
Plasma boronizing of Ti6AlV4 using solid precursors
by double glow plasma alloying technique
QIN Lin(秦 林) 1, QU Jun-zhi(曲均志)1, LIN Nai-ming(林乃明)1,
FAN Ai-lan(范爱兰)1, CHANG Da-qing(常大庆)2, TANG Bin(唐 宾)1
1. Research Institute of Surface Engineering, Taiyuan University of Technology, Taiyuan 030024, China;
2. Pingshuo Coal Industrial Company, Shuozhou 038500, China
Received 28 July 2006; accepted 15 September 2006
Abstract: Solid state B-offerings (FeB compound) were used for plasma-assisted boronizing on Ti6Al4V alloy by double glow discharge plasma alloying technique (DGPSA). During all experiment processing there was no harmful and toxic. The thickness and morphology, phase formation, elements concentrations and wear properties of boride layers were examined by means of optical microscopy, Vickers microhardness, X-ray diffraction, glow discharge optical emission spectroscopy and Ball-disk wear tests. The results show that friction coefficient of boride alloying samples versus corundum ball is lower than that of Ti6Al4V samples, as well as wear rate. This indicates that plasma-assisted boronizing of Ti6Al4V alloy has a high potential for industrial applications under tribological conditions.
Key words: plasma boronizing; titanium alloy; precursors; FeB compound
1 Introduction
Titanium alloys offer many attractive properties, which lead to an ever increasing interest in using them in such fields as power generation, medical devices and especially in aerospace. Mainly their excellent specific strength and outstanding corrosion resistance have made them very important. However, their use is currently restricted by their poor tribological applications, owing to their high friction and wear rate and a strong tendency to galling[1]. Therefore, different surface engineering techniques, such as chemical heat treatment, have been developed in recent years[2-4] to overcome these problems.
Titanium borides are well known for their high hardness and excellent wear resistance in many tribological systems. These characteristics make boronizing an excellent candidate for surface modification of titanium alloys[5-6]. The most frequently boronizing process used in industrial applications is pack boronizing using solid precursors in the form of powder or paste[7-8]. However, this technique has several disadvantages, such as a high degree of manual work and the lack of automatic process control. Results from boronizing in the gas phase using diborane and boron halides and triethyl borane as precursors have also been researched. Due to their toxicity or corrosion power these precursors are not used commercially[9].
For boronizing of titanium, the boron precursor has to be free of oxygen, and even hydrogen for avoiding hydrogen embrittlement in titanium alloys. Therefore, a plasma boronizing process for titanium has been developed using solid precursors (FeB compound). In the following, the first results on plasma boronizing of titanium alloys with solid precursors will be presented to demonstrate the potential of this method for industrial applications.
2 Experimental
Boronizing experiments on TiAl6V4 alloy were made by a new plasma boronizing method using solid precursors.
Fig.1 shows a construction of the DGPSA for plasma boronizing with solid boron precursors. The vacuum chamber of the furnace was connected to earth as anode. The sputtering source and sample substrate were cathodes with different negative bias. The sputtering source comprised FeB compound for the surface treatment of sample substrate. The source and the sample substrate were connected to source power supply and cathode power supply, respectively. Argon was used as environmental gases. The flow rate of the gas was controlled by mass flow controller. The vacuum system allowed plasma treatment in 13 Pa to 133 Pa. The principle of the DGPSA technique was a hollow cathode discharge between two parallel electrodes charged negatively in vacuum chamber. Application of a voltage between the sample and the anode produced a current-intensive glow discharge. Both glow discharges reinforced each other. The sample and source were heated due to the discharge.
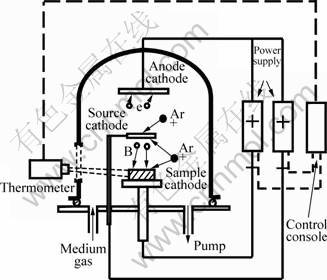
Fig.1 Sketch of double glow plasma surface alloying technique
The surface alloying took place with three possible processes or mechanisms. The first process was to sputter B atoms from the source cathode by positive ion produced in vacuum discharge. The second process comprised the ion cleaning and heating of the sample, depositions, and absorbing sputtered B atoms with the surface atoms of the sample. The third process comprised the diffusion of the B atoms into the solid surface of the sample with consistency difference driving action at the high temperature.
Samples were cut from bar of TiAl6V4 alloy with a 36 mm in diameter and 5 mm in thickness. Grinding and mirror-polishing to an average roughness of 0.08 μm and cleaning with chymic alcohol were performed directly before charging the samples into the treatment chamber to minimize contamination by air. Sputter cleaning was performed in Ar atmosphere during the heating phase prior to plasma boronizing. All samples were cooled after plasma boronizing in vacuum to room temperature. The processing parameters were: source electrode voltage 850 V; sample substrate voltage 600 V; working pressure 50 Pa; treatment time 3 h; and the distance between the two cathodes 15 mm.
The structure of the boronizing layers was analyzed by X-ray diffraction (D/max-RB) and optical microscopy (Zeiss Axiovert 25CA Inverted Reflected-light Micro- scope). Glow discharge optical emission spectroscopy (GDOES―750) was used to determine the depth profiles of boron, oxygen and the elements of the alloy Ti, Al and V after plasma diffusion treatment.
The tribological behavior of the boronizing layers in sliding contact was studied with a ball-on-disc tribometer (manufactured by Xi’an Jiaotong University) in which the friction was continuously recorded with a X-Y recorder.
3 Results and discussion
In plasma boronizing process, there was not any poison or pollution in the chamber, so it was redundant to add some accessories like evaporator and filter. Of course, any contain-H-material wasn’t brought in because it maybe results in H-crack to titanium alloy.
3.1 Concentrations and phase structure
The layers formed during plasma alloying on flat specimens are homogeneous, dense and closed. The metallographic cross-section of the boronizing Ti6Al4V sample is shown in Fig.2. The distribution of alloy elements in the alloying layer is shown in Fig.3. At the interface, a strong decrease in the iron content occurs from the layer to the matrix material. To estimate the depth of the diffusion zone of boron, the decreasing depth resolution of GDOES with the increase of depth of the sputtering crater must be considered. Therefore, it can be concluded that the diffusion zone of boron into titanium is about 2 μm. This is in accordance with the extremely low solubility of boron in titanium. The alloying elements aluminum and vanadium have a concentration maximum below the titanium boride layer. This can be attributed to a displacement of these elements by the boron, as it is found for nitrogen in the case of plasma nitriding.

Fig.2 Cross-section micrograph of boronizing Ti6Al4V specimen

Fig.3 Distribution of alloying elements in boronizing layer
In Fig.4, XRD pattern is given for the boronizing layer on Ti6Al4V alloy. The peaks can be attributed to the appropriate phases of the matrix material and the layer. Plasma boronizing of the titanium alloy TiAl6V4 leads to the formation of a layer on the surface consisting of TiB, TiB2, Ti3B4, Fe3B and FeB, furthermore, there still exist single phases Ti and Fe.
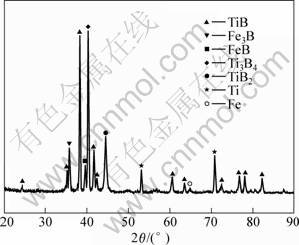
Fig.4 XRD spectrum of boronizing layer
To analyze the layers in detail, glow discharge optical emission spectroscopy was used to determine the depth profiles of boron, iron, oxygen and the elements of the alloy (Ti, Al, V) qualitatively after plasma diffusion treatment. If TiB2 was found at the surface, the boron content would be higher than the content of titanium and was approximately two-fold at the maximum. If only TiB was measured at the surface, the maximum boron content would equal the content of titanium. In particular, the results shown in Fig. 3 indicate that, there mostly is low-Boron compound, such as TiB in the boronizing layer. The formation of low-boron borides formed is a specific feature of the diffusion treatment of titanium.
3.2 Wear properties
The hardness of the surface was measured directly on the alloyed specimen perpendicular to the surface. Don’t like TiB coatings produced by PACVD, the hardness HK0.025 is not very high, only 836. It may be because of the low-boron compounds in boronizing layer.
Fig.5 shows the friction coefficient curves for Ti6Al4V sample and plasma boronizing sample sliding against corundum balls under dry friction. At the beginning of the test, due to the genuine touch of the test pair was only some tiny protruding parts, their baffling action made all the two samples’ coefficient less than 0.05. As tiny protruding was rubbing, a real surface contact begun, the friction coefficient of Ti6Al4V sample was quickly jumping up to about 0.45 and steady at 0.5 at last, however the boronizing layer increased slowly. When sliding distance was 50 m, the friction coefficient became closed to that of bare Ti6Al4V.
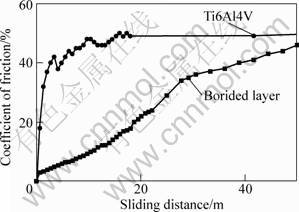
Fig.5 Coefficient of friction vs sliding distance
Fig.6 shows the wear scar of the bare Ti6Al4V sample after sliding distance 100 m vs corundum ball and Fig.7 shows the surface morphology of boronizing Ti6Al4V after 100 m sliding. It can be seen that the friction track of bare Ti6Al4V is wide and deep. The dominant wear mechanisms for the bare Ti6Al4V are extensive abrasive and adhesive wear, which is proved by furrow and middle lacerated marks in Fig.6. The shallower and narrower track of the boronizing layer shows lightly abrasion, because of the improvement of surface hardness and the decrease of friction coefficient after plasma boronizing. The wear volume calculated shows that the wear resistance of the boronizing layer is improved about 36 times. So the wear resistance performance of Ti6Al4V alloy improves evidently after plasma boronizing using solid precursors.

Fig.6 Wear scar of Ti6Al4V sample for 100 m sliding distance

Fig.7 Wear scar of boronizing sample for 100 m sliding distance
4 Conclusions
Plasma boronizing of Ti6AlV4 using solid precursors by double glow technique is successfully carried out in an industrial plant. Depended on the process parameters, a layer is formed which consists of TiB, TiB2, Ti3B4, Fe3B, FeB, and a little Ti and Fe. Due to the most low-boron compounds, the surface hardness HK0.025 is only 836. In comparison with bare Ti6Al4V, the friction coefficient of the boronizing layer decreases, wear resistance of the boronizing layer improves about 36 times. Plasma boronizing of titanium and titanium alloys is a promising method for improving the tribological properties of these metals, thus the field of application of titanium alloys increases.
References
[1] FRIDRICI V, FOUVRY S, KAPSA P H. Effect of shot peening on the fretting wear of Ti-6Al-4V[J]. Wear, 2002, 250(1/12): 642-649.
[2] QIN Lin, FAN Ai-lan, WU Pei-qiang, TANG Bin, XU Zhong. Fretting characters of molybdenum nitride layer on Ti6Al4V alloy[J]. Rare Metal Materials and Engineering, 2006, 35(7): 1053-1056.(in Chinese)
[3] QIN Lin, TANG Bin, LIU Dao-xin, XU Zhong. Friction and wear behavior of mo-n diffusion layer on Ti6Al4V alloy surface[J]. Rare Metal Materials and Engineering, 2005, 34(9): 1465-1468.(in Chinese)
[4] DONG H, BELL T. Enhanced wear resistance of titanium surfaces by a new thermal oxidation treatment[J]. Wear, 2000, 238(2): 131-137.
[5] KAESTNER P, OLFE J, RIE K T. Plasma-assisted boriding of pure titanium and TiAl6V4[J]. Surface and Coatings Technology, 2001, 142/144: 248-252.
[6] RIE K T. Recent advances in plasma diffusion process[J]. Surface and Coatings Technology, 1999, 112(1/3): 56-62.
[7] CAMPOS I, OSEGUERA J, FIGUEROA U, GARC?A J A, BAUTISTA O, KELEMENIS G. Kinetic study of boron diffusion in the paste-boriding process[J]. Materials Science and Engineering A, 2003, 352(1/2): 261-265.
[8] CAMPOS I, TORRES R, BAUTISTA O, RAM??REZ G, Z????IGA L. Effect of boron paste thickness on the growth kinetics of polyphase boride coatings during the boriding process[J]. Applied Surface Science, 2006, 252(6): 2396-2403.
[9] KUPER A, QIAO X, STOCK H R, MAYR P. A novel approach to gas boronizing[J]. Surface and Coatings Technology, 2000, 130: 87-94.
(Edited by CHEN Can-hua)
Foundation item: Project(50501016) supported by the National Natural Science Foundation of China; Projects(2006021023, 20051049) supported by the Natural Science Foundation of Shanxi province; Project supported by Young Scholar of University of Shanxi Province.
Corresponding author: TANG Bin; Tel: +86-351-6010540; E-mail: tangbin@tyut.edu.cn