Trans. Nonferrous Met. Soc. China 23(2013) 271-275
Measurement of binary phase diagram of Cu2S-MoS2 system
Xing-yu CHEN1, Zhong-wei ZHAO1,2, Ming-ming HAO1, Xu-heng LIU1
1. School of Metallurgical Science and Engineering, Central South University, Changsha 410083, China;
2. Key Laboratory for Metallurgy and Material Processing of Rare Metals, Central South University, Changsha 410083, China
Received 24 December 2012; accepted 8 January 2013
Abstract: A novel method, bath smelting process, was developed to treat molybdenite concentrate aiming at the existing problems of traditional process. To understand the dissolving behavior of MoS2 in white matte, the binary phase diagram of Cu2S-Mo2S was measured by the cooling curve method. The result shows that this system is a simple binary eutectic with a eutectic temperature of (1117.0±3.0) °C and a eutectic composition of (1.70±0.20)% MoS2 in mass fraction. When the MoS2 addition exceeds 4.48%, MoS2 and Cu2S can form the ternary compound containing CuMo2S3 or Cu2Mo6S8. In the temperature range of copper smelting, 1200-1300 °C, molybdenite can dissolve in the cuprous sulfide. At 1200 °C, the solubility of molybdenite can reach 14.8%.
Key words: binary system; Cu2S-MoS2; bath smelting process; solubility; eutectic; molybdenite
1 Introduction
Molybdenum is an important rare refractory metal and a strategic storage metal. It is applied widely in steel, glass, electronic, chemical, mechanical processing, aerospace and national defense, etc [1]. The metallurgical process of Mo mainly includes the process of transiting Mo from tetravalent MoS2 to hexavalent MoO3 by the oxidation. Currently, the mainstream technology for treating molybdenite includes oxidizing roasting, ammonia leaching, ion-exchange, evaporative crystallization at home and abroad. The obtained ammonium tetrathiomolybdate can be further treated for preparing molybdenum product.
In the traditional oxidizing roasting process, multiple hearth furnace, rotary kiln, fluidized bed roaster and reverberatory furnace are often adopted [2,3]. However, the production efficiency of multiple hearth furnace and fluidized bed roaster is lower. Especially, the producitity of multiple hearth furnace is only 0.08-0.12 t/(m2・d). Temperature control is also a hard task. Once it exceeds 600 °C, the loss caused by molybdenum sublimation will increase drastically. Meanwhile, the MoO3 produced easily melts with the molybdate, which affects the further desulfurization of ores. In addition, the SO2 flue gas from these roasting processes, 3% from fluidized bed roaster, 0.8%-3% from multiple hearth furnace and 0.8%-2% or even lower from rotary kiln, is hard to be treated and will cause serious air pollution since it is unsuitable for preparation of high quality sulfuric acid [4,5].
In view of the above problems, a novel method, bath smelting process, was developed to treat molybdenite by our research group. The first step was dissolving molybdenum in the molten white matte to form molybdenum-copper matte. Then, air was blasted into the molybdenum-copper matte, molybdenum was preferentially oxidized into MoO3 and entered into flue gas. Meanwhile, MoO3 was recycled by the dust collection after condensation. Like the bath smelting process, this method can treat the molybdenite continuously and has a higher production efficiency. Through converting the molybdenum-copper matte, high-concentration SO2 flue gas and relatively high purity MoO3 can be produced. In addition, the produced copper matte can be recycled.
The key of this process is that whether or not MoS2 can dissolve in Cu2S and how the dissolving behavior of MoS2 at different temperature is.
In the research process of superconducting materials CuMoSx, GROVER and MOH [6,7] found that liquid phase appeared once the melted blend containing Cu, Mo and S was heated to 1080 °C. Unfortunately, the defined composition was not reported. In addition, GULYANITSKAYA et al [8] adopted DTA to research the Cu2-xS-MoS2 melt, and it was found that the eutectic temperature of melt was unexpectedly only 992 °C. Though the reported temperature had a difference of about 100 °C, it at last revealed that MoS2 can dissolve in Cu2S.
In order to accurately understand the Cu-Mo-S system and the dissolving behavior of MoS2 in white matte, it is necessary to study the binary phase diagram of Cu2S-MoS2 system. In this research, the cooling curve method was adopted to measure and research the binary phase diagram of Cu2S-MoS2 system.
2 Experimental
2.1 Preparation of binary Cu2S-MoS2 system
The MoS2 with analytical grade and the prepared Cu2S were used. The preparation method of Cu2S was as follows. First, copper powder (99.7%, 74 μm, analytical grade) and sulfur powder (w(S)>99.5%, chemical grade) were added into a quartz tube on mole ratio of 2:1. Then, the quartz tube was vacuumized up to 10-2 Pa and sealed. The sealed quartz tube was well mixed by shaking and placed into the furnace for melting. The temperature- raising system was as follows: 1) Rising from room temperature to 200 °C within 1 h and preserving heat for 10 h; 2) Rising from 200 °C to 600 °C within 2 h and preserving heat for 35 h; 3) Rising from 600 °C to 800 °C within 1 h, preserving heat for 23 h and finally cooling with furnace. As the quartz tube was cooled to room temperature, the black schistous substance was obtained (in Fig. 1). The XRD pattern (Fig. 2) of black substance shows that the phase composition is Cu1.97S and Cu1.96S, and the chemical composition includes 77.90% Cu and 22.10% S.

Fig. 1 Photo picture of prepared Cu2S in quartz tube
MoS2 and the prepared Cu2S were mixed by grinding in an agate mortar and then added into a graphite crucible (d60 mm×100 mm). The graphite crucible was placed in the vacuum induction furnace which was first vacuumized to 5 Pa and then charged into argon. The mixture was heated for 1 h. High vacuum degree is helpful to reduce the oxygen partial pressure and decrease the oxidation of sulfide. In addition, graphite crucible can consume part of oxygen at high temperature and further decrease the oxidation of sulfide. After cooling, the obtained fusion sample was easily taken out from the graphite crucible and then was ground into powders for measuring the binary phase diagram of Cu2S-MoS2 system.

Fig. 2 XRD pattern of prepared Cu2S
2.2 Measurement of binary phase diagram of Cu2S-MoS2 system
Fusion samples of 14 g with different MoS2 contents were sealed into vacuumized quartz tubes (d20 mm×100 mm). The sealed quartz tubes were placed in a crucible resistor furnace (Yuandong Experimental Furnace Plant of Changsha, SG2-10-13), and the experimental schematic diagram is presented in Fig. 3. The temperature-raising procedure is as follows: 1) Rising from room temperature to 600 °C at 10 °C/min; 2) Rising from 600 °C to 1200 °C at 5 °C/min and preserving heat for 240 min; 3) Cooling to 1000 °C at 2 °C/min. During the cooling process, the temperature variation along with time was measured, and the cooling curve (T―t) was obtained.
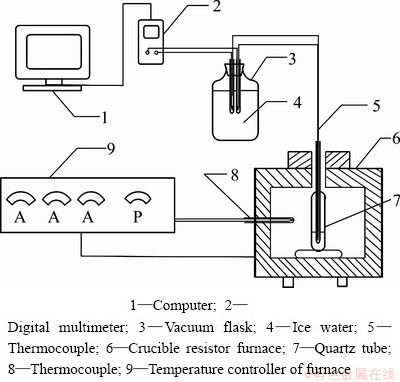
Fig. 3 Experimental schematic diagram
3 Results and discussion
3.1 Cu2S-MoS2 binary melt
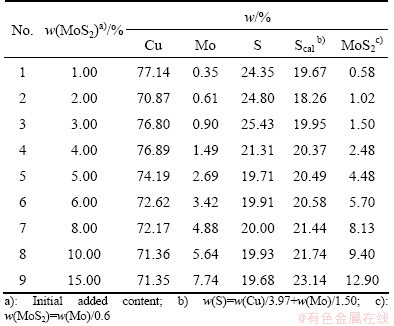
The compositions of the initial composition of Cu2S-MoS2 binary melt are shown in Table 1. From Table 1, MoS2 contents of the fusion samples (5-9) exceed 5%, below the initial addition of MoS2, but the two values are very close, which indicates that the effect of oxidation on the melting is very small. However, the difference between MoS2 and initial addition of MoS2 is relatively large for the fusion samples (1-4) with MoS2 content below 5%. In addition, similar results can also be obtained from the comparison between the S content calculated from the Cu and Mo contents and the assayed S content in the melting product. The results show that vacuumizing and using graphite crucible can restrain the oxidation of Mo and S during the melting process.
Table 1 Initial composition of Cu2S-MoS2 binary melt
3.2 Measurement of melting point for Cu2S-MoS2 binary melt
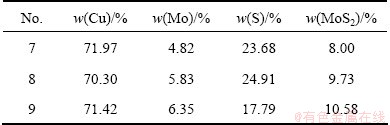
Figure 4 shows the cooling curve of Cu2S-MoS2 binary melt samples 6-9. Through the measurement of cooling curve, after the melt was crushed, it was found that the powders of Cu2S-MoS2 binary melt samples 1-6 completely melted, and the compositions of melts accorded with those in Table 2. However, there was still some unmelted powders in the melt samples 7-9 and the of unmelted amount gradually increased in order. Therefore, the compositions of melt samples 7-9 need to assay again. The melting parts of 7-9 were assayed again, and their compositions are shown in Table 2. From Table 2, the composition of melt sample 7 was almost the same as former assay result. But to melt samples 8 and 9, the MoS2 content was below the initial addition of MoS2. The main reason is that the vacuum induction furnace can provide a higher melting temperature, good stirring effect and good diffusion conditions, so the Cu2S-MoS2 binary melt can completely melt. During the measurement of cooling curve, the Cu2S-MoS2 binary melt was placed in the quartz tube, lack of stirring and easily sticking to the melting.

Fig. 4 Cooling curves of Cu2S-MoS2 binary melt samples
Table 2 Composition of melt samples 7-9 after assay
During the cooling process of Cu2S, the temperature of melt first rose and then decreased. The phenomenon was called “recalescence”. In this period, phase-transition was generated and heat was released. The temperature range of “recalescence” was close to the melting point of Cu2S. The highest temperature of “recalescence” process was 1141.0 °C. Therefore, “recalescence” can be regarded as the solidification process of Cu2S. Generally, the highest temperature of “recalescence” was considered the melting point of substance. So, the measured melting point of Cu2S was 1141.0 °C. Actually, the thermal effect also existed in the cooling process of fusion samples 1-9. But the thermal effect was very weak. Therefore, there was no “recalescence” phenomenon. The cooling rate of furnace was set as 2 °C/min, and the cooling process of melt should also be linear. If the cooling curve deviated from the linear, it can be regarded as the phase-transition, along with the thermal effect. Therefore, the starting temperature deviating linear was regarded as the initial phase-transition temperature, and the temperature regaining linear relationship was regarded as the final phase-transition temperature. Based on this, the initial and final phase-transition temperatures for fusion samples 1-9 are listed in Table 3.
3.3 Binary phase diagram of Cu2S-MoS2 system
According to the phase-transition temperatures of Cu2S-MoS2 melt samples with different compositions (Table 3), the binary phase diagram of Cu2S-MoS2 system is plotted in Fig. 5. In addition, the XRD patterns of samples 1-9 are shown in Fig. 6.
Table 3 Phase-transition temperature of fusion samples with different compositions
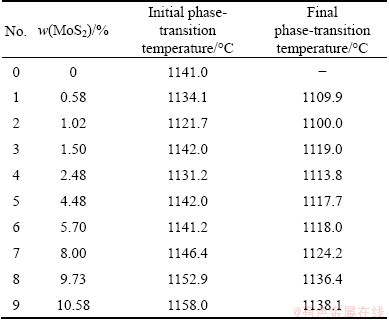

Fig. 5 Binary phase diagram of Cu2S-MoS2 system
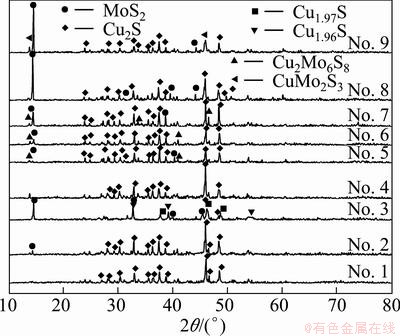
Fig. 6 XRD patterns of Cu2S-MoS2 melt samples with different compositions
From Fig. 5 and Fig. 6, it can be seen that as the MoS2 content of Cu2S-MoS2 melt is 0-4.48%, the plot is the simple eutectic binary phase diagram, and the eutectic temperature is (1117.0±3.0) °C and the MoS2 content of eutectic composition is (1.70±0.20)%. As the MoS2 content exceeds 4.48%, MoS2 and Cu2S can form ternary compound containing CuMo2S3 or Cu2Mo6S8. The melting points of CuMo2S3 and Cu2Mo6S8 are respectively (1770±10) °C and (1750±30) °C [7,9], which are far below the melting point of MoS2, 2050.0 °C. Therefore, the formation of ternary compound is helpful to Mo entering into matte phase. According to the trend of curve, the melting line and liquidoid can be drawn from 1160 °C to 1200 °C. From the figure, the two curves rose with increasing MoS2 content, meanwhile its interval also increased, which indicated that the dissolved Mo as the ternary compound in the Cu2S-MoS2 melt increased gradually. In addition, according to the rising trend of melting line, it can estimate that Cu2S-MoS2 melt can dissolve about 14.80% MoS2 at 1200 °C. The research results are different from the results of GULYANITSKAYA et al [8]. GULYANITSKAYA et al thought that the eutectic composition of Cu2-xS-MoS2 system is 20% MoS2 and the eutectic temperature is 992 °C. It is found that Cu2S-MoS2 binary system with different composition cannot melt at so low temperature. As the MoS2 content is between 4.00%-10.00%, the temperature of melting line should be kept at 1140-1150 °C. The temperature range is suitable for treating molybdenite using the bath smelting process in industry. Although there is some temperature fluctuation during the converting process, MoS2 is also not precipitated.
4 Conclusions
The binary phase diagram of Cu2S-MoS2 system was obtained. MoS2 can dissolve in the Cu2S to form molybdenum-copper matte. As the MoS2 content was 0-4.48%, the Cu2S-MoS2 binary system was the simple binary eutectic. Its eutectic temperature was (1117.0±3.0) °C, and the MoS2 content of eutectic composition was (1.70±0.20)%. As the MoS2 content exceeded 4.48%, MoS2 and Cu2S can form the ternary compound containing CuMo2S3 or Cu2Mo6S8. The dissolved Mo as the ternary compound in the Cu2S-MoS2 melt increased gradually with increasing MoS2 content. At 1200 °C, the solubility of MoS2 in the white matte was 14.8%. The melting temperature (<1200 °C) and the corresponding solubility of MoS2 indicated that the bath smelting process for treating molybdenite seems to be feasible.
References
[1] JOHN S. Application of Mo metal and its alloys [EB/OL]. International molybdenum association [1995]. http://www.imoa.info/ _files/brochures_articles/Applications_Mo_Metal.pdf.
[2] FU Jian-gang, ZHONG Hong. The latest development and current status of molybdenite roasting process [J]. Rare Metals and Cemented Carbides, 2005, 33(2): 43-46. (in Chinese)
[3] GUPTA C K. Extractive metallurgy of molybdenum [M]. Boca Raton: CRC Press, 1992: 225-251.
[4] TANG Jun-li, LIU Dong-xin, TANG Li-xia, GUO Jun-gang. Analyzing the manufacturing technique difference at home and abroad by comparing the quality of molybdenum concentrate, technical molybdenum oxide and ammonium molybdate [J]. China Molybdenum Industry, 2008, 32(2): 42-44. (in Chinese)
[5] XIANG Tie-gen. Molybdenum metallurgy [M]. Changsha: Central South University Press, 2002: 35-56. (in Chinese)
[6] GROVER B, MOH G H. Phase equilibrium relations in the Cu-Mo-S system with reference to natural minerals [J]. Neues Jahrbuch fuer Mineralogie, Monatshefte, 1969, 12: 529-544.
[7] MOH G H. High temperature metal sulfide chemistry [J]. Topics in Current Chemistry, 1978, 76: 107-151.
[8] GULYANITSKAYA Z F, ZVIADADZE G N, POVLYUCHENKO N M. Study of the rhenium sulphide-copper sulphide-molybdenum sulphide (ReS2-Cu2-xS-MoS2) system [R]. Moskava, Russian: Deposita Documents, 1979.
[9] FLUKIGER R, BAILLIF R, MULLER J. The constitution diagram of the system CuxMo6S8 in the temperature range 11-2000 K [J]. Journal of the Less-common Metals, 1980, 72(2): 193-204.
Cu2S-MoS2二元系相图的测定
陈星宇1,赵中伟1,2,郝明明1,刘旭恒1
1. 中南大学 冶金科学与工程学院,长沙 410083;
2. 中南大学 稀有金属冶金与材料制备湖南省重点实验室,长沙 410083
摘 要:针对现有辉钼矿氧化焙烧工艺生产效率低、设备结构复杂、炉料容易烧结、二氧化硫污染严重等不足,提出一种熔池熔炼辉钼矿的新方法。为了得到不同温度下辉钼矿在白冰铜中的溶解度,用干化学法合成硫化亚铜,并用步冷曲线法测定了Cu2S-MoS2 二元系相图。实验结果表明,在MoS2质量分数为0~4.48%的范围内,Cu2S-MoS2 二元系为简单的二元共晶体系,共晶温度为(1117.0±3.0) °C,共晶组成为(1.70±0.20)%MoS2;当MoS2含量超过4.48%时MoS2和Cu2S形成固溶体。在铜熔炼温度范围(1200~1300 °C)内,白冰铜能够溶解辉钼矿,在1200 °C时溶解度达到14.8%。
关键词:二元系;Cu2S-MoS2;熔池熔炼;溶解度;共晶;辉钼矿
(Edited by Hua YANG)
Foundation item: Project (50974137) supported by the National Natural Science Foundation of China
Corresponding author: Zhong-wei ZHAO; Tel: +86-731-88830476; E-mail: zhaozw@csu.edu.cn
DOI: 10.1016/S1003-6326(13)62456-5