
Contact reactive brazing of Al alloy/Cu/stainless steel joints and dissolution behaviors of interlayer
WU Ming-fang1, 2, SI Nai-chao1, CHEN Jian2
1. School of Material Science and Engineering, Jiangsu University, Zhenjiang 212013, China;
2. School of Material Science and Engineering, Jiangsu University of Science and Technology,
Zhenjiang 212003, China
Received 30 June 2010; accepted 24 November 2010
Abstract: Contact reactive brazing of 6063 Al alloy and 1Cr18Ni9Ti stainless steel was researched by using Cu as interlayer. Effect of brazing time on microstructure of the joints, as well as the dissolution behaviors of Cu interlayer was analyzed. The results show that the product of reaction zone near 1Cr18Ni9Ti is composed of Fe2Al5, FeAl3 intermetallic compound (IMC), and Cu-Al IMC; the near by area is composed of Al-Cu eutectic structure with Al (Cu) solid solution. With increasing the brazing time, the thickness of IMC layer at the interface increases, while the width of Al-Cu eutectic structure with Al(Cu) solution decreases. Calculation shows the dissolution rate of Cu interlayer is very fast. The complete dissolution time is about 0.47 s for Cu interlayer with 10 μm in thickness used in this study.
Key words: Al alloy; stainless steel; contact reactive brazing; microstructure; dissolution of interlayer
1 Introduction
Contact reactive brazing (CRB) is a kind of liquid diffusion welding process. After the heating temperature reaches the eutectic temperature, eutectic liquid will be formed between the base metal and reactive material through contact reaction, and then the liquid wets and spreads on the surface of base metal to form joints[1-3]. CRB has been widely used because of it’s low heating temperature and no requiring the brazing flux[4-6], mainly in the joining between aluminum and steel[7-8].
From Al-Cu binary phase diagram, it is known that there is a eutectic reaction on Al-Cu with the composition of 66.8% Cu (mass fraction) and 33.2% Al. The contact reactive brazing between Al and Cu has been researched using this eutectic reaction[9-11], which indicates that the interdiffusion of Al and Cu atoms was generated by chemical potential, and Cu atom diffused preferentially into Al alloy/oxidation film interface. After the temperature reached 548 °C, Al-Cu eutectic reaction occurred to form Al-Cu eutectic liquid at the interface. Therefore, the real reactive spreading process of Cu on the Al surface is that Cu atoms diffuse into the Al alloy/oxide film interface preferentially and participate in the eutectic reaction continuously. Also, the growth on the intermetallic compounds at the interface is diffusion controlled[12].
In this work, the contact reactive brazing between 6063Al alloy and 1Cr18Ni9Ti stainless steel was researched with Cu as the contact reactive interlayer. Due to the compact oxidation layer at 6063 Al alloy surface, the key factor using contact reactive brazing between Al alloy and stainless steel is whether the eutectic liquid will be produced between 6063 Al alloy and Cu. Also, the effect of brazing time was researched and the dissolution rate of Cu interlayer was calculated.
2 Experimental
The base materials used in this study were commercial 6063 Al alloy and 1Cr18Ni9Ti stainless steel with dimensions of 15 mm×15 mm×2 mm. Cu with 10 ?m in thickness was used as the reactive interlayer between them. The materials were polished to 0.05 mm with diamond compounds and then colloidal silica solution, and then assembled, as shown in Fig.1. The load with the pressure of 0.1 MPa was applied to the specimen for contact reactive brazing.
To minimize the oxidation on the 6063 Al alloy, the contact reactive brazing on the specimen was finished in the vacuum chamber. The brazing temperature was selected as 570 °C according to the Al-Cu eutectic temperature (548 °C) and liquid solid phase line temperature of Al alloy (615-655 °C). The holding time for contact reactive brazing varied from 1 to 60 min. The microstructure at the interface and the solution behavior of Cu interlayer were characterized using scanning electron microscopy (SEM) and electron probe micro- analysis (EPMA). The phase structure at the interface was characterized using X-ray diffractometry (XRD).

Fig.1 Schematic diagram of specimen test
3 Results and discussion
3.1 Effect of brazing time on microstructure of joints
In this work, the contact reactive brazing temperature was selected as 570 °C, while the effect of brazing time was researched. Figure 2(a) shows the typical SEM secondary electron image of the joint in which the holding time was 20 min. Figure 2(b) is the enlarged area of zone A in Fig.2(a). In Fig.2, it seems that the whole reaction zone at the interface during contact reactive brazing includes the reactive layers near stainless steel side and the Al-Cu eutectic structure near Al side.
From Fig.2(b), it is easy to find two reactive layers with about 10 ?m in thickness and each formed at the interface between stainless steel and the eutectic structure, marked as zone D and zone E. The wetting and spreading behavior of Al-Cu eutectic liquid formed by contact reactive brazing on Al alloy and stainless steel surface was excellent, and the microstructures of the interfaces were compact without any micro-defects.
To understand the dissolution behavior of Cu atoms during the brazing process, the EPMA results for each characteristic point in Fig.2(b) are listed in Table 1. The results shows that the reactive zone could be divided into four different regions, namely, dark grey particle region B, light grey large block region C, and interfacial reactive layers D and E. Region B mainly contained Al and Cu, with the compositions of 67.46% Al (molar fraction) and 32.54% Cu. Region C contained Al. In region D near steel side, the concentrations of Al and Fe were up to 67.63% and 23.59%, respectively, with minor Ni and Cr left. Region E mainly contained Al, Fe and Cu elements. The phase structure of interface reactive layer D at the side of stainless steel was analyzed with XRD, as shown in Fig.3. It is shown that the reactive layer D was composed of Fe2Al5 and FeAl3 intermetallic compounds (IMCs). Based on XRD result, microstructure, EPMA results, and Al-Cu and Al-Fe binary phase diagrams, the microstructure at the interface was composed of Fe2Al5, FeAl3 IMC, Cu-Al IMC, and Al-Cu eutectic with Al solid solution from stainless steel side to Al alloy side.

Fig.2 SEM images on specimen brazed at 570 °C with time of 20 min (a), and enlarged microstructure of zone A (b)
Energy spectrum line scanning analyses at the joint interface are shown in Fig.4. The results demonstrated the diffusion behaviors of Fe and Al elements in 1Cr18Ni9Ti and 6063 Al alloy respectively. The diffusion range of Fe elements was mainly in two IMC layers, while Al reached the stainless steel and reacted with Cu and Fe elements to produce Fe-Al and Cu-Al IMCs.
Table 1 EMPA results of regions in Fig.2(b)

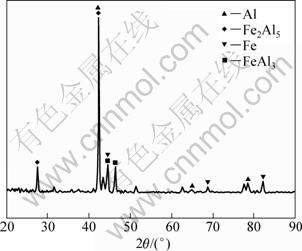
Fig.3 XRD result of region D marked in Fig.2(b)
Concentration distribution of Al element was non-uniform, the content of Al element at peak of line scanning curve was consistent with that of base metal 6063 Al alloy, which presented the large block Al solid solution, while the low point corresponded to Al-Cu eutectic structure.

Fig.4 SEM image of joints (a) and EPMA test results of main elements (b)
Based on the above qualitative and quantitative analyses, as the heating temperature was up to 570 °C, large quantity of Al-Cu eutectic liquid was produced at the interface of 6063 Al alloy and Cu interlayer during contact reactive brazing, which played an active role in breaking oxidation of base metal. Al-Cu eutectic liquid reacted with stainless steel to produce Fe-Al IMCs, and diffused into Al alloy to produce Al-Cu eutectic with Al solid solution. In the holding stage at 570 °C, because diffusion coefficient of Cu in Al was 2000 times as quick as that of Al in Cu [12], Cu atoms diffused into the base metal, which caused the rising of melting point of the liquid phase and then the isothermal solidification.
Figure 5 shows the effect of contact reactive brazing time on the SEM microstructure of 6063 Al alloy/ Cu plating layer/1Cr18Ni9Ti joint at the brazing temperature of 570 °C. With increasing the holding time, the thickness of IMC layer increased, while the width of Al-Cu eutectic structure decreased.

Fig.5 Effect of holding time on microstructure at 570 °C: (a) 30 min; (b) 60 min
3.2 Dissolution rate of Cu interlayer
To decide the suitable thickness of the interlayer and the contact reactive brazing parameters, it is very necessary to know the dissolution rate of Cu interlayer.
The dissolution of Cu interlayer is a diffusion controlled process. Cu concentration gradient would be formed during heating, which provides a precondition for the eutectic reaction between Cu and Al. Once the temperature was up to or over the temperature, the eutectic reaction was triggered to produce the contact reactive liquid. With the nucleation of eutectic liquid at the Cu/6063 Al interface, Cu atoms diffused into it quickly, thus, the thickness of the liquid phase would increase rapidly. The solid/liquid interface moved on until the complete dissolution of Cu interlayer. This dissolution was controlled by Cu diffusion in the Al-Cu liquid phase because the diffusion rate of atom in liquid phase was far quicker than in solid phase. ISAAC et al[13] pointed out that the dissolution of Cu interlayer followed the Fick’s second law in Ag/Cu/Ag contact melting system. In this work, the dissolution of Cu also follows the Fick’s equation:
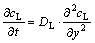 (1)
(1)
where cL is the concentration of Cu in the liquid; DL is diffusion coefficient; and y is the distance across the contact reactive region (the initial position of the solid/liquid interface is chosen as the origin of the y axis). The well-known solution for Eq.(1) is
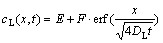 (2)
(2)
where E and F are constants, determined by the specific boundary conditions. Eq.(2) implies that the interface displacement obeys a general square root law[14-15]:
 (3)
(3)
where Y(t) is solid/liquid interface position at time t, and K1 is a constant dictated by Al-Cu binary phase diagram. When the width of the liquid zone reached the initial width W0, the time t was required for complete dissolution.
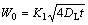 (4)
(4)
 (5)
(5)
Based on above analysis, the dissolution time was correlated with the initial width W0 of interlayer, diffusion coefficient of Cu in Al-Cu liquid and constant K1. DL was determined by[16]
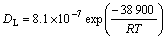 (6)
(6)
where R is molar gas constant and T is temperature.
In this work, W0 is 10 μm, T is 843 K, and K1 is 0.13 determined by Al-Cu binary phase diagram. The dissolution of such Cu layer will be 0.47 s.
By differentiating Eq.(3), the dissolution rate of Cu interlayer would be written as
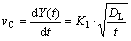 (7)
(7)
where vC is the dissolution rate of Cu interlayer.
The dissolution rate of Cu (vC) is 3.364 47×10-4 m/s in this work. Thus, for the contact reactive brazing of 6063Al alloy/Cu interlayer/1Cr18Ni9Ti stainless steel, the dissolution process of Cu interlayer was an instantaneous process. However, it was difficult to observe the complete liquidation of interlayer in test. According to Eq.(5), the relationship between the initial thickness and dissolution time at temperatures of 843 K and 873 K were obtained, as shown in Fig.6.
From Fig.6, the time required for the complete dissolution of Cu interlayer increased with increasing the thickness of interlayer. Also, it seems that the effect of temperature on dissolution rate is negligible.
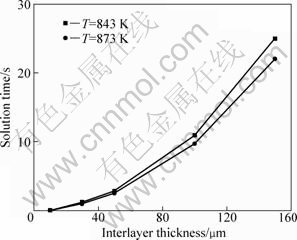
Fig.6 Relationship between thickness and complete solution time of Cu interlayer
4 Conclusions
1) Interfacial microstructure shows that the reaction layers near 1Cr18Ni9Ti side are composed of Fe2Al5, FeAl3 and Cu-Al IMCs, while the structure near Al alloy is Cu-Al eutectic structure with Al solid solution.
2) With increasing the brazing time, the thickness of IMC layer increases, while the width of eutectic structure decreases at the interface.
3) Calculation shows the dissolution of Cu interlayer is a very short process. The dissolution time is only about 0.47 s for a 10 μm-Cu interlayer.
References
[1] LIU L M, TAN J H, LIU X J. Reactive brazing of Al alloy to Mg alloy using zinc-based brazing alloy [J]. Mater Lett, 2007, 61: 2373-2377.
[2] L? Xue-qin, YANG Shang-lei, WU Yi-xiong. Transitional layers brazing of Al-alloy and stainless steel [J]. Transactions of the China Welding Institution, 2004, 25(1): 95-102. (in Chinese)
[3] DONG Zhan-gui, QIAN Yi-yu, MA Xin, SHI Su-qin. Law for selecting interlayer in contact reactive brazing [J]. The Chinese Journal of Nonferrous Metal, 2002, 12(2): 241-244. (in Chinese)
[4] YIN Shi-qiang, QIU Xiao-ming, SUN Da-qian. Effect of compound layer on process and joint properties of Al contact eutectic reaction brazing [J]. The Chinese Journal of Nonferrous Metal, 2002, 12(2): 323-326. (in Chinese)
[5] HE Peng, FENG Ji-cai, QIAN Yi-yu. Resolve of fragile and defect problem for a new contact reaction brazing aluminum to stainless steel [J]. Materials Science and Technology, 2005, 13(1): 82-85. (in Chinese)
[6] LIU S, SUZUMURA A, IKESHOJI T T, YAMAZAKI T. Brazing of stainless steel to various aluminum alloys in air [J]. JSME International Journal Series A, 2005, 48(4): 420-425
[7] IKESHOJI T T, LIU S, SUZUMURA A, YAMAZAKI T. Effect of isothermal solidification behavior of Al-Si brazing filler layer on the braze ability of Al-alloy to stainless steel in the air [J]. Quarterly Journal of the Japan Welding Society, 2006, 24(4): 362-367. (in Japanese)
[8] SONG W, SAIDA K, ANDO A, NISHIMOTO K. Brazability of aluminum alloy to steels using aluminum filler metal dissimilar laser brazing of aluminum alloy and steels (Report 1) [J]. Quarterly Journal of the Japan Welding Society, 2004, 22(2): 315-322. (in Japanese)
[9] SAIDA K, KURODA S, NISHIMOTO K. Improvement in interfacial reactivity of A6061 by eutectic reaction between A6061 and pre-coated metals [J]. Quarterly Journal of the Japan Welding Society, 2005, 23(3): 405-411. (in Japanese)
[10] NISHIMOTO K, SAIDO K, KURODA S. Removing of surface oxide film of SUS316 and improvement of diffusion bondability of A6061 aluminum alloy to SUS316 stainless steel using surface-activated pre-coating technique [J]. Quarterly Journal of the Japan Welding Society, 2000, 18(4): 563-571. (in Japanese)
[11] CHEN Ding-hua, QIAN Yi-yu, LIAN Yan. The mechanism of flow of filler metal into brazing space: Investigating the flow characteristics of liquid filler metal in an unparallel clearance [J]. Transactions of the China Welding Institution, 1981, 2(1): 1-8. (in Chinese)
[12] VIKAS J, SRIVASTAVA V C, ARPAN D, GHOSH R N. Reactive diffusion in the roll bonded iron-aluminum system [J]. Mater Lett, 2006, 60: 1758-1761.
[13] ISAAC T, DOLLAR M, MASSALSKI T B. A study of transient liquid phase bonding process applied to a Ag/Cu/Ag sandwich joint [J]. Metall Mater Trans A, 1988, 19(3): 675-686.
[14] VIKAS J, SRIVASTAVA V C. Growth of intermetallic layer at roll bonded IF-steel/aluminum interface [J]. Journal of Materials Processing Technology, 2008, 195: 88-93.
[15] KOBAYASHI S, YAKOU T. Control of intermetallic compound layers at interface between steel and aluminum by diffusion-treatment [J]. Mater Sci Eng A, 2002, 23(6): 44-53.
[16] DU Y, CHANG Y A, HUANG B Y. Diffusion coefficients of some solutes in fcc and liquid Al: Critical evaluation and correlation [J]. Mater Sci Eng A, 2003, 363: 140-151.
铝合金/Cu/不锈钢接触反应钎焊及中间层溶解行为
吴铭方1, 2,司乃潮1,陈 健2
1. 江苏大学 材料科学与工程学院,镇江 212013;
2. 江苏科技大学 材料科学与工程学院,镇江 212003
摘 要:以Cu作为接触反应材料连接6063铝合金与1Cr18Ni9Ti不锈钢,探讨焊接工艺参数对接头组织的影响规律,分析中间反应层Cu的溶解特性。结果表明:在1Cr18Ni9Ti不锈钢一侧界面反应层由Fe2Al5、FeAl3金属间化合物和Cu-Al金属间化合物构成,与之相邻区域主要含Cu-Al金属间化合物,焊缝组织由Al-Cu共晶及大块状的Al固溶体组成;随着保温时间的延长,焊缝组织最为显著的变化是在1Cr18Ni9Ti不锈钢一侧界面的金属间化合物层厚度增加,共晶组织宽度逐渐减小;中间反应层Cu的溶解速度非常迅速,是以秒为计量单位的快速过程,厚度为10 μm的Cu溶解时间仅为0.47 s。
关键词:铝合金;不锈钢;接触反应钎焊;组织;中间层溶解
(Edited by YANG Hua)
Corresponding author: WU Ming-fang; Tel: +86-511-84401185; E-mail: Wu_mingfang@163.com
DOI: 10.1016/S1003-6326(11)60818-2