������ʱ��: 2019-05-17 10:48
ϡ�н��� 2021,45(01),93-105 DOI:10.13373/j.cnki.cjrm.xy19040039
����Ⱦ�������������о���չ
�Ӿ� ������ ������ ���� ��ѧ��
ժ Ҫ��
�˿�ұ��ҵ��Ѹ�ٷ�չ�������β��(��)�Ĵ����ѻ�������Χ�����͵���ˮ����Ⱦ�������أ������ཡ������ᰲȫ���DZ����в������Ⱦ�������ѳ�Ϊؽ������Ļ������⡣�������Ⱦ��������������ʵ��֤��ֻʹ�ô�ͳ������ѧ����������Ⱦ��ʱ���Ѵﵽ�������Ч�����������ɱ�ͨ���dz����������������ij��֣�Ϊ����Ⱦ�����ṩ�˸���ɫ�����ã���Ч���ȶ����ֶΣ��������о�ѧ���ǵĹ㷺��ע�����нϴ���о���ֵ������Ӧ��ǰ�������ȶ��ֽ�����Ⱦ�������������˼��ܣ����ص�����������Ⱦ�����������������ڹ������о���չ��״�����Ž���������Ⱦ���������û����Լ��������Ӷ�����Ⱦ��Ч����Ӱ�죬��������Ŀǰ����Ⱦ�������������ڵ����⣬�����δ���д������о��ķ���Ϊ����Ⱦ�����ṩ����˼·��
�ؼ��ʣ�
����Ⱦ ;������ ;���ﻹԭ ;������ ;Ӱ������ ;
��ͼ����ţ� X172;X591
����飺 �Ӿ�(1995-)��Ů�����Ϻ����ˣ�˶ʿ�о������о���������Ⱦ��������E-mail:15707332838@163.com��; *��������ڣ��绰��010-82241312,E-mail:wellwoodliu@163.com;
�ո����ڣ� 2019-04-19
���� ������Ȼ��ѧ������Ŀ(U1402234,41573074); �����ص��з��ƻ���Ŀ(2018YFC1802702,2018YFC1801803); ������ѧ�о��뼼�������ƻ���Ŀ(���AB16380287�����AB17129025); �����ƹ��ֺ���ʩ���ۼ������Է�������������Ŀ(�ƹ���˾[2018]1521��)����;
Research Progress of Bioremediation Technology for Uranium Contamination
Zhong Juan Liu Xingyu Zhang Mingjiang Yan Xiao Hu Xuewu
National Engineering Laboratory of Biohydrometallury,GRIMAT Engineering Institute Co.,Ltd
Abstract��
Anthropogenic activities such as uranium mining activities,primarily associated with decades of nuclear fuel production andweapon making,had accumulated a huge amount of uranium tailings(residue)and abundant hazardous uranium waste.Thus,caused anincreasingly serious uranium contamination in surrounding soil and under ground water,which posed a potential threat to the humanhealth and social security around the world.In particular,radioactive uranium commonly present as the uranyl cation,is highly solubleand mobile under oxidizing conditions and poses great danger to human health.Therefore,it is significant to focus on developing efficientremediation and long-term remediation strategies of increasingly severe uranium contamination.Various methods for the remediation ofuranium were emerged in an endless stream.Traditional physical and chemical remediation approaches based on pump and treat practice,such as lime neutralization,anion exchange,activated aluminum and biosorption,were not only prohibitively expensive but could also belimited by poor extraction efficiency,inhibitory competing ions and massive waste production.Besides,bringing the radioactive contami-nants up to the surface could increase health and safety risks for cleanup workers and the public.So,there was a great need for cost-effec-tive alternatives to treat uranium-contaminated groundwater and prevent its further migration and spread through the deep subsurface.However,the emergency of bioremediation technologies provided an eco-friendly,high-efficiency,stably to solve this problem.Bioreme-diation technologies had attracted extensive attention of researchers,which had great research value and broad application prospect.It wasfound that under the stress of uranium,microorganisms used the deposit or other external substances to gain energy for metabolism in orderto grow and survive,and deposited the dissolved U(VI)through biological reduction,biomineralization and other ways,reducing the con-centration of U(VI)in the solution.At present,the mechanisms of microbial remediation of uranium contamination mainly included biore-duction,biomineralization,biosorption and bioaccumulation.Uranium bioreduction had been proposed as a bioremediation technique,stimulated by adding an electron donor to promote enzymaticreduction of aqueous U(VI)to insoluble U(IV).The speciation of bioreduceduranium was often stated to be uraninite(UO2).Bioreduction was widely used in field tests for its high efficiency,low cost and simple oper-ation.However,potential concerns associated with the use of bioreduction as a remediation technique from whether reduced U(IV)wouldbe stable over long time periods,especially when the environment changed,such as the presence of oxygen and nitrate,solidified U(IV)would be reoxidized to the dissolved U(VI).Biomineralisation referred to the process by which metals precipitated with microbially gener-ated ligands such as sulfide or phosphate,or ascarbonates or hydroxides in response to localized alkaline conditionsat the cell surface.Adding inorganic phosphate directly into the contaminated area could also precipitate U(VI).However,due to its high activity,it was like-ly to precipitate rapidly with metal ions,resulting in a decrease in the permeability coefficient of the adding point.Compared with bioreduc-tion,biomineralization of U(VI)phosphate was observed over a wide p H range and in the presence of high uranium and nitrate concentra-tions,and might be a complementary approach to bioreduction.The cost of organophosphate was the biggest obstacle to the wide applica-tion of biomineralization remediation technology.In addition,some researchers believed that the rapid precipitation of metals on the cellsurface could hinder the cell metabolism and reduced the remediation effect.Biosorption described the passive uptake of uranium to thesurface of living or dead microbial cells.Despite the potential for bacteria to biosorb uranium,it was unlikely to be useful in the context ofbioremediation.Problems associated with biosorption were that desorption from cell surfaces could be as rapid assorption,and other cat-ions competed for binding site.Cell surfaces could also quickly become saturated,preventing further biosorption.Sorbed material couldbe re-released to solution when cells died and decomposed.Microbial cells were also able to accumulate a broad range of metalions via��bioaccumulation��mechanisms.Although of academic interest,there was scant evidence suggesting bioaccumulation of uranium wouldbe a viable technique for bioremediating contaminated land or water.Uranium remediation rates and effects had been extensively studied,with most studies conducted in microcosms containing suspended cells.However,the results of remediation rates and effects between stud-ies or between different microbial species were often difficult to predict because of the large variation in the experimental conditions.Manyphysical,geochemical and biological factors could affect the remediation effects,including aqueous uranyl speciation and uranium initialconcentration,cell concentration,temperature and p H,electron donor,bicarbonate,competing electron acceptors and other compounds.Besides,these factors and their effects were often closely coupled.The mobility and bioavailability of uranium varied with the type andcomplexity of the complex formed by uranium.And other culture parameters could substantially influence the reduction effects,becausethey influenced U speciation in aqueous environments.Thus,an understanding of U speciation was pivotal when considering the designand operation of bioremediation systems for U(VI)removal.In addition,many of the parameters,such as p H,temperature and high con-centrations of other metal ions could have direct effects on the growth and activity of U(VI)-remediation microorganisms.Overall,it wasclear that microbial remediation had a significant impact on uranium contamination across a wide range of environments and would be im-portant in managing contaminated land sites.But there were also some shortcomings that needed to be solved urgently.The long-term sta-bility of the bioreduced U(IV)solids was still questionable and further research was needed to underst and the mechanisms that led to theformation of most stable U(IV)products to effectively decrease U(IV)reoxidation and ensure longevity and environmental safety.Re-search worth further exploration in the future included:(1)Screening and cultivating the strains with higher remediation efficiency;(2)In the premise of high effect of remediation,choose cheaper carbon and phosphorus sources to promote the remediation of uranium contam-ination by target strains;(3)Molecular biological research methods were used to understand the metabolic pathways and figured out thesuccession of microbial community in the whole process of remediation.(4)Combined remediation method was adopted to make up for thepossible deficiency of single remediation method,so as to obtain better remediation effect and shorten remediation period.
Keyword��
uranium contamination; bioremediation; biological reduction; remediation mechanisms; influence factor;
Received�� 2019-04-19
���Ÿ����˵�Ĵ�����չ������Դ�������ӣ��˿�Ŀ��ɡ�ұ��Ѹ�ٷ�չ������ȫ���˲����Ծ��˵��ٶ���������֮����������β�������Ĵ����ѻ�
[1 ]
���ݲ���ȫͳ�ƣ�ȫ������β���ܲ����Ѹߴ�200���ڶ֡��ҹ�����β��ȹ������ѷų�Լ��200�����ֲ���14��ʡ��30�������
[2 ]
������ҹ�ʵ�ʹ��飬�йز����ƶ��ˡ��˿�ұ��ʩ�����������������涨�������˰�ȫ���������Ⱦ���Ρ�ʮ����滮����2025��Զ��Ŀ�꡷�����л������˰�ȫ������һϵ�����˿�ұ���������йصı��ͷ��ɡ�
�ѻ�����β���������绯���������ˡ�����ȹ�ͬ�����£����еķ��������ʺ��ؽ������Ӳ��Ͻ�����������ˮ���ﳾǨ����ɢ�������ܱ����������������ˮ���ڱ���Ⱦˮ���У���ͨ�����������ӣ�UO2 2+ ����̬���ڣ�UO2 2+ �ܺ������������Լ�̼��������������������γɸ����������
[3 ]
��������̬�˵Ļ�����ͨ���ܽ�ȸߣ����������ˮǨ�������������ཡ���ͻ�����ɳ���DZ����в���˶����彡����Σ����Ҫ�������ؽ�����ѧ���Ժͷ����Է���Σ��������ʽ�����л�ѧ���Զ������Σ��Զ���ڷ����Զ������á��������������Ҫ����ڸ��ࡢ��������У��������Ի������ж����շ����ּ�������ͻ�䡢������������
[4 ]
����ˣ�����Ⱦ�����ͷ����ѳ�Ϊؽ������Ļ������⡣
����������Ⱦ������һ����������չDZ�������������������Ҫͨ�����ﻹԭ������ȷ�ʽ���˷�������ã��ı��˵ĸ���״̬���������ڻ����е�Ǩ���ʣ��Լ����䶾������
[5 ]
���÷����й�����Ӧ��ǰ���������������о�ѧ�߹�ע���������Ƚ���������Ⱦ��������״�������ص�����������Ⱦ������������������������������û����Լ�����Ⱦ������Ӱ�����أ������������������ٵĻ�������ս������չ��������Ϊ�ҹ�����Ⱦ���о����������ṩ�������ݡ�
1 ����Ⱦ�������о�����
����Ⱦ�����������·�Ϊ��������ѧ�Լ����3�֡���ͳ������ѧ����ָ����������ѧ��ԭ�����������ӽ�������ʯ���кͷ����������ȣ������Ҫ����ֲ������
[6 ,7 ]
��Ŀǰ���ҹ��˿�ұ�������ʵ���У�һ��������ʯ���к�β��������������p Hֵ�������˺Ͳ�����Ⱦ����ܳ�����Դͷ������Ⱦ��IJ��������Ѳ�������ˮ���ռ�����Ҫ��ȡ��ѧ���������������ķ�ʽ��������Щ������ʽ��Ч�죬���ܽ���Ⱦ�����ˮ�г���ȥ������Ҳ����һ���ıˣ���ʯ���кͷ����ڷ��ᡢ�˳����ܳ������⣻����������������Ч�����ȶ���������ȥ��Ч�ʵ͡�ѡ��������
����������������������Ҫ��ɲ��֣���ָ������Ȼ���ڵĻ��������Ĺ�����������˵Ļ��������£�ͨ������ķǴ�л�����������ʹ�л��������ԭ���õ�һ������������
[8 ]
������Ⱦ��������������Ҫ��ͨ������Ⱦ�����м�����ӹ��弰������ѧ���ʴٽ���Ⱦ�����ض�������������Լӿ���Ⱦ���صĻ�ԭ�̶�����Щ�����������������Ӿ����Լ����̾������������гɱ��ͣ��������Ի���������С��������Ⱦ���ŵ㡣���⣬����Խ����Ĺ̻�Ч���ȶ��ҿɳ�������ʹβ�����ٲ����µ��ܽ�̬U(VI������Դͷ�Ͻ���˵��ܳ����⣬����DZ�ڻ�����Ⱦ���ա�
Ŀǰ�����ҹ��ѹ������ض����ܾ���ɸѡ������ɸѡ�����ܻ�ԭ�������˵�����
[9 ]
���о���ͨ�������л�̼Դ���л���Դ�ļ���̼������U(VI���Ļ�ԭ�Ϳ��Լ��������������ؼ��������ӵ�Ӱ��Ч��
[10 ,11 ,12 ]
�������˾�����������ϼ���ǿ��ֲ�����������Ⱦ�ļ���
[8 ]
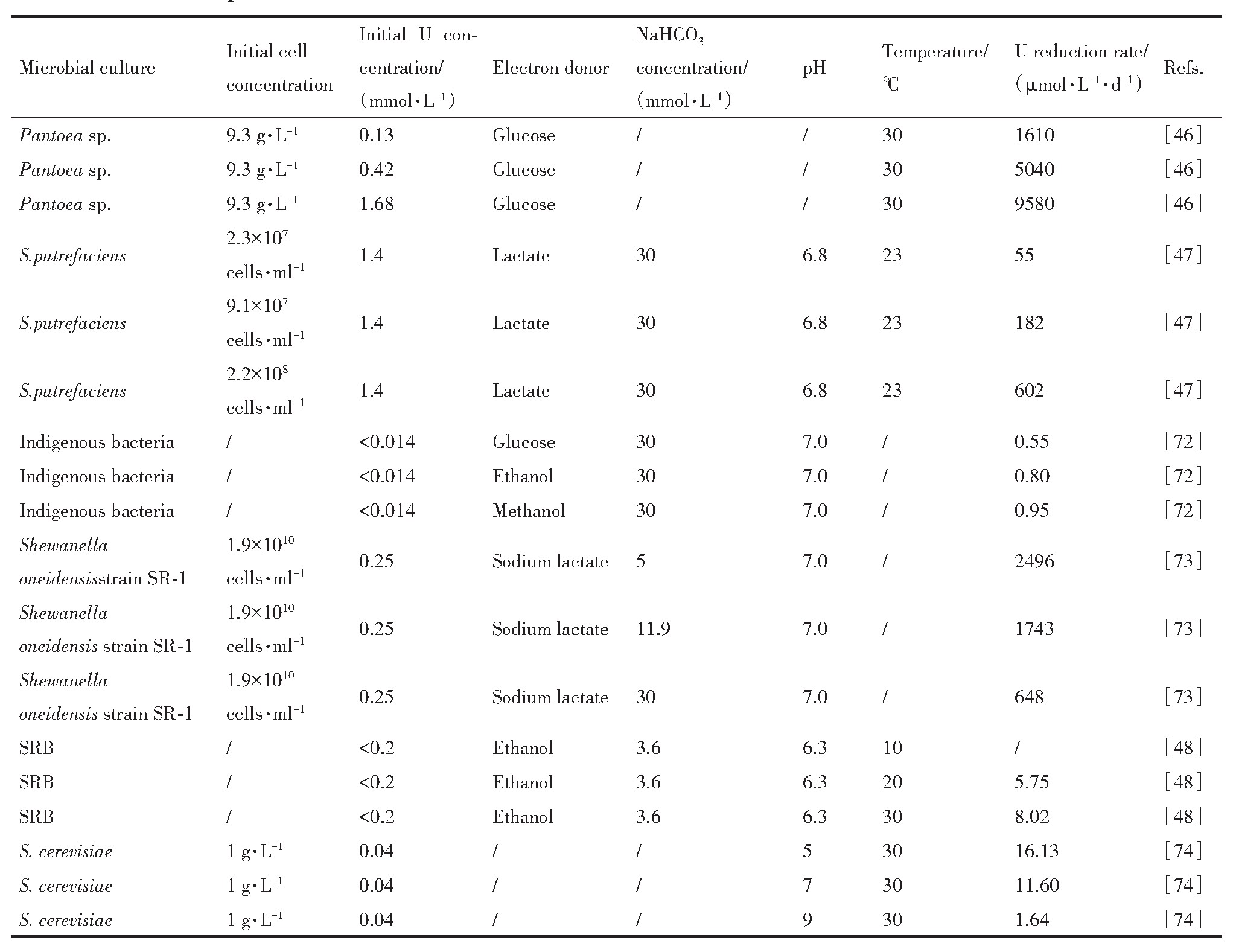
�������ϣ�����������������Ⱦ���о�������Ҫ�����ڣ�1��ɸѡ��ѱ�������Ը�Ч������ꣻ��2��ͨ�����������߹�����������Ⱦ�����еĻ��ԣ���3����������֮�������ú�������˵ijɿ��������4�������̲������Ż����¶ȡ�p H��̼Դ�ȹؼ����ӵĵ��صȷ��档
�ӹ�������Ⱦ������״�����������������ں�����Ⱦ�����������������õ�Ӧ��ǰ���������������������ڹ��ڶ�����˽�������Ⱦ�����л��Ӧ�ã���������Ⱦ�����е�Ӧ�û������о��Σ���δ�й���ʵ�����������Ѿ�������������չ�˶�ι������顣���磬Anderson��
[13 ]
�������
[14 ]
�ֱ����������������ݺ������뿪չ����Ⱦ�����飬ͨ������Ⱦ����ע�������Ҵ�ԭλ�̼��컯������ԭ�����������о�����U(VI����Ũ�Ⱦ��½������ﵽԤ�ڱ���
��������������������ﴦ���������������γ����볣�����������ѧ�����棬����䡢��ͬ���ơ�Э����չ���¸�֡���������������Ⱦ���η�����ʾ�����Եļ������ƣ��������\����������Ⱦ��������Ӧ��ǰ����
2 ����������Ⱦ�����û����о���չ
�о����֣����˵�в���£�����Ϊ�������ʹ���������Ӧ�����仯�����ÿ�������ʽ����³´�л��ͬʱ��ͨ�����ﻹԭ������ȷ�ʽʹ�ܽ�̬��U(VI������������������Һ��U(VI����Ũ�ȡ�Ŀǰ����������Ⱦ�����û�����Ҫ�������ﻹԭ����������������Լ����︻��4�֣���ͼ1��ʾ��
2.1 ���ﻹԭ
���ﻹԭ����ȱ�������£��������ô����Ρ������Ρ�H2 ����Ϊ���ӹ��壬����Ǩ�Ƶ�U(VI����ԭΪ���ȶ���U(IV������ԭ�õ���U(IV����Ҫ�Ծ����˿�UO2 ������̬����
[16 ]
��20����60�����Woolfolk��Whiteley
[17 ]
֤�������ﻹԭU(VI����������Ȼ��ֱ��30���Lovely��
[18 ]
���״������������ԭ��������ˮ�еĿ�����U������ת��Ϊ�ȶ��ġ��ܽ�ȵ͵�U��������������ֹ��Ǩ����ɢ�����롣
�о�����������ԭ����Geobactermetallireducens�������λ�ԭ����Desulfovibriovulgaris��Desulfovibriodesulfuricans strain G20��������ɻ�ԭU(VI)
[19 ]
�������λ�ԭ����SRB����������Ҫ�������л��ʻ�ԭSO4 2- ΪH2 S��Ȼ����������SO4 2- �������ṩ����ĵ�������ʱ��SRB��ͨ�������л��U(VI����ԭΪU(IV���������л�ȡ��л����������Zhou��
[20 ]
�о�����ʵ��5 h��Desulfovibrio vulgaris��U(VI���Ļ�ԭ�ʸߴ�80%��ͬʱ�����ʵĺ���������19%��֤ʵ��D.vulgaris�ܹ��ڻ�ԭU(VI����ͬʱ�ٽ������������������ڽӽ����Ի����£�U(VI����Fe3+ �����Ƶ�������ԭ��λ����Fe3+ ����������д��ڴ���U(VI���������£�����ԭ����Fe RB��Ҳ�ܽ�U(VI����л�Ի�ԭΪU(IV)
[21 ]
�����⣬����ϸ�����������ճϸ����Ҳ�������˵Ļ�ԭ
[22 ]
��U(VI�������ﻹԭ����ͨ��ΪUO2 ����Ҳ���о���Ա������������̬���ڵ�U(VI���Ļ�ԭ������磬Thermoterrabacterium ferrireducens���Ի�ԭU(VI��Ϊ[��NH4 )(UO2 )(PO4 ����3H2 O]����Shewanellaputrefaciens CN32�ܽ���������е�U(VI����ԭΪ��UO3 ��2H2 O)
[10 ]
��
ͼ1 ����������Ⱦ�����û���ʾ��ͼ
Fig.1 Schematic diagram of bioremediation mechanisms of uranium contamination
[15]
�����о�����������Ի�ԭU(VI�������仹ԭ������δ��ȫ���ա�������о�����ֱ��ø�ٷ�Ӧ�ǽ鵼U(VI����ԭ����������
[23 ,24 ]
����Ҳ������ΪU(VI���Ļ�ԭ����Fe2+ ,H2 �Ƚ鵼�ķ����ﻹԭ����
[25 ]
�����ﻹԭ��Ч�ʸߣ��ɱ��ͣ����������㷺Ӧ�����ֳ������С������ﻹԭ���U(VI���������Գ�ʱ��ά���ȶ����ر��ǵ����������仯ʱ�������������������δ���ʱ�̻���U(IV������������Ϊ�ܽ�̬��U(VI����
2.2 �����
�����Ҳ�����������ָ����ͨ��ϸ������ֲ������ʹU(VI����������̼���λ���������
[26 ]
����U(VI��������ø���������ɵ���λ���������Σ������εȹ���������HUO2 PO4 ,[Ca(UO2 )2 (PO4 )2 ]���ȶ��������
[27 ]
��20����90������������״η���Serratia�ܽ�U(VI�������������ο������̬������������ʱ�������Ǽ�����������κ���Citrobacter sp.�������·���ΪSerratia�����ڳ�������ø�ֽܷ���л������Σ��ͷų�������������U(VI����Ӧ���ɰ���HUO2 PO4
[28 ]
��������������ʵ�����о�������Ca2+ Ũ�ȱȽϸߵ�����»��γ�[Ca(UO2 )2 (PO4 )2 ]���������⣬˿״�����Aspergillus niger�����ں����������������������������䷢��ľ�˿Ҳ�ܳ��������������������Liang��
[29 ]
�ڸ��Բ����������м��������������л������Σ�������Aspergillus niger��Paecilomycesjavanicus������Թ۲쵽�˵������γ�����
ֱ������Ⱦ���������������Ҳ�ܳ���U(VI������������dz����ã��ܿ������������Ѹ�����ɳ��������¼�������ϵ���½����谭������ļ���Ǩ��
[30 ]
����ͨ������ø�ٷ�Ӧˮ���л��Ĺ����ǻ������еģ��Ӷ�������ע��λ�ñ������ķ��ա����⣬ϸ���ܳ�Ϊ�����ɺ˺��ģ���˸÷������˵�Ч�ʸ��ߡ���������ﻹԭ����������ڸ�����p Hֵ��Χ�ڡ��ϸߵ�������������Ũ����������Ⱦ
[31 ]
��ͬʱ���ɵ��������������ڽϿ���p Hֵ��Χ�����ܽ�ȵͣ��ܳ��ڱ����ȶ���Ŀǰ�谭������������㷺Ӧ�õ�����ϰ����л������εijɱ�����
[22 ]
�����⣬���о�ѧ����Ϊϸ����������Ŀ��ٳ���ԭ���ϻ��谭ϸ���Ĵ�л��������Ч����
2.3 ��������
����������ָ�˱�����������ϸ�����档��������ͨ���dz�Ѹ�٣�����ϸ����������л�ء���ϸ����ϸ���ȱ������ϸ���ڱ��溬�еĹ����ţ����ǻ����Ȼ����ϻ��ȣ�������ɣ����Խ�����������������ϸ���ڱ���
[32 ,33 ]
��Ŀǰѿ�߸˾����ظ˾�����״ѿ�߸˾�����ù���ͽڸ˾�����֤��������Ϊ�˵�����������������ѿ�߸˾��Ѷ����������Ⱦ��������Gorman-Lewis��
[34 ]
�о��˿ݲ�ѿ�߸˾����˵���������������������κ�ʵ�������¶��ܹ۲쵽����ϸ������Ĵ��������������������Ƚ��ʺϴ����е�Ũ�ȵ��ؽ�����ˮ���ӵ�Ʒ�ˮ�з������Pseudomonas MGF-48����p HΪ6.5ʱ���þ���5 min�ڿ�������86%����
[35 ]
�������о��������߾�����ĸ����˿״�����Ҳ�������������˵�DZ�������磬Zinjard��
[36 ]
��ʯ����Ⱦ��ˮ�з�����Ľ�ĸ����p HΪ7.5ʱ�ܹ�������Һ��50%���ˡ�
��Ȼ������������˵�DZ�������������Ӧ��������Ⱦ��ȴ����ʵ�֡�������Ϊ�����е�����������Ҳ�Ὰ��ϸ��Ĥ��������λ�㣬��Щλ������״ﵽ���ͣ��Ӷ���ֹ�������ļ������С�ͬʱ��ϸ��������ֽ⣬���������ӻᱻ�����ͷš���Ҳ�Ǿ���������������ȥ��ˮ��Һ�е��ˣ����伸��û�й�ҵӦ�õ�ԭ��
2.4 ���︻��
�������ͨ�����︻�����ն����ؽ������ӽ���ϸ�����ڡ�Ȼ���˲�ͬ��ϸ����л�������Ԫ�أ���Fe,Zn�ȣ�������ͨ��һ��������;����ϸ���ڸ������о�����������ϸ�����ڵĸ�����������Ϊ�˵Ķ����ƻ���ϸ��Ĥ��ͨ��
[37 ]
�����⣬Ҳû��ֱ��֤�ݱ����������ڴ����˵�ת�˵��ף�������︻���˱���Ϊ����ϸ��������л�ص�
[38 ]
������һ������ϸ���ڻ��ۣ�ϸ�����ܻ�ͨ�����ַ�ʽ���̶��ˣ�����������ζ��˵��������ã�����һ�ֱ����Ľ������ܻ��ƣ������ϸ�����˵�������
[39 ]
�����ֻ�������������п��Թ۲쵽����A.ferroxidans
[40 ]
��Sphingomonas sp.S15��S1
[41 ]
�������о�ѧ�߶����︻����չ������о���������û��֤�ݱ������︻��������Ⱦ�Ƿ���С�
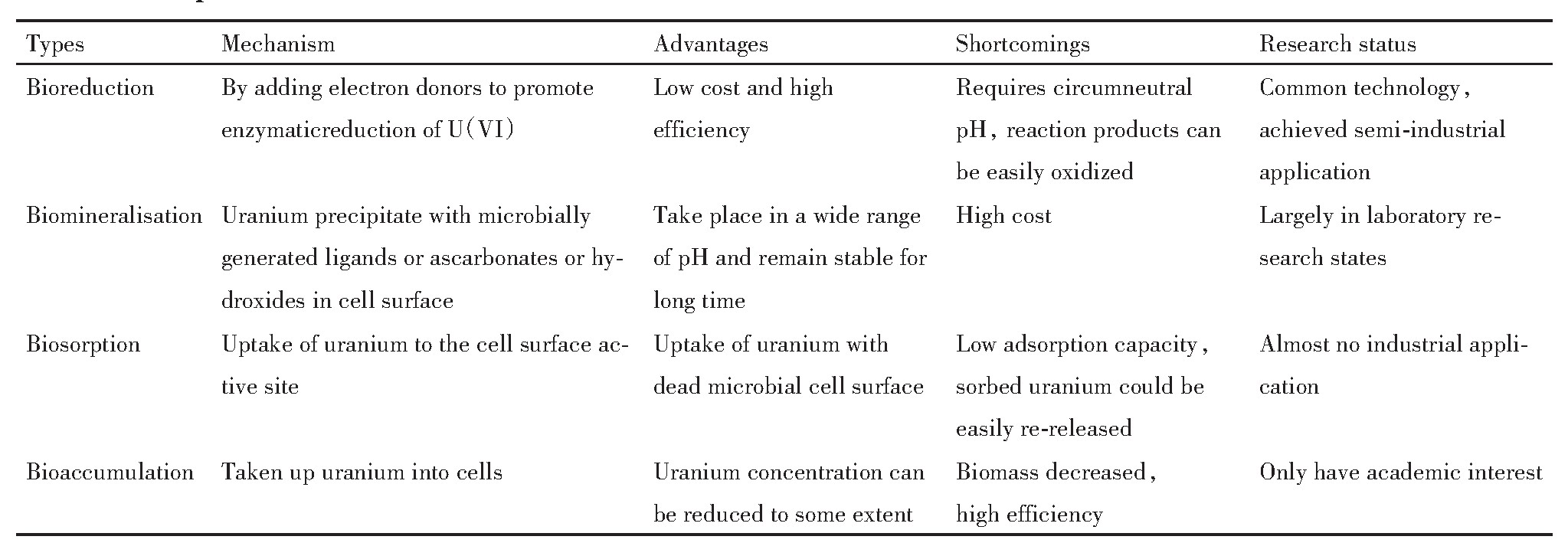
����ۺϱ����������ݶ�����Ⱦ���������û��Ƶ�ԭ������ȱ���Լ��о���״�����˹����ܽᣬ���1��ʾ��
3 �������Ӷ�����Ⱦ������Ч����Ӱ��
����Ⱦ������Ч����������Ԥ�⣬���ض������»����ض������õĽ����һ�����������������������塣����Ҫ����Ϊ��������ض���Ӱ����Ч���������¶ȡ����ȡ����������˵�Ũ�Ⱥ���״̬�Լ�����������Ĵ��ڵȵȡ�
��1 ����Ⱦ���������û��ƶԱ� ����ԭͼ
Table 1 Comparison of bioremediation mechanisms of uranium contamination
3.1 �˵�Ũ�ȼ��丳����̬
������ø�ٷ�Ӧ���ƣ�����Ũ������ʱ���˵Ļ�ԭ�������ӣ���Ũ�ȹ���ʱ���˵Ļ�ԭ�ʷ������Խ��ͣ�������Ϊ��Ũ�ȵ��˶������ж������á����˶�����Ķ�������ǿ������ȡ�����˵�Ũ�ȣ���ȡ��������������Լ��˵ĸ���״̬�ȡ��о�������ͬ��������˵������������Բ�ͬ�����磬���˵�Ũ�ȷֱ�ﵽ50,1000,1190 mg��L-1 ʱ������Clostridium sp.ATCC 53464,Pseudomonas aeruginosa,Thermoterrabacterium ferrireducens������
[42 ]
���ڲ�ͬ�����У��˿��벻ͬ�����γɶ���������������������ˮ���������������������������������̼��������Ϊ��������Щ�������ܽ�ȴ������Բ���
[43 ,44 ]
���˵�Ǩ���ʺ���������ö������γɵ���������ͺ��ӳ̶ȶ��죬���˵���������ö������䶾��������أ�����˵ĸ���״̬��һ���̶��Ͼ������˵Ķ���ǿ����Belli��
[45 ]
�о��������˶�S.putrefaciens�Ķ������Է�̼������̬���ڵ���������Ũ��ֱ����أ�������Ũ�Ȳ���ֱ������Ԥ���˵Ķ��ԡ����⣬���ǻ���ΪCa�Ĵ����ܽ����˵Ķ��ԣ�������ΪCa?UO2 ?CO3 �������γɽ������˵���������öȡ�
3.2 ���������¶Ⱥ�p Hֵ
�����о����������ӳ�ʼ������Ũ�ȿ�������˵�ȥ���ʡ����磬Chabalala��Chirwa���о���
[46 ]
����ʼ������Ũ�ȸߴ�9.3 g��L-1 ������һ���̶��Ͽ��Խ�����ʵ��24 h���˵Ļ�ԭ�ʸߴ�100%��ԭ�����⣬Senko��
[47 ]
���о���������S.putrefaciens��ʼ�����������ӣ�U(VI���Ļ�ԭ����Ҳ������ߡ�Ȼ�����Ⲣ����ζ����ʵ��Ӧ���У�Ӧ�ü������������ΪΪ��ά�ָߵ�������Ũ����ҪͶ�������Ӫ��Դ���������ɴ�����������ɱ������⣬��������������ܻ����β���϶���ر�������������ֳ������С�
�¶Ⱥ�p Hֵ������Ӱ�������ø����˵ĸ���״̬���Ӷ�Ӱ������Ⱦ����Ч�ʡ���Ȼ�¶ȵ�����ͨ����ӿ컯ѧ��Ӧ�����ʣ������������ֻ�ܴ����ij���ض����¶Ⱥ�p Hֵ��Χ�ڡ����˵�p Hֵ���¶Ȼ�Ӱ�������л���ԡ�����Boonchayaanant��
[48 ]
�����Ƶ��¶ȴ�20��������30��ʱ��U(VI����ԭ������ߣ�����10��ʱ�����۲첻�����ϵͻ�ϸߵ�p H�ή��������ԣ�����U(VI���Ļ�ԭ�����о�����ijЩϸ�����ڽϵ͵�p Hֵ�����»�ԭU(VI������Gao��Francis
[49 ]
��������Ⱦ���з����Clostridium sp.�Խϵ͵�p Hֵ���нϸߵ������ԣ�����p HֵԼΪ4ʱ��ԭU(VI����
��ͼ2���Կ�����p HֵС��5�����7.5ʱ������Ҫ�����ܽ��UO2 2+ ��̼����������̬���ڣ������Һ��U(VI����Ũ�����ӡ�ͬʱ�ڽϵ�p Hֵ�£�����H+ �����С��Ũ�Ƚϸ߶�����ռ��ϸ��ϸ��Ĥ�������λ�㣬�Ӷ���ֹ���������ӵĻ�ԭ����������ˣ������U(VI�������ﻹԭ����Ҫp Hֵ�ӽ��������������⣬������p Hֵ�Ե������Ի�����5.7��6.2�������ܻ����ƾ����������������������������л���ӹ�������������δ֤������л�ԭU(VI��������
[50 ]
��
3.3 ���ӹ���
����ͨ���̼�����������������ɹ�ʵ��ԭλ���ﻹԭU(VI����ѡ����ʵĵ��ӹ����DZز����ٵġ������������£�������ԭϸ���Ե����ʡ���ά�ػ���֬����ȸ߷����л�������Ĵ�л��������
[52 ]
����ˣ�U(VI�������ﻹԭͨ������ѡ��ͷ������л�����Ϊ���ӹ��塣Ŀǰ�о����ְ����������Ҵ��ȴ��ࡢ�����ǵ����ࡢ�����ε��л����ࡢ�Լ��ױ��ȷ���������������Ϊ���ӹ���ٽ�U(VI�������ﻹԭ
[53 ]
���ֳ������й㷺Ӧ�ô�������Ϊ���ӹ���ʵ��U(VI�������ﻹԭ
[54 ,55 ]
��N�@Guessan��
[56 ]
�о����ֵ���������������ӹ��壬U(VI���Ļ�ԭ��������ˣ����һ�ԭ���U(IV���������ٶȴ�������Ȼ����Ҳ���о����ּ��࣬�ر��Ǽ״����Ҵ����DZȴ����θ����˵ĵ��ӹ���
[57 ]
����˵�����ӹ����ѡ�������Ҫ����һϵ�����أ�������Ⱦ����ԭ������Ⱥ��ṹ����������Լ�����ѧ�����ȡ����磬��ij����Ⱦ������ô����κ������Ƕ�U(VI�����ﻹԭ��Ч�����Ժ��ڼ����λ�������
[58 ]
������һ�о���ȴ�����Ҵ��ȴ����λ��������ܸ���ػ�ԭU(VI)
[57 ]
��
ͼ2 U-CO2-H2OϵU(VI���ĸ���״̬��p Hֵ�Ĺ�ϵ
Fig.2 Distribution of U(VI)species as a function of p H in U-CO2 -H2 O system
[51]
���ü��л�����ٽ�����ԭλ������Ⱦ�������ǿ��еģ������ȴ̼�ע��㸽���������������ܻ�������Ⱦ����ֲ�����������U(VI���Ļ�ԭ���ʡ��о����֣����ø����ӵĵ��ӹ����������Ρ�ֲ���͡�������֬��Ӧ����ԭλ��������Ч�����ܸ��ã���Ϊ���ǵ����ͷ��ٶ����������ڸ���Χ��ά�ֻ�ԭ��Χ
[53 ]
��
3.4 ̼������
�о�������̼�����ζ�����Ⱥ��ṹ��U(VI����ȥ����������Ӱ�졣���벻ͬŨ�ȵ�̼�����οɵ�����Һp Hֵ��Һ����U(VI���ķ�������仯
[59 ]
��Lovehy��Phillips
[60 ]
��20����90����״������̼������Ӧ��������Ⱦ�������������о�����̼�����ζ�U(VI�����Ѹ�����������U(VI���Ļ�ԭ���Ͽ��Դٽ���������Ч��������������ֳ�����Ҳ֤��̼�������ܴٽ�U(VI���Ļ�ԭ�����磬Long��
[54 ]
����������Ⱦ���龮�м�������Լ�̼�����Σ���U(VI���Ļ�ԭ�������Ը���ֻ�Ӵ���Ķ����顣���о�����Ũ�ȹ��ߵ�̼�����ο��ܻή������Ļ��ԣ��Ӷ�����U(VI���Ļ�ԭ������D.desulfuricans��̼������Ũ��Ϊ30mmol��L-1 ��Һ�л�ԭU(VI�������ʱ�100 mmol��L-1 �Ŀ�
[60 ]
��
���⣬������ϵ�л�����һ������Caʱ��Ca����U(VI����̼�������γ�Ca?UO2 ?CO3 �����Ӷ�����Fe RB��SRB��U(VI���Ļ�ԭ
[61 ]
��
3.5 �����Ե�������
�����Ρ������Ρ��̺���������Ⱦ�����еĵ�����֣�ͬʱҲ�������������������˵������塣���ǵĴ��ڿ����ӻ�������ֹU(VI����ԭ��
SO4 2- �ܶ�U(VI���Ļ�ԭ����Ҫ���á�SO4 2- �ܴٽ���ɻ�ԭU(VI����Desulfovibrio spp.��������Ҳ���Դٽ���Desulfobacter spp.�����������Dz��ܻ�ԭU(VI����������U(VI����ԭ���������ӹ��塣Spear��
[62 ]
���о����������εĴ��������U(VI���Ļ�ԭ�ʣ����������ΪSO4 2- ��Ϊ���������ܴٽ�SRB����������ǿϸ�����ԡ�����SO4 2- ��ԭ���ɵ�����Ҳ���л�ԭU(VI��������
[63 ]
����Ҳ���о�ѧ����ΪU(VI���Ļ�ԭ�ٶȲ���SO4 2- Ũ�ȵ�Ӱ�죬��SO4 2- ������U(VI���Ļ�ԭ�ٶȣ������������SO4 2- ����U(VI���������ӹ��塣
NO3 - �ձ����������Ⱦ�����У����о�������NO3 - �ĺ����ߴ�50 mg��L-1 ʱ������D.desulfuricans��U(VI���Ļ�ԭ
[64 ]
��NO3 - ��U(VI����ԭ��Ӱ����Ҫ�����������棺һ����U(VI���������ӹ��壬����U(VI���Ļ�ԭ��������ʹ��ԭ���ɵ�U(IV����������Istok��
[65 ]
��һ��NO3 - Ũ�Ⱥܸߵĺ�ˮ��������̼Դ�̼���������ԭU(VI�������鷢�ּ����̼Դ��NO3 - �ܿ챻���ģ�����һ�β�δ����U(VI���Ļ�ԭ��ֱ��NO3 - ����ȫ���IJ����¼������̼Դ��ŷ���U(VI���Ļ�ԭ������ֽε��ֳ������飬ͨ������ͨ����ѧ�Լ���ϴ
[66 ]
������������
[67 ]
�ȷ�ʽ��ȥ��Ⱦ�����NO3 - ������������ȥ���ˡ�
���ݱ�2���������ۻ�ԭ��λ��֪��Mn(IV���Ļ�ԭ��λ��ߣ�Fe(III����֮��U(VI����ĩ����˵�ϸ�����л�̼Դ��H2 �����ӹ���ʱ��Mn(IV����Fe(III��Ӧ����U(VI��֮ǰ����ԭ�����⣬��������ѧ����Mn(IV����Fe(III������ʹ��ԭ��U(IV�����±�������DZ������p Hֵ��5��8֮��ʱ��Fe(III��һ�����������ˮ�������̬���ڣ����ܽ�Ⱥܵͣ�Fe(III����U(IV����ԭ��Ӱ��ɺ��Բ��ơ�Ȼ������p HֵС��5ʱ��Fe(III���ڳ������е�Ũ��ͨ��Զ����U(VI),U(VI���Ļ�ԭͨ����Fe(III���Ļ�ԭͬʱ����
[68 ,69 ]
��Ŀǰ��Mn(IV����U(IV����ԭ��Ӱ���о����١�Fredrickson��
[70 ]
���������������ܽ���U(VI����ԭ���ʣ���U(VI�����ܻ�ԭ�ʲ�δ���١�
��2 �缫��Ӧ�����ۻ�ԭ��λ��E0 ) ����ԭͼ
Table 2 Electrode reactions and theoretical reduction po-tentials(E0 )
3.6 ����������
����Ⱦ������ڵĽ������ӣ�����Zn,Ni,Cu,Ca��Mg������������������Ⱦ��Zn,Ni��Cu���������ÿ����������������Ķ������¡��о�������Ni��11.7 mg��L-1 ,Zn��25 mg��L-1 ��Cu��15 mg��L-1 ʱ������ȫ����U(VI����ȥ��
[19 ]
����Ca��Mg����U(VI����̼�����γ�����ȶ������ܵ���Ԫ̼�����������Ӷ�����U(VI��������Brooks��
[71 ]
�о����ֵ���Һ�д���Caʱ��U(VI���Ļ�ԭЧ�ʴ�͡����������������Ҫԭ������ǵ���Һ�д��ڸ�ʱ��Ca2 UO2 (CO3 )3 ������Һ��ռ��Ҫ�ɷ֡�Ca2 UO2 (CO3 )3 ��ԭ���Ƶͣ����ױ���ԭ������Ϊһ�־������Ƽ�������������̬���˱���ԭ��
�о�ѧ�߶Ի�������Ӱ������Ⱦ��������Ч�����˴����о�����������ѡȡ�����д�����ʵ�飬�����˲��ֻ���Ӱ�����Ӷ��˻�ԭ�ʵ�Ӱ�죬���3��ʾ��
��3 ���ֻ���Ӱ�����Ӷ��˻�ԭ�ʵ�Ӱ�� ����ԭͼ
Table 3 Influence of partial environmental factors on uranium reduction rates
4 ���ڵ����⼰չ��
��������������������Ⱦ������ɱ��ͣ���������Ч���ã�������Ⱦ���ŵ��������о�ѧ�ߵĹ�ע��ȡ����һЩͻ���Ե��о���չ����Ҳ������һЩ���㣬ؽ������ȥ��������磬��������ʱ��Խϳ�����Ч����һ���IJ�ȷ���ԣ����ҵ����������ı�ʱ������IJ���������Գ�ʱ��ά���ȶ���
�ֽε��о���Ҫ������ʵ����С���о������ģ�ֳ������������٣���Щ������ʵ�������нϺõ�Ч������һ��Ӧ����ʵ�ʹ��̣���Ч����������ۿۡ���������������Ҫԭ��һ�Ƕ�����������Ⱦ�����û����о����������룬δ����Ū������Ⱦ�������Լ���������һ����������ε��ز��������õģ������ֳ��������Ӷ�䣬����Ⱦ�����ͣ��¶ȡ����ȣ��˵ĸ���״̬��������ԣ�����������Ĵ��ڵ���һϵ�����ض���Ӱ������Ч�������������ҪӦ�����ֳ����飬�����һ�����о���̽����δ��ֵ������̽�����о����������
1.ɸѡ��������Ч�ʸ��ߵ����ƾ��꣬��Ҫʱ������л�����죬�����Ի����������Ժ���Ч�ʡ�
2.�ڱ�֤��Ч����ǰ���£�Ѱ��۸������̼Դ����Դ���ٽ�Ŀ�����ƾ��������Ⱦ������
3.���÷�������ѧ�о��������˽�����Ⱦ��������ϸ����л;���͵��ػ��ƣ����պ͵�����ȫ�����е�����Ⱥ�����棬���������Ч�����ȶ��ԡ�
4.�о���������������������ֲ�����������������Ӧǽ���������������绯ѧ�������������ȷ������ֲ���һ���������ܴ��ڵIJ��㣬��ø��õ���Ч�������������ڡ�
�����
[1] L��Y,L��J W,Zhou J L,Shen J.Surfactant study on promoting leaching rate of uranium[J].Chinese Journal of Rare Metals,2016,41(2):182.(���������ģ��ܽ������꽨.������Լ�����˿�ʯ�������о�[J].ϡ�н�����2016,41(2):182.)
[2] Zhang B,Zhang X W,Li M,Tang D S,Lv J W,Tan WF.The characteristics and research progress of uranium tailings comprehensive treatment techniques[J].Journal of China Mining Magazine,2015,24(4):58.(�ű룬�����ģ����ܣ��ƶ�ɽ�������ģ�̷�ķ�.��β����Ⱦ�������ۺ����������о���չ[J].�й���ҵ��2015,24(4):58.)
[3] Cheng C J.Experimental study of contaminants Release from Dump Leaching Tailings-Water Interaction in Some Uranium Mine of Guangdong[D].Hengyang:University of South China,2014.5.(�̴���.�㶫ij�˿�ѽ�β��-ˮ�������Ⱦ��Ǩ�ƹ���ʵ���о�[D].�������ϻ���ѧ��2014.5.)
[4] Shin W,Oh J,Choung S,Cho B W,Lee K S,Yun U,Woo N C,Kim H K.Distribution and potential health risk of groundwater uranium in Korea[J].Chemosphere,2016,163:108.
[5] Acharya C.Uranium bioremediation:approaches and challenges[J].Environmental Microbial Biotechnology,2015,45:119.
[6] George C E,Lightsey G R,Jun I,Fan J.Soil decontamination via microwave and radio frequency co-volatilization[J].Environmental Progress&Sustainable Energy,2010,11(3):216.
[7] Bhalara P D,Punetha D,Balasubramanian K.A review of potential remediation techniques for uranium(VI)ion retrieval from contaminated aqueous environment[J].Journal of Environmental Chemical Engineering,2014,2(3):1621.
[8] Rong L S.Plant-microbial Remediation of Uranium-Contaminated Soil and the Mechanism Analysis[D].Hengyang:University of South China,2015.5.(����ɼ.����Ⱦ������ֲ��-��������������о�[D].�������ϻ���ѧ��2015.5.)
[9] Gu Z H.Experimental study on Efficiency and Mechanism of Removal U(��)by Humic-Reducing Bacteria[D].Hengyang:University of South China,2013.6.(���л�.��ֳ�ʻ�ԭ��ȥ���˿�ұ��ˮ��U(��)��Ч������������о�[D].�������ϻ���ѧ��2013.6.)
[10] Li D X.Experimental Studies on Bioreduction of U(VI)in Ground Water in a Uranium Tailings Reservoir by Indigenous Functional Microbial Communities[D].Hengyang:University of south China,2018.3.(�����.������������Ⱥ�仹ԭij��β������ˮ��U(��)��ʵ���о�[D].�������ϻ���ѧ��2018.3.)
[11] Huang C.An Experimental Study on Remediation of Uranium Contaminated Groundwater by��-glycerophosphate[D].Hengyang:University of South China,2018.6.(�Ƴ�.��-���������������˵���ˮ��ʵ���о�[D].�������ϻ���ѧ��2018.6.)
[12] Yan X,Liu X Y,Zhang M J,Cui X L,Zhong J,Hu XW.Research progress of biosorption technology for chromium contamination[J].Chinese Journal of Rare Metals,2019.DOI:10.13373/j.cnki.cjrm.XY19060028.(���죬����������������������Ӿ꣬��ѧ��.����Ⱦ���������������о���չ[J].ϡ�н�����2019.DOI:10.13373/j.cnki.cjrm.XY19060028.)
[13] Anderson R T,Vrionis H A,Ortiz-Bernad I,Resch CT,Long P E,Dayvault R,Karp K,Marutzky S,Metzler D R,Peacock A,White D C,Lowe M,Lovley D R.Stimulating the in situ activity of geobacter species to remove uranium from the groundwater of a uranium-contaminated aquifer[J].Applied and Environmental Microbiology,2003,69(10):5884.
[14] Wu W M,Carley J,Watson D,Gu B H,Scott B,Shelly D,Kenneth K,Joy D.Van N,Wu L Y,Xu MY,Zhou J Z,Luo J,Erick C,Huang J Q,Matthew W,Terence L,James M,Stefan J,Joel E,Peter K,Philip M,Craig S.Bioreduction and immobilization of uranium in situ:a case study at a USA department of Energy radioactive waste site,Oak Ridge,Tennessee[J].Acta Scientiae Circumstantiae,2011,31(3):449.(��Ψ��Carley Jack,Watson David���˱�����Brooks Scott,Kelly Shelly D,Kemner Kenneth,Joy D.Nostrand Van�������Σ���õӢ���ܼ��У�����Cardenas Erick���S������Fields Matthew W,Terence L.Marsh,James M.Tiedje,Stefan J.Green,Joel E.Kostka,Peter K.Kitanidis,Philip M.Jardine,Craig S.Criddle.����ˮ����Ⱦ��ԭλ���ﻹԭ��̶�����������Դ���������������������Ⱦ�ֳ�������[J].������ѧѧ����2011,31(3):449.)
[15] Newsome L,Morris K,Lloyd J R.The biogeochemistry and bioremediation of uranium and other priority radionuclides[J].Chemical Geology,2014,363:164.
[16] Lovley D R,Phillips E J.Reduction of uranium by Desulfovibrio desulfuricans[J].Applied&Environmental Microbiology,1992,58(3):850.
[17] Woolfolk C A,Whiteley H R.Reduction of inorganic compounds with molecular hydrogen by Micrococcus lactilyticus.I.Stoichiometry with compounds of arsenic,selenium,tellurium,transition and other elements[J].Journal of Bacteriology,1962,84:659.
[18] Lovley D R,Phillips E J.Reduction of uranium by Desulfovibrio desulfuricans[J].Applied and Environmental Microbiology,1992,58(3):850.
[19] Lakaniemi A,Douglas G B,Kaksonen A H.Engineering and kinetic aspects of bacterial uranium reduction for the remediation of uranium contaminated environments[J].Journal of Hazardous Materials,2019,371:198.
[20] Zhou C,Vannela R,Hyun S P,Hayes K F,Rittmann B E.Growth of Desulfovibrio vulgaris when respiring U(VI)and characterization of biogenic uraninite[J].Environmental Science&Technology,2014,48(12):6928.
[21] Liu J S,Zhang Y H,Bang-Mei L I,Xie X H.The study kinetic for growth of Acidithiobacillus ferrooxidans[J].Journal of Microbiology,2006,26(2):9.
[22] Newsome L,Morris K,Lloyd J R.The biogeochemistry and bioremediation of uranium and other priority radionuclides[J].Chemical Geology,2014,363:164.
[23] Williams K H,Bargar J R,Lloyd J R,Lovley D R.Bioremediation of uranium-contaminated groundwater:a systems approach to subsurface biogeochemistry[J].Current Opinion in Biotechnology,2013,24(3):489.
[24] Bargar J R,Williams K H,Campbell K M,Long P E,Stubbs J E,Suvorova E I,Lezama-Pacheco J S,Alessi D S,Stylo M,Webb S M,Davis J A,Giammar D E,Blue L Y,Bernier-Latmani R.Uranium redox transition pathways in acetate-amended sediments[J].Proceedings of the National Academy of Sciences,2013,110(12):4506.
[25] Fox P M,Davis J A,Kukkadapu R,Singer D M,Bargar J,Williams K H.Abiotic U(VI)reduction by sorbed Fe(II)on natural sediments[J].Geochimica et Cosmochimica Acta,2013,117:266.
[26] Van Roy S,Peys K,Dresselaers T,Diels L.The use of an Akaligenes eutrophus biofilm in a membrane bioreactor for heavy metal recovery[J].Research in Microbiology,1997,6(148):526.
[27] Newsome L,Morris K,Lloyd J R.Uranium biominerals precipitated by an environmental isolate of serratia under anaerobic conditions[J].PLOS ONE,2015,10(7):e132392.
[28] Macaskie L,Empson R,Cheetham A,Grey C,Skarnulis A.Uranium bioaccumulation by a Citrobacter sp.as a result of enzymically mediated growth of polycrystalline HUO2 PO4 [J].Science,1992,257(5071):782.
[29] Liang X,Csetenyi L,Gadd G M.Uranium bioprecipitation mediated by yeasts utilizing organic phosphorus substrates[J].Appl Microbiol Biotechnol,2016,100(11):5141.
[30] Wellman D M,Icenhower J P,Owen A T.Comparative analysis of soluble phosphate amendments for the remediation of heavy metal contaminants:effect on sediment hydraulic conductivity[J].Environmental Chemistry,2006,3(3):219.
[31] Beazley M J,Martinez R J,Webb S M,Sobecky P A,Taillefert M.The effect of p H and natural microbial phosphatase activity on the speciation of uranium in subsurface soils[J].Geochimica et Cosmochimica Acta,2011,75(19):5648.
[32] Bader M,M��ller K,Foerstendorf H,Schmidt M,Simmons K,Swanson J S,Reed D T,Stumpf T,Cherkouk A.Comparative analysis of uranium bioassociation with halophilic bacteria and archaea[J].PLOS ONE,2018,13(1):e190953.
[33] Meng C Y,Jing Q K,Ma J,Liu W Y,Liu X Y,Wen JK.Overview of microbiological technology for recovery of rare metal resources[J].Chinese Journal of Rare Metals,2015,39(4):371.(�ϴ�褣���Ǭ���������������壬������½���.���\����ϡ�н�����Դ�����е��о��ſ�[J].ϡ�н�����2015,39(4):371.)
[34] Gorman-Lewis D,Elias P E,Fein J B.Adsorption of aqueous uranyl complexes ontobacillus subtilis cells[J].Environmental Science&Technology,2005,39(13):4906.
[35] Malekzadeh F,Farazmand A,Ghafourian H,Shahamat M,Levin M,Colwell R R.Uranium accumulation by a bacterium isolated from electroplating effluent[J].World Journal of Microbiology and Biotechnology,2002,18(4):295.
[36] Zinjarde S S,Pant A A.Hydrocarbon degraders from tropical marine environments[J].Marine Pollution Bulletin,2002,44(2):118.
[37] Suzuki Y,Banfield J F.Geomicrobiology of uranium[J].Reviews in Mineralogy&Geochemistry,1999,38(4):393.
[38] Suzuki Y,Banfield J F.Resistance to,and accumulation of,uranium by bacteria from a uranium-contaminated site[J].Geomicrobiology Journal,2004,21(2):113.
[39] Van Engelen M R,Field E K,Gerlach R,Lee B D,Apel W A,Peyton B M.UO2 2+ speciation determines uranium toxicity and bioaccumulation in an environmental Pseudomonas sp.isolate[J].Environmental Toxicology and Chemistry,2010,29(4):763.
[40] Merroun M,Hennig C,Rossberg A,Reich T,SelenskaPobell S.Characterization of U(VI)-Acidithiobacillus ferrooxidans complexes using EXAFS,transmission electron microscopy,and energy-dispersive X-ray analysis[J].Radiochimica Acta,2009,91(10):583.
[41] Merroun M,Nedelkova M,Rossberg A,Hennig C,Scheinost A C,Selenskapobell S.Interaction mechanisms of uranium with bacterial strains isolated from extreme habitats[J].Special Publication-Royal Society of Chemistry,2006,94:723.
[42] Tapia-Rodr��guez A,Luna-Velasco A,Field J A,SierraAlvarez R.Toxicity of uranium to microbial communities in anaerobic biofilms[J].Water,Air,&Soil Pollution,2012,223(7):3859.
[43] Schindler M,Legrand C A,Jr M F H.Alteration,adsorption and nucleation processes on clay-water interfaces:mechanisms for the retention of uranium by altered clay surfaces on the nanometer scale[J].Geochimica Et Cosmochimica Acta,2015,153:15.
[44] Sharp J O,Lezama-Pacheco J S,Schofield E J,Junier P,Ulrich K U,Chinni S,Veeramani H,Margot-Roquier C,Webb S M,Tebo B M.Uranium speciation and stability after reductive immobilization in aquifer sediments[J].Geochimica Et Cosmochimica Acta,2011,75(21):6497.
[45] Belli K M,Dichristina T J,Cappellen P V,Taillefert M.Effects of aqueous uranyl speciation on the kinetics of microbial uranium reduction[J].Geochimica Et Cosmochimica Acta,2015,157(15):109.
[46] Chabalala S,Chirwa E M N.Uranium(VI)reduction and removal by high performing purified anaerobic cultures from mine soil[J].Chemosphere,2010,78(1):52.
[47] Senko J M,Kelly S D,Dohnalkova A C,Mc Donough JT,Kemner K M,Burgos W D.The effect of U(VI)bioreduction kinetics on subsequent reoxidation of biogenic U(IV)[J].Geochimica et Cosmochimica Acta,2007,71(19):4644.
[48] Boonchayaanant B,Kitanidis P K,Criddle C S.Growth and cometabolic reduction kinetics of a uranium-and sulfate-reducing Desulfovibrio/Clostridia mixed culture:temperature effects[J].Biotechnology and Bioengineering,2008,99(5):1107.
[49] Gao W,Francis A J.Reduction of uranium(VI)to uranium(IV)by clostridia[J].Applied and Environmental Microbiology,2008,74(14):4580.
[50] Finneran K T,Anderson R T,Nevin K P,Lovley D R.Potential for bioremediation of uranium-contaminated aquifers with microbial U(VI)reduction[J].Journal of Soil Contamination,2002,11(3):339.
[51] Xie Y,Chen C,Ren X,Wang X,Wang H,Wang X.Emerging natural and tailored materials for uranium-contaminated water treatment and environmental remediation[J].Progress in Materials Science,2019,103:180.
[52] Kourtev P S,Nakatsu C H,Konopka A.Responses of the anaerobic bacterial community to addition of organic C in chromium (VI)-and iron(III)-amended microcosms[J].Applied and Environmental Microbiology,2006,72(1):628.
[53] Barlett M,Moon H S,Peacock A A,Hedrick D B,Williams K H,Long P E,Derek,Jaffe L P R.Uranium reduction and microbial community development in response to stimulation with different electron donors[J].Biodegradation,2012,4(23):535.
[54] Long P E,Williams K H,Davis J A,Fox P M,Wilkins M J,Yabusaki S B,Fang Y,Waichler S R,Berman ES F,Gupta M,Chandler D P,Murray C,Peacock A D,Giloteaux L,Handley K M,Lovley D R,Banfield J F.Bicarbonate impact on U(VI)bioreduction in a shallow alluvial aquifer[J].Geochimica et Cosmochimica Acta,2015,150:106.
[55] Williams K H,Long P E,Davis J A,Wilkins M J,N'Guessan A L,Steefel C I,Yang L,Newcomer D,Spane F A,Kerkhof L J.Acetate availability and its influence on sustainable bioremediation of uranium-contaminated groundwater[J].Geomicrobiology Journal,2011,28(5-6):519.
[56] N'Guessan A L,Moon H S,Peacock A D,Tan H,Sinha M,Long P E,Jaffe P R.Postbiostimulation microbial community structure changes that control the reoxidation of uranium[J].FEMS Microbiol Ecol,2010,74(1):184.
[57] Wu W,Carley J,Gentry T,Ginder-Vogel M A,Fienen M,Mehlhorn T,Yan H,Caroll S,Pace M N,Nyman J,Luo J,Gentile M E,Fields M W,Hickey R F,Gu B,Watson D,Cirpka O A,Zhou J,Fendorf S,Kitanidis P K,Jardine P M,Criddle C S.Pilot-scale in situ bioremedation of uranium in a highly contaminated Aquifer.2.reduction of U(VI)and geochemical control of U(VI)bioavailability[J].Environmental Science&Technology,2006,40(12):3986.
[58] Finneran K T,Anderson R T,Nevin K P,Lovley D R.Potential for bioremediation of uranium-contaminated aquifers with microbial U(VI)reduction[J].Journal of Soil Cont amination,2002,11(3):339.
[59] Zhou P,Gu B.Extraction of oxidized and reduced forms of uranium from contaminated soils:effects of carbonate concentration and p H[J].Environmental Science&Technology,2005,39(12):4435.
[60] Lovley D R,Phillips E J.Reduction of uranium by Desulfovibrio desulfuricans[J].Applied&Environmental Microbiology,1992,58(3):850.
[61] Luo W,Wu W,Yan T,Criddle C S,Jardine P M,Zhou J,Gu B.Influence of bicarbonate,sulfate,and electron donors on biological reduction of uranium and microbial community composition[J].Applied Microbiology and Biotechnology,2007,77(3):713.
[62] Spear J R,Figueroa L A,Honeyman B D.Modeling reduction of uranium U(VI)under variable sulfate concentrations by sulfate-reducing bacteria[J].Applied&Environmental Microbiology,2000,66(9):3711.
[63] Hua B,Xu H,Terry J,Deng B.Kinetics of uranium(VI)reduction by hydrogen sulfide in anoxic aqueous systems[J].Environmental Science&Technology,2006,40(15):4666.
[64] Tucker M D,Barton L L,Thomson B M.Removal of Uand Mo from water by immobilized desulfovibrio desulfuricans in column reactors[J].Biotechnology&Bioengineering,2015,60(1):88.
[65] Istok J D,Senko J M,Krumholz L R,Watson D,Bogle M A,Peacock A,Chang Y J,White D C.In situ bioreduction of technetium and uranium in a nitrate-contaminated aquifer[J].Environmental Science&Technology,2004,38(2):468.
[66] Gu B,Wu W,Ginder-Vogel M A,Yan H,Fields MW,Zhou J,Fendorf S,Criddle C S,Jardine P M.Bioreduction of uranium in a contaminated soil column[J].Environmental Science&Technology,2005,39(13):4841.
[67] Wu W,Gu B,Fields M W,Gentile M,Ku Y,Yan H,Tiquias S,Yan T,Nyman J,Zhou J,Jardine P M,Criddle C S.Uranium(VI)reduction by denitrifying biomass[J].Bioremediation Journal,2005,9(1):49.
[68] Fredrickson J K,Zachara J M,Kennedy D W,Duff MC,Gorby Y A,Li S W,Krupka K M.Reduction of U(VI)in goethite(a-Fe OOH)suspensions by a dissimilatory metal-reducing bacterium[J].Geochimica et Cosmochimica Acta,2000,64(18):3085.
[69] Stewart B D,Amos R T,Fendorf S.Effect of uranium(VI)speciation on simultaneous microbial reduction of uranium(VI)and iron(III)[J].Journal of Environmental Quality,2011,40(1):90.
[70] Fredrickson J K,Zachara J M,Kennedy D W,Liu C,Duff M C,Hunter D B,Dohnalkova A.Influence of Mn oxides on the reduction of uranium(VI)by the metal-reducing bacterium Shewanella putrefaciens[J].Geochimica et Cosmochimica Acta,2002,66(18):3247.
[71] Brooks S C,Fredrickson J K,Carroll S L,Kennedy DW,Zachara J M,Plymale A E,Kelly S D,Kemner KM,Fendorf S.Inhibition of bacterial U(VI)reduction by calcium[J].Environmental Science&Technology,2003,37(9):1850.
[72] Madden A S,Palumbo A V,Ravel B,Vishnivetskaya TA,Phelps T J,Schadt C W,Brandt C C.Donor-dependent extent of uranium reduction for bioremediation of contaminated sediment microcosms[J].Journal of Environment Quality,2009,38(1):53.
[73] Sheng L,Fein J B.Uranium reduction by shewanella oneidensis MR-1 as a function of Na HCO3 concentration:surface complexation control of reduction kinetics[J].Environmental Science&Technology,2014,48(7):3768.
[74] Zheng X Y,Shen Y H,Wang X Y,Wang T S.Effect of p H on uranium(VI)biosorption and biomineralization by Saccharomyces cerevisiae[J].Chemosphere,2018,203:109.