A comparison of sulfur and chlorine gas species in pulverized-coal, air- and oxy-combustion[J]. Combustion and Flame, 2013, 160(11): 2529-2539.
[29] LIU Wei, XU Haomiao, GUO Yongfu, et al. Immobilization of elemental mercury in non-ferrous metal smelting gas using ZnSe1-xSx nanoparticles[J]. Fuel, 2019, 254: 115641.
[30] QUAN Zongwen, HUANG Wenjun, LIAO Yong, et al. Study on the regenerable sulfur-resistant sorbent for mercury removal from nonferrous metal smelting flue gas[J]. Fuel, 2019, 241: 451-458.
[31] LIU Wei, XU Haomiao, LIAO Yong, et al. Recyclable CuS sorbent with large mercury adsorption capacity in the presence of SO2 from non-ferrous metal smelting flue gas[J]. Fuel, 2019, 235: 847-854.
[32] LIU Wei, ZHOU Yongxian, HUA Yinfeng, et al. A sulfur-resistant CuS-modified active coke for mercury removal from municipal solid waste incineration flue gas[J]. Environmental Science and Pollution Research, 2019, 26(24): 24831-24839.
[33] United Nations Environment Programme. Global mercury assessment 2018[R].
[34] SHEN Haotian, WANG Hui, SHEN Chang, et al. Effect of atmosphere of SO2 coexisted with oxidizing gas on mercury removal under oxy-fuel condition[J]. Chemosphere, 2020, 127525.
[35] YANG Jianping, ZHU Wenbing, ZHANG Shibo, et al. Role of flue gas components in Hg0 oxidation over La0.8Ce0.2MnO3 perovskite catalyst in coal combustion flue gas[J]. Chemical Engineering Journal, 2019, 360: 1656-1666.
[36] LIU Jian, GUO Ruitang, GUAN Zhenzhen, et al. Simultaneous removal of NO and Hg0 over nb-modified MnTiOx catalyst[J]. International Journal of Hydrogen Energy, 2019, 44(2): 835-843.
[37] LI Honghu, WANG Yuan, WANG Shengkai, et al. Removal of elemental mercury in flue gas at lower temperatures over Mn-Ce based materials prepared by co-precipitation[J]. Fuel, 2017, 208: 576-586.
[38] XU Haomiao, ZHANG Hongbo, ZHAO Songjian, et al. Elemental mercury(Hg0) removal over spinel LiMn2O4 from coal-fired flue gas[J]. Chemical Engineering Journal, 2016, 299: 142-149.
[39] MENG Jialin, DUAN Yufeng, HU Peng, et al. Simultaneous removal of elemental mercury and NO from simulated flue gas at low temperatures over Mn-V-W/TiO2 catalysts[J]. Energy & Fuels, 2019, 33(9): 8896-8906.
[40] YANG Ru, MEI Congli, WU Xiushan, et al. Mn-Cu binary metal oxides with molecular-scale homogeneity for Hg0 removal from coal-fired flue gas[J]. Industrial & Engineering Chemistry Research, 2019, 58(41): 19292-19301.
[41] LIU Miao, LI Caiting, ZENG Qiang, et al. Study on removal of elemental mercury over MoO3-CeO2/cylindrical activated coke in the presence of SO2 by Hg-temperature-programmed desorption[J]. Chemical Engineering Journal, 2019, 371: 666-678.
[42] CHEN Chuanmin, JIA Wenbo, LIU Songtao, et al. Simultaneous NO removal and Hg0 oxidation over CuO doped V2O5-WO3/TiO2 catalysts in simulated coal-fired flue gas[J]. Energy & Fuels, 2018, 32(6): 7025-7034.
[43] LI Guoliang, WU Qingru, WANG Shuxiao, et al. Promoting SO2 resistance of a CeO2(5)-WO3(9)/TiO2 catalyst for Hg0 oxidation via adjusting the basicity and acidity sites using a CuO doping method[J]. Environmental Science & Technology, 2020, 54(3): 1889-1897.
[44] YANG Zequn, LI Hailong, LIU Xi, et al. Promotional effect of CuO loading on the catalytic activity and SO2 resistance of MnOx/TiO2 catalyst for simultaneous NO reduction and Hg0 oxidation[J]. Fuel, 2018, 227: 79-88.
[45] YUE Huifang, LU Peng, SU Wei, et al. Simultaneous removal of NOx and Hg0 from simulated flue gas over CuaCebZrcO3/r-Al2O3 catalysts at low temperatures: performance, characterization, and mechanism[J]. Environmental Science and Pollution Research, 2019, 26(13): 13602-13618.
[46] ZHANG Jie, LI Caiting, DU Xueyu, et al. Promotional removal of gas-phase Hg0 over activated coke modified by CuCl2[J]. Environmental Science and Pollution Research, 2020, 27: 17891-17909.
[47] 赵波. 钼负载型催化剂对燃煤烟气汞催化氧化作用的实验研究[D]. 武汉: 华中科技大学能源与动力工程学院, 2015: 76-80.
ZHAO Bo. Experimental study on flue gas mercury oxidized by molybdenum modified catalysts[D]. Wuhan: Huazhong University of Science and Technology. School of Energy and Power Engineering, 2015: 76-80.
[48] ZHANG Zhen, WU Jiang, LI Bin, et al. Removal of elemental mercury from simulated flue gas by ZSM-5 modified with Mn-Fe mixed oxides[J]. Chemical Engineering Journal, 2019, 375: 121946.
[49] LI Honghu, ZHANG Jingdong, CAO Yanxiao, et al. Enhanced activity and SO2 resistance of Co-modified CeO2-TiO2 catalyst prepared by facile co-precipitation for elemental mercury removal in flue gas[J]. Applied Organometallic Chemistry, 2020, 34(4): e5463.
[50] ZHANG Huawei, ZHANG Mingzhu, HAO Lifeng, et al. Enhanced SO2 tolerance of FeCeOx/CNTs catalyst for NO and Hg0 removal by coating shell SiO2[J]. Fuel Processing Technology, 2020, 201: 106342.
[51] ZHANG Dan, HOU Li′an, CHEN Guanyi, et al. Cr doping MnOx adsorbent significantly improving Hg0 removal and SO2 resistance from coal-fired flue gas and the mechanism investigation[J]. Industrial & Engineering Chemistry Research, 2018, 57(50): 17245-17258.
[52] LI Guoliong, WU Qingru, WANG Shuxiao, et al. The influence of flue gas components and activated carbon injection on mercury capture of municipal solid waste incineration in China[J]. Chemical Engineering Journal, 2017, 326: 561-569.
[53] XIE Yine, LI Caiting, ZHAO Lingkui, et al. Experimental study on Hg0 removal from flue gas over columnar MnOx-CeO2/activated coke[J]. Applied Surface Science, 2015, 333: 59-67.
[54] CHI Guilong, SHEN Boxiong, YU Ranran, et al. Simultaneous removal of NO and Hg0 over Ce-Cu modified V2O5/TiO2 based commercial SCR catalysts[J]. Journal of Hazardous Materials, 2017, 330(15): 83-92.
[55] WU Xiang, DUAN Yufeng, LI Na, et al. Regenerable Ce-Mn/TiO2 catalytic sorbent for mercury removal with high resistance to SO2[J]. Energy & Fuels, 2019, 33(9): 8835-8842.
[56] LI Hailong, LIU Suojiang, YANG Jianping, et al. Role of SO2 and H2O in the mercury adsorption on ceria surface: a DFT study[J]. Fuel, 2020, 260: 116289.
[57] CHEN Guanyi, ZHANG Dan, ZHANG Anchao, et al. CrOxMnOx-TiO2 adsorbent with high resistance to SO2 poisoning for Hg0 removal at low temperature[J]. Journal of industrial and engineering chemistry, 2017, 55(25): 119-127.
[58] GAO Lei, LI Caiting, LI Shanhong, et al. Superior performance and resistance to SO2 and H2O over CoOx-modified MnOx/biomass activated carbons for simultaneous Hg0 and NO removal[J]. Chemical Engineering Journal, 2019, 371: 781-795.
[59] YANG Shu, LIU Zhilou, YAN Xu, et al. Catalytic oxidation of elemental mercury in coal-combustion flue gas over the CuAlO2 catalyst[J]. Energy & Fuels, 2019, 33(11): 11380-11388.
[60] LI Guoliang, WANG Shuxiao, WU Qingru, et al. Exploration of reaction mechanism between acid gases and elemental mercury on the CeO2-WO3/TiO2 catalyst via in situ DRIFTS[J]. Fuel, 2019, 239: 162-172.
[61] LIU Huan, ZHAO Yongchun, ZHOU Yuming, et al. Removal of gaseous elemental mercury by modified diatomite[J]. Science of the Total Environment, 2019, 652(20): 651-659.
[62] WANG Fangjun, TAN Shaoqing, CAO Yinxia, et al. Experimental study on the influence of surface characteristics of activated carbon on mercury removal in flue gas[J]. Energy & Fuels, 2020, 34(5): 6168-6177.
[63] ZOU Sijie, LIAO Yong, TAN Wei, et al. H2S-modified natural ilmenite: a recyclable magnetic sorbent for recovering gaseous elemental mercury from flue gas[J]. Industrial & Engineering Chemistry Research, 2017, 56(36): 10060-10068.
[64] YANG Shu, WANG Dongli, LIU Hui, et al. Highly stable activated carbon composite material to selectively capture gas-phase elemental mercury from smelting flue gas: copper polysulfide modification[J]. Chemical Engineering Journal, 2019, 358: 1235-1242.
[65] MEI Jian, WANG Chang, KONG Lingnan, et al. Outstanding performance of recyclable amorphous MoS3 supported on TiO2 for capturing high concentrations of gaseous elemental mercury: mechanism, kinetics, and application[J]. Environmental Science & Technology, 2019, 53(8): 4480-4489.
[66] YUAN Yong, XU Haomiao, LIU Wei, et al. Morphology-controlled synthesis and sulfur modification of 3D hierarchical layered double hydroxides for gaseous elemental mercury removal[J]. Journal of Colloid and Interface Science, 2019, 536: 431-439.
[67] XU Haomiao, MA Yongpeng, MU Bailong, et al. Enhancing the catalytic oxidation of elemental mercury and suppressing sulfur-toxic adsorption sites from SO2-containing gas in Mn-SnS2[J]. Journal of Hazardous Materials, 2020, 392: 122230.
[68] LIU Wei, XU Haomiao, LIAO Yong, et al. Co-doped ZnS with large adsorption capacity for recovering Hg0 from non-ferrous metal smelting gas as a co-benefit of electrostatic demisters[J]. Environmental Science and Pollution Research, 2020, 27: 20469-20477.
[69] KONG Lingnan, ZOU Sijie, MEI Jian, et al. Outstanding resistance of H2S-modified Cu/TiO2 to SO2 for capturing gaseous Hg0 from nonferrous metal smelting flue gas: performance and reaction mechanism[J]. Environmental Science & Technology, 2018, 52(17): 10003-10010.
[70] HUO Qihuang, WANG Yahui, CHEN Huijun, et al. ZnS/AC sorbent derived from the high sulfur petroleum coke for mercury removal[J]. Fuel Processing Technology, 2019, 191: 36-43.
[71] SHE Min, JIA Charles Q, DUAN Yufeng, et al. Influence of different sulfur forms on gas-phase mercury removal by SO2-impregnated porous carbons[J]. Energy & Fuels, 2020, 34(2): 2064-2073.
[72] CHEN Cong, DUAN Yufeng, ZHAO Shilin, et al. Experimental study on mercury removal and regeneration of SO2 modified activated carbon[J]. Industrial & Engineering Chemistry Research, 2019, 58(29): 13190-13197.
[73] YANG W, ADEWUYI Y G, HUSSAIN A, et al. Recent developments on gas-solid heterogeneous oxidation removal of elemental mercury from flue gas[J]. Environmental Chemistry Letters, 2019, 17(1): 19-47.
[74] YANG Yingju, LIU Jing, WANG Zhen. Reaction mechanisms and chemical kinetics of mercury transformation during coal combustion[J]. Progress in Energy and Combustion Science, 2020, 79: 100844.
[75] GOMEZ-GIMENEZ C, BALLESTERO D, JUAN R, et al. Mercury capture by a regenerable sorbent under oxycoal combustion conditions: effect of SO2 and O2 on capture efficiency[J]. Chemical Engineering Science, 2015, 122(27): 232-239.
[76] GOMEZ-GIMENEZ C, IZQUIERDO M T, DELASOBRAS-LOSCERTALES M, et al. Mercury capture by a structured Au/C regenerable sorbent under oxycoal combustion representative and real conditions[J]. Fuel, 2017, 207: 821-829.
[77] ZHAO Songjian, XU Haomiao, MEI Jian, et al. Ag-Mo modified SCR catalyst for a co-beneficial oxidation of elemental mercury at wide temperature range[J]. Fuel, 2017, 200: 236-243.
[78] 李成伟, 张安超, 宋军, 等. Ag/BiOI光催化剂湿法脱除烟气中气态单质汞性能及机理[J]. 化工进展, 2018, 37(4): 1442-1450.
LI Chengwei, ZHANG Anchao, SONG Jun, et al. Mechanism and performance of wet process to remove gaseous elemental mercury from flue gas using Ag/BiOI photocatalyst Ag/BiOI[J]. Chemical Industry and Engineering Progress, 2018, 37(4): 1442-1450.
[79] HUO Qihuang, YUE Caixia, WANG Yahui, et al. Effect of impregnation sequence of Pd/Ce/γ-Al2O3 sorbents on Hg0 removal from coal derived fuel gas[J]. Chemosphere, 2020, 249: 126164.
[80] 张志会, 张安超, 安飞跃, 等. 抗硫型Ru-Mn-Ti吸附剂脱汞实验研究[J]. 燃烧科学与技术, 2015, 21(6): 543-548.
ZHANG Zhihui, ZHANG Anchao, AN Feiyue, et al. Hg0 removal by Ru-Mn-Ti adsorbent with higher SO2 resistance[J]. Journal of Combustion Science and Technology, 2015, 21(6): 543-548.
[81] ZHANG Anchao, ZHANG Zhihui, LU Hao, et al. Effect of promotion with Ru addition on the activity and SO2 resistance of MnOx-TiO2 adsorbent for Hg0 removal[J]. Industrial & Engineering Chemistry Research, 2015, 54(11): 2930-2939.
[82] LIU Z, SRIRAM V, LEE J Y. Heterogeneous oxidation of elemental mercury vapor over RuO2/rutile TiO2 catalyst for mercury emissions control[J]. Applied Catalysis B: Environmental, 2017, 207: 143-152.
(编辑 刘锦伟)
收稿日期: 2020 -06 -01; 修回日期: 2020 -09 -05
基金项目(Foundation item):国家自然科学基金资助项目(51706104);江苏省自然科学基金资助项目(BK20170849);江苏省六大人才高峰项目(XNY-026);南京理工大学大型仪器设备开放基金资助项目(60)(Project(51706104) supported by the National Natural Science Foundation of China; Project(BK20170849) supported by the Natural Science Foundation of Jiangsu Province; Project(XNY-026) supported by the Peak of the Six Talents Program of Jiangsu Province; Project(60) supported by the Open Fund for Large-scale Equipments of NUST)
通信作者:王卉,博士,副教授,从事燃煤汞污染物控制研究;E-mail: wanghui22@njust.edu.cn
引用格式: 沈畅, 王卉, 沈昊天, 等. 烟气抗硫脱汞材料的研究进展[J]. 中南大学学报(自然科学版), 2021, 52(1): 133-143.
Citation: SHEN Chang, WANG Hui, SHEN Haotian, et al. Research progress on flue gas mercury removal materials with SO2resistance[J]. Journal of Central South University(Science and Technology), 2021, 52(1): 133-143.

