Cu-BTCǰ�����Ʊ�̼�������Ľṹ����������
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2018���5��
�������ߣ����� ���� ����� �°���
����ҳ�룺980 - 988
�ؼ��ʣ��л������Ǽܣ�Cu-BTCǰ���壻̼������������������CO
Key words��metal organic frameworks; Cu-BTC precusor; carbon-based catalyst; low-temperature denitraion; CO
ժ Ҫ����Cu-BTCΪǰ���壬����ˮ�ȷ��Ʊ�̼�������������е������������о���ͨ��X��������(XRD)����������(Raman)��ɨ��羵(SEM)��������ɢX������(EDS)�ȼ�����̼�������Ľṹ���б��������������̼����������������Cu-BTCǰ����İ�������ò������Ҫ������������ͭ�ṹ��ͨ��̼�����������������о����֣�CuOx/C���ֳ���ѵĴ����ԣ������������Ag�IJ���õ���һ������ߡ���Cu/AgĦ����Ϊ6:1����¶�Ϊ600 ��Cʱ��̼�������������ŵĴ����ԣ�������Ч�ʴﵽ100%ʱ�¶ȿɽ�����235 ��C���ô���Ӧ�����У���Ҫ��Cu+������á�
Abstract: Using Cu-BTC prepared by hydrothermal method as precursor, carbon-based catalysts were obtained as model materials for low-temperature DeNOx. These catalysts were characterized by X-ray diffractometry (XRD), Raman spectroscopy, scanning electron microscopy (SEM) and energy dispersive X-ray spectrometry (EDS). The results showed that all carbon-based catalysts held the octahedron shape of Cu-BTC in most parts, and they mainly consisted of face-centered cubic copper. CuOx/C exhibited excellent catalytic activity, and such catalytic activity was further improved with the introduction of Ag. The catalyst with a Cu to Ag mole ratio of 6:1 and an activated temperature of 600 ��C showed the best catalytic performance, and its catalytic denitration rate reached 100% at a temperature as low as 235 ��C. During the catalytic reaction process, Cu+ mainly played a catalytic role.
Trans. Nonferrous Met. Soc. China 28(2018) 980-988
Li ZHANG, Lei HUANG, Yi-hong QIN, Bai-zhen CHEN
School of Metallurgy and Environment, Central South University, Changsha 410083, China
Received 23 November 2016; accepted 22 May 2017
Abstract: Using Cu-BTC prepared by hydrothermal method as precursor, carbon-based catalysts were obtained as model materials for low-temperature DeNOx. These catalysts were characterized by X-ray diffractometry (XRD), Raman spectroscopy, scanning electron microscopy (SEM) and energy dispersive X-ray spectrometry (EDS). The results showed that all carbon-based catalysts held the octahedron shape of Cu-BTC in most parts, and they mainly consisted of face-centered cubic copper. CuOx/C exhibited excellent catalytic activity, and such catalytic activity was further improved with the introduction of Ag. The catalyst with a Cu to Ag mole ratio of 6:1 and an activated temperature of 600 ��C showed the best catalytic performance, and its catalytic denitration rate reached 100% at a temperature as low as 235 ��C. During the catalytic reaction process, Cu+ mainly played a catalytic role.
Key words: metal organic frameworks; Cu-BTC precusor; carbon-based catalyst; low-temperature denitraion; CO
1 Introduction
As a major kind of air pollutants, nitrogen oxides are very harmful to human health since they may lead to photochemical smog, acid rain, ozone depletion and greenhouse effect [1-3]. The selective catalytic reduction (SCR) of NO has been considered as a promising method for NO reduction due to its significant environmental impact. Currently, one of the leading technologies is selective catalytic reduction by ammonia (NH3-SCR) [4-6]. However, the NH3-SCR process is typically expensive due to the high capital investment requirement, injection of ammonia, catalytic toxicity, equipment corrosion and NH3 slip issue [7-9]. As one of the contaminants in exhaust, carbon monoxide is a promising agent showing good reducing effect as well as cost-reduction potential in SCR processes [10,11], thus CO-SCR can be considered as an advantageous technology in the future.
Selective catalytic reduction of NO with CO has been demonstrated over so-called three-way Rh-Pt-Pd catalysts. In the past decade, many studies have been performed to replace noble metal catalysts in view of their high cost and scarce resources [12-14]. Thereafter, various base metal catalysts with high SCR activity at lower temperatures have been found, which are more suitable for coal-fired power plants and industrial application [15-17]. YU et al [18] obtained Cu-catalysts supported with different Ce/Zr mole ratios for the reduction of NO with CO, and their results showed that the dispersion of CuO was influenced by the crystal structure of CeO2-ZrO2 support: once Cu atoms were located in oxygen vacancies, the reduction performance of CuO over NO was improved [18]; though Cu-containing catalysts performed poorly at low temperature, their activity could be enhanced with the addition of other elements, such as Ag atoms. This phenomenon could be a good basis for a new type of bimetallic system if the selectivity of system for NO reduction could be enhanced by the presence of a second metal, such as Ag or Cu.
Metal oxide-supported carbonaceous materials, such as activated carbon (AC), activated carbon fibers (ACFs), carbon nanotubes (CNTs), and ordered mesoporous carbon (OMC), among others, have shown high activity and resistance to SO2 [19-23]. The performance of carbon-supported catalysts is significantly affected by the pore structure and surface properties of the supports. At present, metal organic frameworks (MOFs) consisted of metal ions and polyfunctional organic ligands have attracted considerable attention due to their splendid structures and outstanding properties [24,25], which have been investigated for potential applications in gas storage, separation, catalysis, drug delivery, etc. [26-28]. From the point of crystallology, MOFs may be an affordable approach to generate metal oxides and carbon-based materials [29-31]. For instance, Zn-based MOFs were used to obtain highly porous carbon [32], and Fe-based MOFs were applied as precursor to prepare Fe2O3 as possible anode material for Li-ion batteries [33]. Compared with other means, this synthetic route is advantageous because it is free of surfactant, other template or other complicated additives [34].
Among reported MOFs materials, Cu-benzene- 1,3,5-tricarboxylic acid (Cu-BTC) shows better potential. Because it is formed by paddlewheel secondary builing units which contain plenty of Cu2+ dimers coordinatively, Cu-BTC can not only exhibit high porosity, but also possess a large number of unsaturated metal sites, providing it with powerful catalytic performance in low temperature range [35,36]. WANG et al [37] have studied the potential use of Cu-BTC for gas purification and separation. Recently, Cu-BTC metal-organic framework as a novel catalyst was used for low temperature selective catalytic reduction of NO by NH3, and showed a good catalytic activity with operating-temperature below 300 ��C [38]. However, there has been no report on Cu-BTC used for low-temperature DeNOx by CO.
In this work, we focus our attention on CuOx/C and a series of Cu-AgyOx/C obtained from Cu-BTC. These materials were applied in the selective catalytic reduction of NO with CO. As far as we know, this was the first time that the sample method was used to obtain carbon-based catalysts in the field of denitration. In the present study, carbon-based catalysts were characterized by different instrumental techniques and the effects of reaction conditions (such as mole ratio of Cu to Ag and activated temperature) on the catalytic performance of cabon-based catalysts were also investigated.
2 Experimental
2.1 Preparation of catalysts
2.1.1 Cu-BTC precursor
Cu-BTC was synthesized by the hydrothermal method according to literatures [39,40]. Briefly, 4 mmol of benzebe-1,3,5-tricarboxylic acid (H3BTC, 98%) was dissolved in 20 mL of ethyl alcohol (CH3CH2OH, 99%), and 6 mmol of copper nitrate trihydrate (Cu(NO3)2, 99%) was dissolved in 20 mL of ultrapure water. Then, they were mixed together and stirred for 30 min. Afterwards, the mixture was poured into a 50 mL stainless steel reactor with a polytetrafluoroethylene liner, and the autoclave was put into a 120 ��C oven for 24 h. The precipitated crystals from the solution were allowed to settle down as precipitates. After the removal of top solution, the solid precipitates were purified in 50 mL of ethyl alcohol to remove unreacted chemicals and the process lasted for 30 min. The purified products were filtered and washed with ethyl alcohol (50 mL �� 3) and ultrapure water (50 mL �� 3) repetitively, then dried at 100 ��C for 12 h before test.
2.1.2 CuO catalysts
CuO catalyst was prepared by putting copper nitrate trihydrate at 500 ��C in air for 3 h. Cu-BTC was put in a quartz tube and roasted at 500 ��C for 3 h in air. The two materials were named as CuOn and CuOc.
2.1.3 CuOx/C catalysts
Cu-BTC was put in a quartz tube and roasted at 500 ��C for 3 h in a nitrogen atmosphere and reducing atmosphere (1% of CO in volume), and they were named as CuOx/Cn and CuOx/Cr, respectively.
2.1.4 Cu-AgyOx/C catalysts
Furthermore, Ag element was directly loaded by synthetic exchange solvothermal method. Briefly, 4 mmol of benzebe-1,3,5-tricarboxylic acid (H3BTC, 98%) was dissolved in 20 mL of ethyl alcohol (CH3CH2OH, 99%), 6 mmol of copper nitrate trihydrate (Cu (NO3)2, 99%) and different amounts (2, 1, 0.5, 0.25 mmol) of silver nitrate (AgNO3, 99%) were dissolved in 20 mL of ultrapure water. Then, they were mixed together and stirred for 30 min. The mixture was put in a 50 mL stainless steel reactor with a poly- tetrafluoroethylene liner, and the autoclave was put into a 120 ��C oven for 24 h. The purification process of the precipitation was the same as that of Cu-BTC. The precipitation was decomposed in a reducing atmosphere (1% CO in volume) at 500 ��C for 3 h. Then, the four samples were prepared for test, and they were named as Cu-AgyOx/C (y= 2, 1, 0.5, 0.25).
2.2 Catalyst characterization
X-ray diffraction data were collected on a Rigaku D/Max 2200 diffractometer employing Cu K�� radiation. Raman spectra were recorded by using HR-8 micro Raman spectrometer from French HORIBA Jobin Yvon Company. The 532 nm wavelength laser source was adopted to investigate the M��O bonding situation in MOFs. The scanning electron microscopy (SEM) images were taken on JEOL 3600 electron microscope equipment using a 20 kV energy source under vacuum, coupled with energy dispersive X-ray spectrometry (EDS).
2.3 Catalytic tests
The NO reduction reaction in specific stream was carried out at a fixed bed quartz reactor. For every experiment, 0.5 g of catalyst was put in a quartz tube with a diameter of 12 mm. The space velocity was 128000 mL/(h��g). The experimental equipment consists of three sections: a gas feeding system, a reactor, and a gas analyzer. The gas was mixed through a MF-4B flue gas flow meter. The reaction mixture consisted of 0.04% NO, 0.05% CO, 82.85% N2, and 17.06% O2 in volume. The reactant and product mixtures were analyzed by an on-line gas analytical device made by Germanic MRU Company. The NO conversion was calculated as follows:
XNO={([NO]in-[NOX]out)/[NO]in}��100%
(NOX includes NO and NO2)
3 Results and discussion
3.1 Catalyst characterization
3.1.1 XRD analysis
Figure 1 shows the XRD results of Cu-BTC, CuOn, CuOc, CuOx/Cn, CuOx/Cr and Cu-Ag1Ox/C samples. The characteristic peaks of Cu-BTC were roughly identical to the characteristic peaks of Cu3(BTC)2��3H2O (JCPDS-ICDD 39-1962), confirming the formation of Cu-BTC phase [41]. CuOn and CuOc exhibited the structure of monoclinic CuO (JCPDS-ICDD 44-0706) with their characteristic peaks at 32.4��, 35.5��, 38.7�� and 48.8��. However, the peaks of CuO disappeared since Cu-BTC was decomposed to CuOx/Cn and CuOx/Cr in nitrogen atmosphere and reducing atmosphere. The new positions of the main reflections at 43.6��, 50.7�� and 74.4�� corresponded to the structures of (111), (200) and (220) of face-centered cubic copper (JCPDS-ICDD 04-0836). There were no characteristic diffraction peaks of Cu2O (2�ȡ�36.4��, 42.3�� and 43.3��) and C in the XRD patterns of CuOx/Cn and CuOx/Cr, perhaps because the carbon element and Cu2O were well dispersed. No characteristic peaks of Ag appeared in the XRD patterns of Cu-Ag1Ox/C, perhaps because silver atoms were not densely distributed on the surface of carbon structure, but were well dispersed in the carbon structure.

Fig. 1 XRD patterns of catalysts
3.1.2 Raman spectroscopy analysis
In order to further determine the existing states of copper, silver and carbon element in the carbon-based materials, Raman spectra analyses of CuO/Cr and Cu-Ag1Ox/C were carried out and the results were shown in Fig. 2. The weak Raman peaks of CuO and Cu2O [42] appeared in the wavelength ranging from 100 to 1000 cm-1, demonstrating that a small amount of Cu2+ and Cu+ were well dispersed in the structure of CuOx/Cr and Cu-Ag1Ox/C.
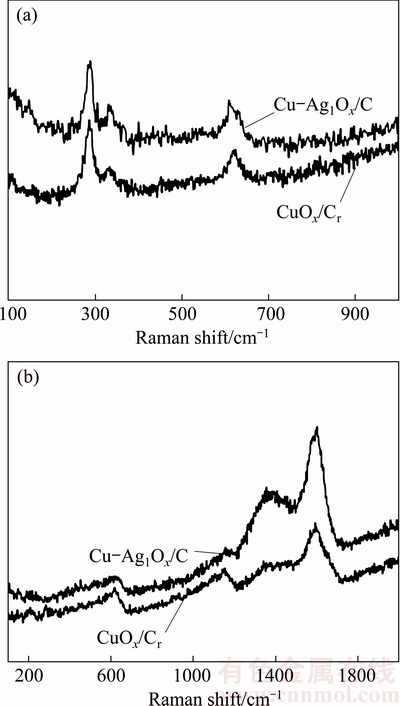
Fig. 2 Raman spectra at wavenumber of 100-1000 cm-1 (a) and 100-2000 cm-1 (b)
The D band at 1361 cm-1 (Fig. 2(b)) is a common feature for the lattice defects of carbon atoms, and the stretching vibrations in sp3 hybridization surface of carbon atoms with disordered structure belong to amorphous carbon. The G band at 1594 cm-1 (Fig. 2(b)) provided information on the stretching vibrations in sp2 hybridization surface of carbon atom with graphitization degree, which belongs to graphite carbon [43]. The calculated IG/ID ratio of carbon in CuOx/Cr was 1.33:1, hence, the carbon material in CuOx/Cr was determined as graphite carbon. The Raman spectra of Cu-Ag1Ox/C showed the same peak positions as those of CuO/Cr, but the strength of peaks was enhanced obviously. It was easy to find that no Raman spectra peak was associated with Ag appeared in Fig. 2.
3.1.3 SEM analysis
The SEM images of the six catalysts were shown in Fig. 3. Cu-BTC had a uniform octahedron shape. CuOn had different sizes of round shapes, which were directly made by Cu (NO3)2��3H2O in the air. The morphology of CuOc obtained by using Cu-BTC as precursor maintained the topography of Cu-BTC in some parts, and tended to be octahedron in shape. Carbon-based catalysts, including CuOx/Cn, CuOx/Cr and Cu-Ag1Ox/C, all held the topography of Cu-BTC in more parts. It can be inferred that metal ions and carbon still played a structure supporting role during the disintegration process of Cu-BTC.
3.1.4 EDS analysis
The results of energy dispersive spectrometer analysis on Cu-Ag1Ox/C and CuO/Cr were shown in Fig. 4. It can be determined that Ag atoms were doped successfully in Cu-Ag1Ox/C by comparing the EDS results of Cu-Ag1Ox/C with those of CuO/Cr (Fig. 4). It can be seen that Ag atoms in Cu-Ag1Ox/C did not produce new Raman spectra peaks, thus Ag atoms only played surface enhancing role in the Raman spectra. Therefore, the speculation that Ag atoms were well dispersed in the carbon structure was correct.

Fig. 3 SEM images of catalysts
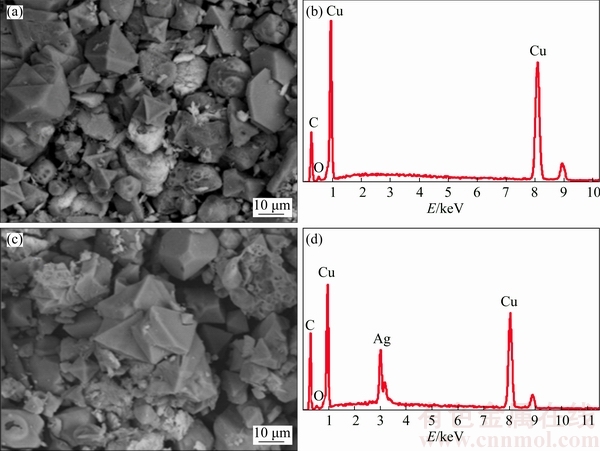
Fig. 4 SEM images (a, c) and corresponding EDS analyses (b, d) of CuOx/Cr (a, b) and Cu-Ag1Ox/C (c, d)
3.2 Catalytic performance
3.2.1 Catalytic activity of different catalysts
NO reduction reaction was performed by putting these catalysts in the quartz tube and letting specific gas flow through the tube. Through programmed temperature control, the temperature went up to 500 ��C from 50 ��C at 2.5 ��C/min. The on-line gas analytical device took notes of the gas composition minute-by-minute after the catalytic reaction began. The NO conversions over CuOn, CuOc, CuOx/Cn, CuOx/Cr and Cu-Ag1Ox/C were described in Fig. 5. CuOn and CuOc had the similar catalytic performance at 500 ��C (87.1% CuOn, 88.6% CuOc). The NO conversions over CuOx/Cn and CuOx/Cr were obviously better than that over single CuO, and it reached 100% at 317 and 310 ��C, respectively. It was clear that carbon-based materials had much better catalytic activity, mainly because carbon, as a carrier, was beneficial for the dispersion of catalytic active sites.
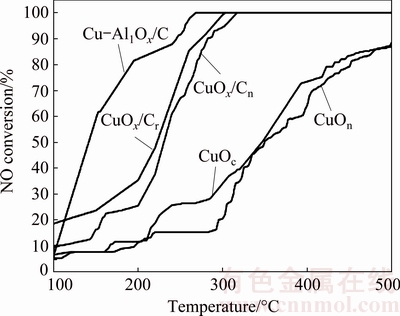
Fig. 5 NO conversion over CuOn, CuOc, CuOx/Cn, CuOx/Cr and Cu-Ag1Ox/C
On the other hand, reducing atmosphere was more prone to produce Cu species in low valence, which was beneficial for forming more oxygen vacancies; hence, the CuOx/Cr synthesized under reducing atmosphere exhibited better catalytic denitration performance when compared with CuOx/Cn synthesized under nitrogen atmosphere. Cu-Ag1Ox/C reached a catalytic denitration rate of 100% at 268 ��C, and its catalytic denitration activity was superior to CuOx/Cr and CuOx/Cn in the whole temperature interval. Thus, it can be inferred that the introduction of Ag to carbon-based copper oxide could improve the catalytic denitration activity, which endowed the catalysts with stronger adaptability and better flexibility at the processing temperature of flue gas.
3.2.2 Effect of different mole ratios of Cu to Ag
Cu-AgyOx/C (y=2, 1, 0.5, 0.25) was investigated to find the mole ratio of Cu to Ag that offered the best performance. The Cu/Ag mole ratio (6: y) was obtained by direct synthetic exchange solvothermal method. It was demonstrated that the NO conversion could reach 100% at 275, 268, 274, 285 ��C for Cu-AgyOx/C with the Cu/Ag mole ratios of 6:2, 6:1, 6:0.5, 6:0.25, respectively (Fig. 6). The catalysts with the addition of Ag atoms performed better than single CuOx/Cr, and the catalytic performance was improved with the increase of Ag until the Cu to Ag mole ratio reached 6:2; afterwards, with the increase of Ag, the catalytic performance was depressed, probably because too many Ag atoms may restrain the main structure of Cu-BTC under direct solvothermal method, leading to a worse structure of the precursor. Furthermore, Cu-Ag1Ox/C would be the best catalyst for CO-NO reaction in our work.
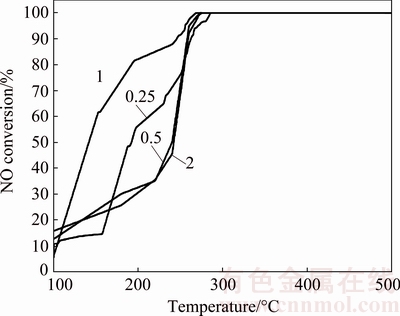
Fig. 6 Denitrification efficiency of Cu-AgyOx/C (y=2, 1, 0.5, 0.25)
3.2.3 Effect of activated temperature
As activated temperature plays an important role for catalysts, it was also considered in this study. The activated CuOx/Cr catalysts were respectively pretreated at 400, 500, 600 ��C for 3 h in advance, and the NO conversions reached 100% at 314, 303, 274 ��C, respectively, as shown in Fig. 7. What��s more, the NO conversion of activated Cu-Ag1Ox/C catalysts reached 100% at 305, 268, 235 ��C respectively under the same pretreated conditions as mentioned above. It was noted that high activated temperature contributed to more activate molecules, therefore, 600 ��C was considered as the best pretreated temperature for our catalysts.
3.3 Reaction mechanism
In order to further understand the catalytic reaction process, CuOx/Cr and Cu-Ag1Ox/C with good catalytic denitration activity were selected, and the changes of their surface topography and structure before and after the catalytic reaction were studied by scanning electron microscopy (SEM) and X-ray diffraction (XRD). There were no obvious changes on the surface topography of CuOx/Cr and Cu-Ag1Ox/C in Fig. 8, and both of them kept a part topography of Cu-BTC with regular octahedrons. However, the structures of catalysts before and after catalytic reaction had changed a lot, as shown in Fig. 9. Zero valence copper was the main state of copper in the catalysts before the reaction. After the catalytic reaction, Cu in zero valence was reduced and Cu2O with cubic structure (JCPDS-ICDD 35-1091) showed XRD peaks at 36��, 38��, 42��, 43��, 61�� and 73��, illustrating that Cu+ mainly played a catalytic role in the CO-NO reaction, in accordance with previous reports [44,45]. The relevant reaction calculations were shown as follows (300 ��C):
CO(g)+2CuO(s)=Cu2O(s)+CO2(g) (1)
��H=145.506 kJ/mol
2NO(g)+2Cu2O(s)=N2(g)+4CuO(s) (2)
��H=-456.071 kJ/mol
CO(g)+Cu2O(s)=2Cu(s)+CO2(g) (3)
��H=-113.56 kJ/mol
2NO(g)+2Cu(s)=N2(g)+2CuO(s) (4)
��H=-488.270 kJ/mol
CO(g)+Ag2O(s)=2Ag(s)+CO2(g) (5)
��H=-254.379 kJ/mol
2NO(g)+4Ag(s)=2Ag2O(s)+N2(g) (6)
��H=-239.602 kJ/mol
2Cu(s)+Ag2O(s)=2Ag(s)+Cu2O(s) (7)
��H=-140.704 kJ/mol
Cu(s)+CuO(s)=Cu2O(s) (8)
��H=-16.015 kJ/mol
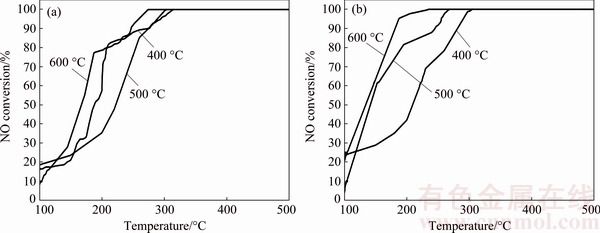
Fig. 7 Denitrification efficiency of CuOx/Cr (a) and Cu-Ag1Ox/C (b) activated at different temperatures

Fig. 8 SEM images of CuOx/Cr and Cu-Ag1Ox/C before and after reaction
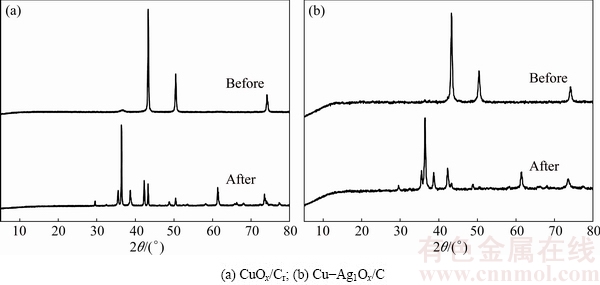
Fig. 9 XRD patterns before and after reaction of catalysts
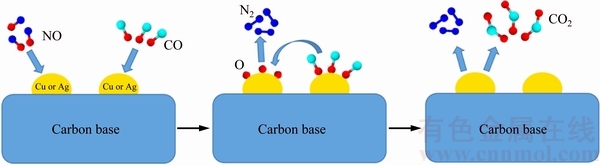
Fig.10 Reaction mechanism of catalytic denitration
The calculations demonstrated that, as an important node in CO-NO reaction, Cu+ could be obtained and react with CO and NO much more easily. Recent research indicated that the interaction between CO and metal oxides only weakened the metal-oxygen bond, thus the catalyst surface was only partially reduced to form activated CO molecule. CO could be adsorbed by Cu+ easily and form Cu+(CO)n (n=1-3) . Additionally, CO could be adsorbed by Ag+ to acquire Ag+(CO)n (n=1-4) or Ag+nCO (n=2-4) [46,47]. Infrared ray study had confirmed that NO was adsorbed on the surface of catalyst, in this case, the N��O bond strength was weakened and even dissociated on the surface, yielding adsorbed O and N atoms that led to the formation of N2 and O2. NO was also absorbed by Cu+ easily to obtain Cu+(NO)n (n=1-2), producing adsorbed oxygen [48]. While adsorbed oxygen was obtained by NO on the surface of Ag atoms, different activated O species (O��, O��, O��) would be produced in agreement with intensive activated sites for low temperature DeNOx [49]. Hence, the reaction mechanism might be Langmuir- Hinshelwood mechanism, as described in Fig. 10.
4 Conclusions
1) Carbon-based catalysts prepared from Cu-BTC precursor mainly consisted of face-centered cubic copper; a small amount of Cu2+ and Cu+ were well dispersed in the carbon skeleton that determined as graphite carbon structure. Cu-BTC had a uniform octahedron shape, and carbon-based catalysts, including CuOx/Cn, CuOx/Cr and Cu-Ag1Ox/C, all held the topography of Cu-BTC in more parts.
2) The NO conversion over CuOx/Cn and CuOx/Cr was obviously better than that over single CuO, and it reached 100% at 317 and 310 ��C, respectively, illustrating that MOFs Cu-BTC is a better precursor candidate in the catalytic field. The introduction of Ag was beneficial for improving the catalytic activity, and the catalytic denitration rate of Cu-Ag1Ox/C reached 100% only at 268 ��C. The mole ratio of Cu to Ag and the activated temperature played important roles on the catalytic performance. The optimized conditions were determined as Cu/Ag ratio of 6:1 and activated temperature of 600 ��C, under which the catalytic denitration rate reached 100% at a temperature as low as 235 ��C.
3) The reaction mechanism was investigated, and Cu+ was found to occupy the main activated sites. The reaction of low temperature DeNOx was greatly improved with the introduction of Ag atoms, because NO would be rapidly consumed on the surface of Ag atoms to produce more activated O species. The reaction mechanism gave a good model for Cu+- and Ag-loaded catalyst in CO-NO reaction. It was noted that MOFs would be a good precursor while investigating multi- metal and carbon-based metal oxides used for the future design materials in many applications.
References
[1] MCDONALD-BULLER E C, KIMURA Y, CRAIG M, MCGAUGHEY G, ALLEN D, WEBSTER M. Dynamic management of NOx and SO2 emissions in the Texas and mid-Atlantic electric power systems and implications for air quality [J]. Environmental Science & Technology, 2016, 50(3): 1611-1619.
[2] STOLARSKI R S, DOUGLASS A R, OMAN L D, WAUGH D W. Impact of future nitrous oxide and carbon dioxide emissions on the stratospheric ozone layer [J]. Environmental Research Letters, 2015, 10(3): 034011.
[3] ZHANG Li, QIN Yi-hong, CHEN Bai-zhen, PENG Ya-guang, HE Han-bing, YUAN Yi. Catalytic reduction of SO2 by CO over CeO2-TiO2 mixed oxides [J]. Transactions of Nonferrous Metals Society of China, 2016, 26(11): 2960-2965.
[4]  N, LOPEZ-ANTON M A,
N, LOPEZ-ANTON M A,  M R. Mercury oxidation in catalysts used for selective reduction of NOx (SCR) in oxy-fuel combustion [J]. Chemical Engineering Journal, 2016, 285: 77-82.
M R. Mercury oxidation in catalysts used for selective reduction of NOx (SCR) in oxy-fuel combustion [J]. Chemical Engineering Journal, 2016, 285: 77-82.
[5] ANDREOLI S, DEORSOLA F A, GALLETTI C, PIRONE R. Nanostructured MnOx catalysts for low-temperature NOx-SCR [J]. Chemical Engineering Journal, 2015, 278: 174-182.
[6] WANG Jian-cheng, TIAN Dong, HAN Li-na, CHANG Li-ping, BAO Wei-ren. In situ synthesized Cu-ZSM-5/cordierite for reduction of NO [J]. Transactions of Nonferrous Metals Society of China, 2011, 21(2): 353-358
[7] SHI Xiao-yan, LIU Fu-dong, XIE Li-juan, HE Hong. NH3-SCR performance of fresh and hydrothermally aged Fe-ZSM-5 in standard and fast selective catalytic reduction reactions [J]. Environmental Science & Technology, 2013, 47(7): 3293-3298.
[8] ZHAO Wei, ZHONG Qin, PAN Yan-xiao, ZHANG Rui. Systematic effects of S-doping on the activity of V2O5/TiO2 catalyst for low-temperature NH3-SCR [J]. Chemical Engineering Journal, 2013, 228(28): 815-823.
[9] SULTANA A, SASAKI M, SUZUKI K, HAMADA H. Tuning the NOx conversion of Cu-Fe/ZSM-5 catalyst in NH3-SCR [J]. Catalysis Communications, 2013, 41(21): 21-25.
[10] KANTCHEVA M, MILANOVA M, MAMETSHERIPOV S. In situ FT-IR spectroscopic investigation of gold supported on tungstate zirconia as catalyst for CO-SCR of NOx [J]. Catalysis Today, 2012, 191(1): 12-19.
[11] LI Jun, LUO Guo-hua, CHU Yue, WEI Fei. Experimental and modeling analysis of NO reduction by CO for a FCC regeneration process [J]. Chemical Engineering Journal, 2012, 184(2): 168-175.
[12] BAIDYA T, BERA P, MUKRI B D, HEGDE M S. DRIFTS studies on CO and NO adsorption and NO plus CO reaction over Pd2+-substituted CeO2 and Ce0.75Sn0.25O2 catalysts [J]. Journal of Catalysis, 2013, 303: 117-129.
[13] C NSUL J M D, COSTILLA I, GIGOLA C E, BAIBICH I M. NO reduction with CO on alumina-modified silica-supported palladium and molybdenum-palladium catalysts [J]. Applied Catalysis A: General, 2008, 339(2): 151-158.
NSUL J M D, COSTILLA I, GIGOLA C E, BAIBICH I M. NO reduction with CO on alumina-modified silica-supported palladium and molybdenum-palladium catalysts [J]. Applied Catalysis A: General, 2008, 339(2): 151-158.
[14] LI Jun, WANG Shan, ZHOU Li, LUO Guo-hua, WEI Fei. NO reduction by CO over a Fe-based catalyst in FCC regenerator conditions [J]. Chemical Engineering Journal, 2014, 255(7): 126-133.
[15] SPASSOVA I, VELICHKOVA N, NIHTIANOVA D, KHRISTOVA M. Influence of Ce addition on the catalytic behavior of alumina-supported Cu-Co catalysts in NO reduction with CO [J]. Journal of Colloid & Interface Science, 2011, 354(354): 777-84.
[16] G RA-MAREK K, DATKA J. IR studies of coadsorption of CO and NO on Co2+ and Cu+ sites in zeolites [J]. Vibrational Spectroscopy, 2011, 57(1): 148-151.
RA-MAREK K, DATKA J. IR studies of coadsorption of CO and NO on Co2+ and Cu+ sites in zeolites [J]. Vibrational Spectroscopy, 2011, 57(1): 148-151.
[17] YU Qiang, WU Xiao-xia, YAO Xiao-jiang, LIU Bin, GAO Fei, WANG Jia-ming, DONG Lin. Mesoporous ceria�Czirconia�Calumina nanocomposite-supported copper as a superior catalyst for simultaneous catalytic elimination of NO-CO [J]. Catalysis Communications, 2011, 12(14): 1311-1317.
[18] YU Qiang, LIU Lian-jun, DONG Li-hui, LI Dan, LIU Bin, GAO Fei, SUN Ke-qin, DONG Lin, CHEN Yi. Effects of Ce/Zr ratio on the reducibility, adsorption and catalytic activity of CuO/CexZr1-xO2/ ��-Al2O3 catalysts for NO reduction by CO [J]. Applied Catalysis B: Environmental, 2010, 96(3): 350-360.
[19] CHEN Jing-huan, CAO Fei-fei, QU Rui-yang, GAO Xiang, CEN Ke-fa. Bimetallic cerium-copper nanoparticles embedded in ordered mesoporous carbons as effective catalysts for the selective catalytic reduction of NO with NH3 [J]. Journal of Colloid and Interface Science, 2015, 456: 66-75.
[20] ZHANG Deng-song, ZHANG Lei, SHI Li-yi, FANG Cheng, LI Hong-rui, GAO Rui-hua, HUANG Lei, ZHANG Jian-ping. In situ supported MnOx-CeOx on carbon nanotubes for the low-temperature selective catalytic reduction of NO with NH3 [J]. Nanoscale, 2013, 5: 1127-1136.
[21] ZHANG Deng-song, ZHANG Lei, FANG Cheng, GAO Rui-hua, QIAN Yi-lei, SHI Li-yi, ZHANG Jian-ping. MnOx-CeOx/CNTs pyridine-thermally prepared via a novel in situ deposition strategy for selective catalytic reduction of NO with NH3 [J]. RSC Advances, 2013, 3: 8811-8819.
[22] HAN Jin, ZHANG Deng-song, MAITARAD Phornphimon, SHI Li-yi, CAI Si-xiang, LI Hong-rui, HUANG Lei, ZHANG Jian-ping. Fe2O3 nanoparticles anchored in situ on carbon nanotubes via an ethanol-thermal strategy for the selective catalytic reduction of NO with NH3 [J]. Catalysis Science & Technology, 2015, 5: 438-446.
[23] CAO Fei-fei, CHEN Jing-huan, LIU Chang-lei, NI Ming-jiang, GAO Xiang, CEN Ke-fa. Synthesis, characterization and catalytic performances of Cu- and Mn- containing ordered mesoporous carbons for the selective catalytic reduction of NO with NH3 [J]. Catalysis Science & Technology, 2015, 5: 1267-1279.
[24] BRADSHAW D, GARAI A, HUO J. Metal-organic framework growth at functional interfaces: Thin films and composites for diverse applications [J]. Chemical Society Reviews, 2012, 41(6): 2344-2381.
[25] AHMED I, JUN J W, JUNG B K, JHUN S H. Adsorptive denitrogenation of model fossil fuels with Lewis acid-loaded metal�Corganic frameworks (MOFs) [J]. Chemical Engineering Journal, 2014, 255(6): 623-629.
[26] GALLO M, GLOSSMAN-MITNIK D. Fuel gas storage and separations by metal organic frameworks: simulated adsorption isotherms for H2 and CH4 and their equimolar mixture [J]. The Journal of Physical Chemistry C, 2009, 113(16): 6634-6642.
[27] FARRUSSENG D, AGUADO S, PINEL C. Metal-organic frameworks: Opportunities for catalysis [J]. Angewandte Chemie International Edition, 2009, 48(41): 7502-7513.
[28] DHAKSHINAMOORTHY A, ALVARO M, GARCIA H. Deactivation of Cu3(BTC)2 in the synthesis of 2-phenylquinoxaline [J]. Catalysis Letters, 2015, 145(8): 1600-1605.
[29] DAS R, PACHFULE P, BANERJEE R, PODDAR P. Metal and metal oxide nanoparticle synthesis from metal organic frameworks (MOFs): Finding the border of metal and metal oxides[J]. Nanoscale, 2012, 4(2): 591-599.
[30] DEKRAFFT K E, WANG Cheng, LIN Wen-bin. Metal-organic framework templated synthesis of Fe2O3/TiO2 nanocomposite for hydrogen production [J]. Advanced Materials, 2012, 24(15): 2014-2018.
[31] KIMITSUKA Y, HOSONO E, UENO S, FUJIHARA S. Fabrication of porous cubic architecture of ZnO using Zn-terephthalate MOFs with characteristic microstructures [J]. Inorganic Chemistry, 2013, 52(24): 14028-14033.
[32] PACHFULE P, BISWAL B P, BANERJEE R. Control of porosity by using isoreticular zeolitic imidazolate frameworks (IRZIFs) as a template for porous carbon synthesis [J]. Chemistry: A European Journal, 2012, 18(36): 11399-11408.
[33] XU Xiao-dong, CAO Rui-guo, JEONG S, CHO J. Spindle-like mesoporous ��-Fe2O3 anode material prepared from MOF template for high-rate lithium batteries [J]. Nano Letters, 2012, 12(9): 4988-4991.
[34] SALAVATI-NIASARI M, DAVAR F, FERESHTEH Z. Synthesis and characterization of ZnO nanocrystals from thermolysis of new precursor [J]. Chemical Engineering Journal, 2009, 146(3): 498-502.
[35] KUMAR R S, KUMAR S S, KULANDAINATHAN M A. Highly selective electrochemical reduction of carbon dioxide using Cu based metal organic framework as an electrocatalyst [J]. Electrochemistry Communications, 2012, 25: 70-73.
[36] SCHLICHTE K, KRATZKE T, KASKEL S. Improved synthesis, thermal stability and catalytic properties of the metal-organic framework compound Cu3(BTC)2 [J]. Microporous and Mesoporous Materials, 2004, 73: 81-88.
[37] WANG Qing-min, SHEN Dong-min, B��LOW M, LAU M L, DENG Shu-guang, FITCH F R, LEMCOFF N O, SEMANSCIN J. Metallo-organic molecular sieve for gas separation and purification [J]. Microporous and Mesoporous Materials, 2002, 55: 217-230.
[38] LI Chun-yan, SHI Yong, ZHANG Hao, ZHAO Qi-dong, XUE Fang-hong, LI Xin-yong. Cu-BTC metal-organic framework as a novel catalyst for low temperature selective catalytic reduction (SCR) of NO by NH3: Promotional effect of activation temperature [J]. Integrated Ferroelectrics, 2016, 172(1): 169-179.
[39] FERREIRA A F P, SANTOS J C, PLAZA M G, RODRIGUES A E. Suitability of Cu-BTC extrudates for propane�Cpropylene separation by adsorption processes [J]. Chemical Engineering Journal, 2011, 167(1): 1-12.
[40] VISHNYAKOV A, RAVIKOVITCH P I, NEIMARK A V, WANG Qing-min. Nanopore structure and sorption properties of Cu-BTC metal-organic framework (the presentation) [J]. Nano Letters, 2003, 3(6): 713-718.
[41] MARX S, KLEIST W, BAIKER A. Synthesis, structural properties, and catalytic behavior of Cu-BTC and mixed-linker Cu-BTC-PyDC in the oxidation of benzene derivatives [J]. Journal of Catalysis, 2011, 281(1): 76-87.
[42] YAO Wei-tang, YU Shu-hong, ZHOU Yong, JIANG Jie. Formation of uniform CuO nanorods by spontaneous aggregation: Selective synthesis of CuO, Cu2O, and Cu nanoparticles by a solid-liquid phase arc discharge process [J]. The Journal of Physical Chemistry B, 2005, 109(29): 14011-6.
[43] KUDIN K N, OZBAS B, SCHNIEPP H C, PRUD��HOMME R K, AKSAY I A, CAR R. Raman spectra of graphite oxide and functionalized grapheme sheets [J]. Nano Letters, 2008, 8(1): 36-41.
[44] LV Yuan-yuan, LIU Li-chen, ZHANG Hong-liang, YAO Xiao-jiang, GAO Fei, YAO Kai-an, DONG Lin, CHEN Yi. Investigation of surface synergetic oxygen vacancy in CuO-CoO binary metal oxides supported on ��-Al2O3 for NO removal by CO [J]. Journal of Colloid and Interface Science, 2013, 390(1): 158-169.
[45] PATEL A, SHUKLA P, RUFFORD T E, RUDOLPH V, ZHU Zhong-hua. Selective catalytic reduction of NO with CO using different metal-oxides incorporated in MCM-41 [J]. Chemical Engineering Journal, 2014, 255(7): 437-444.
[46] MEYER F, CHEN Yu-min, ARMENTROUT P B. Sequential bond energies of Cu(CO) +x and Ag(CO) + x (x=1-4) [J]. Journal of the American Chemical Society, 1995, 117(14): 4071-4081.
[47] ZHOU Ming-fei, ANDREWS L. Infrared spectra and density functional calculations of Cu(CO)+1�C4, Cu(CO)1�C3, and Cu(CO) -1�C3 in solid neon [J]. Journal of Chemical Physics, 1999, 111(10): 4548-4557.
[48] BRAND H V, REDONDO A, HAY P J. Theoretical studies of CO and NO adsorption on Cu+-ZSM [J]. The Journal of Physical Chemistry B, 1997, 101(39): 7691-7701.
[49] QIAN Min, LIAUW M A, EMIG G. Formaldehyde synthesis from methanol over silver catalysts [J]. Applied Catalysis A: General, 2003, 238(2): 211-222.
�� ������ �ڣ�����죬�°���
���ϴ�ѧ ұ���뻷��ѧԺ����ɳ 410083
ժ Ҫ����Cu-BTCΪǰ���壬����ˮ�ȷ��Ʊ�̼�������������е������������о���ͨ��X��������(XRD)����������(Raman)��ɨ��羵(SEM)��������ɢX������(EDS)�ȼ�����̼�������Ľṹ���б��������������̼����������������Cu-BTCǰ����İ�������ò������Ҫ������������ͭ�ṹ��ͨ��̼�����������������о����֣�CuOx/C���ֳ���ѵĴ����ԣ������������Ag�IJ���õ���һ������ߡ���Cu/AgĦ����Ϊ6:1����¶�Ϊ600 ��Cʱ��̼�������������ŵĴ����ԣ�������Ч�ʴﵽ100%ʱ�¶ȿɽ�����235 ��C���ô���Ӧ�����У���Ҫ��Cu+������á�
�ؼ��ʣ��л������Ǽܣ�Cu-BTCǰ���壻̼������������������CO
(Edited by Wei-ping CHEN)
Foundation item: Project (738010004) supported by the Project of Low Concentration Sulfur Dioxide Flue Gas Treatment, China; Project (2017GK4010) supported by the Scientific and Technological Breakthrough and Major Achievements Transformation of Strategic Emerging Industries of Hunan Province in 2017, China
Corresponding author: Yi-hong QIN; Tel: +86-731-88830216; E-mail: qinyihong@163.com
DOI: 10.1016/S1003-6326(18)64734-X

