
Catalytic effect of activated carbon on bioleaching of low-grade primary copper sulfide ores
ZHANG Wei-min(张卫民)1, GU Shi-fei(谷士飞)2
1. School of Civil and Environmental Engineering, East China Institute of Technology, Fuzhou 344000, China;
2. Geological Environmental Monitoring Station of Guangxi Zhuangzu Autonomous Region, Guilin 541004, China
Received 1 February 2007; accepted 5 June 2007
______________________________________________________________
Abstract: The catalytic effect of activated carbon on the bioleaching of low-grade primary copper sulfide ores using mixture of Acidithiobacillus ferrooxidans and Acidithiobacillus thiooxidans was investigated. The results show that the addition of activated carbon can greatly accelerate the rate and efficiency of copper dissolution from low-grade primary copper sulfide ores. The solution with the concentration of 3.0 g/L activated carbon is most beneficial to the dissolution of copper. The resting time of the mixture of activated carbon and ores has an impact on the bioleaching of low-grade primary copper sulfide ores. The 2 d resting time is most favorable to the dissolution of copper. The enhanced dissolution rate and efficiency of copper can be attributed to the galvanic interaction between activated carbon and chalcopyrite. The addition of activated carbon obviously depresses the dissolution of iron and the bacterial oxidation of ferrous ions in solution. The lower redox potentials are more favorable to the copper dissolution than the higher potentials for low-grade primary copper sulfide ores in the presence of activated carbon.
Key words: bioleaching; low-grade primary copper sulfide ores; activated carbon; catalytic effect
______________________________________________________________
1 Introduction
Biohydrometallurgy has become one of the most important alternatives for recovery of low-grade secondary copper sulfide ores and copper oxide ores, and has been extensively applied in industry production due to its priorities in full utilization of resources, lower investment, lower cost, lower energy consumption and being friendly to environment. However, it’s difficult to be applied in recovery of low-grade primary copper sulfide ores due to the longer leaching cycle and lower leaching rate. It is well known that chalcopyrite is the main copper mineral in the low-grade primary copper sulfide ores, and is relatively difficult to be leached by chemical and microbiological oxidation because of its special crystal structure and electrochemistry in contrast to many other copper minerals[1-2]. It is essential to find desirable methods to enhance chalcopyrite bioleaching.
Several catalysts and/or promoters have been proposed, such as surfactants, iron powder, hematite, and silver ions[3-6]. Silver ions are the most effective catalyst that has been proposed in the bioleaching of chalcopyrite, but the processes using silver ion are costly and unless ores contain silver minerals, silver ion would not be used in practice. Therefore, it is essential to develop cheaper and more effective catalysts or methods for improving chalcopyrite bioleaching.
WAN et al[7] proposed that the dissolution rate of chalcopyrite in the ferric sulfate leaching was enhanced by the formation of chalcopyrite/carbon aggregates. They suggested that the conductive carbon particles change the conductivity of the reaction product layer and such a phenomenon accounts for the increase in leaching rate. NAKAZAWA et al[8] and LI et al[9] suggested that the enchanced rate of copper bioleaching from chalcopyrite by adding activated carbon could be attributed to the galvanic interaction between activated carbon and chalcopyrite. The presence of Acidithiobacillus ferrooxidans could accelerate the galvanic interactions.
The purpose of this work is to study the catalytic effect of activated carbon on the bioleaching of low-grade primary copper sulfide ores.
2 Experimental
2.1 Bacteria
The mixture of Acidithiobacillus ferrooxidans and Acidithiobacillus thiooxidans used in this study was isolated from acid mine drainage at Yongping Mine in China and cultured in 9K+S ore-culture medium with pH=1.20 in the constant-temperature shaker with rotation rate of 130 r/min and temperature of 30 ℃ using chalcopyrite ores (10%, mass to volume ratio) as main energy sources. The 9K+S ore-culture medium contained 3 g/L (NH4)2SO4, 0.1 g/L KCl, 0.5 g/L K2HPO4, 0.5 g/L MgSO4?7H2O, 0.01 g/L Ca(NO3)2, 5 g/L FeSO4?7H2O, 1 g/L S and 10 g/L chalcopyrite ores, and was adjusted to pH<1.50 with sulphuric acid. The bacterial populations were about 1×107 cells/mL.
2.2 Samples
The samples of low-grade primary copper sulfide ores used in this study were obtained from Yongping Mine in China. The particle size was less than 5 mm. The X-ray diffraction analysis showed that the most important mineral phases of low-grade chalcopyrite ores are pyrite and chalcopyrite. The chemical composition of low-grade primary copper sulfide ores is as follows: 0.40%Cu, 40.90%SiO2, 14.12%Fe, 6.68%Al2O3, 9.97%CaO, 1.64%MgO, 1.58%K2O, 0.27%Na2O, 0.05%P2O5 and 13%S. The chemical phase analysis of low-grade primary copper sulfide ores showed that primary copper sulfide is 0.38%, secondary copper sulfide 0.007 4%, free copper oxide 0.005 3%, and bonded copper oxide 0.002 6%, respectively.
2.3 Bioleaching experiments
Bioleaching experiments were carried out in 250 mL elenmeryer flasks with 80 mL 9K+S culture medium without ferrous iron and 20 mL inoculation at initial pH= 1.20 (pH values were controlled within 1.50 with sulphuric acid during the bioleaching processes) and 25% pulp density. When activated carbon was added, it was mixed with primary copper sulfide ores in a porcelain bowl and rested for some days as needed. In all bioleaching experiments, sample solutions were withdrawn periodically for analysis and an equal volume of medium was replaced. Any evaporation was compensated with water. The flasks were incubated in a rotary shaker at 130 r/min and 30 ℃.
2.4 Analytical methods
The pH was measured by a pH probe calibrated with a low pH buffer. The Eh was measured with Pt electrode, using a calomel electrode as a reference. The data were converted into the standard hydrogen electron potential(SHE). The Cu2+ concentration was determined by atomic absorption spectrophotometry(AAS). The Fe2+ and Fe3+ concentrations were determined by titration with EDTA (ethylene diamine tetraacetic acid).
3 Results and discussion
Figs.1-4 show the bioleaching results of primary copper sulfide ores in the presence and absence of activated carbon with particle size of less than 7.4 μm. As shown in Fig.1, the rate and efficiency of copper dissolution are greatly accelerated by addition of activated carbon at the initial stage of bioleaching. The yields of copper bioleaching are 11%, 64%, 70%, 79% and 60% under the condition of 0, 0.5, 2.0, 3.0 and 5.0 g/L activated carbon, respectively, after 600 h bioleaching. The results also show the dissolution of copper increases with increasing activated carbon concentration if the concentration of added activated carbon is not higher than 3 g/L. The concentration of 3.0 g/L activated carbon seems to be most beneficial to the dissolution of copper. Under such conditions, the recovery rate of copper is increased from 11% to 79% after 600 h bioleaching, which is 68% higher than that in the absence of activated carbon. More activated carbon addition is of no significance.

Fig.1 Catalytic effect of concentration of activated carbon added on copper bioleaching (Resting time of mixture of activated carbon and ores is 1 d)

Fig.2 Catalytic effect of concentration of activated carbon added on iron bioleaching (Resting time of mixture of activated carbon and ores is 1 d)
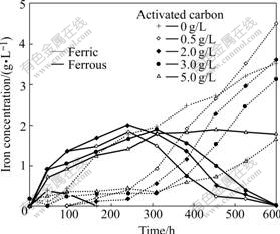
Fig.3 Catalytic effect of concentration of activated carbon added on iron concentration in solution (Resting time of mixture of activated carbon and ores is 1 d)

Fig.4 Catalytic effect of concentration of activated carbon added on Eh in solution (Resting time of mixture of activated carbon and ores is 1 d)
As shown in Figs.2 and 3, the addition of activated carbon obviously depresses the dissolution of iron from primary copper sulfide ores in contrast to the dissolution of copper. The dissolution of iron decreases with increase of the concentration of initial activated carbon added. The addition of activated carbon initially inhibits the bacterial oxidation of ferrous ions in solution. The inhibition increases with increase of the concentration of activated carbon added. The complete oxidation of ferrous ions is achieved after 164 h in the absence of activated carbon while after 600 h in the presence of 0.5-3.0 g/L activated carbon.
At the early beginning of bioleaching of primary copper sulfide ores, the redox potentials in solution are lowered with the addition of activated carbon and maintained under 650 mV for a long time (Fig.4). The Eh values decrease with increasing concentration of initial activated carbon. This phenomenon coincides with the slow bacterial oxidation of ferrous ions. When the redox potentials in solution are maintained under 650 mV by ferrous iron, the rate of copper bioleaching is accelerated, whereas it becomes slow. The enhanced rate of copper bioleaching could be attributed to the galvanic interactions. Activated carbon is electrically conductive and may form a galvanic coupling in contact with chalcopyrite. In the absence of activated carbon, chalcopyrite is directly oxidized by ferric ions and/or dissolved oxygen at higher redox potentials to release cupric ions and a dense elemental sulfur layer is formed on the mineral surface during bioleaching, acting as a diffusion barrier. The cupric ions are extracted according to the following reactions[10]:
CuFeS2+4Fe3+=Cu2++5Fe2++2S0 (1)
CuFeS2+4H++O2=Cu2++Fe2++2S0+2H2O (2)
In the presence of activated carbon, chalcopyrite is reduced by cupric ions or ferrous ions in the presence of cupric ions at lower redox potentials to form secondary chalcocite and the secondary chalcocite is oxidized by ferric ions and/or dissolved oxygen to release cupric ions. The cupric ions are extracted according to the following reactions[10-11]:
CuFeS2+Cu2+=Cu2S+Fe2++S0 (3)
CuFeS2+3Cu2++3Fe2+=2Cu2S+4Fe3+ (4)
Cu2S+4H++O2=2Cu2++S0+2H2O (5)
Cu2S+4Fe3+=2Cu2++S0+4Fe2+ (6)
The oxidation rate of the secondary chalcocite is higher than that of chalcopyrite, which causes the rapid copper extraction at lower redox potentials. The evidence suggests that the lower redox potentials are more favorable to the copper dissolution than the higher potentials for low-grade primary copper sulfide ores in the presence of activated carbon. The presence of Acidithiobacillus ferrooxidans in the leaching system can significantly enhance the galvanic interaction between chalcopyrite and activated carbon because bacteria make Fe2+ and S0 in reactions (3), (5) and (6) oxidized to Fe3+ and 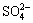 respectively by the following reactions[9, 12-15]:
respectively by the following reactions[9, 12-15]:
2S0+3O2+2H2O=4H2++2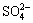 (7)
(7)
4Fe2++O2+4H+=4Fe3++2H2O (8)
Figs.5-7 show the influence of the resting time of the mixture of activated carbon and ores on bioleaching of low-grade primary copper sulfide ores. As shown in Fig.5, the yields of copper dissolution are 65%, 84%, 86%, 72% and 66% under the resting time of 0, 1, 2, 4 and 6 d, respectively, after 600 h bioleaching. The results also show that the dissolution of copper increases with increasing resting time before reaching a limit. The resting time of 2 d is most beneficial to the dissolution of copper, and the copper recovery rate increases from 65% to 86% after 600 h bioleaching, which is 21% higher than that for the resting time of 0 d. However, the resting time more than 2 d is of no significance for copper leaching.
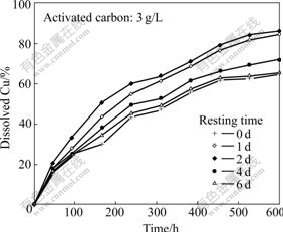
Fig.5 Influence of resting time of mixture of activated carbon and ores on copper bioleaching

Fig.6 Influence of resting time of mixture of activated carbon and ores on iron bioleaching
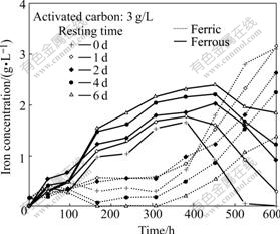
Fig.7 Influence of resting time of mixture of activated carbon and ores on iron concentration in solution
The resting time of the mixture of activated carbon and ores has an impact on the dissolution of iron, too. The results also show the dissolution of iron increases with increasing resting time before reaching a limit (Fig.6). The resting time of the mixture of activated carbon and ores inhibits the bacterial oxidation of ferrous ions in solution. The inhibition increases with increase of the resting time. The complete oxidation of ferrous ions is not achieved after 600 h of bioleaching except under the condition of 0 d resting time (Fig.7).
4 Conclusions
1) The addition of activated carbon can greatly accelerate the rate and efficiency of copper dissolution in the bioleaching of low-grade primary copper sulfide ores using Acidithiobacillus ferrooxidans and Acidithiobacillus thiooxidans. The solution with a concentration of 3.0 g/L activated carbon is most beneficial to the dissolution of copper, whose recovery rate is increased from 11% to 79% after 600 h bioleaching, being 68% higher in the presence of activated carbon than that in the absence of activated carbon.
2) The resting time of the mixture of activated carbon and ores has an impact on the bioleaching of low-grade primary copper sulfide ores. The resting time of 2 d is most beneficial to the dissolution of copper, and the copper recovery rate increases from 65% to 86% after 600 h bioleaching, which is 21% higher than that of the resting time of 0 d.
3) The enhanced rate and efficiency of copper dissolution can be attributed to the galvanic interaction between activated carbon and chalcopyrite. The addition of activated carbon obviously depresses the dissolution of iron in low-grade primary copper sulfide ores and the bacterial oxidation of ferrous ions in solution.
4) The lower redox potentials are more favorable to the copper dissolution than the higher potentials for low-grade primary copper sulfide ores in the presence of activated carbon.
References
[1] PARKER A J, PAUL R L, POWER G P. Electrochemistry of the oxidative leaching of copper from chalcopyrite [J]. J Electroanal Chem, 1981, 118: 305-316.
[2] CRUNDWELL F K. The influence of the electronic structure of solid on the anodic dissolution and leaching of semiconductor sulphide minerals [J]. Hydrometallurgy, 1988, 21: 155-190.
[3] AHONEM L, TUOVINEN O H. Catalytic effects of silver in the microbiological leaching of finely ground chalcopyrite-containing ore material in shake flasks [J]. Hydrometallurgy, 1990, 24: 219-236.
[4] GOMEZ E, BALLESTER A, BLAZQUEZ M L. Silver-catalysed bioleaching of a chalcopyrite concentrate with mixed cultures of moderately thermophilic microorganisms [J]. Hydrometallurgy, 1999, 51: 37-46.
[5] SATO H, NAKAZAWA H, KUDO Y. Effect of silver chloride on the bioleaching of chalcopyrite concentrate [J]. Int J Miner Process, 2000, 59: 17-24.
[6] HU Yue-hua, QIU Guan-zhou, WANG Jun, WANG Diang-zuo. The effect of silver-bearing catalysts on bioleaching of chalcopyrite [J]. Hydrometallurgy, 2002, 64: 81-88.
[7] WAN R Y, MILLER J D, FOLEY J, PONS S. Electrochemical features of the ferric sulfate leaching of CuFeS2/C aggregates [C]//RICHARDSON P E, SRINIVASAN S, WOODS R. Proceedings of Electrochemistry in Mineral and Metal Processing. NJ: PENNINGTON, 1984: 391-416.
[8] NAKAZAWA H, FUJISAWA H, SATO H. Effect of activated carbon on the bioleaching of chalcopyrite concentrate [J]. Int J Miner Process, 1998, 55: 87-94.
[9] LI Hong-xu, QIU Guan-zhou, HU Yue-hua, WANG Dian-zuo. Galvanic effect on mixed sulfide bioleaching [J]. The Chinese Journal of Nonferrous Metals, 2003,13: 1283-1287. (in Chinese )
[10] HIROYOSHI N, ARAI M, MIKI H, TSUNEKAWA M, HIRJIMA T. A new reaction model for the catalytic effect of silver ions on chalcopyrite leaching in sulfuric acid solutions [J]. Hydrometallurgy, 2002, 63: 257-267.
[11] ELSHERIF A E. The influence of cathodic reduction, Fe2+ and Cu2+ ions on the electrochemical dissolution of chalcopyrite in acidic solution [J]. Minerals Engineering, 2002, 15: 215-223.
[12] NATARAJAN K A, IWASAKI I. Role of galvanic interactions in the bioleaching of Duluth Gabbro copper-nickel sulphides [J]. Sep Sci Technol, 1985, 18: 1095-1111.
[13] JYOTHI N, SUDHA K N, NATARAJAN K A. Electrochemical aspects of selective bioleaching of sphalerite and chalcopyrite from mixed sulphides [J]. Int J Miner Process, 1989, 27: 189-203.
[14] SADOWSKI Z, JAZDZYK E, KARAS H. Bioleaching of copper ore flotation concentrates [J]. Minerals Engineering, 2003, 16: 51-53.
[15] LI Hong-xu, QIU Guan-zhou, HU Yue-hua, CANG Da-qiang, WANG Dian-zuo. Electrochemical behavior of chalcopyrite in presence of Thiobacillus ferrooxidans [J]. Trans Nonferrous Met Soc China, 2006, 16: 1240-1245.
__________________
Corresponding author: ZHANG Wei-min; Tel: +86-794-8258535; E-mail: wmzhang@ecit.edu.cn
(Edited by YANG Bing)