
A novel electroless silver depositing method for magnesium alloys
ZHAO Hui(赵 惠), CUI Jian-zhong(崔建忠)
Key Laboratory of Electromagnetic Processing of Materials, Ministry of Education,
Northeastern University, Shenyang 110004, China
Received 28 July 2005; accepted 15 September 2006
Abstract: Depositing silver on magnesium alloy by both electroless plating and organic coatings was studied. The organic coating was made by immersing samples in organosilicon heat-resisting varnish. In this method the organic coating acts as interlayer between the substrate and silver film. When the reaction starts, silver deposits directly on the interlayer. X-ray diffraction and SEM analysis were used to determine the composition and morphology of the interlayer and silver film. The potentiodynamic polarization curves for corrosion studies of coated magnesium alloys were performed in a corrosive environment of 3.5% NaCl(mass fraction) at neutral pH (6.9). The results indicate that compared with the substrate, the corrosion resistance of coated magnesium alloys increases greatly. Moreover, the method proposed in this work is environmentally friendly, non-toxic chemicals were used. In addition, it provides a new concept for the corrosion inhibition of magnesium alloys.
Key words: magnesium alloy; silver film; organic coating; electroless deposition; corrosion
1 Introduction
Magnesium alloys are used in a wide range of structural applications industries such as aircraft, computer, automobile and electronics. It is expected that the applications, particularly those involving motion or need portability, will increase in the near future because of their high strength-to-mass ratio and relatively high stiffness. However, a relatively poor resistance to corrosion is a serious impediment against wider application of magnesium alloys. At room temperature magnesium and its alloys could be easily oxidized by air. Therefore, some methods must be supplied to improve the corrosion resistance of magnesium and its alloys. One of the most significant ways to protect corrosion is to coat the base material[1-6].
Among the various technologies available for coating, organic coatings are the most common, simple and cost effective mode of corrosion protection for metallic objects and structures. Their possible uses as primer or top coatings or blends have been studied by many researchers[7-12]. In recent years, electroless plating has attracted attention due to its virtues. The electroless bath provides a deposit that follows all contours of the substrate exactly. A sharp edge receives the same thickness of deposit as does a blind hole[13-16]. Silver was recently promoted as a potential candidate due to its low room temperature bulk receptivity (1.59 ?Ω?cm) and relatively high melting point in comparison to the common Al and Cu metallization. Silver films have perfectly corrosion resistant, antibacterial and decorative properties. There are many reports about depositing silver on material surfaces[17-21]. Nevertheless, the published literature about electroless depositing silver onto the surfaces of magnesium and its alloys are rather scanty. It is because that electrolessly deposited silver on magnesium directly and its alloys are difficult to carry out due to its high reactivity. However, we successfully obtain a silver film on the magnesium alloys by using electroless plating and organic protection coating. We present experimental data on coating process, microstructure of the interlayer and the silver film, and corrosion resistance of the coated composite.
2 Experimental
2.1 Specimen preparation
In this study the substrates used for the silver films were AZ31 rolling magnesium alloys plates, supplied by Yingkou Magnesium Alloy Factory, China. The organic coating used was a kind of commercial varnish, 8604 organosilicon heat-resisting varnish, which has favorable water proof and salts mist resisting.
The process of sample preparation was as follows.
1) Surface treatment. It was carried out by immersing samples in a 30 mL/L HNO3 aqueous solution at room temperature for 3 min. Then, the samples were cleaned by water and dried completely in the drying oven.
2) Coating interlayer. In this process, the samples were submersed in the 8604 organosilicon heat-resisting varnish; next, hung in air for 30 min; then, dried absolutely at 180 ℃ for 1 h in the drying oven; finally, repeated above-mentioned procedures twice.
3) Surface roughing. Surface roughing was actually a caustic etching process. Roughing solution was constituted by 600 g/L NaOH aqueous solution at 30 ℃. The time of roughing was controlled to 30 min.
4) Surface sensitization. It was conducted by immersing the samples in 10 g/L SnCl2 and 100 mL/L HCl at 30 ℃ for 6 min. Finally, electroless deposition.
In this process silver films were deposited from an AgNO3-based solution. Glucose was used as a reducing agent. The composition of the silver salts solution in the plating bath was as follows: solution A contained 3.5 g AgNO3, 60 mL adequate NH4OH and 0.025 g/mL NaOH. Solution B contained 5 g/L of dextrose, 4 g/L tartaric acid, 200 mL/L ethanol and deionized water. The two solutions A and B were mixed in a 1?1(volume ratio) just before the bath were immersed[22]. To deposit silver, the samples were immersed into the mixture for 30min at room temperature. Then, the samples were rinsed quickly with deionized water and dried in the oven with air blower at 30 ℃.
2.2 Specimen characterization
Morphological analysis was conducted on the surfaces before and after etching the samples coated with interlayer, and electroless metallization with silver using a scanning electron microscope (SEM). The non-conducting specimens were coated with gold prior to the analysis.
X-ray diffraction(XRD) analysis of samples before and after being metallized was preformed with an X-ray diffractometer (X'Pert Pro MPD). The incident radiation was Cu Kα. Data were collected form 20? to 90?.
For studying the corrosion resistance of samples coated with coatings, the potentiodynamic polarization curves were used to investigate the corrosion behavior of coated samples. The samples were prepared by initially attaching an electrical connection wire to the surface with conducting glue. Surface with dimension of 1 cm×1 cm of the sample was exposed and the rest was sealed with silica gel. The experiment was carried out in a corrosive environment of 3.5% NaCl aqueous solution (pH6.9) at room temperature. The solution was prepared with AR grade NaCl. The potential of the sample was swept using an Autolab Pgstat 302 electrochemical measurement system. The polarization started from about -1 750 to 250 mV at a scan rate of 1 mV/s to construct the Tafel plots (logarithmic variation of current as a function of voltage) and derive the anodic and catholic Tafel constants. A standard calomel electrode (SCE) was used as a reference electrode and platinum mesh was used as a counter electrode. The samples were immersed in solution for 15 min prior to each polarization test, allowing the system to be stabilized.
3 Results and discussion
A major source of the loss of corrosion protection in paint films is non-uniform film properties of defects on the films[23]. Fig.1 shows the cross-sectional SEM image of the substrate after coating. The film is compact and symmetrical. The interlayer is about 40?m. The barrier properties of organic coatings are of primary interest in the design of high performance coatings for long-term protection of metal substrates. But it will fail when there is lack of adhesion between the film and the substrate. It means that the properties of the coating/metal substrate interface are closely tied to the barrier properties of the system[23]. As shown in Fig.1, after being polished, there exist no separation and flaw between the interlayer and the substrate. Accordingly, the adhesion between them is excellent.

Fig.1 Cross-sectional SEM image of interlayer/substrate
Fig.2 shows the micrographs of the interlayer surface before and after three etchings. The non-etched sample (Fig.2(a)) presents the surface morphology preferentially amorphous. The surface of the interlayer is hydrophobe and smooth. Nevertheless it is unfavorable to subsequent plating. The aim of the etching is to increase the hydrophilic character and surface roughness of the interlayer on substrates, allowing anchorage of the silver film during the metallization process. After etching, it is obvious that an increase in the micro-roughness occurs in this sample (Fig.2(b)).

Fig.2 SEM images of interlayer surface before (a) and after (b) etching
After etching, the samples are completely immersed in the plating solution. The silver particles are deposited on the samples by reducing Ag+. Fig.3 shows the SEM
micrographs of the samples surfaces plated for different time. It can be seen that the distribution of solid film on the substrate surface is homogeneous on the whole. The sliver film is a uniform crystalline layer, except for some minor pores. The silver grains are of nearly spherical shape, which indicates the similar growth rate in all directions. At the beginning of the plating process, more small silver particles appear (Figs.3(a) and (b)). However, at this time, silver film is not formed though there are many small silver particles on the interlayer. Because of without plating gold on the surface of the sample, the morphology of the surface could not be seen under the scanning electron microscope (Figs.3(a) and (b)). As the plating time prolongs, these grains come in contact with each other and continuous film forms. After the first layer of sliver film forms, some grains grow above it and form silver particle clusters (Figs.3(c) and(d)). During the prolonged plating, silver ions in the solution become diluted and eventually reach a low level at which the whole reaction would be ceased.
X-ray diffraction patterns of the samples before and
after plating are shown in Fig.4. The substrates are plated in the same solution respectively for 2, 6, 15 and 24 min. Fig.4 shows the XRD patens of the sample before plating, silver films plated on the samples for different time. From the image, it can be seen that the silver grains are of perfect crystal structure. The intensities of silver peaks increase with the plating time, witch is caused by the growth of the silver particles. All the peaks other than those of silver are attributed to the interlayer on the substrates. In Fig.4, the intensities of interlayer are decreased with the increasing plating time, indicating the growth of silver film. Furthermore, there is no significant peak shift of silver, suggesting a well-crystallized lattice structure.

Fig.3 SEM images showing growth of silver grains plating with time: (a), (b) 2 min; (c), (d) 25 min
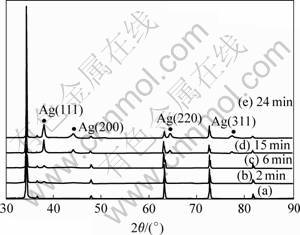
Fig.4 X-ray diffraction patterns of samples: (a) Sample only with interlayer; (b)-(e) Silver films plated on samples for different time
Tafel polarization curves are employed to evaluate the effect of the coatings on the corrosion resistance characteristics of magnesium alloys. The standard electrode potential of pure magnesium is known to be the lowest among engineering metallic materials, implying its poor corrosion resistance. Fig.5 shows the polariz- ation curves of the AZ31 magnesium plate and silver- coated sample in a 3.5% NaCl solution. From these curves, it can be obviously seen that the corrosion potential (φcorr) of the AZ31 magnesium plate and silver coated sample are -1.569 and -1.259 V, respectively. There is a about 300 mV increase in the matter of theφcorr. The effect of coatings on the polarization of AZ31 magnesium plate is clear in moving φcorr to the noble direction. It is also noted that once the scanned potential exceeds -1.57 V, corrosion current density of magnesium plate instantaneously increases. However the corrosion current density of the coated plates is stabilized when the scanned potential exceeds about -0.5 V.

Fig.5 Potentiodynamic polarization curves of non-coated(a) and silver-coated(b) samples in 3.5%NaCl at 30 ℃
4 Conclusions
A novel approach to coat surfaces of magnesium alloys was developed. The optimum silver coating is achieved by using electroless deposition and organic coatings. The micro-morphology of films was studied by SEM and X-ray diffraction investigations. The adhesion between the interlayer and the substrate is good. To improve the adhesion between the silver film and interlayer, caustic etching has been employed to increase surface roughness. Organic protection coating acts as interlayer in this method and silver particles will deposit virtually on the surface of interlayer. Accordingly, once the silver film broken, the interlayer can restrain the occurrence of double action between the silver film and the substrate. The anodic polarization behavior reveals that the coatings enhance the corrosion resistance of AZ31 alloys. In addition, the method proposed can be used for plating the metals with high reactivity.
References
[1] MORDIKE B L, EBERT T. Magnesium: Properties-applications-
potential.[J]. Mater Sci Eng A, 2001, 302: 37-45.
[2] LIU zhen-min, GAO Wei. The effect of substrate on the electroless nickel plating of Mg and Mg alloys[J]. Surface and Coatings Technology, 2006, 200: 3553-3560.
[3] COLE G S, SHERMAN A M. Lightweight materials for automotive applications[J]. Materials Characterization, 1995, 35: 3-9.
[4] MAJUMDAR J J, GALUN R, MORDIKE B L, MANNA I. Effect of laster surface melting on corrosion and wear resistance of a commercial magnesium alloy[J]. Mater Sci Eng A, 2003, A361: 119-129.
[5] Rajan A, ZHOU W. Electroless nickel-plating on AZ91D magnesium alloy: Effect of substrate microstructure and plating parameters[J]. Surface and Coatings Technology, 2004, 179: 124-134.
[6] YFANTIS A, PALOUMPA I, SCHMEIBER D, YFANTIS D. Novel corrosion-resistant films for Mg alloys[J] Surface and Coatings Technology, 2002, 151/152: 400-404.
[7] GRAY J E, LUAN B. Protective coatings on magnesium and its alloys-a critical review[J]. Journal of Alloys and Compounds, 2002, 336: 88-113.
[8] TUN? T. Zinc deposited polymer coatings for copper protection[J]. Progress in Organic Coatings, 2006, 55: 60-65.
[9] LI Yi, LU Qing-hua, QIAN Xue-feng, ZHU Zi-kang, YIN Jie. Preparation of surface bound copper nanoparticles on polyimide by surface modification method and its application on electroless metal deposition[J]. Applied Surface Science, 2004, 233: 299-306.
[10] WANG W C, HVORA R, KANG E T, NEOH G K. Electroless plating of copper on fluorinated polyimide films modified by surface graft copolymerization with 1-vinylimidazole and 4-vinylpyridine[J]. Polymer Engineering and Science, 2004, 44: 362-375.
[11] DOMENECH S C, LIMA E Jr, DRAGO V, De LIMA J C. Electroless plating of nickel-phosphorous on surface-modified poly (ethylene terephthalate) films[J]. Applied Surface Science, 2003, 220: 238-250.
[12] Pérez P A, Jorge A R, Hugo S A. Synthesis and water ultrafiltration properties of copper membrane supported on porous ceramics[J]. Desalination, 1997, 114: 203-208.
[13] CHOI S J, CHOI J, SEO C Y, PARK C N. An electroless copper plating method for Ti. Zr-based hydrogen storage alloys[J]. Journal of Alloys and Compounds, 2003, 356-357: 725-729.
[14] LIU R S, YOU C C, TSAI M S, HU S F, LI Y H, LU C P. An investigation of smooth nano-sized copper seed layers on TiN and TaSiN by new non-toxic electroless plating[J]. Solid State Communications, 2003, 125: 445-448.
[15] GONG Ying-peng, GUO Zhang-cheng, LU Wei-chang, Electroless copper coating on nickel foils in super-gravity field [J]. Materials Letters, 2005, 59: 667-670.
[16] EUGENIJUS N, ALGIRDAS V, JAN? J, IRENA S, GIEDRIUS S. Obtaining of high surface roughness copper deposits by electroless plating technique[J]. Electrochemical Acta, 2006, 51: 3495-3499.
[17] LI Yi, LU Qing-hua, QIAN Xue-feng, ZHU Zi-kang, YIN Jie. Preparation of surface bound silver nanoparticles on polyimide by surface modification method and its application on electroless metal deposition[J]. Applied Surface Science, 2004, 233: 299-306.
[18] GRAY J E, GRIFFITHS K. Mechanism of adhesion of electroless- deposited silver on poly (ether urethane) [J]. Thin solid films, 2005, 484: 196-207.
[19] MEI Fang, SHI Dong-lu. Electroless plating of thin silver films on porous Al2O3 substrate and the study of deposition kinetics [J].Tsing-
hua Science and Technology, 2005, 10: 680-689.
[20] HUANG T C, WEI M C, CHEN H I. Preparation of hydrogen- permselective palladium-silver alloy composite membranes by electroless co-deposition[J]. Separation and Purification Technology, 2003, 32: 239-245.
[21] BOGUSH V, INBERG A, CROITORU N, DUBIN V, SHACHANM- DIAMAND Y. Electroless deposition of novel Ag-W thin films[J]. Microelectronic Engineering, 2003, 70: 489-494.
[22] LI Ning. Electroless Plating Utility Technology[M]. Beijing: Chemical Technology Press, 2004.
[23] BIERWAGEN G P. Reflections on corrosion control by organic coatings [J]. Progress in Organic Coatings, 1996, 28: 43-48.
(Edited by LONG huai-zhong)
Corresponding author: CUI Jian-zhong; Tel: +86-24-83681738; E-mail: jzcui@mail.neu.edu.cn