J. Cent. South Univ. Technol. (2010) 17: 498-503
DOI: 10.1007/s11771-010-0513-5 
Preparation and electrochemical properties of Co3O4/graphite composites as anodes of lithium ion batteries
GUO Hua-jun(������), LI Xiang-qun(����Ⱥ), LI Xin-hai(���º�), WANG Zhi-xing(��־��),
PENG Wen-jie(���Ľ�), SUN Qian-ming(��Ǭ��), XIE Jie(л��)
School of Metallurgical Science and Engineering, Central South University, Changsha 410083, China
? Central South University Press and Springer-Verlag Berlin Heidelberg 2010
Abstract: Co3O4/graphite composites were synthesized by precipitation of cobalt oxalate on the surface of graphite and pyrolysis of the precipitate, and the effects of graphite content and calcination temperature on the electrochemical properties of the composites were investigated. The samples were characterized by thermogravimetry and differential thermal analysis (TG/DTA), X-ray diffractometry (XRD), scanning electron microscopy (SEM), cyclic voltammetry (CV), electrochemical impedance spectroscopy (EIS) and charge/discharge measurements. With increasing the graphite content, the reversible capacity of the Co3O4/graphite composites decreases, while cycling stability improves dramatically, and the addition of graphite obviously decreases the average potential of lithium intercalation/deintercalation. The reversible capacity of the composites with 50% graphite rises from 583 to 725 mA?h/g as the calcination temperature increases from 300 to 500 ��, and the Co3O4/graphite composites synthesized at 400 �� show the best cycling stability without capacity loss in the initial 20 cycles. The CV profile of the composite presents two couples of redox peaks, corresponding to the lithium intercalaction/deintercalation for graphite and Co3O4, respectively. EIS studies indicate that the electrochemical impedance decreases with increasing the graphite content.
Key words: composite materials; cobalt oxides; lithium ion batteries; graphite; electrochemical properties; precipitation
1 Introduction
Graphite has been commercially used as the anode material of lithium ion batteries owing to its excellent charge/discharge behavior. However, with the growing demand for high capacity secondary batteries, the low capacity of graphite (theoretical capacity: 372 mA?h/g) has become one of the limiting factors. Much work was done to develop alternative anode materials with high capacity and many achievements were made [1-3].
Transition metal oxides, such as nickel oxides, cobalt oxides, tin oxides and iron oxides, were proposed as potential alternatives to carbon anode materials for lithium ion batteries [1, 4-6]. Among which cobalt oxides demonstrated a large reversible capacity as lithium storage materials. A variety of methods such as direct decomposition of cobalt compounds [7], chemical vapor deposition [8], ultrasonic spray pyrolysis [9], hydrothermal synthesis [10] and electrolytic deposition [11] were used in the preparation of cobalt oxides. However, the cobalt oxides usually undergo a relatively fast capacity fade during cycling because of their low conductivity, large volume change, serious aggregation or pulverization in the charge/discharge process [12-14]. To further improve the performance of cobalt oxides, YAO et al [12, 15] developed cobalt oxides/carbon nanofiber (16.7%-24.3%, mass fraction) composites with a large reversible capacity above 900 mA?h/g in the initial 50 cycles. NUNI et al [13] synthesized nickel- cobalt oxides/carbon nanoflake (11.6%-20.1%, mole fraction) composites with an initial reversible capacity of 792 mA?h/g and a capacity retention ratio of 88.3% after 30 cycles.
The present work aims to develop a new Co3O4/C composite using graphite as carbon material instead of nanosized carbon. Compared with the nanosized carbon materials, graphite is much cheaper and more abundant in resource. Furthermore, graphite has larger tap density and better lubricating ability, which facilitate the fabrication of anode electrode. In this work, the Co3O4/ graphite composites were synthesized by precipitation of cobalt oxalate on the surface of graphite and pyrolysis of the precipitate. The effects of graphite content and calcination temperature on properties of the composites were investigated.
2 Experimental
Cobalt chloride (CoCl2��6H2O) and different amounts of artificial graphite were added into distilled water and mixed by stirring for 10 min, and then ammonium oxalate solution was added into the mixture with stirring at 50 �� in a water bath, where the mole ratio of cobalt chloride to ammonium oxalate was 1:1.2. After stirring for 1 h, the precipitates were collected by filtration and washed with distilled water, and then dried in an oven at 120 �� for 6 h. The Co3O4/graphite composites were obtained by calcining the precipitates in air at different temperatures for 2 h.
TG/DTA data for decomposition and oxidation of cobalt oxalate (CoC2O4��2H2O) and CoC2O4��2H2O/ graphite composite precursors were obtained at a heating rate of 5 ��/min in air with a Mettler Toledo TGA/ SDTA851e thermal gravimetric analyzer. Powder X-ray diffraction analysis was conducted on a Rigaku diffractometer. Scanning electron micrographs were obtained with a JEOL JSM-5600LV spectrometer.
The Co3O4/graphite composite (or pure Co3O4), acetylene black and poly-vinylidene difluoride (PVDF) were mixed in a mass ratio of 8:1:1 and emulsified in N-methyl-2-pyrrolidone. The cathode was prepared by spreading the resulting slurry on a copper foil and dried overnight at 120 ��. The coin cell was prepared in Ar atmosphere glove box using Li foil as an anode and UP 3025 porous membrane as a separator. The electrolyte was 1 mol/L LiPF6 dissolved in a mixture of ethylene carbonate (EC), dimethyl carbonate (DMC) and methyl-ethyl carbonate (EMC) with a volume ratio of 1:1:1. The cell was charged and discharged from 0.01 to 3.0 V at a current density of 120 mA/g.
Cyclic voltammetery was carried out on a CHI660A electrochemical work-station between 0 and 3.0 V at a scan rate of 0.1 mV/s and electrochemical impedance spectra were measured between 0.001 and 100 kHz.
3 Results and discussion
3.1 Thermal analysis of precursors
The TG/DTA measurements of cobalt oxalate and cobalt oxalate/graphite composite precursors were carried out to find out the suitable temperature for calcination of the precursors. The results are shown in Fig.1. In the TG/DTA profiles of cobalt oxalate shown in Fig.1(a), a mass loss rate of 18.1% can be observed from 148 to 193 �� on the TG curve, which corresponds to a broad endothermic peak at around 180 �� on the DTA curve. This is due to the dehydration of chemically bonded water in the CoC2O4��2H2O sample. The second mass loss process and the corresponding sharp exothermic peak occur at around 283 ��, indicating the decomposition and oxidation reaction of CoC2O4 as follows [11]:

Fig.1 TG/DTA profiles of cobalt oxalate (a) and cobalt oxalate/graphite composite precursors (b) at heating rate of 5 ��/min in air
3CoC2O4��2O2��Co3O4��6CO2�� (1)
The mass loss rate in this process is 37.08%, which is consistent with the throretcial value of 36.46%.
As shown in Fig.1(b), the cobalt oxalate/graphite composite precursors present similar TG/DTA profiles to those of cobalt oxalate between room temperature and 450 ��, where the graphite content in the composite precursor is 30.48 % and the mass ratio of Co3O4 to graphite in the final product is 1:1. The sample mass remains 61.08% after the second mass loss process, which is consistent with the throretcial value of 60.96%. However, another drastic mass loss occurs from 450 to 670 �� on the TG curve and corresponds to a broad and high exothermic peak on the DTA curve, indicating the oxidation of graphite in the composite precursor.
Based on the above results, a calcination temperature range of 300-500 �� is determined in order to decompose cobalt oxalate completely and avoid dramatic oxidation of graphite.
3.2 Effects of graphite content on properties of Co3O4/ graphite composites
Pure Co3O4 and Co3O4/graphite composites with different mass ratios of Co3O4 to graphite were prepared by calcining their precursors at 300 �� for 2 h. XRD patterns of the samples are shown in Fig.2. The diffraction peaks of pure Co3O4 represent the typical character of a face-centered cubic (FCC) Co3O4 phase of fd3m space group (JCPDS 74-1656). Diffraction peaks of graphite can be observed at around 2��=26.5? and 54.5? in the XRD patterns of the composites. With increasing the graphite content, the intensity of the diffraction peaks for graphite increases, while the diffraction peaks for Co3O4 become low and broad. This can be attributed to the decrease in graphite content and crystallinity of Co3O4 phase.
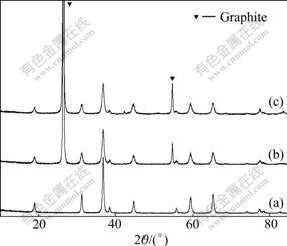
Fig.2 XRD patterns of pure Co3O4 and Co3O4/graphite composites: (a) Pure Co3O4; (b) Mass ratio of Co3O4 to graphite is 1:1; (c) Mass ratio of Co3O4 to graphite is 1:2
Fig.3 shows SEM images of pure Co3O4 and Co3O4/graphite composites with different mass ratios of Co3O4 to graphite. Powders of all the three samples consist of aggregates of primary particles and present similar morphology of stick clusters. Compared with pure Co3O4 sample, the composites have thin and short sticks, indicating that the primary particle size decreases with increasing the graphite content. During the preparation of the composites, cobalt oxalate deposits on the surface of graphite, so tiny cobalt oxalate particles can be obtained for the composite precursors, and the excessive growth and aggregation of Co3O4 are suppressed by the graphite particles during the calcination.
Fig.4 shows electrochemical performances of the pure Co3O4 and Co3O4/graphite composites charged/ discharged between 0.01 and 3 V versus Li/Li+ at a current density of 120 mA/g. As shown in Fig.4(a), the initial discharge (lithium intercalation) profile of pure Co3O4 presents a long voltage plateau (about 820 mA?h/g) at around 1.05 V and then a sloping voltage profile from 1.05 to 0.01 V, and the charge (lithium deintercalation) process exhibits a higher and more sloping voltage

Fig.3 SEM images of pure Co3O4 and Co3O4/graphite composites: (a) Pure Co3O4; (b) Mass ratio of Co3O4 to graphite is 1:1; (c) Mass ratio of Co3O4 to graphite is 1:2
profile, with a charge voltage plateau at about 2.0 V. The initial discharge capacity of the pure Co3O4 reaches 1 317 mA?h/g and the charge capacity (reversible capacity) amounts to 972 mA?h/g. The Co3O4/graphite composites present similar discharge/charge curves at above 0.25 V, but another couple of discharge/charge voltage plateau corresponding to the lithium intercalation/ deintercalaction can be observed in the voltage curves. With increasing the graphite content, the plateau in the low voltage range rises while that in the high voltage range decreases. The Co3O4/graphite composites with 50.0% and 66.7% graphite show reversible capacities of 583 and 457 mA?h/g, and the charge capacities below 0.25 V for them are 136 and 206 mA?h/g, respectively. The results indicate that the addition of graphite can obviously decrease the average lithium intercalation/ deintercalation potential, which facilitates to improve the voltage of lithium ion batteries with Co3O4 composite anodes. This is due to the increase in capacity corresponding to lithium storage of graphite, decrease in Co3O4 primary particle and increase in conductivity of the Co3O4/graphite composites.
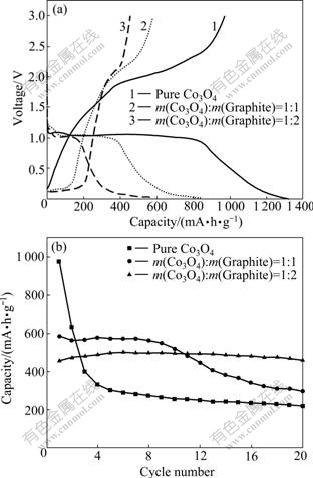
Fig.4 Initial charge/discharge curves (a) and cycling performance profiles (b) of pure Co3O4 and Co3O4/graphite composites
Fig.4(b) shows the cycling performance profiles of pure Co3O4 and Co3O4/graphite composites. The reversible capacity of pure Co3O4 declines quickly during the first several cycles and a capacity retention ratio of 22.6% can be observed after 20 cycles, while the Co3O4/graphite composite with 66.7% graphite shows good cycling stability with a capacity retention ratio of 91.8% after 20 cycles, indicating that the cycling stability is improved dramatically with increasing the graphite content. This can be attributed to the facts that the addition of graphite decreases the particle size, the graphite acts as the framework of the composite and depresses the aggregation or pulverization of Co3O4 particles during the cycling.
Based on the above results, with combined consideration for preferable capacity and cycling performance, the Co3O4/graphite composite with 50% graphite is used in this work on effects of calcination temperature.
3.3 Effects of calcination temperature on properties of Co3O4/graphite composites
Fig.5 shows XRD patterns of Co3O4/graphite composites synthesized at different temperatures, where

Fig.5 XRD patterns of Co3O4/graphite composites synthesized at different temperatures: (a) 300 ��; (b) 400 ��; (c) 500 ��
the mass ratio of Co3O4 to graphite is 1:1. With increasing calcination temperature, the diffraction peaks for Co3O4 become intense and sharp, while those for graphite become low, indicating that the crystallinity of Co3O4 increases and graphite content decreases due to partial oxidation of graphite.
Fig.6 shows SEM images of Co3O4/graphite composites synthesized at different temperatures. Compared with the composites synthesized at 300 �� shown in Fig.3(b), those synthesized at 400 and 500 �� present tighter and smoother surface, and the particle size rises with the increase of calcination temperature. This is due to better growth of Co3O4 crystal and particles at higher temperatures.
Fig.7 shows the charge/discharge performances of Co3O4/graphite composites synthesized at different temperatures. The initial discharge/charge profiles of

Fig.6 SEM images of Co3O4/graphite composites synthesized at different temperatures: (a) 400 ��; (b) 500 ��

Fig.7 Initial charge/discharge curves (a) and cycling performance profiles (b) of Co3O4/graphite composites synthesized at different temperatures
Co3O4/graphite composites synthesized at different temperatures are similar, but the capacity and the cycling stability are different. Discharge/charge plateau in high voltage range rises with increasing the calcination temperature. The Co3O4/graphite composites synthesized at 300, 400 and 500 �� deliver initial discharge capacities of 850, 884, 973 mA?h/g, and reversible capacities of 583, 658 and 725 mA?h/g, respectively. The capacity increase for the composites synthesized at higher temperature may be attributed to the higher crystallinity of Co3O4 and lower graphite content resulting from partial oxidation of graphite. As shown in Fig.7(b), the composites synthesized at 400 and 500 �� show greatly improved cycling performance compared with that at 300 ��. The capacity retention ratios after 20 cycles for Co3O4/graphite composites synthesized at 300 and 500 �� are 50.6% and 92.0%, respectively. While Co3O4/graphite composites synthesized at 400 �� show the best cycling stability without capacity loss during the initial 20 cycles. This may be due to the preferable combination of good crystallinity and small particle size of Co3O4 for Co3O4/graphite composites synthesized at 400 ��.
3.4 Cyclic voltammetry and electrochemical impedance spectroscopy
Fig.8 shows the cyclic voltammogram of Co3O4/ graphite composites synthesized at 300 �� and a scan rate of 0.1 mV/s, where the mass ratio of Co3O4 to graphite is 1:1. During the discharge process, a large cathodic current peak corresponding to the reduction (lithium intercalation) of Co3O4 appears at 0.8 V. And then the cathodic current increases with descending the potential of Co3O4/graphite composite to 0 V (vs Li+/Li), which is due to the lithium intercalation into the graphite. Two anodic current peaks at around 0.2 and 2.2 V are observed in the charge process, which correspond to the lithium deintercalation from the lithiated graphite and Co3O4, respectively. The two couples of redox peaks are consistent with the voltage plateau in the discharge/discharge curves.

Fig.8 Cyclic voltammogram of Co3O4/graphite composite with mass ratio of 1:1 at scan rate of 0.1 mV/s
AC impedance measurement was carried out between 0.001 and 100 kHz. The results are shown in Fig.9. The impedance plot consists of a depressed semicircle and an inclined line. The intercept on Z�� axis in a high frequency range corresponds to the ohmic resistance (Re), which represents the resistance of the electrolyte. The semicircle in the middle frequency range indicates the charge transfer resistance (Rct). The inclined line in the low frequency range represents the Warburg impedance (Zw), which is associated with lithium ion diffusion in the cathode particles [16]. In this work, the Re values are almost the same throughout the experiments due to the same electrolyte and fabrication parameters, and they are around 5 �� and much smaller than the values of Rct. Rct values for Co3O4 electrodes with a graphite content of 0, 50.0% and 66.7% can be evaluated to be about 370, 85 and 75 ��, respectively, indicating that Rct decreases with increasing the graphite content. This can be attributed to the decrease in particle size of Co3O4, higher reactivity and conductivity of graphite compared to those of Co3O4.
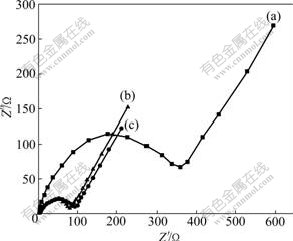
Fig.9 Nyquist impedance spectra of different samples: (a) Pure Co3O4; (b) Mass ratio of Co3O4 to graphite is 1:1; (c) Mass ratio of Co3O4 to graphite is 1:2
4 Conclusions
(1) Co3O4/graphite composites are synthesized by precipitation of cobalt oxalate on the surface of graphite and pyrolysis of the precipitate. Cobalt compound exists as Co3O4 phase of fd3m space group in the composites.
(2) With increasing the graphite content from 0 to 66.7%, the excessive growth and aggregation of Co3O4 are suppressed, the particle size of Co3O4 decreases, the reversible capacity decreases from 972 to 457 mA?h/g, while the capacity retention ratio after 20 cycles is greatly improved from 22.6% to 91.8%. The addition of graphite decreases the average potential of lithium intercalation/deintercalation, which facilitates to improve the voltage of lithium ion batteries with Co3O4 composite anodes.
(3) As the calcination temperature increases from 300 to 500 ��, the crystallinity of Co3O4 and the particle size rise, and the reversible capacity increases. The capacity retention ratios after 20 cycles for the Co3O4/ graphite composites synthesized at 300 and 500 �� is 50.6% and 92.0%, respectively, while the composite synthesized at 400 �� shows the best cycling stability without capacity loss during the initial 20 cycles.
(4) Two couples of redox peaks are observed in the CV profiles of Co3O4/graphite composite. The redox peaks can be attributed to the lithium intercalaction/ deintercalation for the graphite and Co3O4, respectively. The charge transfer resistance Rct for Co3O4 electrodes with graphite contents of 0, 50% and 66.7% can be evaluated to be about 370, 85 and 75 ��, respectively, indicating that Rct decreases with increasing the graphite content.
References
[1] Poizot P, Laruelle S, Grugeon S, Dupont L, Tarascon J M. Nano-sized transition-metal oxides as negative- electrode materials for lithium-ion batteries [J]. Nature, 2000, 407: 496-499.
[2] HE Ze-qiang, XIONG Li-zhi, LIU Wen-ping, WU Xian-ming, CHEN Shang, HUANG Ke-long. Synthesis and electrochemical properties of SnO2-polyaniline composite [J]. Journal of Central South University of Technology, 2008, 15(2): 214-217.
[3] Kim S S, Ikuta H, Wakihara M. Synthesis and characterization of MnV2O6 as a high capacity anode material for a lithium secondary battery [J]. Solid State Ionics, 2001, 139: 57-65.
[4] Nuli Y N, Zhao S L, Qin Q Z. Nanocrystalline tin oxides and nickel oxide film anodes for Li-ion batteries [J]. Journal of Power Sources, 2003, 114: 113-120.
[5] Belliard F, Irvine T S. Electrochemical performance of ball-milled ZnO-SnO2 systems as anodes in lithium-ion battery [J]. Journal of Power Sources, 2001, 97/98: 219-222.
[6] WANG G X, CHEN Y, KONSTANTINOV K, YAO J, AHN J h, LIU H K, DOU S X. Nanosize cobalt oxides as anode materials for lithium-ion batteries [J]. Journal of Alloys and Compounds, 2002, 340: L5-L10.
[7] Wang G X, Chen Y, Konstantinov K, Lindsay M, Liu H K, Dou S X. Investigation of cobalt oxides as anode materials for Li-ion batteries [J]. Journal of Power Sources, 2002, 109: 142-147.
[8] BAHLAWANE N, RIVERA E F, KOHSE-HOINGHAUS K, BRECHLING A, KLEINEBERG U. Characterization and tests of planar Co3O4 model catalysts prepared by chemical vapor deposition [J]. Appl Catal B: Environmental, 2004, 53(4): 245-255.
[9] Oh S W, Bang H J, Bae Y C, Sun Y K. Effect of calcination temperature on morphology, crystallinity and electrochemical properties of nano-crystalline metal oxides (Co3O4, CuO, and NiO) prepared via ultrasonic spray pyrolysis [J]. Journal of Power Sources, 2007, 173: 502-509.
[10] Liu Y, Mi C, Su L, Zhang X. Hydrothermal synthesis of Co3O4 microspheres as anode material for lithium-ion batteries [J]. Electrochimica Acta, 2008, 53: 2507-2513.
[11] LIU H C, YEN S K. Characterization of electrolytic Co3O4 thin films as anodes for lithium-ion batteries [J]. Journal of Power Sources 2007, 166: 478-484.
[12] Yao W L, Wang J L, Yang J, Du G D. Novel carbon nanofiber-cobalt oxide composites for lithium storage with large capacity and high reversibility [J] Journal of Power Sources, 2008, 176: 369-372.
[13] Nuli Y, Zhang P, Guo Z, Liu H, Yang J, Wang J. Nickel- cobalt oxides/carbon nanoflakes as anode materials for lithium-ion batteries [J]. Mater Res Bull, 2009, 44: 140-145.
[14] Nam K W, Lee E S, Kim J H, Lee Y H, Kim K B. Synthesis and electrochemical investigations of Ni1�CxO thin films and Ni1�CxO on three-dimensional carbon substrates for electrochemical capacitors [J]. J Electrochem Soc, 2005, 152: A2123-A2129.
[15] Yao W L, Yang J, Wang J, Tao L. Synthesis and electrochemical performance of carbon nanofiber-cobalt oxide [J]. Electrochimica Acta, 2008, 53: 7326-7330.
[16] Gao F, Tang Z Y. Kinetic behavior of LiFePO4/C cathode material for lithium-ion batteries [J]. Electrochimica Acta, 2008, 53: 5071-5075.
Foundation item: Project(2007CB613607) supported by the National Basic Research Program of China; Projects(2009FJ1002, 2009CK3062) supported by the Science and Technology Program of Hunan Province, China
Received date: 2009-09-12; Accepted date: 2010-01-15
Corresponding author: GUO Hua-jun, PhD, Associate professor; Tel: +86-731-88836633; E-mail: ghj.csu@163.com
(Edited by CHEN Wei-ping)