Effects of hydrogenation on ambient deformation behaviors of Ti-45Al alloy
来源期刊:中国有色金属学报(英文版)2009年增刊第2期
论文作者:苏彦庆 刘鑫旺 赵龙 王亮 郭景杰 傅恒志
文章页码:403 - 408
Key words:hydrogen; TiAl alloy; hydride; dislocation; fracture
Abstract: Effects of hydrogenation on ambient deformation behaviors of Ti-45Al alloy were studied. The stress―strain curves demonstrate that the plastic deformation of the hydrogenated alloys becomes more remarkable than that of the unhydrogenated alloy. Meanwhile, the compression strength and maximum strain are reduced. Both the hydride and hydrogen atoms in the interstices affect the compression deformation behaviors. The reason of the hydrogen-induced embrittlement is that the hydride is easy to become the nucleus of the cracks. And the variation of plastic deformation process is attributed to hydrogen-promoted emission, multiplication and motion of dislocation.
基金信息:the National Natural Science Foundation of China

SU Yan-qing(苏彦庆), LIU Xin-wang(刘鑫旺), ZHAO Long(赵 龙),
WANG Liang(王 亮), GUO Jing-jie(郭景杰), FU Heng-zhi(傅恒志)
School of Materials Science and Engineering, Harbin Institute of Technology, Harbin 150001, China
Received 10 June 2009; accepted 15 August 2009
Abstract: Effects of hydrogenation on ambient deformation behaviors of Ti-45Al alloy were studied. The stress―strain curves demonstrate that the plastic deformation of the hydrogenated alloys becomes more remarkable than that of the unhydrogenated alloy. Meanwhile, the compression strength and maximum strain are reduced. Both the hydride and hydrogen atoms in the interstices affect the compression deformation behaviors. The reason of the hydrogen-induced embrittlement is that the hydride is easy to become the nucleus of the cracks. And the variation of plastic deformation process is attributed to hydrogen-promoted emission, multiplication and motion of dislocation.
Key words: hydrogen; TiAl alloy; hydride; dislocation; fracture
1 Introduction
Effects of hydrogen on TiAl alloys have been studied extensively for several decades, for hydrogen can be easily absorbed by TiAl-based alloys. Many researchers focused on hydrogen-induced embrittlement [1-2]. Several mechanisms have been put forward to explain the hydrogen embrittlement, theories of the hydride-induced cleavage mechanism[3] and decohesion [4]. But researchers found that hydrogen could produce positive effects on TiAl alloys[5]. The hydrogen also has interaction with dislocation, which can facilitate the dislocation mobility and multiplication so as to improve the plastic deformation[6]. And when it is used appropriately, hydrogen can promote the hot workability, superplastic forming, powder consolidation, and composite processing of TiAl alloys[7]. So hydrogen effects on TiAl alloys are very complicated. The effects of hydrogen on ambient compression properties are also important for the applications of TiAl-based alloys. In the conventional researches, hydrogen was introduced into TiAl alloys by permeating[8-10] or cathodic charging[11]. The process needs long time for hydrogen to permeate into the alloys homogeneously. Normally, more than 24 h was spent even for small specimens[12-13]. In this study, hydrogen was introduced into the TiAl alloy melt directly. The diffusion velocity of hydrogen in alloy melt is much higher than that in solid state. So this method is much more efficient than conventional one.
The aim of the present work is to examine the effects of hydrogen on ambient compression properties of Ti-45%Al (all are molar fraction in this work, %). Meanwhile, the mechanisms of effects of hydrogen on deformation behaviors of TiAl alloys are also discussed.
2 Experimental
2.1 Ingots preparation and hydrogenation process
TiAl ingots in button form were prepared in non-consumable electrode arc furnace under argon atmosphere. 25-30 g alloy buttons were melted 4-5 times to improve the chemical homogeneity. Chemical analysis indicates that the composition is Ti-45Al.
The process of melt hydrogenation was performed in our hydrogen charging system based on Sivert’s method[14] similar to many researches[15-16], which consists of a non-consumable electrode arc melting furnace with a water-cooled copper hearth and a hydrogen analyzer. The hydrogen analyzer can detect volume fraction of hydrogen and the pressure in the melting chamber. The TiAl ingots were melted under the gaseous mixture of hydrogen and argon in the non-consumable electrode arc melting furnace. Infrared thermoscope indicates that the temperature of the alloy melt is about 2 000 K. Hydrogen diffused into the melt until saturation, and furnace-cooled. The hydrogen would remain in the alloys without escaping because the hydrogen in the alloy would be in dynamical equilibrium with the hydrogen in the chamber. During the whole process, the hydrogen fraction in the gaseous mixture and total pressure were monitored by the hydrogen analyzer. The hydrogen content of the alloys can be determined by the decrease of hydrogen in the melting chamber. The compositions of the alloys containing hydrogen are Ti-45Al-0.5H and Ti-45Al-2.1H by calculation according to the data of the hydrogen analyzer.
2.2 Ambient compression test and analysis methods
Specimens for ambient compression tests were cut from the hydrogenated and unhydrogenated alloys by electric discharge machine. They were cylinders of 10 mm in diameter and 5 mm in height. The compression tests were preformed on an Instron 5500 machine with an initial strain rate of 1.4×10-4 s-1 at room temperature.
The X-ray diffraction (XRD) analysis was carried out by Philip X’Pert with Cu Kα radiation operated at 40 kV and 35 mA. The SEM analysis was operated by FEI Quanta 200 at 25 kV for microstructure observation.
3 Results and discussion
3.1 Stress―strain behaviors
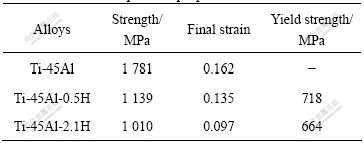
The stress―strain curves pertaining to the unhydrogenated and hydrogenated alloys are shown in Fig.1. Table 1 shows the ambient compression properties of Ti-45Al-xH (x=0, 0.5, 2.1). It can be concluded that:
1) With the increase of hydrogen content, the compression strength and the final strain decrease.
2) There is not evident yield point during deformation process of Ti-45Al. But the yield point can be observed clearly in Ti-45Al-0.5H and Ti-45Al-2.1H. The yield strength decreases with the increase of hydrogen content.
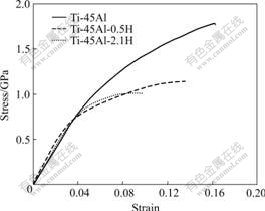
Fig.1 Stress―strain curves of the hydrogenated and unhydro- genated alloys deforming at 1.4×10-4 s-1
Table 1 Ambient compression properties of Ti-45Al-Xh
3) Ti-45Al is of brittle-failure. Plastic deformation stage cannot be distinguished obviously. But after hydrogenation, plastic deformation becomes remarkable relatively. It seems that hydrogen addition improves the plastic deformation behaviors.
3.2 Microstructures
Fig.2 presents the microstructures of the unhydrogenated and hydrogenated alloys. They are all of typical dendrite structures with secondary arms, which are the typical structures of TiAl alloys by melting in the water-cooled copper hearth. The dendrite of Ti-45Al is very coarse, but the dendrites of the hydrogenated alloys become finer. It may be due to the refinement effects of hydrogen on the alloys during the solidification process.

Fig.2 Microstructures of Ti-45Al(a), Ti-45Al-0.5H(b) and Ti-45Al-2.1H(c)
3.3 Hydrogen in TiAl alloys
Hydrogen solubility in TiAl alloys increases with the increase of temperature, and hydride precipitates after hydrogen becomes saturated[17-18]. So hydrogen becomes supersaturated and then hydride precipitates during the solidification and cooling process. According to Ti-H phase diagram, titanium hydride forms under relatively low temperature of about 797 ℃[19]. But the hydrides are not detected in the hydrogenated alloys in XRD analysis (Fig.3). It may be due to that hydrides are too few. There are not yet precise data for the solubility of hydrogen in Ti-45Al at room temperature. It is thought that the hydrides will form very easily in TiAl alloys when the hydrogen content exceeds the solubility. The hydride will form in the TiAl alloys easily because the hydrogen solubility is less than 0.5% at room temperature [17]. So hydrides form in the hydrogenated alloys during the cooling process and the hydride content increases with the increase of hydrogen addition. It is considered that some hydrogen forms hydrides and the others are dissolved in the interstices. The hydride and hydrogen atoms both affect the compression behaviors of hydrogenated alloys.
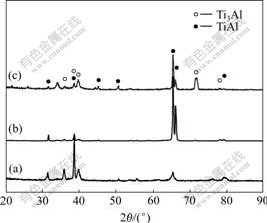
Fig.3 XRD patterns of Ti-Al-xH: (a) x=0; (b) x=0.5; (c) x=2.1
3.4 Mechanism analysis
It is well known that hydrides can lead to embrittlement in many metals including TiAl alloys[20]. A qualitative mechanism for stress-induced hydride embrittlement was firstly proposed by WESTLAKE[21] which has been widely recognized. During the deformation process, the phase transformation is accompanied by the decrease in the critical stress intensity for the crack propagation because the hydride is the crack source. The original crack then propagates through the hydride, the formation of which is accompanied by a high-compressive local stress field resulting from the increase of the volume. So hydride affects the fracture process. The crack propagates and extends following the formation of crack source and the alloy fractures before sufficient plastic deformation. That is the reason for hydrogen-induced embrittlement.
Plastic deformation occurs in the alloys accompanied with the dislocation evolvement. Then the first crack appears and extends, and failure will take place along the cracks which extends the fastest. So the failure is determined by the formation and development of crack. Hydrides are prone to be the source of nucleation of cracks, for the hydrides are incoherent with the matrix. So the hydrides facilitate the fracture of TiAl alloys remarkably.
Hydrides have great effect on the failure of TiAl alloys, which is considered to be the nucleus of cracks and can accelerate the extension of cracks. Thus the crack formation of the alloys containing hydrides is much earlier than those without hydrides. So the deformation rate of hydrogenated alloys is smaller than that of the unhydrogenated alloy. The crack nucleus is more prone to form in the alloys containing more hydrides, therefore the final strain of Ti-45Al-2.1H is less than that of Ti-45Al-0.5H.
The hydrogen atoms in the interstices have strong interaction with the dislocation[6], thus the hydrogen atoms act mainly on the plastic deformation. Plastic deformation is always related to the dislocation, including emission, multiplication and movement. Hydrogen effect on the dislocation emission is determined qualitatively by the following formula[22]:
![]()
where KIe and KIe(H) are the stress intensity factor of the dislocation emission without and with hydrogen respectively; n is the number of hydrogen atoms remaining behind the dislocation line after the dislocation of per unit length moves per interatomic distance b; k is a constant; T is temperature; A is a constant related to the strain energy of the dislocation of per unit length around the crack tip; f * is the fugacity of hydrogen diffusing along the dislocation pipe and f is the fugacity of hydrogen diffusing along the lattices.
The fugacity f * decreases to f when the hydrogen exists in the alloys. Consequently the energy barrier for dislocation emission of per unit length decreases. Because f * >f, KIe>KIe(H). In other words, the critical stress intensity factor of dislocation emission is decreased by hydrogen. That means hydrogen promotes the dislocation emission.
The critical shear stress for dislocation multiplication is: τ=2UD/Lb, which is also the stress for start of Frank-Read source. UD is strain energy of dislocation; and L is length of Frank-Read source. The strain field of hydrogen is non-spherical symmetrical, thereby hydrogen has interaction with both edge dislocation and screw dislocation and forms air mass around the dislocation[23]. The hydrogen air mass can move with the dislocation as a result of the high diffusion coefficient of hydrogen. Because the strain energy of dislocation U(H) with the hydrogen air mass is lower than UD without hydrogen, besides the hydrogen can move with the dislocation, the external stress for the dislocation multiplication with the hydrogen air mass is reduced remarkably. So the dislocation multiplication is promoted by hydrogen, which is also reported in Ref.[24]. The hydrogen air mass around screw dislocation redistributes and becomes non-spherical symmetrical under external stress. During this process, hydrogen air mass will give birth an additional force FH=τHb acting on the dislocation, which can assist external force ![]() to promote the dislocation motion. The resultant force to impel the
to promote the dislocation motion. The resultant force to impel the
dislocation motion is ![]() . If the
. If the
resistance force of dislocation motion is τ, the dislocation motion can take place when τ=kτEX=τs. In other words, the external force for dislocation motion is τEX(H)= τs/k with hydrogen, and it is τEX=τS without hydrogen. For k>1, τEX=τS, which means hydrogen promotes the dislocation motion.
Hydrogen promotes the emission, multiplication and motion of dislocation as described above. So emission, multiplication and movement of dislocation can perform at lower stress for the action of hydrogen. In other words, hydrogen facilitates the dislocation mobility[25]. Therefore, the plastic deformation stages of the hydrogenated alloys become more notable compared with the unhydrogenated alloy. Refinement of the microstructures may also contribute to the plastic deformation.
Fig.4 shows the typical TEM images of both unhydrogenated and hydrogenated alloys. It is clear that the initial dislocation in Ti-45Al alloy is chaotic and there are many dislocation tangles. The dislocation density in and around the α2 phase is much higher than that in the γ phase, indicating that the deformation is not homogeneous. But with the increase of hydrogen addition, the deformation in the hydrogenated alloys becomes more homogeneous. This is due to the fact that hydrogen induces the dislocation activity and deformation compatibility. The plastic deformation is improved before hydride-induced failure.

Fig.4 Typical TEM micrographs of substructures of Ti-45Al(a), Ti-45Al-0.5H(b) and Ti-45Al-2.1H(c) after deforming at 1.4×10-4 s-1
Lattice decohesion theory that hydrogen will weaken the cohesive force between the atoms has been accepted extensively[26-27]. The local stress concentration for nucleation of microcrack is reduced in the hydrogenated alloys in terms of lattice decohesion theory. So, it is also a reason why the compression strength of the hydrogen-contained alloys decreases.
3.5 Fracture analysis
The Ti-45Al alloy fractures along the direction oriented 45? to the load axis; however, the hydrogenated alloys comminuted fracture. Fig.5 shows the fractographs of the hydrogenated and unhydrogenated alloys. The fracture of the unhydrogenated and hydrogenated alloys exhibits river-liked patterns and they are typically cleavage-fractured. But there are some arrises distributed on the fracture of the hydrogenated alloys. This indicates that plastic deformation takes place in more regions of hydrogenated alloys. It is in respect that hydrogen promotes plastic deformation and more regions deform around the crack tip.

Fig.5 Fractographs of Ti-45Al(a), Ti-45Al-0.5H(b) and Ti-45Al-2.1H(c)
4 Conclusions
1) The microstructures of Ti-Al alloy become finer during solidification process after hydrogenation.
2) With the increase of hydrogen addition, the compression strength and the final strain decrease. The hydrides form in the alloys during the cooling process. The embrittlement is mainly caused by the formation of hydrides.
3) The plastic deformation of the hydrogenated alloys is more remarkable than that of the unhydrogenated alloy, because hydrogen can promote the emission, multiplication and motion of dislocation. And microstructure refinement also contributes to the plastic deformation.
4) The deformation becomes more homogeneous with the increase of hydrogen addition. And plastic deformation takes place in many regions of hydrogenated alloys.
References
[1] RONNIE H R. Role of hydrogen environment induced hydrogen embrittlement of Ti-8Al-1Mo-2V alloy[J]. Materials Science and Engineering A, 2008, 494(1/2): 143-146.
[2] LIU Y Z, ZU X T, LI C, QIU S Y, LI W J, HUANG X Q. Hydrogen embrittlement of a Ti-Al-Zr alloy evaluated by impact test method[J]. Scripta Materialia, 2005, 52(9): 821-825.
[3] HERRERA-BARROS E C, SUNDARAM P A, FOUNTZOULAS C. Process and properties of hydride formed on gamma titanium aluminide during cathodic charging[J]. Journal of Alloys and Compounds, 2005, 400(1/2):125-130.
[4] ORIANI R A, JOSEPHIC P H. Testing of the decohesion theory of hydrogen-induced crack propagation[J]. Scripta Metallurgica, 1972, 6(8): 681-688.
[5] FROES F H, SENKOV O N, QAZI J I. Hydrogen as a temporary alloying element in titanium alloys: thermohydrogen processing[J]. International Material Review, 2004, 49(3/4): 227-245.
[6] ROBERTSON I M. The effect of hydrogen on dislocation dynamics[J]. Engineering Fracture Mechanics, 2001, 68(6): 671-692.
[7] SENKOV O N, FROES F H. Thermohydrogen processing of titanium alloys[J]. International Journal of Hydrogen Energy, 1999, 24(6): 565-576.
[8] SUARDI K, HAMZAH E, OURDJINI A, VENKATESH V C. Effect of heat treatment on the diffusion coefficient of hydrogen absorption in gamma-titanium aluminide[J]. Journal of Materials Processing Technology, 2007, 185(1/3): 106-112.
[9] LOPEZ-SUAREZ A, RICKARDS J, TREJO-LUNA R. Mechanical and microstructural changes of Ti and Ti-6Al-4V alloy induced by the absorption and desorption of hydrogen[J]. Journal of Alloys and Compounds, 2008, 457(1/2): 216-220.
[10] SUN Z G, ZHOU W L, HOU H L. Strengthening of Ti-6Al-4V alloys by thermohydrogen processing[J]. International Journal of Hydrogen Energy, 2009, 34(4): 1971-1976.
[11] ESTUPINAN H A, URIBE I, SUNDARAM P A. Hydrogen permeation in gamma titanium aluminides[J]. Corrosion Science, 2006, 48(12): 4216-4222.
[12] IINO Y, GAO K W, OKAMURA K, QIAO L J, CHU W Y. Effects of high-temperature hydrogen exposure on room temperature tensile properties and fatigue crack growth behaviour of TiAl based alloy[J]. Materials Science and Engineering A, 2002, 338: 54-59.
[13] LI K, POLLOCK T M, GRAEF M D, HABEL U, THOMPSON A W. On the effect of low and high pressure hydrogen charging on the oxidation of a Ti-48Al-2Cr-2Nb alloy[J]. Materials Science and Engineering A, 2001, 318(1/2): 224-234.
[14] SIEVERTS A. Absorption of gases by metals[J]. Zeitschrift fur Metallkunde, 1929, 21(1/2): 37-46.
[15] SHAN D B, ZONG Y Y, LU T F, L? Y. Microstructural evolution and formation mechanism of FCC titanium hydride in Ti-6Al-4V-xH alloys[J]. Journal of Alloys and Compounds, 2007, 427(1/2): 229-34.
[16] LAPOVOK R, TOMUS D, SKRIPNYUK V M, BARNETT M R, GIBSON M A. The effect of hydrogenation on the ECAP compaction of Ti-6Al-4V powder and the mechanical properties of compacts[J]. Materials Science and Engineering A, 2009, 513/514: 97-108.
[17] TAKASAKI A, FURUYA Y, OJIMA K, TANEDA Y. Hydrogen solubility of two-phase (Ti3Al+TiAl) titanium aluminides[J]. Scripta Metallurgica, 1995, 32(11): 1759-1764.
[18] LEGZDINA D, ROBERTSON I M, BIRNBAUM H K. Oxidation behavior of a single phase γ-TiAl alloy in low-pressure oxygen and hydrogen[J]. Acta Materialia, 2005, 53(3): 601-608.
[19] OKAMOTO H. H-Ti (Hydrogen-titanium)[J]. Journal of Phase Equilibria, 1992, 13(4): 443.
[20] CHAN K S. A fracture model for hydride-induced embrittlement[J]. Acta Metallurgy Materialia, 1995, 33(12): 4325-4335.
[21] WESTLAKE D G. A generalized model for hydrogen embrittlement[J]. Transaction of the ASM, 1969, 62: 1000.
[22] CHU W Y. Crack and environmental crack[M]. Beijing: Science Press, 2000: 75-85. (in Chinese)
[23] WEN M, FUKUYAMA S, YOKOGAWA K. Atomistic simulations of hydrogen effect on dissociation of screw dislocations in nickel[J]. Scripta Materialia, 2005, 52(10): 959-962.
[24] BARNOUSH A, YANG B, VEHOFF H. Effect of hydrogen and grain boundaries on dislocation nucleation and multiplication examined with a NI-AFM[M]. Advances in Solid State Physics, 2008, 47: 253-269.
[25] NIBUR K A, BAHRA D F, SOMERDAY B P. Hydrogen effects on dislocation activity in austenitic stainless steel[J]. Acta Materialia, 2006, 54(10): 2677-2684.
[26] ALVAREZ A-M, ROBERTSON I M, BIRNBAUM H K. Hydrogen embrittlement of a metastable b-titanium alloy[J]. Acta Materialia, 2004, 52(14): 4161-4175.
[27] ORIANI R A, JOSEPHIC P H. Equilibrium aspects of hydrogen-induced cracking of steels[J]. Acta Metallurgy, 1974, 22(9): 1065-1074.
(Edited by CHEN Can-hua)
Foundation item: Project(50771041) supported by the National Natural Science Foundation of China
Corresponding author: SU Yan-qing; Tel: +86-451-86417395; E-mail: suyq@hit.edu.cn

