稀有金属 2016,40(01),43-47 DOI:10.13373/j.cnki.cjrm.2016.01.008
N,S共掺杂五氧化二铌的光催化性能研究
倪东宁 姜恒 宫红 苏婷婷
辽宁石油化工大学化学与材料科学学院
摘 要:
以硫脲(TU)和草酸铌为原料,采用固相法制备Nb2O5/TU质量比为1∶1,1∶2,1∶3的N,S掺杂五氧化二铌光催化剂。以X射线衍射仪(XRD)、扫描电镜(SEM)、X射线荧光光谱(XRF)、X射线光电子能谱(XPS)和紫外-可见漫反射(UV-Vis)对不同光催化剂进行表征,研究了在紫外光照射条件下对罗丹明B的光催化降解效果。由XRD结果可知N,S掺杂没有改变Nb2O5的晶型结构,Nb2O5和N,S掺杂Nb2O5均以六方晶相结构存在。SEM结果表明,N,S掺杂Nb2O5表面出现孔隙,且催化剂粒径变小,当Nb2O5/TU质量比为1∶1时,催化剂表面孔隙较大且排列规则,在光催化过程中增大了与罗丹明B的反应面积,有助于光催化效果提高。XRF和XPS的表征结果证明,N,S掺杂进入Nb2O5晶体结构中,其中S以S6+形式存在,可看作是以SO42-形式吸附在Nb2O5表面,部分S6+取代了Nb5+,而N取代O存在于O-NbN环境中。UV-Vis结果显示N,S掺杂Nb2O5带隙变窄而出现红移现象,拓宽了光的吸收范围,这有利于光催化效果的提高。光催化效果表明,在紫外光照射条件下降解罗丹明B,N,S掺杂Nb2O5的催化效果比Nb2O5提高约30%,尤其是当Nb2O5/TU质量比为1∶1时制备的光催化剂,3 h对罗丹明B降解效果为92%。
关键词:
五氧化二铌;N,S掺杂;光催化;
中图分类号: O643.36
作者简介:倪东宁(1989-),女,辽宁营口人,硕士研究生,研究方向:绿色化学与催化;E-mail:ndn1989@126.com;;姜恒,教授;电话:024-56860790;E-mail:hjiang78@hotmail.com;
收稿日期:2014-05-29
基金:辽宁省自然科学基金项目(201202122)资助;
Photocatalytic Activity of N,S-Doped Niobium Pentoxide
Ni Dongning Jiang Heng Gong Hong Su Tingting
School of Chemistry and Materials Science,Liaoning Shihua University
Abstract:
N,S-doped Nb2O5 photocatalysts were prepared by solid-state method using thiourea( TU) and niobium oxalate as raw materials,the mass ratio of Nb2O5/ TU was 1 ∶ 1,1 ∶ 2 and 1 ∶ 3. Different photocatalysts were characterized by X-ray diffraction( XRD),scanning electron microscope( SEM),X-ray Fluorescence( XRF),X-ray photoelectron spectroscopy( XPS) and ultravioletvisible diffuse reflectance spectroscopy( UV-Vis). The photocatalytic degeneration of rhodamine B under ultraviolet radiation was investigated. The XRD patterns showed that N and S doping did not change the crystal structure of Nb2O5,and both Nb2O5 and N,Sdoped Nb2O5 existed as hexagonal phase structure. The SEM images showed that there were many pores in the surface of N,S-doped Nb2O5,and the size of catalyst particle became smaller. Many ordinary pores appeared on the surface of N,S-doped Nb2O5 with the mass ratio of 1 ∶ 1( Nb2O5/ TU),which increased the reaction area with rhodamine B. Further characterizations by XRF and XPS showed that N and S was doped into the Nb2O5 structure,S was in the form of S(6 +),which could be regarded as SO42-adsorbed on the surface of Nb2O5,and part of S(6 +)replaced Nb(5 +),and O was replaced by N existing in the environment of O- Nb- N. UV-Vis determinations showed that the band gap of N,S-doped Nb2O5 was narrowed and a red shift appeared,indicating that the range of light absorption was broadened,which improved the photocatalytic performance. The results of photocatalytic degradation of rhodamine B showed that the catalytic effect of N and S doping Nb2O5 was about 30% higher than that of Nb2O5 under the condition of UV irradiation. The degrade ratio for rhodamine B was 92% when the mass ratio of Nb2O5/ TU was 1∶ 1 under UV radiation for 3 h.
Keyword:
niobium pentoxide; N,S doped; photocatalysis;
Received: 2014-05-29
五氧化二铌因具有独特的性能和潜在的应用价值引起人们的关注[1,2,3,4,5,6]。它的应用包括: 作为催化剂,如水合、脱水反应等; 作为载体,如加氢、脱氢反应等[7]; 作为助剂,如氮氧化合物的消除反应等[8]。目前葛素香等[9]对碳掺杂五氧化二铌光催化降解罗丹明B性能进行了研究。然而,其他相关五氧化二铌在光催化降解有机污染物的性能研究方面却鲜有报道。
本实验以预处理后的五氧化二铌和硫脲按不同比例制得前驱体,采用焙烧法制得N,S掺杂五氧化二铌光催化剂。非金属N,S的掺杂有效拓宽了五氧化二铌在紫外光区域的响应范围,提高了光催化活性。N,S掺杂五氧化二铌光催化剂降解有机染料的技术具有绿色环保优势,对于进一步研究铌系催化剂和污水处理领域具有较大意义。
1 实验
1. 1 试剂与仪器
试剂: 硫脲、罗丹明B( 均为分析纯) ,国药集团化学试剂有限公司; 草酸铌( NH4[Nb O( C2O4)2( H2O)2]・3H2O,实测Nb2O5: 30. 545% ,实测Nb:21. 36% ,分析纯) ,宁夏东方钽业股份有限公司。
实验仪器: Rigaku D/max-RB型X射线衍射仪( XRD) ,日本理学公司。JSM-7500F型扫描电子显微镜( SEM) ,配备EDAX-EDS,日本JEOL公司。ZSX-100e型X射线荧光光谱仪( XRF) ,日本理学公司。Multilab 2000X光电子能谱( XPS) ,美国Thermo公司。Lamda 900 紫外-可见-分光光度计( UV-Vis) ,美国Perkin Elmer公司。
1. 2 催化剂的制备
五氧化二铌的制备: 在550 ℃条件下,将一定量的草酸铌在坩埚中焙烧3 h,冷却至室温后,收集Nb2O5。
分别称取一定质量的Nb2O5和硫脲( TU) ,使Nb2O5/ TU质量比为1∶ 1,1 ∶ 2,1 ∶ 3,在玛瑙研钵中充分研磨混合均匀制得光催化剂前驱体。通过程序升温将催化剂前驱体在350 ℃ 下焙烧2 h,得到光催化剂( 标记为NTU-1,NTU-2,NTU-3) 。
1. 3 光催化活性测试
光催化降解罗丹明B的反应是在一个玻璃容器中进行的,以紫外灯( 雪莱特365 nm) 为光源。实验步骤: 称取0. 3 g光催化剂,放入300 ml罗丹明B( 15 mg・L- 1) 溶液中,在磁力搅拌器上剧烈搅拌。首先,在黑暗条件下充分搅拌0. 5 h,使催化剂和溶液达到吸附-脱附平衡,然后,每隔1 h抽取10 ml溶液离心沉降,取上清液在554 nm处通过分光光度计测定吸光度变化来计算降解率。
2 结果与讨论
2. 1 样品XRD分析
图1 给出了Nb2O5和不同比例Nb2O5/ TU的XRD图,N,S掺杂后的催化剂衍射峰与Nb2O5基本一致,均与六方晶相结构的Nb2O5吻合,其pd号为7-61。这说明,N,S掺杂没有影响Nb2O5的晶型结构,同时检索结果并没有其他杂相存在,说明N,S以无定型状态分散在Nb2O5晶体或间隙之间。
2. 2 产物SEM分析
图2 为Nb2O5和不同比例Nb2O5/ TU的SEM图。图2( a) 所示,未掺杂的Nb2O5呈球状,有明显的团聚现象,且分散性较差,粒径大小处于50 ~200 nm范围内。随着N,S掺杂量的增加,催化剂粒径逐渐变小,且在粒子之间形成了不同程度的孔隙,推测是在高温焙烧时硫脲挥发造成的。当Nb2O5/ TU掺杂比为1∶ 1 时,催化剂粒子间形成了大小均一而且较深的孔隙,见图2( b) 。这导致催化剂与染料接触面积增大,将有利于NTU-1 催化效果的提高。当掺杂量继续增加时,空隙变得不规则且逐渐变小变少。
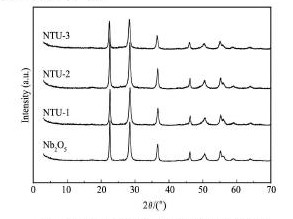
图1 Nb2O5和不同比例Nb2O5/TU的XRD图Fig.1 XRD patterns of Nb2O5and different proportions of Nb2O5/TU

图2 Nb2O5和不同比例Nb2O5/TU的SEM图Fig.2 SEM images of Nb2O5and different proportions of Nb2O5/TU
(a)Nb2O5;(b)NTU-1;(c)NTU-2;(d)NTU-3
2. 3 样品XRF分析
为进一步研究掺杂后催化剂的元素组成,对样品进行了XRF测试。半定量数据分析( SQX) 结果见表1,随着硫脲掺杂比例增大,以SO3计的硫含量增加,说明了S元素掺杂在Nb2O5晶体表面或者结构中。XRF结果没有显示出N含量,这是因为XRF元素分析范围只能涵盖从钠( 11 Na) 到铀( 92 U) 。
2. 4 样品XPS分析
为了进一步研究N,S掺杂Nb2O5情况,选取了NTU-1 的XPS全谱图及其Nb2O5和不同比例Nb2O5/ TU的Nb 3d,O 1s,N 1s图进行说明。从NTU-1 的谱图( 图3 ) 可知,NTU-1 样品含有Nb,O,S,N,C等元素。其中,C 1s在297. 65 e V的峰可能来自环境污染或硫脲中未分解的C。S 2p的峰出现在172. 47 e V,高氧化态的S 2p其电子能级在168 e V以上,可以看作S6 +以SO42-的形式吸附在Nb2O5表层[10],而样品中S 2p的峰比单纯SO42-中S 2p的峰偏移4. 47 e V,可能是部分S6 +取代了Nb5+[11]。另外,从谱图中可以看出N掺杂后的样品Nb 3d和O 1s特征峰均向低结合能方向位移,说明Nb周围环境发生变化[12]。N 1s在402 ~ 403e V处的特征吸收峰表明N取代O存在于O - Nb -N环境中[13,14]。
表1 Nb2O5和不同比例Nb2O5/ TU元素组成及含量Table 1Element composition and content of Nb2O5and different proportions of Nb2O5/ TU ( %,mass fraction) 下载原图

表1 Nb2O5和不同比例Nb2O5/ TU元素组成及含量Table 1Element composition and content of Nb2O5and different proportions of Nb2O5/ TU ( %,mass fraction)
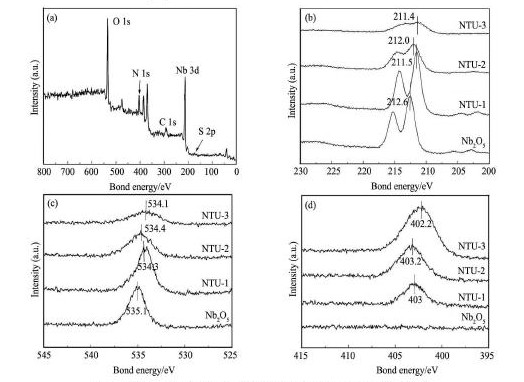
图3 NTU-1 和不同比例Nb2O5/ TU的Nb 3d,O 1s,N 1s的XPS谱图Fig. 3 XPS spectra of NTU-1 ( a) and Nb 3d ( b) ,O 1s ( c) ,N 1s ( d) of different proportions of Nb2O5/ TU
2. 5 样品UV-Vis分析
图4 为Nb2O5和不同比例Nb2O5/ TU的UVVis图。如图4 所示,Nb2O5只在紫外光区有响应,掺杂N,S的样品在400 ~ 650 nm处都有较强的光吸收。Nb2O5,NTU-1,NTU-2 和NTU-3 的紫外吸收边分别为403. 6,423. 1,425. 1 和436. 4 nm,对应的能带间隙分别为3. 07,2. 93,2. 92 和2. 84 e V。由于N,S掺杂Nb2O5催化剂能带变窄,从而可以吸收可见光,实现向可见光区域的红移[15]。随着N,S掺入量的增加红移现象越来越明显,原因可能是N,S掺杂在Nb2O5表面或晶体中,使光吸收能力增强,这与样品的颜色变化相对应,从开始的白色变成淡黄,逐渐加深最后达到土黄,间接而又直观地证明了非金属N,S掺杂进入Nb2O5晶体结构中。
2. 6 光催化评价
图5 是Nb2O5和不同掺杂量的Nb2O5/ TU在紫外光条件下降解罗丹明B的效果图。由图5 可见,未掺杂N,S的Nb2O5也有一定的光催化效果,在紫外照射3 h后降解罗丹明B 48%。N,S掺杂Nb2O5的催化活性均显著提高,光照3 h后,NTU-2和NTU-3对罗丹明B的降解率(η)为82%~84%,而NTU-1的降解率达到92%,这可能与催化剂晶粒大小及掺杂后晶体结构变化等因素有关[16]。
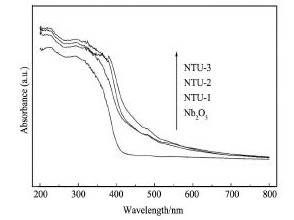
图4 Nb2O5和不同比例Nb2O5/TU的UV-Vis图Fig.4 UV-Vis spectra of Nb2O5and different proportions of Nb2O5/TU
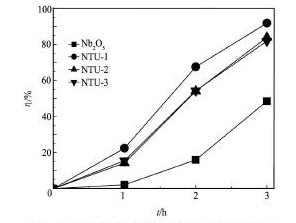
图5 Nb2O5和不同比例Nb2O5/ TU的光催化效果Fig. 5 Photocatalytic results of Nb2O5and different proportions of Nb2O5/ TU
3 结论
以上分析结果表明,本实验制备的N,S掺杂Nb2O5光催化剂,S以S6 +形式存在,可看作是以SO42-形式吸附在Nb2O5表面,部分S6 +取代了Nb5 +; 而N则是取代O存在于O - Nb - N环境中,N,S掺杂使Nb2O5晶体结构发生变化,实现光催化剂从紫外光区域到可见光区域的红移,使光催化效果显著提高。在紫外光照射条件下降解15mg・L- 1罗丹明B时,N,S掺杂Nb2O5比Nb2O5对罗丹明B的光催化降解率提高了30% ~ 40% ;NTU-1 的晶体中出现大量规则孔隙,使其催化效果最佳,紫外照射3 h后罗丹明B的降解率为92% 。
参考文献
[1] Yang K,Yang Q,Bao Y F.Formation of carbonitride precipitates in hardfacing alloy with niobium addition[J].Rare Metals,2013,32(1):52.
[2] Ansanay Y,Kolar P,Sharma-Shivappa R R,Cheng J J.Niobium oxide catalyst for delignification of switchgrassfor fermentable sugar production[J].Industrial Crops and Products,2014,52:790.)
[3] Bertuna A,Comini E,Poli N,Zappa D,Sberveglieri G.Niobium oxide nanostructures for chemical sensing[J].Procedia Engineering,2014,87:807.
[4] Prado da Silva M H,da Rocha D N,de Andrade Gobbo L,dos Santos Azevedo L M,Louro L H L,Machado Costa A,Brant de Campos J.Synthesis of piezoelectric and bioactive Na Nb O3from metallic niobium and niobium oxide[J].Journal of Biomedical Materials Research Part B:Applied Biomaterials,2015,10:1002.
[5] Leindecker G C,Alves A K,Bergmann C P.Synthesis of niobium oxide fibers by electrospinning and characterization of their morphology and optical properties[J].Ceramics International,2014,40(10):16195.
[6] Wang J J,Wang X H,Zhang Y,Zheng S L.Comprehensive recovery of tantalum and niobium from tantalum niobium Residue[J].Chinese Journal of Rare Metals,2015,39(3):251.(汪加军,王晓辉,张盈,郑诗礼.含钽铌废渣中钽铌资源的综合回收工艺研究[J].稀有金属,2015,39(3):251.)
[7] Tanabe K.Catalytic application of niobium compounds[J].Catalysis Today,2003,78:65.
[8] Tanabe K,Okazaki S.Various reactions catalyzed by niobium compounds and materials[J].Applied Catalysis A,1995,133:191.
[9] Ge S X,Zheng Z,Zhang L Z.Research for visible light catalytic performance of C-doped niobium pentoxide[A].The National Academic Meeting of Solar Photochemical and Photocatalytic in 2010[C].Kunming:Yunnan University Press,2010.374.(葛素香,郑直,张礼知.碳修饰五氧化二铌纳米结构可见光光催化性能研究[A].2010年全国太阳能光化学与光催化学术会议论文集[C].昆明:云南大学出版社,2010.374.)
[10] Yao N.Preparation,Characterization and Photocatalytic Activity of N,S-doped Ti O2Powders[D].Hubei:Huazhong University of Science and Technology,2011.39.(姚念.N,S共掺Ti O2粉体的制备、表征及光催化性能研究[D].湖北:华中科技大学,2011.39.)
[11] Bai Y N,Zuo S L,Yu Y C,Liu J J,Li B S.Fabrication of nitrogen and sulfur codoped titanium dioxide and its visible light photocatalytic activity[J].Chemical Research,2013,24(4):355.(白玉娜,左胜利,于迎春,刘建军,李保山.氮-硫共掺杂二氧化钛光催化剂的制备及其可见光催化活性[J].化学研究,2013,24(4):355.)
[12] Ma Y F.Design,Preparation and Performs of Ti O2-Based Composite Photocatalytic Materials[D].Jilin:Northeast Normal University,2012.56.(马延凤.Ti O2基复合光催化材料的设计制备及其性能研究[D].吉林:东北师范大学,2012.56.)
[13] Zhang X R,Lin Y H,Zhang J F,He D Q,Wang D J.Photoinduced charge carrier propertiesand photocatalytic activity of N-doped Ti O2nanocatalysts[J].Acta Physico-Chimica Sinica,2010,26(10):2733.(张晓茹,林艳红,张健夫,何冬青,王德军.N掺杂Ti O2纳米粒子表面光生电荷特性与光催化活性[J].物理化学学报,2010,26(10):2733.)
[14] Tang Y C,Huang X H,Yu H Q,Hu C.Characterization and visible-light-activity of nitrogen-doped Ti O2photocatalyst[J].Chinese Journal of Inorganic Chemistry,2005,21(11):1747.(唐玉朝,黄显怀,俞汉青,胡春.N掺杂Ti O2光催化剂的制备及其可见光活性研究[J].无机化学学报,2005,21(11):1747.)
[15] Xu L,Tang C Q,Dai L,Tang D H,Ma X G.Primary principle electronic structure of N-doped anatase Ti O2[J].Acta Physica Sinica,2007,56(2):1048.(徐凌,唐超群,戴磊,唐代海,马新国.N掺杂锐钛矿Ti O2电子结构的第一性原理研究[J].物理学报,2007,56(2):1048.)
[16] Zhang H M,Zhao L H,Wang D J.Preparation,characterization of visible light S-doped Ti O2and its photocatalytic activity[J].Chemical Research and Application,2009,21(10):1386.(张海明,赵莲花,王德军.可见光响应的硫掺杂Ti O2的制备、表征及其光催化活性[J].化学研究与应用,2009,21(10):1386.)