Pressure acid leaching of black shale for extraction of vanadium
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2010��������1��
�������ߣ���F͢ κ�� ���� ����� ��־�� ���˱�
����ҳ�룺112 - 117
Key words��pressure acid leaching; black shale; vanadium; catalyst
Abstract: Extraction of vanadium from black shale was attempted in pressure acid leaching. The chemical components of the sample obtained from Guizhou Province of China show that it contains 3.258% V2O5, 52.880% SiO2 and 16.140% Al2O3. Phase analyses of vanadium indicates vanadium mainly exists in the free oxide and mica. Vanadium contents in the two phases are 18% and 53%, respectively. The contents of V3+, V4+ and V5+ are almost equal. Under the optimum parameters of one-step leaching (reaction time of 3 h, sulfuric addition of 25%, temperature of 150 ��, liquid to solid ratio of 1.2 mL/g, catalyst (FeSO4) addition of 5% and size of 85% particle 0.074 mm), about 77% of vanadium is recovered. After two-step countercurrent leaching, the leach recovery of vanadium can reach above 90%. Air replacing oxygen is completely feasible. The impurity metals can dissolve into solution in different degrees.
������Ϣ��the Hi-tech Research and Development Program of China
Yunnan Provincial Science and Technology Agency

LI Min-ting(��F͢), WEI Chang(κ ��), FAN Gang(�� ��), LI Cun-xiong(�����),DENG Zhi-gan(��־��), LI Xin-bin(���˱�)
Faculty of Metallurgical and Energy Engineering, Kunming University of Science and Technology,Kunming 650093, China
Received 6 July 2009; accepted 30 December 2009
____________________________________________________________________________________________
Abstract: Extraction of vanadium from black shale was attempted in pressure acid leaching. The chemical components of the sample obtained from Guizhou Province of China show that it contains 3.258% V2O5, 52.880% SiO2 and 16.140% Al2O3. Phase analyses of vanadium indicates vanadium mainly exists in the free oxide and mica. Vanadium contents in the two phases are 18% and 53%, respectively. The contents of V3+, V4+ and V5+ are almost equal. Under the optimum parameters of one-step leaching (reaction time of 3 h, sulfuric addition of 25%, temperature of 150 ��, liquid to solid ratio of 1.2 mL/g, catalyst (FeSO4) addition of 5% and size of 85% particle 0.074 mm), about 77% of vanadium is recovered. After two-step countercurrent leaching, the leach recovery of vanadium can reach above 90%. Air replacing oxygen is completely feasible. The impurity metals can dissolve into solution in different degrees.
Key words: pressure acid leaching; black shale; vanadium; catalyst
____________________________________________________________________________________________
1 IntroductionVanadium is currently recovered as a by-product or a best co-product from mineral resource[1]. Many industrial sub-products have been investigated and used for vanadium recovery, including oil fly ash[2-4], spent catalyst[5-8] and petroleum coke[9]. HOLLOWAY and ETSELL[3] reported the vanadium (V2O5) content of oil sands fly ash from Aberta in Canada is 3.6%-6.2%. This is thus an interesting resource for the recovery of vanadium. MISHRA et al[6] reported the vanadium level of spent hydro-processing catalyst from the SK Petroleum Corp in South Korea is 7.7%-10.3%.
Vanadium-bearing black shale (also called stone coal) is an alternative resource of vanadium in China in addition to vanadium-titanomagnetite. It extensively exists in Yunnan, Guizhou, Hunan, Zhejiang, Jiangxi, Guangxi, Gansu and Xinjiang Province or autonomous regions. The gross reserve of V2O5 in black shale is 110 Mt, accounting for more than 87% of the domestic reserve of vanadium and black shale containing less than 0.5% V2O5 accounts for 60% of the total reserve[10]. The mineral compositions of black shale are very complicated and inconstant. Generally, more than five kinds of vanadium-bearing minerals are present in black shale such as mica, kaolin, free oxide, taltalite and garnet[10]. XU[11] found that for the black shale in south of China the most vanadium is present in the form of isomorphism where V3+ substitutes for Al3+ in the crystal lattice of aluminosilicate, for instance, mica, taltalite and garnet. Vanadium (V5+) exists in free oxide and is present in the absorption form. Vanadium (V4+) might exist in the form of isomorphism and absorption. The leachability of vanadium in different minerals is quite different, and vanadium in free oxide is more easily leached than that existing in aluminosilicate.
For the recent twenty years, the vanadium recovery from black shale has received considerable attention. Many studies have focused on the recovery of vanadium from black shale using several processes: roast leaching[12-15] or direct acid leaching[16-20]. AMER[21] studied the pressure acid leaching of Egyptian black shale of the Quseir-Safaga region and obtained a vanadium recovery of up to 95% using 10% H2SO4 solution with an oxygen partial pressure of 8��10 Pa for 40 min.
In this work, the feasibility of extracting vanadium from black shale obtained from Guizhou was analyzed in pressure acid leaching.
2 Experimental2.1 Materials
Black shale was collected from Guizhou Province of China. It was dried to a constant mass at 75 ��, and 85% of black shale was subsequently ground to 0.074 mm which was used in most of the experimental work. Table 1 depicts the main chemical components of the sample. It can be seen that content of vanadium in form of V2O5 is 3.258%. Vanadium phase analysis was carried out according to the sequential extraction procedures[22-23]. The vanadium phase and valence state of vanadium are given in Table 2. Vanadium mainly exists in mica (80.530%) and next in free oxide (18.00%).
Table 1 Main chemical components of studied sample (mass fraction, %)

Table 2 Distribution of vanadium in different phases
2.2 Method
In the leaching experiments, a titanium autoclave with an agitation stirrer (speed was kept at 580 r/min in all leaching experiments) was used. Sample (200 g) was mixed with a measured amount of distilled water and calculated amounts of sulfuric acid (AR). The slurry was added to the autoclave and then was heated. At the same time, oxygen was continuously aerated into the reactor at 1.2 MPa (total pressure). When temperature reached the specified value, the reaction was initiated. At the end of each leaching test, the insoluble leach residue was separated from the pregnant leach solution by vacuum filtration and the solids were washed with distilled water. The washed leach residues were dried, weighed and analyzed by ICP-AES for vanadium content. The leach recovery of vanadium was calculated using the analysis of V2O5 content in the black shale and leach residue and their respective masses.
3 Results and discussion3.1 Effect of reaction time on leach recovery of vanadium
Reaction time was varied between 1 h and 5 h. As shown in Fig.1, the leach recovery of vanadium increases sharply with the increase in reaction time from 1 h to 3 h and is 75.79% at a reaction time of 3 h. This is probably due to vanadium in the form of adsorption easily dissolved into solution, so before 3 h the dissolution of vanadium is quick. However, for 3-5 h reaction time, the leach recovery of vanadium seemingly has a gradually declined trend. With the increase in reaction time, the more aluminosilicate reacts with H2SO4 to produce a condensation product of silicon which can absorb the positive cations of vanadium such as VO2+ and VO2+. So, in the subsequent experiments, reaction time is chosen to be 3 h.

Fig.1 Effect of reaction time on leach recovery of vanadium at 180 ��, 25% addition of H2SO4, 1.2 mL/g L/S and 85% particle of 0.074 mm
3.2 Effect of temperature on leach recovery of vanadium
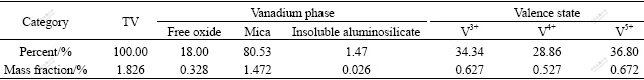
Fig.2 shows the leach recovery of vanadium obtained by varying temperature from 120 to 180 ��, using an addition of H2SO4 of 25% with a liquid to solid ratio (L/S) of 1.2 mL/g for 3 h. It can be seen from Fig.2 that the leaching temperature obviously affects the recovery of V. Up to 75% of vanadium can be recovered when the temperature reaches 180 ��. In the subsequent experiments, temperature is chosen to be 180 ��.
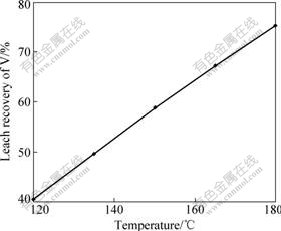
Fig.2 Effect of temperature on the leach recovery of vanadium at 3 h, 25% addition of H2SO4, 1.2 mL/g L/S and 85% particle size of 0.074 mm
3.3 Effect of addition of sulfuric acid on leach recovery of vanadium
The effect of H2SO4 addition (mass fraction of material) on the leach recovery of vanadium was studied in the range from 15% to 40%, as shown in Fig.3. The leach recovery of vanadium increases from 51.1% to 91.02 % with an increase in H2SO4 addition from 15% to 40%, but there is an increase in residual concentration of H2SO4 in leachate with increased addition of H2SO4. When the addition of H2SO4 is 40%, the residual concentration of H2SO4 reaches above 65 g/L for the addition of H2SO4 of 40%. This needs a more addition of ammonia the in adjusting the pH process and adds the production cost during the solvent extraction. So, an optimum H2SO4 addition of 25% is chosen.

Fig.3 Effect of H2SO4 addition on leach recovery of vanadium at 3 h, 180 ��, 1.2 mL/g L/S and 85% particle size of 0.074 mm
3.4 Effect of liquid solid ratio on leach recovery of vanadium
A series of five experiments were carried out by varying L/S in the range of 1.1-3.0 mL/g. The results are shown in Fig.4. It can be seen from Fig.4 that the leach recovery of vanadium decreases with the increase of L/S, due to the fact that the concentration of H2SO4 decreases with the increase of L/S under the same H2SO4 addition of 25%. However, the leach recovery at L/S of 1.1 mL/g is lower than that at L/S of 1.2 mL/g. The reason is that ore slurry is very ropy at L/S of 1.1 mL/g and is unfavorable for the diffusion and mass transfer. The leach recovery of vanadium reaches the highest value (75.3%) at L/S of 1.2 mL/g. The optimum liquid solid ratio is chosen to be 1.2 mL/g.
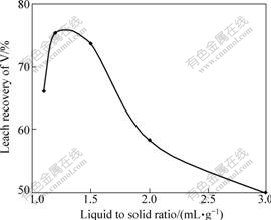
Fig.4 Effect of L/S on leach recovery of vanadium at 3 h, 180 ��, 25% addition of H2SO4 and 85% particle size of 0.074 mm
3.5 Effect of size of particle on leach recovery of vanadium
The effect of size of particle on the leach recovery of vanadium is shown in Fig.5. It can be seen form Fig.5 that leach recovery of vanadium is not evidently changed in the range of 0.095-0.048 mm. However, when the size of particle is less than 0.048 mm, the leach recovery of vanadium sharply decreases. This might be due to the fact that the carbonaceous components in the sample, which is exposed to surface in the grinding process, might cover the vanadium-bearing mineral, and makes it become more difficult when leaching agent reaches the mineral surface to react. Meanwhile, the smaller the size of particle, the higher the energy consumption in grinding process. So, optimum particle size of 85% particle is chosen to be 0.095 mm.
Fig.5 Effect of size of particle on leach recovery of vanadium at 3 h, 180 ��, 25% addition of H2SO4 and L/S of 1.2 mL/g
3.6 Effect of catalyst on leach recovery of vanadium
To improve the leach recovery of vanadium and decrease the leaching temperature, the two kinds of catalysts (ferrous sulfate and copper sulfate) were used. Four experiments were carried out under the following conditions: reaction time of 3 h, leaching temperature of 150 ��, addition of H2SO4 of 25%, L/S of 1.2 mL/g and size of 85% particle of 0.074 mm. As shown in Fig.6, compared with no catalyst (150 ��) and no catalyst (180 ��), the leach recovery increases by about 7.7% and 2.5% for FeSO4, respectively. This indicates that the addition of FeSO4 contributes to an increase in recovery of vanadium. However, addition of CuSO4 does not affect leach recovery compared with no catalyst (150 ��). So the catalyst is chosen to be FeSO4.
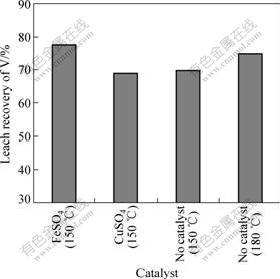
Fig.6 Effect of catalyst kinds on leach recovery of vanadium at 3 h, 25% addition of H2SO4, 1.2 mL/g L/S and 85% particles size of 0.074 mm
The effect of the addition of ferrous sulfate on the leach recovery of vanadium is shown in Fig.7. It can be seen form Fig.7 that the leach recovery of V increases from 70.4% to 75.2% with the increase of FeSO4 addition from 0 to 0.8%. While with a further increase in catalyst addition, the leach recovery increases slightly and only increases from 76.1% at 2.5% FeSO4 to 78.1% at 5% FeSO4. This result may be related to the leachability of iron compound in material. In the acid leaching process, iron compound reacts with sulfuric acid to produce dissolvable species (Fe3+ or Fe2+), which might also play an important role during transferring oxygen process in the presence of the dissolved oxygen. So, the catalyst addition does not significantly affect the leach recovery of vanadium.
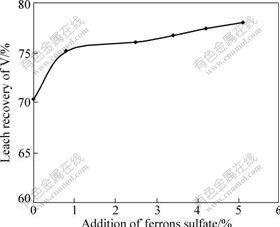
Fig.7 Effect of addition of ferrous sulfate on leach recovery of vanadium at 3 h, 150 ��, 25% addition of H2SO4, 1.2 mL/g L/S and 85% particle size of 0.074 mm
According to the above results, the optimum processing parameters for the one-step leaching of vanadium are as follows: reaction time of 3 h, sulfuric addition of 25%, leaching temperature of 150 ��, liquid/solid ratio of 1.2 mL/g, FeSO4 addition of 5% and size of 85% particle of 0.074 mm. Three experiments were carried out under these optimum conditions. The mean leach recovery of vanadium is around 77%.
3.7 Two-step countercurrent leaching of black shale
3.7.1 Leaching of vanadium
To further improve the leach recovery of vanadium, the two-step countercurrent leaching experiment which consisted of five unit experiments was performed. The flow sheet is given in Fig.8, and the results are shown in Fig.9. The leach recovery of the first-step leaching is only about 51% and decreases by 24% compared with one-step leaching (77%), but the total (sum of first-step and second-step) recovery reaches 90% and increases by 15% compared with one-step leaching.
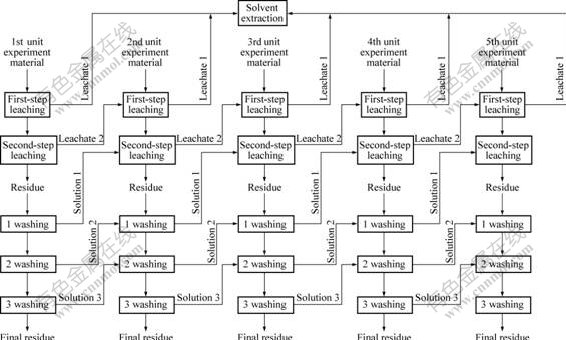
Fig.8 Flow sheet of two-step countercurrent leaching

Fig.9 Vanadium recovery of two-step countercurrent leaching experiment (First-step leaching: 2 h, 150 ��, 25% addition of H2SO4, 1.2 mL/g L/S, 85% particle size of 0.074 mm and 5% addition of FeSO4; Second-step leaching: 3 h, 150 ��, 35% addition of H2SO4, 1.2 mL/g L/S and 5% addition of FeSO4)
3.7.2 Feasibility of air replacing oxygen
The feasibility of substituting air for oxygen was investigated under the same conditions (as shown in Fig.9). As shown in Fig. 10, the leach recovery of first-step, second-step and the total (sum of first-step and second-step) does not appear an obvious change (88%-89%) and only decreases by 1%-2% compared with that of oxygen. This shows that the method substituting air for oxygen is absolutely feasible.

Fig.10 Vanadium recovery using air under same condition of two-step countercurrent leaching (Conditions as Fig.9)
3.7.3 Enrichment behavior of vanadium in two-step countercurrent leaching process
The enrichment behavior of vanadium in the two-step countercurrent leaching process was investigated. The results are shown in Fig.11. It can be observed that the concentration trend of vanadium in the first-step leachate is consistent with that in the second- step leachate. The concentration of V2O5 in the first-step leachate can reach above 16 g/L after 4 cycles. In theory, this is a maximum for the two-step countercurrent leaching process used in this work due to the fact that a complete cycle is achieved after 4 cycles. And then this leachate is used to extract vanadium pentoxide through solvent extraction. Similarly, the concentration of V2O5 in the different washing liquors increases with an increase in the cycles. After 4 cycles, the mass concentration of V2O5 is only 1.95 g/L for the 3 washing liquor.
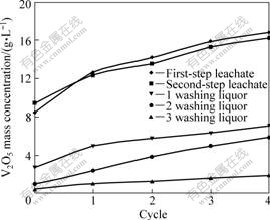
Fig.11 Enrichment behavior of vanadium in different solutions during two-step leaching
2.7.4 Leaching of impurity metals
Due to the poor selectivity of acid leaching, the impurity metals dissovle into solution in different degrees. Consequently, on one hand, this is a main reason that causes the high acid consumption for the direct acid leaching of black shale. On the other hand, the higher concentration of these impurity metals brings some difficulties to the subsequent solvent extraction procedure, specially the ferric iron. The leaching percent of the main impurity metals is shown in Fig.12. About 45% Fe, 64% Al2O3, 50% K2O, 77% Mg, 15% Na2O and 38% CaO are removed from black shale.
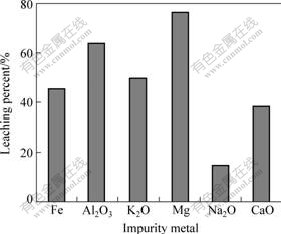
Fig.12 Leaching percent of impurity matal in two-step countercurrent leaching process
4 Conclusions
1) The leaching of vanadium from black shale obtained from Guizhou Province of China is achieved using pressure acid leaching process. The leach recovery of vanadium is about 77% at the following optimum leaching parameters of one-step leaching: reaction time of 3 h, temperature of 150 ��, 25% addition of H2SO4, L/S of 1.2 mL/g, 85% particle size of 0.074 mm, and 5% addition of FeSO4.
2) The extraction yield of vanadium reaches 90% for the two-step countercurrent leaching process. The feasibility substituting air for oxygen is well demonstrated and the recovery of vanadium using air is above 88%. The main impurity metals, such as Fe, Al2O3, K2O, Mg, Na2O and CaO dissolve into solution in different degrees.
References[1] NAVARRO R, GUZMAN J, SAUCEDO I, REVILLA J, GUIBAL E. Vanadium recovery from oil fly ash by leaching, precipitation and solvent extraction processes [J]. Waste Management, 2007, 27(3): 425-438.
[2] HOLLOWAY P C, ETSELL T H. Alternative reagents for roasting Suncor oil sands fly ash [J]. Trans Inst Min Metall C, 2004, 113(3): 153-160.
[3] HOLLOWAY P C, ETSELL T H. Process for the complete utilization of oil sands fly ash [J]. Canadian Metallurgical Quarterly, 2006, 45(1): 25-32.
[4] VITOLO S, SEGGIANI M. Recovery of vanadium from heavy oil and Orimulsion fly ashes[J]. Hydrometallurgy, 2000, 57(1/2): 141-149.
[5] MISHRA D, KIM D J, RALPH D E, AHM J G, RHEE Y H. Bioleaching of vanadium rich spent refinery catalysts using sulfur oxidizing lithotrophs [J]. Hydrometallurgy, 2007, 88(1/4): 202-209.
[6] MISHRA D, KIM D J, RALPH D, AHM J D, RHEE Y H. Bioleaching of spent hydro-processing catalyst using acidophilic bacteria and its kinetics aspect [J]. Journal of Hazardous Materials, 2008, 152(3): 1082-1091.
[7] LAI Y D, LIU J C. Leaching behaviors of Ni and V from spent catalyst [J]. Journal of Hazardous Materials, 1997, 53(1/3): 213-224.
[8] LOZANO L J, JUAN D. Technical note leaching of vanadium from spent sulphuric acid catalysts [J]. Mineral Engineering, 2001, 14(5): 543-546.
[9] SHLEWIT H, ALIBRAHIM M. Extraction of sulfur and vanadium from petroleum coke by means of salt-roasting treatment [J]. Fuel, 2006, 85(5/6): 878-880.
[10] BIN Zhi-yong. Progress of the research on extraction of vanadium pentoxide from stone coal and the market of the V2O5 [J]. Hunan Nonferrous Metals, 2006, 22(1): 16-20. (in Chinese)
[11] XU Guo-zhen. Guidance functions of vanadium valence and material composition in stone coal to extracting vanadium process [J]. Coal Processing & Comprehensive Utilization, 1989(5): 5-8. (in Chinese)
[12] DONG Jian-hua, YANG Zhi-lan, FENG Ji-yan.. Study on extracting pure vanadium pentoxide (V2O5) from the stone coal vanadium ore [J]. J Xi��an Inst of Metal & Cons Eng, 1993, 25(1): 105-110. (in Chinese)
[13] FAN Bi-wei, LIN Hai-ling. Effects of roasting process on extraction of vanadium from stone coal at Fangshankou [J]. Hydrometallurgy of China, 2001, 20(2): 79-83. (in Chinese)
[14] LU Zhi-hua, ZHOU Bang-na, YU Zhong-xing, CHEN Shi-guan. Craft study on extracting vanadium form stone coal by means of oxidation roasting-diluted alkali solution [J]. Chinese Journal of Rare Metals, 1994, 18(5): 321-327. (in Chinese)
[15] ZOU Xiao-yong, OUYANG Yu-zhu, PENG Qing-jing, TIAN Ren-guo. Study on the process of producing vanadium pentoxide from bone coal vanadium mine by no-salt-roasting and acid leaching [J]. Chemical World, 2001, 42(3): 117-119. (in Chinese)
[16] LU Zhao-ling. Investigation and industrial practice on extraction of V2O5 from stone coal containing vanadium by acid process [J]. Hydrometallurgy of China, 2002, 21(4): 175-183. (in Chinese)
[17] LIANG Jian-long, LIU Hui-juan, SHI Wen-ge, HU E-ming, LI Xi-qi, PENG Jun. A study of a new technology leaching of vanadium ores with hydrometallurgy [J]. China Mining Magazine, 2006, 15(7): 64-66. (in Chinese)
[18] CAO Jian-bing, OUYANG Yu-zhu, LI Zhi-ping, MA Cheng-jin. Study on technological conditions for the extraction of vanadium from stone coal residue [J]. Journal of Hunan City University: Natural Science, 2005, 14(4): 54-56. (in Chinese)
[19] YANG Jing-ling, JIN Xin. A new way of recovering vanadium from iron/vanadium slag [J]. Journal of Beijing University of Chemical Technology, 2007, 30(3): 254-257. (in Chinese)
[20] CHANG Na, GU Zhao-lin, LI Yun. Study on leaching vanadium from stone coal [J]. Inorganic Chemicals Industry, 2006, 38(7): 57-59. (in Chinese)
[21] AMER A M. Hydrometallurgical processing of Egyptian black shale of the Quseir-Safaga region [J]. Hydrometallurgy, 1994, 36(1): 95-107.
[22] TESSIER A, CAMPBELL P G C, BISSON M. Sequential extraction procedures for the speciation of particulate trace metals [J]. Anal Chem, 1979, 51(7): 844-851.
[23] JIN Xiao-zhu. Phase Analysis of vanadium in carbonaceous shale [J]. Chinese Journal of Analysis Laboratory, 2000, 19(2): 50-52. (in Chinese)
______________________________
Foundation item: Project(2006AA06Z130) supported by the Hi-tech Research and Development Program of China; Project(2007GA010) supported by Yunnan Provincial Science and Technology Agency
Corresponding author: WEI Chang; Tel: +86-871-5188819; E-mail: weichang2008@sina.com

