Kinetic process of oxidative leaching of chalcopyriteunder low oxygen pressure and low temperature
来源期刊:中国有色金属学报(英文版)2007年第2期
论文作者:邱廷省 聂光华 王俊峰 崔立凤
文章页码:418 - 418
Key words:chalcopyrite; kinetics; pressure oxidation; sodium chloride hydroxide medium; leaching
Abstract: Kinetic process of oxidative leaching of chalcopyrite in chloride acid hydroxide medium under oxygen pressure and low temperature was investigated. The effect on leaching rate of chalcopyrite caused by these factors such as ore granularity, vitriol concentration, sodium chloride concentration, oxygen pressure and temperature was discussed. The results show that the leaching rate of chalcopyrite increases with decreasing the ore granularity. At the early stage of oxidative reaction, the copper leaching rate increases with increasing the oxygen pressure and dosage of vitriol concentration, while oxygen pressure affects leaching less at the later stage. At low temperature, the earlier oxidative leaching process of chalcopyrite is controlled by chemical reactions while the later one by diffusion. The chalcopyrite oxidative leaching rate has close relation with ion concentration in the leaching solution. The higher ion concentration is propitious for chalcopyrite leaching.
基金信息:the National Natural Science Foundation of China
the Natural Science Foundation of Jiangxi Province, China

QIU Ting-sheng(邱廷省)1, NIE Guang-hua(聂光华)2, WANG Jun-feng(王俊峰)1, CUI Li-feng(崔立凤)1
1. School of Environmental and Architectural Engineering, Jiangxi University of Science and Technology,Ganzhou 341000,China;
2. School of Mineral Engineering, Guizhou University, Guiyang 550003, China
Received 20 July 2006; accepted 28 November 2006
Abstract: Kinetic process of oxidative leaching of chalcopyrite in chloride acid hydroxide medium under oxygen pressure and low temperature was investigated. The effect on leaching rate of chalcopyrite caused by these factors such as ore granularity, vitriol concentration, sodium chloride concentration, oxygen pressure and temperature was discussed. The results show that the leaching rate of chalcopyrite increases with decreasing the ore granularity. At the early stage of oxidative reaction, the copper leaching rate increases with increasing the oxygen pressure and dosage of vitriol concentration, while oxygen pressure affects leaching less at the later stage. At low temperature, the earlier oxidative leaching process of chalcopyrite is controlled by chemical reactions while the later one by diffusion. The chalcopyrite oxidative leaching rate has close relation with ion concentration in the leaching solution. The higher ion concentration is propitious for chalcopyrite leaching.
Key words: chalcopyrite; kinetics; pressure oxidation; sodium chloride hydroxide medium; leaching
1 Introduction
As a traditional metallurgical technology of the sulfide copper mines, copper pyrometallurgy craft called melting-converting-refining-electrolyzing has produced 75%-80% copper among the total in the world. But the copper ore grade becomes lower and lower because of copper resources exploited excessively. Complicate copper ores bearing multi-metals need exploiting urgently, and there is strict environmental act in every country in the world, so this traditional metallurgical method meets tremendous challenge[1-2]. Recently, copper hydrometallurgy technology, especially called leaching-volume extraction-electrodeposit has developed so rapidly that this method research has drawn worldwide attention. It is estimated that it will bring innovation to copper industry. Compared with copper pyrometallurgy, it has many advantages such as effective recycling valuable metals, easy operating, low depletion, environmental-friendship and low cost[3-5].
As the key step of this method, the sulfide concentrate leaching becomes the important process that is paid much attention to the whole craft. Usually, chalcopyrite concentrate is the main raw material that produces copper. There are many chalcopyrite hydrometallurgy methods such as acid leaching, vitriol ferrate leaching, chloride leaching, ammonia leaching, bio-leaching, ozone leaching, peroxide leaching, acid dichromate leaching and reductive leaching after being studied for 30 years[6-13]. But all these focus on the chalcopyrite leaching. The method that cupric sulfide concentrate learning-chalcopyrite was leached in the chloride acid hydroxide medium under low oxygen pressure and low temperature was studied by XIE et al[14] and QIU et al[15]. In this paper, kinetic process of oxidative leaching of chalcopyrite in chloride acid hydroxide medium under oxygen pressure and low temperature was studied in order to provide some new theoretical references and help for copper hydrometallurgy craft in this field.
2 Experimental2.1 Sample
Man-selected crystal-chalcopyrite supplied by a mine in Tongling of Anhui Province, China underwent a series of processes such as crushing, milling and sieving, then distilled water was used to clean it, at last it was stored in bottles for experiment.
2.2 Method
All the experiments of oxidative leaching under oxygen pressure were conducted in the autoclave called FCH whose volume was 2 L. After milled in the pin crusher, chalcopyrite granularities became eligible. And then these ores were mixed with water by appropriate liquid-to-solid ratio. After some vitriol and sodium chloride were added into the autoclave, the full pasty ore slurry was put into the autoclave. Then the autoclave was sealed absolutely. After the temperature was up to request degree, the oxygen was added into the autoclave. Simultaneously, time began to be counted. At the end of the reaction, the apparatus was cooled quickly. The reacted dry slurry was sent to analyze (Fig.1).

Fig.1 Copper oxidative leaching flow under oxygen pressure
2.3 Principle of copper oxidative leaching under oxygen pressure
The ion was separated from the chalcopyrite crystal lattice. Subsequently many interim matters came into being, such as Cu9Fe9S16 and Cu39S28 during the whole oxidative leaching process. The chemical reaction can be shown by the following equation:
CuFeS2→Cu1-XFe1-YS2-Y-X-Z+XCu2++YFe2++
(X+Y+Z)S0+2(X+Y+Z)e (1)
where the value of coefficient Y>X,the coefficient Z is something to do with oxidative electric potential of the whole system. With the reaction going, the X value is close to Y step by step. After ions are released from the chalcopyrite crystal lattice thoroughly, the interim matters composed by Cu and S begin to come into being. Their structure can be shown by CuxS. Here, 1≤x≤2.
The process that Cu and S compound is further oxidized to Cu2+ is influenced by oxidative electric potential of the whole system greatly. Under the experimental conditions, the CuxS oxidative reaction is
CuxS+xCl-=xCuCl+S0+xe (2)
The sediment CuCl is dissolved by Cl- complexation, then oxidized to Cu2+. The main reactions are

Subsequently, carphosiderite and natrojarosite sediment by is formed hydrolyzing. The main reactions are

During the process that chalcopyrite was leached in chloride acid hydroxide medium under oxygen pressure and low temperature, oxygen worked as an oxidant, and catalysis occurred with Fe(Ⅲ) and Cu(Ⅱ) from the reaction. Both the vitriol and sodium chloride were solvents. A large amount of S and natrojarosite sediment was generated from the oxidative and solvable process of chalcopyrite. This indicated that there were many factors that affected oxidative and solvable kinetic process of chalcopyrite.
3 Results and discussionIn the chloride acid hydroxide medium under oxygen pressure, the chalcopyrite leaching process relates to the complicated system formed by gas-liquid-solid actually. While during the oxidative leaching process, leaching rate has something to do with these factors such as thickness of diffusion layer, area of phase interface, concentrate of extractor, system temperature and catalyser property. In this experiment, except stirring intension and slurry concentrate, all of the parameters such as ore granularity, temperature, partial oxygen pressure, H2SO4 concentration and NaCl concentration which affected the chalcopyrite leaching rate were investigated, respectively.
3.1 Effect of ore granularity
Ore granularity is one of the important factors influencing the ore dissolving and leaching. Generally speaking, small ore granularity is propitious for leaching. The leaching speed has direct proportion to the reciprocal value of ore granularity when the leaching speed accords with the regularity of core shrinkage model. So it is important to investigate the effect of ore granularity on the chalcopyrite leaching speed. The results are shown in Fig.2.
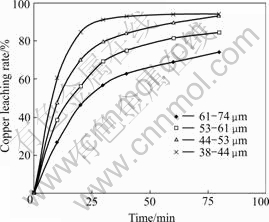
Fig.2 Effect of different ore granularity on copper leaching rate (t=110 ℃; p(O2)=0.45 MPa; [H2SO4]=36 g/L; [NaCl]=60 g/L; m(solution)?m(ore)=20?1; N=750 r/min)
From Fig.2, it can be seen that ore granularity influences the copper leaching rate greatly. At the first stage, the smaller the ore granularity, the faster the copper leaching rate. After oxidized for 20 min, the copper leaching rate can reach 80% when the ore granularity is 44-53 μm. Then oxidative speed turns from quick to slow obviously. Therefore, in order to gain the higher copper leaching rate, ore granularity of 44-53 μm in most experiments is controlled.
3.2 Effect of H2SO4 concentration
Like ore granularity, H2SO4 concentration is also an important factor during the oxidative pre-treatment process under oxygen pressure. It is not only a type of dissolving agent of sulphide ore, but influences directly the outcome and the process of pre-treatment. Therefore, it is necessary to investigate H2SO4 concentration influencing the copper leaching rate. The results are shown in Fig.3.

Fig.3 Effect of different H2SO4 concentrations on copper leaching rate (t=110 ℃; p(O2)=0.45 MPa; D=44-53 μm; [NaCl]=60 g/L; m(solution)?m(ore)=20?1; N=750 r/min)
Fig.3 shows that with the increasing of H2SO4 concentrate, at the initial stage, the copper leaching rate increases rapidly and after 20 min oxidation it becomes slowly. Subsequently H2SO4 concentration continues to increase with H2SO4 concentration 36 g/L. So the increase of H2SO4 concentration can speed up the copper leaching rate at the early stage.
3.3 Effect of NaCl concentration
As one of dissolving agents of sulphide ore, Cl- influencing the copper leaching rate was investigated during the oxidative pre-treatment process under oxygen pressure. The results are shown in Fig.4.
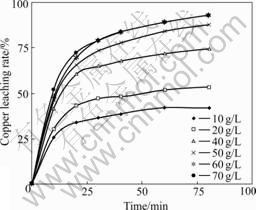
Fig.4 Effect of different NaCl concentrations on copper leaching rate (t=110 ℃; p(O2)=0.45 MPa; D=44-53 μm; [H2SO4]=36 g/L; m(solution)?m(ore)=20?1; N=750 r/min)
It can be seen from Fig.4 that NaCl concentration influences the copper leaching speed greatly. With the increase of NaCl concentration, the copper leaching rate increases. When it is greater than 60 g/L, the rate begins to decrease. So it is necessary to keep up NaCl concentration at 60 g/L during the chalcopyrite oxidative leaching process.
3.4 Effect of partial oxygen pressure
Firstly, O2 diffuses into water from the gas/liquid interface and then diffuses furthermore. It participates in reaction after it touches the ore surface. It is known that the influence of O2 on the chemical reaction is dependent on each O2 diffusible step during the above process. The gas solubility in the water is affected by the temperature and p(O2). The pressure of O2 has direct proportion to the O2 concentration in the water. Enhancing p(O2) promotes the O2 solubility and the oxidation speed is increased. The results are shown in Fig.5.
Fig.5 shows that at the initiative stage leaching reaction speed increases with the rising of p(O2) while at the later stage the speed keeps higher in despite of the lower p(O2). But after 80 min, all the copper leaching rate values keep in linearity. It is estimated that the leaching solvent depletes quickly at high rate under high p(O2) at the first stage. During the later stage, the rate keeps highly because of small amount of leaching solvent existing in the solution. Therefore it is proper that p(O2) is ascertained to be 0.45 MPa.
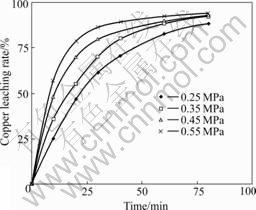
Fig.5 Effect of different p(O2) on copper leaching rate (t=110 ℃; [NaCl]=60 g/L; D=44-53 μm; [H2SO4]=36 g/L; m(solution)? m(ore)=20?1; N=750 r/min)
3.5 Effect of temperature
The system temperature also influences the copper leaching rate greatly. According to the Van’t Hoff regularity, the rate increases by 2-4 times with every increase of 10 K. The results are shown in Fig.6.
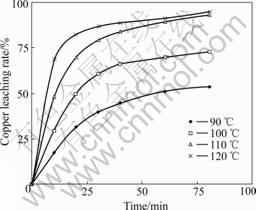
Fig.6 Effect of different temperatures on copper leaching rate (p(O2)=0.45 MPa; [NaCl]=60 g/L; D=44-53 μm; [H2SO4]=36 g/L; m(solution)?m(ore) =20?1; N=750 r/min)
From Fig.6, it can be seen that temperature is another important factor considered in this work. Increasing temperature improves the copper leaching rate. Based on the experiment results of Fig.6, the average reaction speed rate (min-1) at different temperatures and time is summarized in Table 1. According to Eqns.(7)-(8) and Table 1, the apparent activation energies (kJ/mol) at 90-120 ℃ and in 0-10 min and in 60-80 min are listed in Table 2.
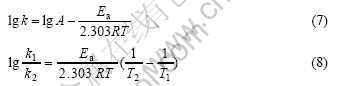
Table 1 Average reaction speed rate at different temperatures and time (min-1)
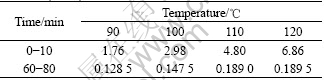
Table 2 Calculated apparent activation energy at different temperatures and time (kJ/mol)
It can be seen from Table 1 and Table 2 that all of the Ea values exceed 42 kJ/mol at different temperatures in 0-10 min. Therefore the oxidative leaching process is controlled by chemical reaction mainly. During this process, all of these such as increasing temperature, more solvents and reducing particle size can reinforce the copper leaching. All of the Ea values exceeding 42 kJ/mol in 60-80 min are less than 24 kJ/mol, and its average value is only 15.22 kJ/mol. It is considered that diffusion predominates in the whole reaction. The temperature influencing the copper leaching in 10-60 min is relatively complicated. Meanwhile leaching rate increase changes from quick to slow at 20 min oxidation.
In sum, it can be seen that each of temperature, Cl- concentration and p(O2) influences the chalcopyrite leaching rate greatly. Besides that, a change of leaching rate appears from quick to slow at 20 min, especially at relative higher temperature, p(O2) and relative higher H2SO4 concentration. According to the analysis, both of new S generated from reaction and carphosiderite and natrojarosite cannot wrap the ore. At the same time, the reason that makes chalcopyrite oxidative leaching rate decreasing dramatically is that a lot of Fe(Ⅲ) form the sediments by hydrolyzing.
4 Analysis on relation of leaching rate between ion and copper
From Fig.7, it can be seen that the ion content in the leaching solution reaches the maximum quickly at the initiative oxidation stage of 10 min. Just at this stage, the oxidative leaching speed exceeds the ion precipitating rate remarkably. The increase of Fe(Ⅲ) in the solution that has strong oxidation and catalysis power enhances the chalcopyrite oxidative rate. Meanwhile from Fig.1 to Fig.6, it can be seen that the copper leaching rates are all high in the leaching process controlled by chemical reaction. At the middle oxidation stage, the chalcopyrite oxidative speed descends rapidly because Fe(Ⅲ) is largely hydrolyzed to sediment. At the same time, lots of O2 are depleted during the process that Fe(Ⅱ) is oxidized to Fe(Ⅲ) rapidly. This means that the oxidant reaching the ore surface reduces largely so that chalcopyrite oxidative and dissolvable speed descend rapidly. At the later oxidation stage, the chalcopyrite oxidative and dissolvable speed keep constant because the Fe((Ⅲ) concentration in the solution is very low and constant. During this stage, chalcopyrite is oxidized and dissolved by the Cu(Ⅱ) and soluble O2 in the solution.
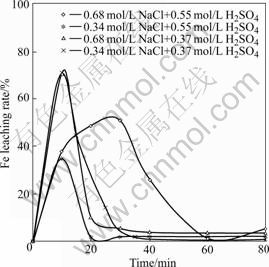
Fig.7 Relation between ion leaching rate and time under relatively low t and low p(O2) (t=110℃, p(O2)=0.45 MPa)
5 Conclusions1) The smaller the ore granularity, the higher the chalcopyrite leaching rate, so the ores are milled fully before the experiment. In initiative oxidative stage, high p(O2) and relatively high H2SO4 concentration enhance the rate, while in later stage effect of p(O2) and H2SO4 concentration is inconspicuous. With the NaCl concentration increasing, copper leaching rate increases. It keeps constant though the NaCl concentration is high.
2) Temperature is another important factor influencing the copper leaching rate greatly. In 0-10 min and at 90-120 ℃, the Ea value is 53.54 kJ/mol. The oxidative leaching process is controlled by the chemical reaction. The situation that temperature influences the copper leaching rate in 10-60 min is relatively complicated. The diffusion predominates in the whole reaction when the Ea is 15.22 kJ/mol.
3) The Fe ion concentration influences the chalcopyrite leaching rate greatly. In the initiative stage, because of the fast iron leaching, the chalcopyrite is oxidized and catalyzed by Fe(Ⅲ) in the solution whose concentration is higher. In the middle stage, the chalcopyrite leaching rate decreases rapidly because Fe(Ⅲ) is largely hydrolyzed to sediment. At the same time, lots of O2 is depleted during the process that Fe(Ⅱ) is oxidized to Fe(Ⅲ) rapidly. In the later oxidation stage, the effect of Fe(Ⅲ) on chalcopyrite oxidative leaching is subtle because the Fe(Ⅲ) concentration in the solution is very low and constant.
References[1] FAN Xing-xiang, PENG Jin-hui, HUANG Meng-yang, ZHANG Shi-min, ZHANG Li-bo. Study on innovative process of leaching of chalcopyrite [J]. Mining and Metallurgical Engineering, 2005, 25(3): 58-62.
[2] WANG Hai-bei, JIANG Kai-xi, ZHANG Bang-shen, WANG Yu-fang, LIN Jiang-shun, WANG Chun. A novel hydrometallurgical process with low temperature and pressure for complicated copper concentrate from XinJiang of China [J]. Nonferrous Metals, 2004, 56(3): 52-56.
[3] ZHU Tun. Modern Hydrometallurgy of Copper [M]. Beijing: Metallurgical Industry Press, 2002.
[4] DUTRIZAC J E. The leaching of sulphide minerals in chloride media [J]. Hydrometallurgy, 1992(29): 1-45.
[5] LOTENS J P, WESKER E. The behaviour of sulphur in oxidative the leaching of sulphidic minerals [J]. Hydrometallurgy, 1987(18): 39-45.
[6] CHO E H. Leaching studies of chalcopyrite and sphalerite with hypochlorous acid [J]. Metallurgical1 Transactions B, 1987, 18(4): 315-316.
[7] HIROYOSHI N, MIKI H, HIRAJIMA T. Enhancement of chalcopyrite leaching by ferrous ions in acidic ferric sulfate solution [J]. Hydrometallurgy, 2001, 60: 185-l97.
[8] GOMEZ E, BALLESTER A, BLOZQUEZ M L. Silver-catalyzed bioleaching of a chalcopyrite concentrate with mixed cultures of moderately thermophilic microorganisms [J]. Hydrometallurgy, 1999, 51: 37-46.
[9] DEVI N B, MADHUECHHANDA M, SRINIVASA RAO K. Oxidation of chalcopyrite in the presence of manganese dioxide in hydrochloric medium [J]. Hydrometallurgy, 2000, 57: 57-76.
[10] HAVLIK T, DVORSCIKOVA J, LVANOVA Z. Sulphuric acid chalcopyrite leaching using ozone as oxidant [J]. Metal1, 1999, 53(1/2): 57-60.
[11] ANTONIJEVIC M M, JANKOVIC Z D, DIMITRIJERIC M D. Kinetics of chalcopyrite dissolution by hydrogen peroxide in sulphuric acid [J]. Hydrometallurgy, 2004, 71: 329-334.
[12] DREISING D, ABED N. A fundamental study of the reductive leaching of chalcopyrite using metallic iron (part I): Kinetics analysis [J]. Hydrometallurgy, 2002, 66: 37-57.
[13] LAN Xing-hua. Technological evolvement of copper leaching from copper concentration [J]. World Nonferrous Metals, 2004(11): 23-27.
[14] XIE Hong-zhen, HUANG Huai-guo, JIANG Cheng, LIN Hong-han, SUN Peng, LUO Ji-su, ZOU Lai-chang, CHEN Jing-he. A study on copper extraction from chalcopyrite concentrate by acidic hot-pressure oxidation [J]. Mining and Metallurgical Engineering, 2003, 23(4): 54-56.
[15] QIU Ting-sheng, NIE Guang-hua, ZHANG Qiang, CHEN Jing-he, ZOU Lai-chang. Mechanism of oxidation and leaching for copper-bearing gold ores [J].The Chinese Journal of Nonferrous Metalls, 2005, 15(12): 2028-2033. (in Chinese)
Foundation item: Project(50664002) supported by the National Natural Science Foundation of China; Project(00650043) supported by the Natural Science Foundation of Jiangxi Province, China
Corresponding author: QIU Ting-sheng; Tel: +86-797-8312351; E-mail: qiutingsheng@163.com

