
Preparation and characterization of new photocatalyst combined MWCNTs with TiO2 nanotubes
ZHU Zhi-ping(朱志平)1, 2, HUANG Ke-long(黄可龙)1, ZHOU Yi(周 艺)2
1. School of Chemistry and Chemical Engineering, Central South University, Changsha 410083, China;
2. School of Chemistry and Environment Engineering, Changsha University of Science and Technology, Changsha 410076, China
Received 15 July 2007; accepted 10 September 2007
Abstract: A new type of photocatalysts MWCNTs/TiO2-NTs nanocomposites prepared by combining multi-walled carbon nanotube (MWCNTs) with TiO2-derived nanotubes were synthesized by a modified hydrothermal method. The SEM, XRD, UV-Vis and TG-DTG were used to characterize its property. The produced MWCNTs/TiO2-NTs nanocomposites were used as the catalysts for photo-degradation of aquatic humic substances, Their photocatalytic efficiency was evaluated by the photodegradation of humic acid in an aqueous solution under ultraviolet light irradiation. The results show MWCNTs could enhance the activity of the photocatalysts; the best degradation efficiency was obtained by using MWCNTs/TiO2-NTs nanocomposites containing 20% MWCNTs as the photo-catalyst.
Key words: carbon nanotubes(CNTs); titanium dioxide; photocatalysis; humic acid
1 Introduction
How to combine carbon nanotubes(CNTs) with TiO2 and let them become a new photocatalyst is of great interesting work due to it concerns with serious pollution problems. The CNTs has many special characteristics, such as high chemical stability, extraordinary mechanical and unique electronic properties[1-2]; furthermore, it can absorb organic substance strongly with particular p-electron structure[3]; TiO2 has been widely used in solving various environmental problems such as wastewater treatment, air clean, as a photocatalyst[4-6]. Provided that CNTs is used as support material and all those advantages of both CNTs and TiO2 are utilized a powerful photocatalyst could be produced.
There are some endeavors to combine CNTs with TiO2 to form nanocomposites. LI et al[7] has reported TiO2 nanoparticle could chemically absorb at single- walled carbon nanotubes(SWCNTs) defeat sites via an ester-type linkage between carboxylic acid group at the defeat sites and hydroxyl groups at the surface of the TiO2 nanoparticle. LEE et al[8] reported that anatase TiO2 nanoparticles with a size range of 2-10 nm had been formed on CNTs by control hydrolysis and condensation of TALH (titanium bis-ammonium lactato dihydroxide). SUN et al[9] reported that amine- terminated TiO2 nanoparticles were coated on SWCNTs using TiCl4 as starting materials. JITIANU et al[10] reported that anatase TiO2 were coated on MWCNTs by sol-gel method using Ti(OEt)4 and Ti(OPri)4 and by hydrothermal hydrolysis of TiOSO4.
The common characteristics of the above nano- composites are the TiO2 nanoparticles linked or coated on CNTs, in this work, we establish a new type of photocatalyst, the MWCNTs is coupled with TiO2- derived nanotubes(TiO2-NTs) to form MWCNTs/ TiO2-NTs nanocomposite, which can be used as a photocatalyst to degraded humic substances in environmental protection and water supply industry. To our best knowledge, this is the first report dealing with the use of MWCNTs/TiO2-NTs nanocomposite to eliminate aquatic humic substance.
2 Experimental
2.1 Catalyst preparation
Titania nanoparticles were produced by sol-gel method similar with Refs.[11-12].
MWCNTs(made by Sigma-Aldrich Inc., outer diameter is 20-50 nm, length 200 nm-2 mm) was modified as following procedure: 1.0 g MWCNTs were suspended in 40 mL concentrated sulfuric acid and concentrated nitric acid (H2SO4?HNO3=3?1), sonictreated for 2 h, refluxed for 8 h in oil bath at 80 ℃, then filtered (by Millipore 0.45 mm membrane filter) and washed with deionized water until its pH value is adjusted to about 7; then put into 100 mL deionized water, and used in synthesis of CNTs and TiO2 nanocomposites.
The 0.6 g titania nanoparticles, different amount of modified MWCNTs (MWCNTs/TiO2 mass fraction being controlled at 5%-30%), and 10 mol/L NaOH were transferred to a Teflon lined stainless steel autoclave of 100 mL capacity, then heated in oven at 125 ℃ for 24 h, similar to previous reports[13-14]. After cooled down to room temperature, filtered by Millipore 0.45 mm membrane filter, washed with 0.1 mol/L HCl aqueous solution and then with deionized water until its pH value equal to about 7, dried at 105 ℃ for 6 h and calcined at 350 ℃ for 8 h in atmosphere ambient, obtained the black MWCNTs/ TiO2-NTs nanocomposite samples.
2.2 Characterization
The morphology and microstructure of TiO2 nanoparticles, modified MWCNTs, and samples of MWCNTs/TiO2-NTs nanocomposites were characterized with SEM (scanning electron microcopy, HITACHI S-4800 instrument, Japan).
XRD patterns were analyzed by using X-ray diffractometer (i.e. SIEMENS Diffraktometer D5000, Germany)using Cu Kα radiation source at 35 kV, with a scan rate of 0.02?. Crystallite size of anatase TiO2 can be determined from the line broadening by using Scherrer’s formula.
The UV-Vis spectra were measured by a JASCO V-570 UV/Vis/NiR spectrophotometer (made by JASCO Corporation, Japan).
The thermogravimetry-differential thermogravity analysis(TG-DTG) was conducted in N2 and O2 at a heating rate of 10 ℃/min on a Labsys TM 1600 (Setaram Scientific & Instrument Equipment, France).
2.3 Photodegradation experiment
The photodegradation experiments were carried out in closed box, whose UV radiation source is 100 W high-pressure mercury lamp, its wavelength range is 290-450 nm, the peak intensity is 365 nm (Model B 100 A, UVP. Inc, USA); a photoreactor is installed on the inside. The photoreactor consisted of two 100 mL beakers, which filled with 80 mL aquesous humic acid solution and photocatalysts suspension (including TiO2 nanoparticles, modified MWCNTs and samples of MWCNTs/TiO2-NTs), and magnetically stirred. No pure oxygen was supplied because it has enough oxygen for oxidation photodegradation under continuously stirring in atmosphere in previous experiment[15].
The initial humic acid concentration(ρ) was 30 mg/L, the photocatalyst concentration was 1 g/L, and the pH of the solution was adjusted to 4.0 with ammonia and nitric acid solution. Before mercury lamp switched on, the mixture solution of humic acid and photocatalyst was sonic treated for 5 min and then magnetically stirred in a dark condition for 30 min to achieve adsorption- desorption balance, then take out sample, centrifuged (Eppendorf, Centrifuge 5417, Germany) and filtrated with 0.45 mm Millpore membrane filters, and measured clear solution concentration(ρ0) by JASCO V-570 UV/Vis/NiR spectrophotometer at 436, 400, 365, 280 and 254 nm wavelength, the ρ0 was considered as beginning concentration after dark adsorption[16]. The samples were withdrawn regularly (in general, every 30 or 1 h for each sample) from the beakers. The humic acid concentrations(ρi) in different times were obtained and the removal rate could be expressed as following formula.
Removal rate: R=ρi/ρ0×100%
3 Results and discussion
3.1 Catalyst characterization
The SEM images of CNTs and TiO2 nanocomposite are given in Fig.1. There are two kinds of nanotubes in the nanocomposite, one is TiO2 nanotube whose diameter is about 10 nm (Fig.1(a)), and another is carbon nanotube Fig.1(b). The TiO2 nanotubes have much higher specific surface area than TiO2 nanoparticles and many hydroxyl group excelling in ion absorption[17], but in general, the TiO2 nanotubes is composed of H2Ti3O7 [18], and H2TinO2n+1?xH2O (3<n<6)[19] as well as H2Ti2O5? H2O[20], which are amorphous with poor photocatalysis activities; if calcined at 350 ℃ in air, a parts of them would changed into anatase and maybe used to be a photocatalyst[21], meanwhile, if temperature is larger than 493 ℃, the MWCNTs could be disappear[22], so in our experiment, 350℃ was chosen as the calcinations temperature, and the samples obtained has photocatalysis activities.
The XRD patterns of MWCNTs, raw material TiO2, and MWCNTs/TiO2-NTs were given in Fig.2. The most intense two peaks of MWCNTs correspond with (002) and (100) reflection, respectively. The raw material TiO2 was in anatase phase; the successful conversion of

Fig.1 SEM images of MWCNTs/TiO2-NTs nanocomposite (Bar full size: 500 nm, MWCNTs of 20%)

Fig.2 XRD patterns of MWCNTs, raw material TiO2, and nanocomposites of MWCNTs/TiO2-NTs: (a) 5% MWCNTs/ TiO2; (b) 20% MWCNTs/TiO2; (c) 30% MWCNTs/TiO2; (d) MWCNTs; (e) TiO2
MWCNTs/TiO2-NTs composites to anatase was demonstrated by XRD analysis using the calcined MWCNTs/TiO2-NTs composites.
The TG-DTG curves of MWCNTs, MWCNTs/ TiO2-NTs in O2 ambient are given in Figs.3 and 4. Pure MWCNTs in Fig.3 exhibit a sharp mass loss at 500- 620 ℃, indicating carbon nanotube disappeared, Note
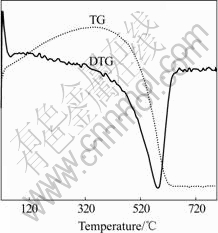
Fig.3 TG-DTG curve of pure MWCNTs in O2 ambient
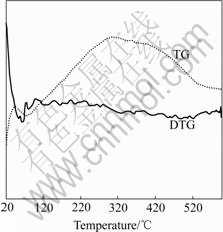
Fig.4 TG-DTG curves of 20% MWCNTs/TiO2-NTs in O2 ambient
that MWCNTs disappeared at the calcination temperature higher than 495 ℃.
The UV-Vis spectra of nanocomposites of MWCNTs/TiO2-NTs are given in Fig.5. The absorption edge shifted to a shorter wavelength (Blue shift) corresponding to the order: (d)>(c)>(b)>(a); which means more higher percent MWCNTs, more stronger shift ability.
3.2 Photocatalysis result
The percent of removal rate of humic acid at different UV light irradiation time over MWCNTs, raw material TiO2, and MWCNTs/TiO2-NTs with different ratios were shown in Fig.6. The MWCNTs shows high adsorbing capacity at the beginning of irradiation time, which has some connection with its special structure[23]; but has poor removal rate because of without photocatalysis ability. The TiO2 particles has a higher
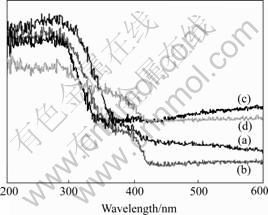
Fig.5 UV-Vis spectra of MWCNTs/TiO2-NTs: (a) 5% MWCNTs/TiO2; (b) 10% MWCNTs/TiO2; (c) 20% MWCNTs/ TiO2; (d) 30% MWCNTs/TiO2

Fig.6 Removal rate of humic acid by using different catalysts
removal rate than MWCNTs, but lower than all MWCNTs/TiO2-NTs, because the TiO2 nanotubes are more active than pure TiO2 nanoparticles for photodegradation of organic substance[24]. At the same time, the diffuse reflectance UV-vis spectra of the MWCNTs/TiO2-NTs composites gives new insights into the photo-degradation mechanism, they show the characteristic spectrum of anatase with its fundamental absorption edge rising at 400 nm. The MWCNTs/ TiO2-NTs composites, on the other hand, can absorb at higher wavelength than the sole TiO2. The absorption is observed even totally over the whole range of the UV-vis spectrum for the MWCNTs/ TiO2-NTs composites of 20% and 30% of MWCNTs. It is noticeable that the MWCNTs/TiO2-NTs composites of 20% MWCNT provides the largest change of the UV-vis spectrum, and be followed by 30%, 10%, and 5% MWCNTs.
4 Conclusions
1) The UV-Vis spectra of the nanocomposites samples indicates the enhancement of absorption increase with MWCNTs percent from 5% to 30%.
2) The synergetic effect has observed for nano- composites of MWCNTs/TiO2 on the photocatalytic degradation of humic acid, which increases with MWCNTs percent from 5% to 20%, the 20% MWCNTs/TiO2 has optimum synergetic effect and has the highest removal rate.
3) The MWCNTs/TiO2-NTs could be utilized with success in other removals of organic pollutants and further studies should be carried out.
References
[1] DURAIRAJ B, JIMMY W M, MATTHEW S B. Noncovalent and nonspecific molecular interactions of polymers with multi-walled carbon nanotubes [J]. Chem Mater, 2005, 17: 3389-3397.
[2] GE J J, ZHANG D, LI Q, HOU H. Multiwalled carbon nanotubes with chemically grafted polyetherimides [J]. J Am Chem Soc, 2005, 127: 9984-9985.
[3] FUGETSU B, SATOH S, SHIBA T, MIZUTANI T. Caged multiwalled carbon nanotubes as the adsorbents for affinity-based elimination of ionic dyes [J]. Environ Sci Technol, 2004, 38: 6890-6896.
[4] MASAKAZU A. Preparation, characterization, and reactivities of high functional tianium oxide-based photocatalyst able to operate under UV-visible light irradiation: Approaches in realizing high efficiency in the use of visible light [J]. Bull Chem Soc Jpn, 2004, 77: 1427-1442.
[5] MILLS A, HUNTE S L. An overview of semiconductor photocatalysis [J]. Photochem Photobiol, 1997, A108: 1-35.
[6] MICHAEL R H, SCOT T M, CHOI WON-YONG, DETLEF W B. Environmental applications of semiconductor photocatalysis [J]. Chem Rev, 1995, 95: 69-96.
[7] LI Xiao-hong, NIU Jia-li, ZHANG Jin, LI Hu-lin. Labeling the defects of single-walled carbon nanotubes using titanium dioxide nanoparticles [J]. J Phys Chem B, 2003, 107: 2455-2458.
[8] LEE S, WOLFGANG M S. Formation of anatase TiO2 nanoparticles on carbon nanotubes [J]. Chem Commun, 2003, 780-781.
[9] SUN Jing, MIKIO I, GAO Lian, ZHANG Qing-hong. Single-walled carbon nanotubes coated with titania nanoparticles [J]. Carbon, 2004, 42: 885-901.
[10] JITIANU A, CACCIAGUERRA T, BENOIT R, DELPEUX S. Synthesis and characterization of carbon nanotubes-TiO2 nanocomposites [J]. Carbon, 2004, 42: 1147-1151.
[11] LIU Xiao-heng, YANG Juan, WANG Ling, YANG Xu-jie. An improvement on sol-gel method for preparing ultrafine and crystallized titania powder [J]. Materials Science and Engineering A, 2000, 289: 241-245.
[12] COL?N G, HIDALGO M C, NAV?O A. A novel preparation of high surface area TiO2 nanoparticles from alkoxide precursor and using active carbon as additive [J]. Catalysis Today, 2002, 76: 91-101.
[13] KASUGA T, HIRAMATSU M, HOSON A, SEKINO T. Formation of tianium oxide nanotube [J]. Langmuir, 1998, 14: 3160-3163.
[14] KASUGA T, HIRAMATSU M, HOSON A, SEKINO T. Titania nanotube prepared by chemical processing [J]. Adv Mater, 1999, 11: 1307-1311.
[15] ZHOU Yi, XU Xie-wen, LIU Qi-cheng. Catalyzed oxidation capability of nanometer grain under natural light [J]. Journal of Central South University of Technology, 2002, 33: 371-373.
[16] RADWAN A R, DAVID J C. Photocatalytic degradation of humic acid in saline waters (Part 2): Effects of various photocatalytic materials [J]. Applied Catalysis A: General, 2003, 246: 39-48.
[17] TOMOKO K. Formation of titanium oxide nanotubes using chemical treatments and their characteristic properties [J]. Thin Solid Films, 2006, 496: 141-145.
[18] CHEN Q, ZHOU W Z, DU G H. Trititanate nanotubes made via a single alkali treatment [J]. Adv Mater, 2002, 14: 1208-1211.
[19] LAN Ying, GAO Xue-ping, ZHU Huai-yong, ZHENG Zhang-feng. Titanate nanotubes and nanorodes prepared from rutile powder [J]. Adv Funct Mat, 2005, 15: 1310-1318.
[20] SONG Zhi-qiang, XU Hai-yan, LI Kun-Wei, WANG Hao. Hydrothermal synthesis and photocatalytic properties of titanium acid H2Ti2O5・H2O nanosheets [J]. Journal of Molecular Catalysis A: Chemical, 2005, 239: 87-91.
[21] RYUHEI Y, YOSHIKAZU S, SUSUMU Y. Effects of synthetic conditions and heat-treatment on the structure of partially ion-exchanged titanate nanotubes [J]. Materials Chemistry and Physics, 2005, 91: 409-416.
[22] LEE S H, SMITHI P, BRIJ M, WOLFGANG S. Inactivation of bacterial endospores by photocatalytic nanocomposites [J]. Colloids and Surfaces B: Biointerfaces, 2005, 40: 93-98.
[23] XU Ji-chuan, LU Mei, GUO Xin-yong, LI Hu-lin. Zinc ions surface-doped titanium dioxide nanotubes and its photocatalysis activity for degradation of methyl orange in water [J]. Journal of Molecular Catalysis A: Chemical, 2005, 226: 123-127.
[24] WANG Wen-dong, PHILIPPE S, PHILIPPE K, JOAQUIM L F. Visible light photodegradation of phenol on MWNT-TiO2 composite catalysts prepared by a modified sol-gel method [J]. Journal of Molecular Catalysis A: Chemical, 2005, 235: 194-199.
(Edited by LAI Hai-hui)
Foundation item: Project(03JJY4046) supported by the Natural Science Foundation of Hunan Province, China
Corresponding author: ZHU Zhi-ping; Tel: +86-731-2618234; E-mail: zzp8389@163.com