
Activated nitrogen-enriched carbon/carbon aerogel nanocomposites for supercapacitor applications
QIN Chuan-li(秦川丽)1,2, LU Xing(卢 幸)2, YIN Ge-ping(尹鸽平)1, BAI Xu-duo(白续铎)2, JIN Zheng(金 政)2
1. Research Station on Material Science and Engineering for Postoral Fellows, School of Materials Science and Engineering, Harbin Institute of Technology, Harbin 150001, China;
2. Key Laboratory of Functional Polymer Materials, School of Chemistry and Material Sciences,
Heilongjiang University, Harbin, 150080, China
Received 10 August 2009; accepted 15 September 2009
Abstract: Activated nitrogen-enriched carbon/carbon aerogel nanocomposites (ANC/ACA) were prepared by synthesis of melamine resin/carbon aerogel composites, carbonization and KOH activation. Novel asymmetric supercapacitors consisting of Ni(OH)2/Co(OH)2 as anode and ANC/ACA with different composite ratios as cathode were assembled. The influence of composite ratio on electrochemical performances of materials was detected by cycle voltammetry (CV) and galvanostatic charge/discharge methods. The results of XPS and SEM show that N atoms exist in the ANC/ACA and ANC/ACA shows nanometer and honeycomb structure with more pores. When the composite ratio of ANC/ACA is 12?1, the ANC/ACA shows the highest Cp1 (312.8 F/g) vs 103.4 F/g of ACA and 230.1 F/g of ANC. And the optimal asymmetric supercapacitor with the ANC/ACA as cathode also shows the best electrochemical performances. The optimal supercapacitor is stable over 100 cycles. When the current density is 50 mA/cm2, the Cp2, Ep and P of the optimal supercapacitor are still 57.3 F/g, 9.0 W?h/g and 1 302.1W/kg, respectively.
Key words: supercapacitor; melamine; carbon aerogels; nanocomposites; nitrogen
1 Introduction
Supercapacitors have received considerable amount of attention over the last few years because of their use in high specific power storage devices[1-4]. Now, kinds of carbon materials were widely studied as electrode materials for supercapacitors[5-8], but their capacitance (Cp) is not high because of the limit of the carbonic structure. In order to improve their Cp, in recent studies the nitrogen functional groups were introduced into carbon materials, and these electrochemically active centers will contribute to the overall Cp with pseudocapacitance that generally originates from the faradaic interactions between the ions of electrolytes and carbon electrode surface[9-13]. LOTA et al[14] reported on capacitors, of which electrodes were prepared by pyrolysis of nitrogen-containing polymers blended with coal tar pitch and subsequent activation. JUREWICZ et al[15] also observed the improved capacitive behavior of various carbon materials subjected to the ammoxidation process, i.e. a treatment of nitrogen-free carbons with an ammonia/air mixture, in which the nitrogen groups were introduced mainly to the surface of carbon at a concentration of about 2% (mass fraction).
Now, the obtained nitrogen-enriched carbon (NC) has relatively low nitrogen content. How to take full advantage of nitrogen atoms of NC and improve its Cp is very significant. In this work, carbon aerogels(CA) with high specific surface and high conductivity were introduced as the carrier of NC and novel activated nitrogen-enriched carbon/carbon aerogel nanocomposites (ANC/ACA) with different composite ratios were prepared. Novel asymmetric supercapacitors consisting of Ni(OH)2/Co(OH)2 as anode and ANC/ACA as cathode were assembled. The electrochemical performances of ANC/ACA and their supercapacitors were detected in order to study the possibility of ANC/ACA as electrode materials of supercapacitors.
2 Experimental
2.1 Preparation of activated carbon aerogels(ACA), ANC and ANC/ACA nanocomposites
Resorcinol (R), formaldehyde (F), distilled water, NaCO3 were mixed at certain mass ratio, then the mixture was cured at room temperature for 24 h and at 85 ℃ for 96 h. The solvent of got gels was exchanged with the acetone and then gels were dried at ambient temperature and pressure. Subsequently, the resultant RF aerogels were heated (5 ℃/min) to 900 ℃ and carbonized at 900 ℃ for 3 h and then cooled in a nitrogen atmosphere to form CA.
Melamine, distilled water, formaldehyde and NaCO3 were mixed at a certain mass ratio. The mixture was heated to 80 ℃ for 0.5 h after being stirred and cured for 24 h at room temperature. The white precipitates were filtered, washed and dried at 100 ℃ to form the melamine resin. The melamine resin and CA were evenly mixed in the distilled water by ultrasonic wave for about 20 min. The obtained melamine resin and melamine resin /CA composites were heated (10 ℃/min) to 750 ℃ for 1 h and carbonized, then cooled in a nitrogen atmosphere to form NC and NC/CA.
Finally, activation was applied to obtain CA, NC and NC/CA. CA, NC and NC/CA were respectively mixed with KOH at a mass ratio of 5?1 and some distilled water was added into the mixture slowly. After being heated at 120 ℃ for 48 h, the ground mixture was heated (10 ℃/min) to 800 ℃ and was activated at 800 ℃ for 1 h in a nitrogen atmosphere. The obtained ACA, ANC and ANC/ACA were washed and dried. ANC/ACA nanocomposites with different composites ratios (mass ratios) were obtained by changing the mass ratio of melamine resin/CA composites.
2.2 Preparation of Ni(OH)2/Co(OH)2 composites
NiCl2・6H2O and Co(NO3)2・6H2O at certain mole ratio were solved in the distilled water at room temperature. The calculated amount of NaOH solution was added slowly into the mixed solution until pH=8 to produce precipitates. The blackish green precipitates were filtrated, washed and dried at 60 ℃ to obtain Ni(OH)2/Co(OH)2 composites.
2.3 Preparation of asymmetric supercapacitors
Ni(OH)2/Co(OH)2 (65%, mass fraction), graphite (30%) and polytetrafluoroethylene binder (PTFE, 5%) and some water were mixed to form the active paste. Then the paste was coated on the Ni-foam current collector (40 mm×10 mm). Finally, the anode was obtained by drying the sample at 60 ℃ for 24 h and pressing it with double roller machine. And the mass of active substances on one electrode was (80±4) mg.
ANC/ACA(ANC or ACA), PTFE and water were mixed at the mass ratio of 8?1?1. Then the cathode (40 mm×10 mm) was prepared by the same method as anode.
Anode, cathode and battery separator were immersed in aqueous solution of 6 mol/L KOH. An asymmetric supercapacitor was assembled by putting battery separator between two electrodes.
3 Measurements
3.1 X-ray photoelectron spectroscopy (XPS) test
XPS analysis was performed using an ESCALAB 250 (Thermo VG Scientific) equipped with an Al Kα monochromatic source (120 μm spot).
3.2 Scanning electron microscopy (SEM) test
The microstructure was observed by scanning electronic microscope (MX2600FE).
3.3 Cyclic voltammetry(CV) test
A type of LK98BII Computer Electroanalytical System was used for CV test. The platinum electrode was used as the auxiliary electrode, HgO/Hg electrode as the reference electrode and the measured electrode (10 mm×10 mm) as working electrode, respectively. The measurement was carried out in 6 mol/L KOH solution. The specific capacitance of electrode materials is calculated using the following equation.
Cp1=(|Ia|+ |Ic|) /(2×m×v) (1)
where Cp1 is the specific capacitance of electrode materials, F/g; Ia is the current of oxidation peak, A; Ic is the current of reduction peak, A; m is the mass of active substances on the measured electrode, g; v is the scanning rate of voltage, V/s.
3.4 Galvanostatic charge/discharge test
The galvanostatic charge/discharge test of asymmetric supercapacitors was performed with a PCBT-138-8D-A Battery Programmed Test Instrument (Wuhan Lixing Instruments Company). The Cp is calculated using the following equation.
Cp2=I×Δt/(ΔV×m) (2)
where Cp2 is the specific capacitance of supercapacitor, F/g; I is the discharge current, A; Δt is the time difference in discharge process, s; ΔV is the potential difference in discharge process, V; and m is the mass of active substances on all electrodes, g.
The Ep and P of supercapacitors are calculated by
Ep=ΔV×I×Δt/(7.2×m) (3)
P=ΔV×I/m (4)
where Ep is the specific energy of supercapacitors, W?h/kg; P is the specific power of supercapacitors, kW/kg; Δt is the time difference in discharge process, s.
3.5 Cycle performance test
The cycle performance test was performed by the PCBT-138-8D-A Battery Programmed Test Instrument and using the computer to control galvanostatic charge and discharge cycles. Then the changes of Cp with the cycle numbers were analyzed.
4 Results and discussion
4.1 XPS analysis
The results of XPS are shown in Table 1. As shown in Table1, N atoms do not exist in the CA, but they exist in the ANC and ANC/ACA and the content of N atoms is 2.22% and 2.12% respectively which verifies that N atoms are introduced into carbon materials by the method.
Table1 Results of XPS test
4.2 SEM analysis
The SEM photographs of ANC, ACA and ANC/ACA are shown in Fig.1. As shown in Fig.1, the ANC shows nanometer and honeycomb structure with more pores. ACA shows nanometer and near-spherical structure whose surface is not smooth due to KOH corrosion. The ANC/ACA shows the same nanometer structure with more pores as that of ACA. It seems that the pores result from the removal of impregnated KOH and KOH-etched compounds, leaving the space previously occupied by the compounds. The well-developed pore structure is beneficial for the adsorption of electrolyte and the improvement of Cp.
4.3 CV analysis
The CV results of ANC, ACA and ANC/ACA with different composite ratios tested in the potential range of -1 to 0 V are shown in Table 2 (the scanning rate of voltage was 1 mV/s).
As shown in Table 2, the Cp1 of supercapacitors with ANC/ACA and ANC as cathode is greater than that of supercapacitor with ACA as cathode. This is due to the fact that nitrogen atoms of ANC/ACA and ANC contribute to the overall Cp with pseudocapacitance that generally originates from the faradaic interactions between potassium cations and the nitrogen atoms of carbon. When the composite ratio of ANC/ACA is 12?1, the ANC/ACA shows the highest Cp1 (312.8 F/g) vs 103.4F/g of ACA and 230.1 F/g of ANC. This is due to the fact that, on one hand, pseudocapacitance of ANC can be utilized well because ANC can be dispersed well by the high specific surface of carbon aerogels which act as the carrier of ANC and a kind of charge storage material; on the other hand, the introduction of carbon aerogels with high conductivity makes the conductivity of the cathode material improved which improves its Cp.
Table 2 Cp1 of different electrode materials tested by cyclic voltammetry


Fig.1 SEM images of ANC (a), ACA (b) and ANC/ACA (c)
4.4 Galvanostatic charge/discharge analysis of supercapacitors
The galvanostatic charge/discharge data of asymmetric supercapacitors consisting of Ni(OH)2/ Co(OH)2 as anode and ANC/ACA with different composite ratios as cathode are shown in Table 3 (the current density was 7.5 mA/cm2). As shown in Table 3, the Cp2, Ep and P of the optimal asymmetric supercapacitor (the ANC/ACA with composite ratio of 12?1 was used as cathode) are the greatest, which is accordance with the result of Table 2.
Table 3 Electrochemical performances of asymmetric super- capacitors with different composite ratios of ANC/ACA as cathodes

The galvanostatic discharge curves of the optimal supercapacitor at different current densities are shown in Fig.2. It indicates that the supercapacitor shows superior quick dischargeability at high current density. The test data are given in Table 4. The Cp2 and Ep of the supercapacitor decrease with the increase of discharge current density, while the P increases. The reason why Cp2 and Ep decreases obviously with the increase of current density is that, when charge or discharge is carried out at high current density and charge or discharge is finished in a short time, there is no enough time for ions to enter the interior minipores of electrode materials when ions transfer from the solution to the electrode surface, so some surface areas and function of electrode materials cannot be utilized well. When the current density is 50 mA/cm2, the Cp2, Ep and P of the supercapacitor is still 57.3 F/g, 9.0 W?h/kg and 1 302.1 W/kg, and the comprehensive properties of the supercapacitor are excellent.
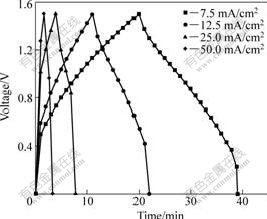
Fig.2 Galvanostatic discharge curves of optimal supercapacitor at different current densities
Table 4 Galvanostatic discharge data of optimal supercapacitor at different current densities

4.5 Cycle performance analysis
The cyclic performance of the optimal asymmetric supercapacitor was further examined by galvanostatic charge/discharge method and the data obtained during 100 cycles are shown in Fig.3.
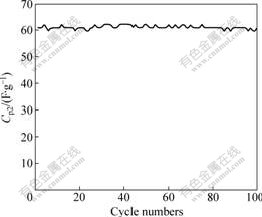
Fig.3 Cyclic performance of optimal asymmetric supercapacitor
As shown in Fig.3, after 100 cycles the Cp2 is invariant and the asymmetric supercapacitor is stable over 100 cycles. The ANC/ACA is a kind of suitable electrode material of supercapacitors.
5 Conclusions
1) N atoms exist in the ANC/ACA and ANC/ACA shows nanometer and honeycomb structure with more pores.
2) When the composite ratio of ANC/ACA is 12?1, the ANC/ACA shows the highest Cp1 (312.8 F/g) vs 103.4 F/g of ACA and 230.1 F/g of ANC and the optimal asymmetric supercapacitors with the ANC/ACA as cathode also show the best electrochemical performances (Cp2, Ep and P is respectively 99.7 F/g, 15.6 W?h/kg, 195.3 W/kg). When the current density is increased to 50 mA/cm2, Cp2, Ep and P of the optimal supercapacitor is still 57.3 F/g, 9.0 W?h/kg, 1 302.1 W/kg.
3) The optimal asymmetric supercapacitor is stable over 100 cycles and its comprehensive properties are excellent. The synthetical ANC/ACA is a kind of suitable electrode material of supercapacitors.
References
[1] LOKHANDE C D, GUJAR T P, SHINDE V R, RAJARAM S M, HAN S H. Electrochemical supercapacitor application of pervoskite thin films [J]. Electrochem Commun, 2007, 9(7): 1805-1809.
[2] PHATIPHAT T, STEPHANE R, BERNARD D. Energy management of fuel cell/battery/supercapacitor hybrid power source for vehicle applications [J]. J Power Sources, 2009, 193(1): 376-385.
[3] WEI Sun, CHEN Xu-yuan. Preparation and characterization of polypyrrole films for three-dimensional micro supercapacitor [J]. J Power Sources, 2009, 193(2): 924-929.
[4] DUFFY N W, BALDSING W, PANDOLFO A G. The nickel-carbon asymmetric supercapacitor―Performance, energy density and electrode mass ratios [J]. Electrochim Acta, 2008, 54(2): 535-539.
[5] JISHA M R, HWANG Y J, SHIN J S, NAHM K S, KUMAR T P, KARTHIKEYAN K, DHANIKAIVELU N, KALPANA D, RENGANATHAN N G, STEPHAN A M. Electrochemical characterization of supercapacitors based on carbons derived from coffee shells [J]. Mater Chem Phys, 2009, 115: 33-39.
[6] FUERTES A B, LOTA G, CENTENO T A, FRACKOWIAK E. Templated mesoporous carbons for supercapacitor application [J]. Electrochim Acta, 2005, 50(14): 2799-2805.
[7] LIU N, ZHANG S T, FU R W, DRESSELHAUS M S, DRESSELHAUS G. Fabrication and structure of carbon aerogel spheres prepared by inverse suspension/emulsion polymerization and ambient pressure drying [J]. J Appl Polym Sci, 2007, 104(5): 2849-2855.
[8] RUCH P W, K?TZ R, WOKAUN A. Electrochemical characterization of single-walled carbon nanotubes for electrochemical double layer capacitors using non-aqueous electrolyte [J]. Electrochim Acta, 2009, 54(19): 4451-4458.
[9] LOTA G, LOTA K, FRACKOWIAK E. Nanotubes based composites rich in nitrogen for supercapacitor application [J]. Electrochem Commun, 2007, 9: 1828-1832.
[10] KIM N D, KIM W, JOO J B, OH S, KIM P, KIM Y, YI J. Electrochemical capacitor performance of N-doped mesoporous carbons prepared by ammoxidation [J]. J Power Sources, 2008, 180: 671-675.
[11] DENISA H J, MYKOLA S, GAO QING-LU, TERESA J B. Combined effect of nitrogen- and oxygen-containing functional groups of microporous activated carbon on its electrochemical performance in supercapacitors [J]. Adv Funct Mater, 2009, 19(3): 438-447.
[12] DENISA H, MASAYA K, HIROAKI H. Electrochemical performance of nitrogen-enriched carbons in aqueous and non-aqueous supercapacitors [J]. Chem Mater, 2006, 18 (9): 2318-2326.
[13] LI Wen-rong, CHEN De-hong, LI Zheng, SHI Yi-feng, WAN Ying, WANG Guan, JIANG Zhi-yu, ZHAO Dong-yuan. Nitrogen-containing carbon spheres with very large uniform mesopores: The superior electrode materials for EDLC in organic electrolyte [J]. Carbon, 2007, 45(9): 1757-1763.
[14] LOTA G, GRZYB B, MACHNIKOWSKA H, MACHNIKOWKI J, FRACKOWIAK E. Effect of nitrogen in carbon electrode on the supercapacitor performance [J]. Chem Phys Lett, 2005, 404: 53-58.
[15] JUREWICZ K, BABEL K, ZIOLKOWSKI A, WACHOWSKA H. Capacitance behaviour of the ammoxidised coal [J]. J Phys Chem Solids, 2004, 65(2/3): 269-273.
(Edited by YANG You-ping)
Foundation item: Project (LBH-Z07090) supported by the Postdoctoral Foundation of Heilongjiang Province, China; Project(11541267) supported by the Education Department of Heilongjiang Province, China; Project(QL200422) supported by Young Science Foundation of Heilongjiang University, China
Corresponding author: QIN Chuan-li; Tel: +86-451-86608131; E-mail: chuanliqin@163.com