PREPARATION AND CLEAVAGE MECHANISM OF CHELATES OF METAL IONS WITH EDTA LINKED TO OLIGODEOXYRIBONUCLEOTIDES
来源期刊:中南大学学报(英文版)1998年第1期
论文作者:Li Xiaoru Wen Zhibin
文章页码:47 - 50
Key words:Oligodeoxyribonucleotide; metal ions; chelate,cleavage
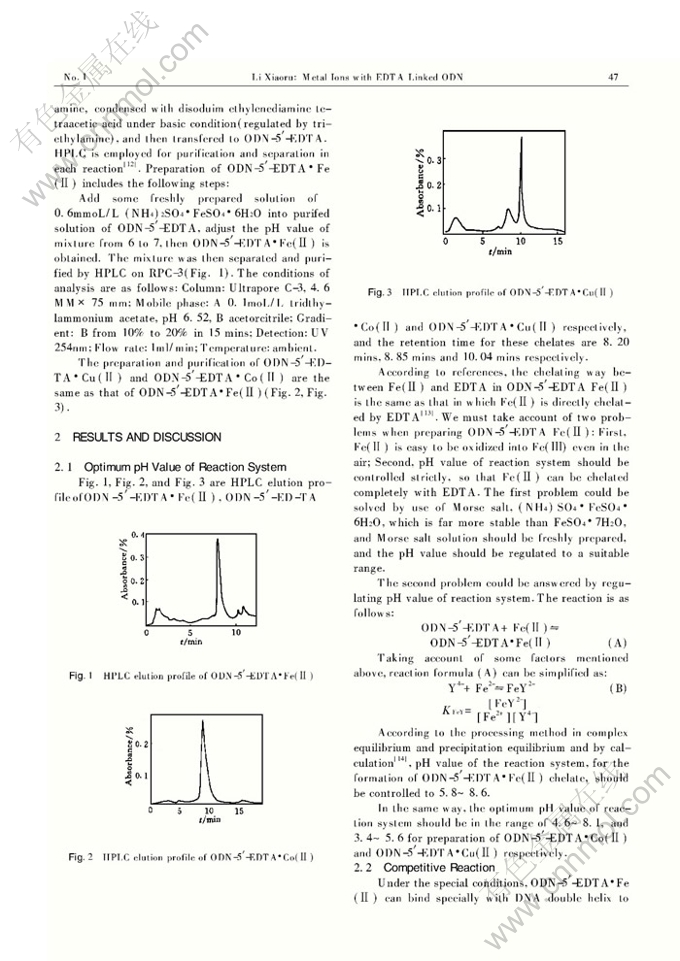
Abstract: The chelates of metal ions with EDTA covalently linked to the 5′-end of oligodeoxyribonuclotides (ODN),i.e,ODN-5′-EDTA・M(Ⅱ),are prepared,in which M(Ⅱ) is Fe(Ⅱ),Co(Ⅱ) or Cu(Ⅱ).The optimum pH value for forming these three chelates is calculated.For ODN-5′-EDTA Fe(Ⅱ) pHvalue is 5.8 to 8.6,pH 4.6~8. 1 for ODN-5′-EDTA Co(Ⅱ),and pH 3.4~5.7 for ODN-5′-EDTA Cu(Ⅱ).Under such pH value conditions neither can Mg(Ⅱ) ion,necessary for cleavage reaction,be competitive with Fe(Ⅱ),Co(Ⅱ) or Cu(Ⅱ) to form ED- TA chelate,nor can it be precipitated.The cleavage mechanism of ODN-5′-EDTA Fe(Ⅱ) for DNA duplex is discussed.Modified ODN binds with DNA duplex in the major groove via hydrogen bond to form triple helix.In the presense of oxygen and reducing agent dithiothreitol,hydroxyl radicals species are generated as intermediates by catalysis of metal ions,and then oxidize the ribo ring and cut the double-stranded DNA at the sites close to the ED- TA・Fe(Ⅱ).