
In situ synthesized Cu-ZSM-5/cordierite for reduction of NO
WANG Jian-cheng1, TIAN Dong1, HAN Li-na1, 2, CHANG Li-ping1, BAO Wei-ren1
1. Key Laboratory of Coal Science and Technology of Ministry of Education and Shanxi Province,
Taiyuan University of Technology, Taiyuan 030024, China;
2. College of Materials Science and Engineering, Taiyuan University of Technology, Taiyuan 030024, China
Received 30 November 2009; accepted 8 August 2010
Abstract: ZSM-5 zeolites were directly synthesized on the surface of honeycomb cordierite substrates by hydrothermal method and certified by XRD and SEM techniques; the adhesion of ZSM-5 coatings was evaluated by ultrasonic vibration. Cu-ZSM-5/cordierite monolithic catalyst was prepared by ion-exchange and impregnation method and applied for the selective catalytic reduction (SCR) of NO by NH3 using a simulated diesel exhaust. The results show that the cordierite surface is almost completely covered by ZSM-5 crystals and the crystallization time greatly impacts the loadings and adhesion of ZSM-5 coatings on substrate, the NOx removal rate over Cu-ZSM-5/cordierite is above 90% in a temperature range of 240-480 °C. Moreover, Cu-ZSM-5/cordierite prepared by different methods shows a wide temperature window (240-540 °C) with high NO removal activities.
Key words: in situ synthesis; Cu-ZSM-5/cordierite; selective catalytic reduction; NH3
1 Introduction
With the increasing number of motor vehicle, the emission of nitrogen oxides into atmosphere has aroused widespread concern. Three-way catalyst converter (TWC) has been successfully developed and widely applied for the gasoline engine exhaust gas cleaning system[1]. However, TWC is not used in dealing with diesel-engine exhaust gas cleaning system because the diesel-engine works under a condition with excess oxygen for the fuel to burn more completely[2-3]. Therefore, the preparation of catalyst with high efficiency for diesel-engine exhaust gas purification has become a pressing task.
Supported metal monolith catalysts, as a combination of high mechanical strength, thermal shock resistance of monolith and specific catalytic properties of the metal component, have been used in automotive emission control reactors for the selective catalytic reduction (SCR) of nitrogen oxides[4-5]. Honeycomb cordierite[6], stainless steel[7] and aluminum alloy[8] etc were used as substrate. Especially, cordierite (2MgO・2Al2O3・5SiO2) monoliths were widely used due to the superior hydrothermal stability, plasticity, good washcoat adherence and compatibility with washcoat[9]. But the specific surface area of most monolithic substrate is below 1 m2/g which is too low for metal to fully disperse onto the surface of catalyst. To improve the surface area of monolithic substrate, Al2O3 is usually coated onto the surface of substrate by dip coating, slip coating or spin coating[10]. Moreover, ZSM-5 has been used as catalysts, catalyst carriers and adsorbents in the environmental protection due to its special pore structure and performance[11]. The study of in situ synthesis indicates that ZSM-5 can be loaded on the cordierite surface to increase the specific surface area. LACHMAN and PATIL[12] synthesized high-silica ZSM-5 zeolite onto honeycomb ceramic supports. Since then, LI et al[13] and WANG et al[14-15] prepared ZSM-5/cordierite and SAPO-34/cordierite by in situ synthesis. LI et al[3] and LACHMAN and PATIL[12] prepared MFI layers on monolithic supports to remove NOx using simulated exhaust gas. The growth mechanism of zeolites on cordierite supports was proposed and the support synthesis was systematically described. In spite of some meaningful results obtained, there are still many problems to overcome. For example,the combining properties of molecular sieve layer and carrier, the uniform distribution of molecular sieve and the amount of molecular sieve on supports, which are crucial for the practical application of the monolithic catalysts.
In this work, ZSM-5/cordierite was prepared by in situ hydrothermal synthesis and Cu-ZSM-5/cordierites were prepared by ion-exchange or impregnation method. The performance of Cu-ZSM-5/cordierite monolithic catalysts in NO removal by NH3 in simulated diesel- engine exhaust conditions was investigated.
2 Experimental
2.1 Synthesis of catalysts
Honeycomb cordierite monoliths were used as substrates. Firstly, the monoliths were calcined at 600 °C to remove organic impurities absorbed in the surface. Subsequently, the monoliths were dealt with 15% nitric acid solution at 80 °C for 4 h in order to increase the surface roughness of cordierite, and then washed by deionized water and calcined at 500 °C. ZSM-5/ cordierite was synthesized using sodium hydroxide as Na source, sodium aluminate as Na and Al sources, silica as Si source and deionized water. The reactants were mixed in a molar ratio of 15(Na2O):(Al2O3): 68(SiO2):3200(H2O). After vigorous stirring for 24 h, the aged synthesis gel (28 mL) and one cordierite monolith (about 3.45 g) were put into a stainless steel autoclave with teflon liner for static crystallization at 180 °C. Crystallization time was controlled in a range from 24 to 60 h. In order to obtain the coatings repeatedly, five samples were prepared for each set of conditions. Then the autoclave was cooled to room temperature and the sample was taken out from the synthesis gel and washed using the deionized water to remove the residues from the solution. Finally, the sample was dried at 120 °C for 5 h. It should be noted that no template agents were used in this process.
Cu-ZSM-5/cordierite catalyst was prepared by ion-exchange or impregnation method. The main process of the ion-exchange was that ZSM-5/cordierite supports were dipped in the solution of ammonium nitrate (0.5 mol/L) at 80 °C for 24 h, and then the supports were calcined at 500 °C. After that, the sample was impregnated with Cu(NO3)2 solution (0.1 mol/L or 0.25 mol/L) under slow stirring at room temperature for 12 h. So the copper species not only provided compensation cations in zeolite but also impregnated into the pores of the sample. The main process of impregnation method was that ZSM-5/cordierite was directly impregnated with Cu(NO3)2 (0.1 mol/L or 0.25 mol/L) at room temperature under slow stirring for 12 h, and then the sample was calcined at 700 °C in air for 12 h.
2.2 Catalysts characterization
The mass increase rate of ZSM-5 on cordierite surface was obtained by calculating the mass increase of cordierite before and after hydrothermal synthesis. The mechanical strength of ZSM-5/cordierite and adhesion degree of ZSM-5 onto cordierite were tested by ultrasonic vibration (300 W, 20 kHz). ZSM-5/cordierite monoliths were treated three times by ultrasonic vibration, each time for 20 min. The samples were dried at 120 °C. The mass increase rate and adhesion degree are calculated as

where ω is the mass increase rate; Δω is the adhesion degree; m0 is the mass of raw cordierite; m1 is the mass of ZSM-5/cordierite; m2 is the mass of ZSM-5/cordierite treated after ultrasonic vibration.
The synthesized ZSM-5/cordierite samples was investigated with a Rigaku D/max2500 diffractometer, using a graphite monochromator and Cu Kα radiation sources (λ=0.154056 nm), tube voltage of 40 kV, tube current of 100 mA. The scanning rate was 6(°)/min with 2θ values from 5° to 35°. In order to analyse accurately, the ZSM-5/cordierite samples were cut into small pieces with size of 10 mm×10 mm.
The morphology of samples was examined with a scanning electron microscope (SEM, Jeol Jsm-6700F). The samples were covered with a thin gold layer to increase the conductivity. An accelerating voltage of 10 kV was used. The surface area of the samples was determined by N2 adsorption on a TriStar 3000 facility (Micromeritics Instrument Corporation).
2.3 Catalytic test
The SCR reaction of NO by NH3 at atmosphere pressure was carried out in a fixed bed microreactor. The gas composition was 0.05% NO, 0.05% NH3, 6.0% O2 and 2.0% H2O (volume fraction) in a N2 balance gas and the total gas flow rate was set at 420 mL/min. 12 g catalyst for each experiment was put into the reactor. The reactor was heated from room temperature to 600 °C at a heating rate of 10 °C/min. The gas composition was analyzed on-line by a gas analyzer (British, Kane-9106). The NO conversion is calculated as:

where X is the conversion rate of NO; c1 is the concentration of NO in the raw gas; c0 is the concentration of NO after reaction at a certain reaction time.
3 Results and discussion
3.1 Effect of crystallization time on ZSM-5/cordierite
The mass increase rate and BET surface areas of monoliths coated for different crystallization time in situ synthesis are shown in Table 1. It should be noted that CT24 stands for the ZSM-5/cordierite with crystallization time of 24 h, and other codes are analogized. When crystallization time was extended from 24 to 60 h, the mass of cordierite increased from 2.63% to 17.36%. The results indicate that ZSM-5 loadings increased gradually and high yields may be gained with increasing crystallization time. Besides, the specific surface area of blank cordierite (0.7 m2/g) significantly increased to 41.7 and 79.2 m2/g after crystallization for 48 and 60 h, respectively.
Table 1 Effect of crystallization time on mass increase and BET surface area of samples

3.2 Adhesion test of ZSM-5/cordierite
Both the mechanical strength of ZSM-5/cordierite and adhesion degree of ZSM-5 onto cordierite were tested by ultrasonic vibration. The effect of ultrasonic time on the mass is shown in Fig.1. The mass loss amount of samples increased gradually with the sample processing time. At the beginning 20 min, the mass loss of zeolite samples were 4.31%, 11.46% and 14.07% corresponding to crystallization time of 24, 48, and 60 h, respectively. With increasing ultrasonic time, the mass loss of ZSM-5 coatings continued to increase, but it tended to equilibrium. When the processing time extended to 60 min, the mass loss amount of the monoliths coating was 4.58%, 14.98% and 19.97% for CT24, CT48 and CT60, respectively. When the monoliths were treated with ultrasonic for 60 min, the mass loss of blank cordierite were 2.5%, 8.68% and 13.89% corresponding to crystallization time of 24, 48 and 60 h, respectively.
3.3 XRD and SEM characterization of ZSM-5/ cordierite
XRD patterns of ZSM-5/cordierite synthesized for different crystallization time are given in Fig.2. To
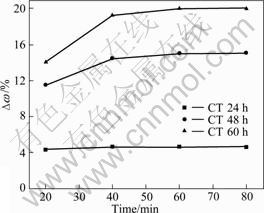
Fig.1 Adhesion of ZSM-5 coating tested with ultrasonic vibration

Fig.2 XRD patterns of ZSM-5/cordierite synthesized for different crystallization time: (a) CT24 powder; (b) CT24; (c) CT48; (d) CT60
compare with ZSM-5 powders, the XRD pattern of ZSM-5 powders obtained from the bottom of autoclave is also given. The characteristic diffraction peaks of ZSM-5 appear at 7.92°, 8.80°, 23.12°, 23.46° and 23.80°. It can be clearly seen that in addition to the characteristic peaks of cordierite, the peaks of CT24, CT48 and CT60 correspond to that of ZSM-5, which indicates that ZSM-5 can be loaded on the cordierite surface by in situ synthesis. However, a small number of other zeolite phase diffraction peaks are also observed in Fig.2. It should be noted that the intensity of characteristic peaks of ZSM-5 in the monolith is quite different from that of pure-phase ZSM-5 peak, and the peak intensity of ZSM-5/cordierite is also different as the crystallization time varies from 24 to 60 h. The peak intensity of samples from CT24, CT48 and CT60 is improved gradually, suggesting that the crystallization time is an important factor. The intensity of ZSM-5 increases with increasing crystallization time, which is consistent with the mass changes of ZSM-5 loading onto the supports.
The XRD patterns of in situ synthesized ZSM-5/ cordierite samples before and after ultrasonic treatment are shown in Fig.3. It can be seen that the intensity of ultrasonic treated ZSM-5 diffraction peaks is weaker and the cordierite peaks become stronger due to the decrease of ZSM-5 loadings.

Fig.3 XRD patterns of CT24 before (a) and after (b) ultrasonic treatment
The SEM images of ZSM-5/cordierite and blank cordierite before in situ synthesis are shown in Fig.4. It is found that the cordierite surface is almost completely covered by ZSM-5 crystalloid (Figs.4(a)-(c)). Moreover, ZSM-5 crystalloid grows not only on the external surface of cordierite (Fig.4(b)) but also on the internal surface
(Fig.4(c)). The size of crystalloid is not uniform. The biggest length of cylinder crystalloid is over 10 μm and the shortest one is about only 1 μm. The size distribution on the external surface and on the internal surface of CT48 is almost the same. It means that in situ synthesis may prepare compact and homogeneous ZSM-5 coatings on different surfaces of cordierite. In addition to the regular ZSM-5 crystalloid, some amorphous crystals are also observed as marked in Fig.4(d), which is consistent with the XRD patterns.
3.4 Catalytic tests of Cu-ZSM-5/cordierite
The performance of NO removing over Cu-ZSM-5/ cordierite is presented in Figs.5-7. SCR of NO by NH3 over Cu-ZSM-5/cordierite prepared by ion-exchange method for resisting water is shown in Fig.5. The catalyst prepared by ion-exchanged method shows a high activity. There is almost no difference between the reaction gas with water and without water. The conversion of NO increased rapidly when the reactor was heated from 160 °C to 260 °C, reached nearly 100% when the temperature rose to 260 °C, and kept stable in the temperature range of 260-460 °C, which indicates that the catalysts have a strong water resistance.
The SCR activities of Cu-ZSM-5/cordierite catalysts prepared by ion-exchange or impregnation method with different concentration all show very high NO conversation (Figs.6 and 7). It is suggested that copper in ZSM-5 is a key factor. The results of SCR

Fig.4 SEM images of surface of blank cordierite (a), internal surface of CT48 (b) and external surface of CT48 (c, d)

Fig.5 SCR of NO by NH3 on Cu-ZSM-5/cordierite prepared by ion-exchange method for resisting water

Fig.6 SCR of NO by NH3 on Cu-ZSM-5/cordierite prepared by ion-exchange method

Fig.7 SCR of NO by NH3 on Cu-ZSM-5/cordierite prepared by impregnation method
activities of Cu-ZSM-5/cordierite obtained from copper precursor solutions with different concentrations indicate that the catalyst obtained from a high concentration solution does not show much better activity (Figs.6 and 7), but the SCR activity of ZSM-5/cordierite is very low. It is suggested that a certain amount copper in Cu-ZSM-5/cordierite plays a key role for SCR-NH3 as reported earlier[16].
4 Conclusions
1) ZSM-5 zeolite coatings were successfully synthesized on cordierite support using in situ hydrothermal synthesis method and ZSM-5 crystalloid almost completely covered the cordierite surface.
2) Crystallization time significantly influences the loading and adhesion of coatings on the surface of cordierite substrate. The cordierite and ZSM-5 have better adhesion strength.
3) Cu-ZSM-5/cordierite is a promising catalyst for the SCR-NH3 process, which was proved to be active in the ambient gas with 2.0% water and 6.0% oxygen. Cu-ZSM-5/cordierite monolithic catalyst prepared by different methods shows high NO removal activities with a wide temperature range of 240-540 °C.
References
[1] KANAZAWA T. Development of hydrocarbon adsorbents, oxygen storage materials for three-way catalysts and NOx storage-reduction catalyst [J]. Catalysis Today, 2004, 96(3): 171-177.
[2] MARTYN V T. Progress and future challenges in controlling automotive exhaust gas emissions [J]. Applied Catalysis B: Environmental, 2007, 70(1-4): 2-15.
[3] LI Lan-dong, CHEN Ji-xin, ZHANG Shu-juan, GUAN Nai-jia, WANG Tian-you, LIU Shu-liang. Selective catalytic reduction of nitrogen oxides from exhaust of lean burn engine over in situ synthesized monolithic Cu-TS-1/cordierite [J]. Catalysis Today, 2004, 90 (3-4): 207-213.
[4] ULLA M A, MALLADA R, CORONAS J, GUTIERREZ L, MIR? E, SANTAMARIA J. Synthesis and characterization of ZSM-5 coatings onto cordierite honeycomb supports [J]. Applied Catalysis A: General, 2003, 253(1): 257-269.
[5] BUENO-LOPEZ A, LOZANO-CASTELLO D, SUCH-BASANEZ I, GARCIA-CORTES J M, ILLAN-GOMEZ M J, DE LECEA C S M. Preparation of beta-coated cordierite honeycomb, monoliths by in situ synthesis utilisation as Pt support for NOx abatement in diesel exhaust [J]. Applied Catalysis B: Environmental, 2005, 58 (1-2): 1-7.
[6] ?HRMAN O, HEDLUND J, STERTE J. Synthesis and evaluation of ZSM-5 films on cordierite monoliths [J]. Applied Catalysis A: General, 2004, 270(1-2): 193-199.
[7] JUAN M Z, MARIA A U, EDUARDO E M. ZSM5 growth on a FeCrAl steel support. Coating characteristics upon the catalytic behavior in the NOx SCR [J]. Microporous and Mesoporous Materials, 2008, 115 (1-2): 113-122.
[8] JUAN M Z, MARIA A U, EDUARDO E M. Improvement in the catalytic performance of in-mordenite through preferential growth on metallic monoliths [J]. Applied Catalysis A: General, 2006, 308(10): 161-171.
[9] JIMMIE L W. Monolith structures, materials, properties and uses [J]. Catalysis Today, 2001, 69(1-4): 3-9.
[10] SU Jun-hua, LIU Zhen-yu, LIU Qing-ya. Honeycomb CuO/ cordierite-Al2O3 catalyst for selective catalytic reduction of NO by NH3-effect of Al2O3 coating [J]. Industrial and Engineering Chemistry Research, 2008, 47(13): 4295-4301.
[11] JANSEN J C, VAN ROSMALEN G M. Oriented growth of silica molecular-sieve crystals as supported films [J]. Journal of Crystal Growth, 1993, 128(1-4): 1150-1156.
[12] LACHMAN I M, PATIL M D. Method of crystallizing a zeolite on the surface of a monolithic ceramic substrate: US 4800187 [P]. 1989.
[13] LI Lan-dong, XUE Bin, CHEN Ji-xin, GUAN Nai-jia, ZHANG Fu-xiang, LIU De-xin, FENG Hong-qing. Direct synthesis of zeolite coatings on cordierite supports by in situ hydrothermal method [J]. Applied Catalysis A: General, 2005, 292(18): 312-321.
[14] WANG An-qin, LIANG Dong-bai, TANG Jun-wang, XU Chang-hai XU Xiao-ying, ZHANG Tao. In-situ synthesis of SAPO-34 molecular sieves on monolithic cordierite substrates [J]. Chemical Journal of Chinese Universities, 2000, 21(12): 1801-1804. (in Chinese)
[15] WANG An-qin, LIANG Dong-bai, TANG Jun-wang, XU Chang-hai, XU Xiao-ying, ZHANG Tao. Catalytic reduction of NO over in situ synthesized Ir/ZSM-5 monoliths [J]. Applied Catalysis B: Environmental, 2001, 32(3): 205-212.
[16] MA A Z, MUHLER M, GR?NERT W. Selective catalytic reduction of NO by ammonia over Raney-Ni supported Cu-ZSM-5 [J]. Applied Catalysis B: Environmental, 2000, 27(1): 37-47.
堇青石基Cu-ZSM-5的原位制备及NO的脱除
王建成1, 田 东1, 韩丽娜1,2, 常丽萍1, 鲍卫仁1
1. 太原理工大学 煤科学与技术教育部和山西省重点实验室, 太原 030024;
2. 太原理工大学 材料科学与工程学院, 太原 030024
摘 要:利用水热合成技术将ZSM-5分子筛直接合成到蜂窝状堇青石上。XRD、扫描电镜观察和超声波处理结果表明,ZSM-5和堇青石能够牢固结合,ZSM-5分子筛涂层质量和牢固度主要取决于晶化时间。采用离子交换或浸渍法制得Cu-ZSM-5/堇青石,利用模拟柴油机车尾气进行选择性催化还原NO。结果表明,在240-480 oC的温度范围内,NO的转化率在90%以上,由离子交换或浸渍法制得的Cu-ZSM-5/堇青石都能在很宽的温度窗口(240-540 oC)保持很高的脱硝转化率。
关键词:原位合成;Cu-ZSM-5/堇青石;选择性催化还原;氨
(Edited by FANG Jing-hua)
Foundation item: Project (20906067) supported by the National Natural Science Foundation of China
Corresponding author: BAO Wei-ren; Tel/Fax: +86-351-6010482; E-mail: baoweiren@tyut.edu.cn
DOI: 10.1016/S1003-6326(11)60721-8