
Pretreatment study on chloridizing segregation and magnetic separation of low-grade nickel laterites
LIU Wan-rong(刘婉蓉)1, LI Xin-hai(李新海)1, HU Qi-yang(胡启阳)1, WANG Zhi-xing(王志兴)1,
GU Ke-zhuan(古可专)1, LI Jin-hui(李金辉)1, 2, ZHANG Lian-xin(张琏鑫)1
1. School of Metallurgical Science and Engineering, Central South University, Changsha 410083, China;
2. School of Materials and Chemical Engineering, Jiangxi University of Science and Technology,Ganzhou 341000, China
Received 6 July 2009; accepted 30 December 2009
____________________________________________________________________________________________
Abstract: The chloridizing segregation and magnetic separation of low-grade nickel laterites from Yunnan province of China was investigated. The nickel laterites were characterized by microscopic investigations, using X-ray diffractometry (XRD) and energy dispersive spectrometry (EDS) techniques. The pellets, which were prepared with magnesium chloride and coke as chloride agent and reductant respectively, were heated to a high temperature, and the pellets after cooling were crushed for magnetic separation. A series of experiments were conducted to examine the effect of chlorinating agent dosage, reductant dosage, chloridizing temperature and chloridizing time on enrichment grade of Ni and Co. The results indicate that the four factors have significant effects on the extractions of Ni and Co. The optimum conditions are as follows: the amounts of magnesium chloride and coke are 6% and 2%, respectively, chloridizing temperature is 1 253 K, and chloridizing time is 90 min. Under the conditions, extractions of Ni and Co reach 91.5% and 82.3%, respectively.
Key words: nickel laterites; nickel; cobalt; chloridizing segregation; magnetic separation
____________________________________________________________________________________________
1 Introduction
Nickel is recognized as an important substance in heavy industry[1]. About 60% of current worldwide production of nickel is derived from sulphide ores, but about 70% of the nickel reserves are present in laterites deposits[2]. Along with the rapid development of nickel industry, the high grade nickel of sulphide ores decreases continually, therefore there is an urgent need for a new and efficient technology to extract Ni and Co from low-grade nickel laterites.
Many conventional methods have been applied for treatment of nickel laterite ores, such as atmospheric acid pressure leaching[3-5], high temperature pressure acid leaching[6-8], biological leaching[9] and pug-roast leaching[10]. The way of atmospheric pressure leaching makes poor Ni and Co extraction[11]. High temperature pressure acid leaching is applicable only for low magnesia/high iron ores in order to minimize acid consumption[12]. The process of biological leaching is too difficult to bring up the efficient bacterium to extract Ni and Co from laterites[13]. In pug-roast leaching process, the consumption of sulphuric acid or sulphur dioxide is less[14], but the Ni and Co extractions are also less. However, in this work, an efficient method to extract Ni and Co from high magnesia low iron laterites was chloridizing segregation and magnetic separation process, which had been heretofore initially applied to copper oxides using coke and sodium chloride as a chlorinating agent[15].
2 Experimental
2.1 Materials
The low-grade nickel laterite ores used in this investigation was obtained from Yunnan province of China. Its chemical composition is shown in Table 1. It indicates that the major species of the laterites are MgO and SiO2, and Ni and Co are in low-grade. The X-ray diffraction (XRD) pattern of the low-grade nickel laterites is shown in Fig.1. It can be seen that Mg3Si2(OH)4O5 and SiO2 are major minerals and a little NiFe2O4 is presented in the ores.
Table 1 Chemical composition of low-grade nickel laterites (mass fraction, %)


Fig.1 X-ray diffraction pattern of low-grade nickel laterites
The analytically pure magnesium chloride was used as chloride agent, and the coke which was sieving to suitable size (200 μm) used as reduction.
2.2 Experimental procedure
The flowchart of chloridizing segregation and magnetic separation of low-grade nickel laterites is shown in Fig.2. The low-grade nickel laterites had been crushed firstly. After drying, grinding and sieving, the ground ore had a grain size of 150 μm. Being mixed with MgCl2, coke and additive together, the ore had been made pellets with diameter of 15-20 mm. The pellets were gradually heated from the room temperature to reach a desired temperature under a slight reducing atmosphere and then roasted at this temperature for a desired time. Then, the roasted pellets were cooled, crushed and sieved. Grinded pellets had a grain size of 74 μm, and subjected to a wet magnetic separation with 0.2 T magnetic field strength to obtain Ni and Co con-centrate. The Ni and Co in the concentrate were analyzed by atomic absorption spectrophotometry (AAS).
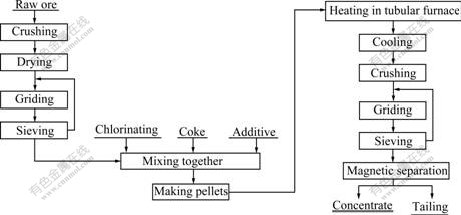
Fig.2 Flowchart of low-grade nickel laterites chloridizing segregation and magnetic separation process
2.3 Reaction mechanism
The process of chloridizing segregation involves chlorinating of the Ni, Co and Fe oxide and subsequently reducing of the Ni, Co and Fe chloride. During the heating stage, the magnesium chloride added to the ore reacts with water vapor to produce hydrochloric acid, and the alkalis and alkali-earth oxides react with the gangue to form complexes of silicates. Then hydrochloric acid reacts with a metal oxide (NiO, CoO) to produce the respective metal chloride (NiCl2, CoCl2). Thereafter, the metal chloride (NiCl2, CoCl2) is reduced to metal in situ on the coke surface and the HCl is regenerated.
Reactions occur during the process of chloridizing segregation: firstly, the magnesium chloride added to the nickel laterites reacts with water vapor to produce hydrochloric acid according to the reaction (1):
MgCl2+SiO2+H2O=MgO・SiO2+2HCl (1)
On the basis of calculation of thermodynamic data [16], the change of Gibbs free energy of reaction (1) can be expressed as  Since
Since 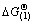 <0 at temperature above 600 K, reaction (1) can proceed. Then NiO and Co2SiO4 react with HCl to generate nickel and cobalt chloride according to the reactions (2) and (3):
<0 at temperature above 600 K, reaction (1) can proceed. Then NiO and Co2SiO4 react with HCl to generate nickel and cobalt chloride according to the reactions (2) and (3):
NiO+2HCl=NiCl2+H2O (2)
Co2SiO4+4HCl=2CoCl2+SiO2+2H2O (3)
Using the thermodynamic data, the change of Gibbs free energy of the above two reactions can be described by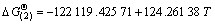 and
and 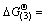 -231 462.86+237.8 T. When the temperature is below 900 K,
-231 462.86+237.8 T. When the temperature is below 900 K, 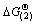 <0 and
<0 and  <0, which means that the two reactions are likely to occur between 600 K and 900 K, the NiCl2 and CoCl2 are generated and then reduced to metal (Ni, Co) according to the reactions (4) and (5):
<0, which means that the two reactions are likely to occur between 600 K and 900 K, the NiCl2 and CoCl2 are generated and then reduced to metal (Ni, Co) according to the reactions (4) and (5):
NiCl2+C+H2O=Ni+2HCl+CO (4)
CoCl2+C+H2O=Co+2HCl+CO (5)
The thermodynamic calculations indicate that the Gibbs free energies of the reactions (4) and (5) can be depicted as 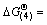 249 695.96-301.87T and
249 695.96-301.87T and 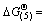 256 875.81-289.29T since
256 875.81-289.29T since 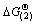 <0 and
<0 and  <0 at temperature above 900 K, coke can react with H2O and generate H2 to reduce nickel and cobalt chloride to metal (Ni, Co). But it usually needs high temperature higher than 1 200 K to obtain high Ni and Co recovery due to their high activity in high temperature.
<0 at temperature above 900 K, coke can react with H2O and generate H2 to reduce nickel and cobalt chloride to metal (Ni, Co). But it usually needs high temperature higher than 1 200 K to obtain high Ni and Co recovery due to their high activity in high temperature.
3 Results and discussion
3.1 Effect of chlorinating agent dosage
The effects of chlorinating agent dosage on the grades of Ni and Co are shown in Fig.3. A series of pellets prepared with varies of chlorinating agent dosage of 2%-12% (mass fraction). It can be observed that the recoveries of Ni and Co increase with chlorinating agent dosage increasing from 2% to 6% (Fig.3). When chlorinating agent is 6%, enough HCl is produced as the reaction (1), and reacts with NiO, Co2SiO4 sufficiently. However, there is not a big change if chlorinating agent dosage is over 6%. Thus the chlorinating agent dosage of 6% was used in the study.
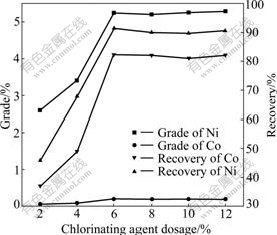
Fig.3 Effects of chlorinating agent dosage on Ni and Co grades and recoveries under conditions of reductant dosage 2%, chloridizing temperature 1 253 K, chloridizing time 90 min and magnetic field strength 0.2 T
3.2 Effect of reductant dosage
The chloridizing products of nickel and cobalt chloride are reduced to simple substance on the coke surface, therefore the reductant dosage can influence the recovery of Ni and Co significantly in the process.
It can be seen in Fig.4 that the recoveries of Ni and Co increase firstly and then decrease with the increase of reductant dosage. The maximum recoveries of Ni and Co can be obtained with 2% reductant dosage in the experiments. Because the more reductant dosage, the higher reducing atmosphere is obtained. So, Fe2O3 can be excessively reduced to Fe, but not FeO. As we know, magnetism of Fe is stronger than that of FeO. Thus, more Fe can be extracted through magnetic separation, leading to that the grades of Ni and Co decrease somewhat.
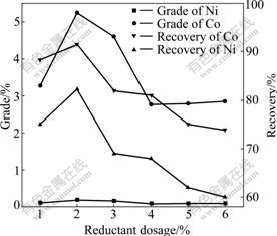
Fig.4 Effect of reductant dosage on Ni and Co grades and recoveries under conditions of chlorinating agent dosage 6%, chloridizing temperature 1 253 K, chloridizing time 90 min and magnetic field strength 0.2 T
3.3 Effect of chloridizing temperature
Fixing the composition of chlorinating agent, additive and coke in the pellets, the effects of chlorinating temperature on recoveries and grades of Ni and Co are shown in Fig.5. It can be seen that with the increase of chloridizing temperature, the recoveries of Ni and Co increase gradually until 1 253 K. It is because below 1 253 K, a new mineralogical component forsterite can be formed, which has been identified to be Ni and Co, and decrease the grades and recoveries of Ni and Co in the magnetic separation[15]. For economic reason, the chloridizing temperature of 1 253 K is suitable.

Fig.5 Effects of chloridizing temperature on Ni and Co grades and recoveries under conditions of chlorinating agent dosage 6%, reductant dosage 2%, chloridizing time 90 min and magnetic field strength 0.2 T
3.4 Effect of chloridizing time
Chloridizing time experiments were conducted to find out the optimum time for extraction of Ni and Co mostly. The recoveries of Ni and Co obtained by chloridizing segregation and magnetic separation can be calculated and are shown in Fig.6. It can be seen that the recoveries of Ni and Co increase gradually with increase of reaction time until 90 min and reach a maximum. Because nickel and cobalt chloride can dissolve in water and lose later in wet magnetic separation. Therefore, it is necessary to control the chloridizing time beyond 90 min at least, the nickel chloride can totally react with the reductant to Ni and avoid loss of Ni and Co.
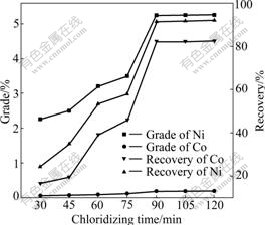
Fig.6 Effects of chloridizing time on Ni and Co grades and recoveries under conditions of chlorinating agent dosage 6%, reductant dosage 2%, chloridizing temperature 1 253 K and magnetic field strength 0.2 T
After four factors considering, chlorinating agent dosage 6%, reductant dosage 2%, chloridizing temperature 1 253 K, chloridizing time 90 min and magnetic field strength 0.2 T are the optimum conditions, and the concentrate with 91.5% of Ni and 82.3% of Co is obtained.
3.5 Analysis of nickel laterites concentrate
The composition of the concentrates of Ni and Co obtained through chloridizing segregation and magnetic separation process is shown in Table 2. The X-ray diffraction (XRD) pattern and EDS spectrum are shown in Fig.7 and Fig.8, respectively.
Table 2 Chemical composition of nickel laterites concentrate (mass fraction, %)
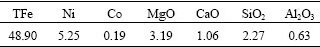
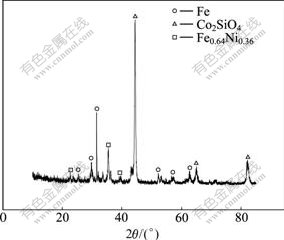
Fig.7 X-ray diffraction pattern of nickel laterites concentrate

Fig.8 SEM (a) and EDS spectrum (b) of nickel laterites concentrate
It can be seen from Fig.2 that the grades of Fe, Ni and Co increase greatly and impurities of Mg and Si decrease obviously in the concentrate. The main components in the concentrate presented through Fig.7 are Fe, Co2SiO4 and Fe0.64Ni0.36. In chloridizing segregation and magnetic separation process, most lizardite in raw ore reacts with chlorinating agent and generate HCl, which is shown in reaction (1). And then the Ni and Co oxides react with HCl and the products of metal chloride are reduced to Fe-Ni alloy which can be proved through Fig.8.
4 Conclusions
1) The recovery and grade of Ni and Co can be influenced by chlorinating agent dosage, reductant dosage, chloridizing temperature and chloridizing time. The optimal conditions of the chloridizing segregation process are obtained as follows: 6% of chlorinating agent (MgCl2), 4% of reductant, chlorinating temperature from the room temperature to 1 253 K and 90 min at least.
2) Under the optimal conditions, the grades of Ni and Co reach 5.25% and 0.19% in the concentrate, and the recoveries achieve 91.5% and 82.3%, respectively.
References
[1] MOSKALYK P R, ALFANTAZI A M. Nickel laterite processing and electrowinning practice [J]. Minerals Engineering, 2002, 15(8): 593-605.
[2] DALVI A D, BACON W G, OSBORN R C. The past and the future of nickel laterites [EB/OL]. [2004-03-10]. http://www.pdac.ca/ pdac/publications/papers/2004/techprgm-dalvi-bacon.pdf.
[3] QUENEAU P B, WEIR W R. Control of iron during hydrometallurgical processing of nickelferrous laterites ores [C]// DUTRIZAC J E, HONHEMIUS A J. Iron Control in Hydrometallurgy. Ellis Horword: Chichester, 1986: 76-105.
[4] SWAMY Y V, KAR B B, MOHANTY J K. Physico-chemical characterization and sulphatization roasting of low-grade nickeliferous laterites [J]. Hydrometallurgy, 2003, 69(1/3): 89-98.
[5] LI Y J, PAPANGELAKIS V G, PAPEREDERIY I. High pressure oxidative acid leaching of nickel smelter slag: Characterization of feed and residue [J]. Hydrometallurgy, 2009, 97(3/4): 185-193.
[6] LEONARDOU S A, ZAFIRATOS I G. Beneficiation of a Greek serpentinic nickeliferous ore Part II. Sulphuric acid heap and agitation leaching [J]. Hydrometallurgy, 2004, 74(3/4): 267-275.
[7] GEORGIOU D, PAPANGELAKIS V G. Sulphuric acid pressure leaching of a limonitic laterite: Chemistry and kinetics [J]. Hydrometallurgy, 1998, 49(1/2): 23-46.
[8] RUBISOV D H, KROWINKEL J M, PAPANGELAKIS V G. Sulphuric acid pressure leaching of laterites―Universal kinetics of nickel dissolution for limonites and limonitic/saprolitic blends [J]. Hydrometallurgy, 2000, 58(1): 1-11.
[9] VALIX M, TANG J Y, CHEUNG W H. The effects of mineralogy on the biological leaching of nickel laterite ore [J]. Minerals Engineering, 2001, 14(12): 1629-1635.
[10] KAR B B, SWAMY Y V, MURTHY V R. Design of experiments to study the extraction of nickel from laterite ore by sulphatization using sulphuric acid [J]. Hydrometallurgy, 2000, 56(3): 387-394.
[11] DAS G K, ANAND S, ACHARYA S, DAS R P, Characterization and acid pressure leaching of various nickel-bearing chromite overburden samples [J]. Hydrometallurgy, 1997, 44(1/2): 97-111.
[12] LIU H X, VLADIMIROS G P. Chemical modeling of high temperature aqueous processes [J]. Hydrometallurgy, 2005, 79(1/2): 48-61.
[13] PATHAK A, DASTIDAR M G, SREEKRISHNAN T R. Bioleaching of heavy metals from sewage sludge: A review [J]. Journal of Environmental Management, 2009, 90(8): 2343-2353.
[14] JOHNSON J A, CASHMORE B C, HOCKRIDGE R J. Optimisation of nickel extraction from laterite ores by high pressure acid leaching with addition of sodium sulphate [J]. Minerals Engineering, 2005, 18(13/14): 1297-1303.
[15] VALIX M, CHEUNG W H, Study of phase transformation of laterites ores at high temperature [J]. Minerals Engineering, 2002, 15(8): 607-612.
[16] LIN Chuan-xian, BAI Zheng-hua, ZHANG Zhe-ru. Thermodynamic handbook of minerals and related compounds [M]. Beijing: Science Press, 1985: 124-167. (in Chinese)
_______________________
Foundation item: Project(2007CB613607) supported by National Basic Research Program of China
Corresponding author: LI Xin-hai; Tel: +86-731-88836633; E-mail: liuwr2003@163.com
(Edited by ZHAO Jun)