
Influence of alloying elements on microstructure and microhardness of Mg-Sn-Zn-based alloys
W. L. CHENG1, 2, S. S. PARK2, W. N. TANG2, B. S. YOU2, B. H. KOO1
1. School of Nano & Advanced Materials Engineering, Changwon National University,
Changwon 641-773, South Korea
2. Light Metals Research Group, Korea Institute of Materials Science (KIMS), Changwon 641-831, South Korea
Received 21 December 2009; accepted 8 April 2010
Abstract: The phase evolution in (88%-91%)Mg-8%Sn-1%Zn-X (X=Al, Mn and/or Ce) system was analyzed via CALPHAD method and simulations were used in precise selection of the chemical composition. The influence of the addition of different alloying elements such as Al, Mn and Ce on the microstructure and microhardness of Mg-8%Sn-1%Zn-based alloys was investigated. Combined addition of Al and Mn shows features distinct from separate addition of Al or Mn. Additions of 1%Al and 1%Mn to base alloy result in the formation of massive Al-Mn phase in ��-Mg matrix grains. Addition of Ce element can refine the second eutectic precipitates and form intermetallic compounds with Sn. Fine rod-like Sn-Ce phase presents mainly on the grain boundaries and plays a role in inhibiting grain growth. The effects of alloying elements on Vickers microhardness and indentation size effect of base alloy were examined.
Key words: magnesium alloys; alloying elements; CALPHAD method; hardness; indentation size effect
1 Introduction
Due to the worldwide emphasis on fuel efficiency in automobiles, there has been increasing interest in the application of magnesium alloys to various structure components. In particular, there is currently much research into the development of Mg-based alloys for hot component such as powertrain system[1-3]. Recently, some Mg-based alloys, such as Mg-Al-RE(AE), Mg-Al-Si(AS) and Mg-Y- RE(WE) systems, have been explored for use at elevated temperatures, but the high cost and low castability restrict these alloys from being extensively used[4]. It has been reported that Mg-Sn-Zn (TZ) is a promising alloy system which exhibits excellent creep resistance and castability comparable to those of Mg-Al-Zn (AZ) and Mg-Al-Mn (AM) systems[5-6].
Alloy development experiments are time and resource consuming, while computational thermo- chemistry (CT) based on the CALPHAD method, can be utilized in the development of alloys or optimization of material dependent process[7]. This method has been successfully applied in the development of Mg alloys[8-9]. In this study, CALPHAD method was used in precise selection of the chemical composition before actual experiment. The results demonstrate that Al, Mn and Ce additions to the Mg-Sn-Zn system result in more thermally stable precipitates such as Al8Mn5 and Ce-containing phase, which can effectively hinder dislocation and grain boundary sliding at elevated temperature.
Generally, aluminum is an effective alloying element in magnesium alloys for improvement in room properties and hardness[10]. Also, some materials show an indentation size effect (ISE) in a decrease in hardness with increase in load[11]. It was not known whether the Mg-Sn-Zn alloys would show such an ISE effect.
The objective of this study is to understand the role of different alloying elements on the microstructure based on the thermodynamic calculation results and microhardness of as-cast Mg-8%Sn-1%Zn alloy.
Furthermore, the effects of combined Al, Mn and/or Ce additions to the base alloy were examined in detail. Any possible ISE was investigated by microhardness testing at different loads for the Mg-8%Sn-1%Zn alloy.
2 Experimental
The formation of different phases during solidification was predicted using Pandat software developed by CompuTherm LLC. The thermo dynamically predicted phase formation was compared with experimental result. Five alloys based on the ternary Mg-8%Sn-1%Zn (A0) alloy were melted in a cemented graphite crucible with the CO2+SF6 gas mixture protection in the melt surface. The elemental Mg, Al, Zn and Ce with 99.9% purity were added to the melt. Mn was added to the melt in the form of Sn-Mn master alloy (17%Mn, mass fraction). The melt was held at 750 ��C for 20 min to ensure the alloying elements were completely dissolved and diffused. For the ingot casting, the molten alloy was poured at 720 ��C into a cylindrical steel mold preheated to 200 ��C with a diameter of 100 mm and length of 200 mm. The nominal chemical compositions and code of the studied alloys are presented in Table 1. All samples for optical microscopy (OM) were sectioned, cold-mounted, polished and then etched in a solution of picric and acetic acid (5 g acid, 10 mL acetic, 10 mL distilled water and 100 mL ethanol) for 5-10s. The average grain size was analyzed from several micrographs of longitudinal sections using a computer-aided liner intercept measurement. Scanning electron microscopy (SEM) and energy dispersive spectroscopy (EDS) were used to study the morphological and micro-chemical characterization of second phases. The phase constitutions were analyzed by an X-ray diffraction instrument with Cu K�� radiation at a rate of 0.02 (��)/s. Thermal analyses were performed using a DSC-TGA SDTBuild 99 differential thermal analyzer at a heating rate of 20 ��C/min. The Vickers hardness was determined by a microhardness tester (JP/FM-7) with a loading force ranging from 0.098 to 2.94 N for a holding time of 10 s. Each measurement was the average value of at least 7 individual measurements.
Table 1 Nominal chemical compositions of studied alloys (mass fraction, %)
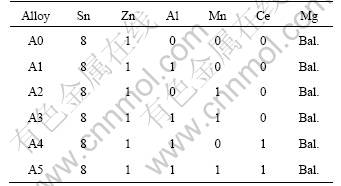
3 Results and discussion
3.1 Microstructure and thermodynamic calculation
The results of the thermodynamic calculation for equilibrium phase evolution during cooling of the base alloy and base alloy with addition of Al, Mn and/or Ce are presented in Fig.1 and Table 2. During the equilibrium solidification of A3 alloy (Fig.1(a)), ��-Mg starts to solidify at the liquids temperature of 699 ��C and coexists with Al8Mn5 at temperatures between 490 and 550 ��C. The main alloying element Sn initiates the formation of Mg2Sn at 525 ��C. The liquidus and solidus temperatures for A4 alloy (Fig.1(b)) are 629 and 538 ��C, respectively. CeMg12-x phase precipitates firstly and then Mg2Sn and MgZn precipitate slightly below 533 and 200 ��C, respectively. The results of other alloys are summarized in Table 2. It should be noted that the solidification range of A5 reaches a minimum value of 70 ��C, which is narrower compared with 136 ��C for Mg-Al binary alloy. As a result, the casting defects such as dispersed shrinkage and hot tearing are less severe in A5 alloy[12].
Table 2 Summary of CT simulation results
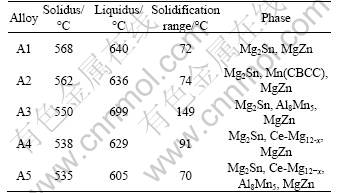
Fig.2 shows the optical micrographs of the as-cast alloys. As seen from Fig.2, these alloys exhibit coarse ��-Mg grains with size ranging from 75 to 200 ��m and a few second phase particles. The microstructure of alloy A1 with addition of 1% (mass fraction) aluminum shows similar characteristic with the base alloy. However, the microstructure of alloys A3 and A5 with addition of both Al and Mn changes slightly. A new massive phase forms in these alloys. As exhibited, the grain refining effect by Ce addition is weak. However, the morphology of the eutectic phases changes from continuous network to dissociative rod-like shape in Ce containing alloys, i.e. alloys A4 and A5. Meanwhile, the size of eutectic phase is also reduced by Ce addition.
To verify the thermodynamically predicted phases and compare them with the phases present in experimental alloys, SEM and EDS analyses were carried out and the results are shown in Figs.3 and 4. SEM examinations reveal that A1 alloy is mainly composed of ��-Mg matrix with dark contrast and second eutectic phase with grey contrast, which is identified as a mixture of ��-Mg and Mg2Sn. The second eutectic phase is relatively unaffected by the separate addition of Al, indicating the alloying element Al is completely dissolved into ��-Mg matrix. When Mn is added to the base alloy, Mn forms a cubic body centered compound (CBCC) phase (predicted phase) occurring as supersaturation of Mn within ��-Mg matrix, which is detected in the experimental alloy (marked dotted triangle in Fig.3(c)). In addition, the cumulative effects of Al and Mn are marked, which results in a new massive Al-Mn phase (marked dotted rectangles in Fig.3(d)). With the addition of Ce, the microstructure of alloys A4 and A5 was changed obviously. Moreover, some rod-like phase (marked dotted rhombus in Figs.3(e) and (f)) accumulates with cluster morphology, and some white massive particles (marked dotted rectangles in Fig.3(f)) are found in alloy A5. The element compositions of the phases in Fig.3 were identified by EDS analysis. The results show that the rod-like phase is Sn7Ce3, the massive phase corresponds to Al8Mn5 and the network phase is Mg2Sn and ��-Mg eutectic.
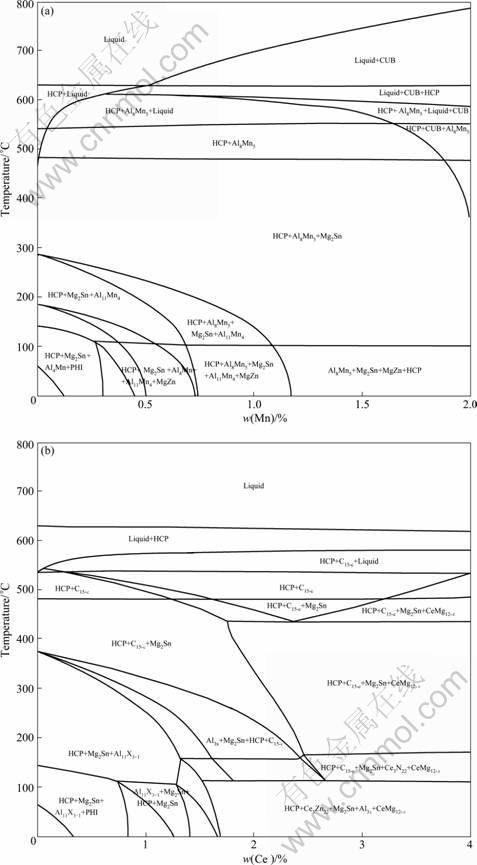
Fig.1 Equilibrium phase diagram of A3 (a) and A4 (b) alloys

Fig.2 Optical micrographs of studied alloys: (a) A0; (b) A1; (c) A2; (d) A3; (e) A4; (f) A5
Fig.5 presents the XRD results of the experimental alloys. As exhibited, there is no difference between alloys A1 and A2 which are mainly composed of ��-Mg and Mg2Sn phases. With the binary combined addition of Al and Mn or ternary combined addition of Al, Mn and Ce, besides Mg2Sn diffraction peaks, new phase diffraction peaks appear, such as the peaks located at 24��, 28�� and 30��. Whereas, the thermodynamically predicted MgZn phase is not detected by XRD.
According to the XRD results and EDS analysis, the alloys consist of ��-Mg, Mg2Sn, Al8Mn5, and Sn7Ce3 phases. These findings are almost in agreement with our theoretical results except Ce containing phase and MgZn phase. The morphology and volume fraction of these intermetallic phases vary with different alloying elements addition.

Fig.3 SEM micrographs of studied alloys: (a) A0; (b) A1; (c) A2; (d) A3; (e)A4; (f) A5
The obtained DTA curves are shown in Fig.6. Endothermic peaks are present in the studied alloys and every peak corresponds to the dissolution of certain phase during heating. There are two peaks present in alloy A3, while only one endothermic peak is present in other alloys. The first peak (441-483 ��C) present in the alloy A3 is due to the dissolution of Al-Mn phase. All the alloys show an endothermic peak, which corresponds to the dissolution of Mg2Sn phase into ��-Mg matrix. The Mg2Sn phase enhances the stability at higher temperature, while the Al-Mn phase degrades that a little.
3.2 Microhardness and ISE
Indentation hardness testing is a convenient technique to investigate the mechanical properties of a local, small volume of material [11]. The Vickers microhardness, HV is defined as[13]:
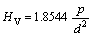 (1)
(1)
where p is the applied load and d is the indentation size. The Vickers hardness of the studied alloys at a load of 1.96 N is shown in Fig.7(a).
As indicated, the highest and lowest microhardness were attained at alloy A2 and A5. The values are HV 47.05 and 57.01, respectively. The microhardness is increased by 16% for alloy A5 compared with HV 49.95 for alloy A0. In addition, the microhardness of alloy A3 is almost unchanged compared with that of alloy A0.
The microhardness change with alloying elements is related to a number of factors including grain size, the volume fraction of the second phase precipitates and solid solution hardening. When Al is added to the base alloy, the hardness is slightly increased through solid solution hardening. However, the addition of Mn decreases the hardness due to its degradation to solid solution hardening. Furthermore, the hardness of alloy A3 is similar to that of base alloy because of the formation of Al-Mn phase, which greatly reduces the solute atoms (Al) in the matrix. Therefore, the solid solution hardening is weakened. On the other hand, the weakened solid solution hardening can be compensated by precipitation hardening. These two contradictory factors result in the hardness of alloy A3 with minor alteration. It is notable that the increase of hardness in alloys A4 and A5 is attributed to the grain refinement by Ce addition and precipitation hardening. The hardness of alloy A5 reaches a maximum.
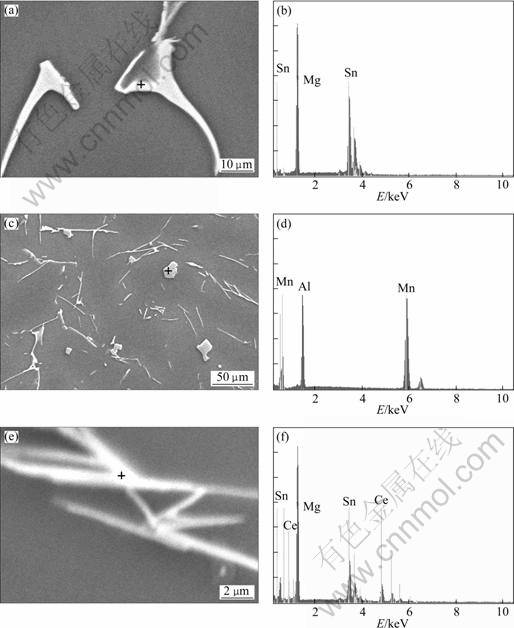
 Fig.4 TEM images and EDS analyses of analyzed phase: (a, b) Mg-Sn phase; (c, d) Al-Mn phase; (e, f) Sn-Ce phase
Fig.4 TEM images and EDS analyses of analyzed phase: (a, b) Mg-Sn phase; (c, d) Al-Mn phase; (e, f) Sn-Ce phase
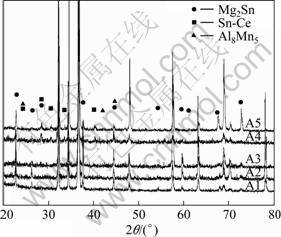
Fig.5 XRD patterns of studied alloys
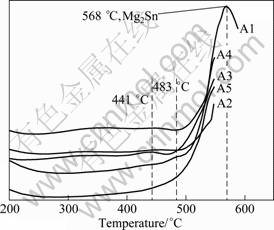
Fig.6 DTA curves of studied alloys at heating rate of 20 oC/min
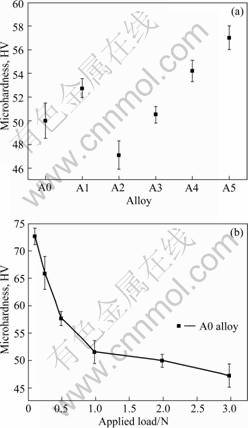
Fig.7 Variations of micro hardness with studied alloys (a) and applied load for A0 alloy (b)
To study the ISE, indentation loads from 0.098 to 2.94 N were applied to the alloy A0. The results are shown in Fig.7(b). At a load of 0.098 N, the microhardness is about HV 72.8, and then decreases to about HV 47.26 as the load increases up to 2.94 N. Fig.7(b) shows that there is a non-linear relationship between the Vickers hardness and the applied load.
An empirical expression, Meyer's Law[14] shown in Eq.(2), can be used to describe the ISE behavior of the base alloy (A0) by correlating the load p and the size of indentation d:
p=kdn (2)
where n is the Meyer exponent and k is the Meyer prefactor. The value of Meyer exponent n is used as a measure of ISE when n��2, the microhardness has a load dependent behavior, i.e. ISE behavior. In this study, the n value is 1.87 which is lower than 1.91 reported by HAN et al[11], indicating a relatively strong ISE behavior.
4 Conclusions
1) The as-cast alloys consist of ��-Mg, Mg2Sn, Al8Mn5 and Sn-Ce phases, which are almost consistent with theoretical simulated results except Ce-containing phase. Changing the alloying elements addition mode can change the phase compositions. With the addition of both Al and Mn in base alloy, Al8Mn5 phase is formed. With the addition of Ce in alloys A1 and A3, the alloys exhibit the existence of Sn-Ce phase and eutectic phase refinement.
2) The microhardness of alloy A5 reaches a maximum. It is attributed to the grain refinement and volume fraction increment of second phase particles by simultaneous addition of Al, Mn and Ce.
3) The as-cast Mg-Sn-Zn alloy exhibits weak ISE, which is well described by Meyer's Law with a Meyer exponent of 1.87.
References
[1] MENDIS C L, BETTLES C J, GIBSON M A, HUTCHINSON C R. An enhanced age hardening response in Mg-Sn based alloys containing Zn [J]. Mater Sci Eng A, 2006, 435/436(1): 163-171.
[2] KANG D H, PARK S S, KIM N J. Development of creep resistant die cast Mg-Sn-Zn-Al alloy [J]. Mater Sci Eng A, 2005, 413/414(1): 555-560.
[3] LIM H K, SOHN S W, LEE J Y, KIM W T, KIM D H. Effect of addition of Sn on the microstructure and mechanical properties of Mg-MM (misch-metal) alloys [J]. J Alloys Compd, 2008, 454(1/2): 515-522.
[4] XIAO Wen-long, JIA Shu-sheng, WANG Jian-li, YANG Jie, WANG Li-min. The influence of mischmetal and tin on the microstructure and mechanical properties of Mg-6Zn-5Al-based alloys [J]. Acta Mater, 2008, 56(5): 934-941.
[5] BOURGEOIS L, MUDDLE B C, NIE J F. The crystal structure of the equilibrium �� phase in Mg-Zn-Al casting alloys [J]. Acta Mater, 2001, 49(14): 2701-2711.
[6] WANG Y X, GUAN S K, ZENG X Q, DING W J. Effects of RE on the microstructure and mechanical properties of Mg-8Zn-4Al magnesium alloy [J]. Mater Sci Eng A, 2006, 416(1/2): 109-118.
[7] AVRAHAM S, KATSMAN A, LEVIATAN T, MAOZ Y, BAMBERGER M. The influence of Zn on the precipitation in Mg-Sn alloys [C]//Magnesium Technology 2008. TMS, 2008: 127-131.
[8] SCHMID-FETZER R, GR?BNER J. Focused development of magnesium alloys using the Calphad approach[J]. Adv Eng Mater 2001, 3(12): 947-961.
[9] BAMBERGER M. Phase formation in Mg-Sn-Zn alloys��Thermodynamic calculations vs experimental verification [J]. J Mater Sci, 2006, 41(10): 2821-2829.
[10] LIM H K, KIM D H, LEE J Y, KIM K W T, KIM D H. Effects of alloying elements on microstructures and mechanical properties of wrought Mg-MM-Sn alloy [J]. J Alloys Compd, 2009, 468(1/2): 308-314.
[11] HAN L, HU H, NORTHWOOD D O. Effect of Ca additions on microstructure and microhardness of an as-cast Mg-5.0 wt.% Al alloy[J].Mater Letter, 2008, 62(3): 381-384.
[12] LIU Hong-mei, CHEN Yun-gui, TANG Yong-bai, WEI Shang-hai, NIU Gao. The microstructure, tensile properties, and creep behavior of as-cast Mg-(1�C10)%Sn alloys [J]. J Alloys Compd, 2007, 440(1/2): 122-126.
[13] K?LEMEN U, UZUN O, YILMAZLAR M, G?CL?N, YAN E. Hardness and microstructural analysis of Bi1.6Pb0.4Sr2Ca2-x- SmxCu3Oy polycrystalline superconductors [J]. J Alloys Compd, 2006, 415(1/2): 300-306.
[14] MANIKA I, MANIK J S. Size effects in micro- and nanoscale indentation [J]. Acta Mater, 2006, 54(8): 2049-2056.
(Edited by FANG Jing-hua)
Foundation item: Project supported by the Fundamental R&D Program for Core Technology of Materials funded by the Ministry of Knowledge Economy, Republic of Korea; Project supported by the Center for Advanced Materials Processing (CAMP) of 21st Century Frontier R&D Program, Republic of Korea
Corresponding author: CHENG W L; Tel: +82-55-280-3303; E-mail:chengweili7@126.com
DOI: 10.1016/S1003-6326(10)60636-X