


Trans. Nonferrous Met. Soc. China 22(2012) 2158-2163
Effect of Sn substitution and heat treatment on microstructure and microhardness of Co38Ni34Al28-xSnx magnetic shape memory alloys
SU Jia-jia1, 2, XIE Zhi-wei1, YANG Yuan-zheng1
1. Faculty of Materials and Energy, Guangdong University of Technology, Guangzhou 510006, China;
2. Guangdong Polytechnic College, Guangzhou 510095, China
Received 16 December 2011; accepted 23 May 2012
Abstract: Sn was used to replace Al in Co38Ni34Al28 alloy. The microstructure and microhardness of Co38Ni34Al28-xSnx (x=0, 1, 2, 3) magnetic shape memory alloys were investigated at different heat treatment temperatures (1373 K, 1473 K, and 1573 K) for 2 h. The results show that more Sn substitution reduces the content of ��-phase and a partial phase of martensite can be obtained in Co38Ni34Al28-xSnx (x=1, 2, 3) alloys after treatment at 1573 K for 2 h. The maximum martensite phase appears when 2% Al is substituted by Sn. The reverse martensitic transformation temperature of Co38Ni34Al28-xSnx alloys increases at x=1 and 2, then decreases as x=3. As the content of Sn and the temperature increase, the microhardness will increase.
Key words: magnetic shape memory alloy; Sn substitution; Co-Ni-Al alloy; microstructure; martensitic transformation
1 Introduction
Ferromagnetic shape memory alloys (FSMAs) have been attracted considerable attention due to their potential applications in magneto-mechanical actuators [1]. Although Ni2MnGa alloy is the most commonly used FSMAs, but other alloys, like Fe-Pd [2], Fe-Pt [3], Ni-Mn-(Al,Sn,Sb,In) [4,5], Co-Ni-(Al,Ga) [6], Ni-Fe-(Al,Ga) [7-9] and Cu-Mn-(Al,Ga) [10,11] alloys, have been also reported as new FSMAs candidates. The martensitic phase transformation in the ferromagnetic state in Co-Ni-Al alloy system was firstly found by Oikawa��s group [12]. These alloys underwent martensitic transformation from B2-type ��-phase to L10-type martensite. The martensitic transformation temperature and magnetic transition temperature can be controlled within a wide range of temperature by adjusting the composition [13], which makes Co-Ni-Al alloys become promising FSMAs. In order to get large magnetic-field-induced-stain (MFIS) in Co-Ni-Al alloys, the fourth element is added to change the phase transformation temperature and the magnetic properties [14]. It was found that the substitution of suitable content of Sn for Ga in Ni-Mn-Ga alloys leads to the saturation magnetization increasing significantly [15]. A recent study showed that the introduction of Sn into Co-Ni-Al alloys could enhance the temperature of martensitic transition. In this work, the effect of Sn on the martensitic phase transformation and the microhardness of Co-Ni-Al alloys was investigated.
2 Experimental
A series of Co38Ni34Al28-xSnx (x=0, 1, 2, 3) alloys were prepared by arc-melting mixture of Co (99.8%), Ni (99.98%), Al (99%) and Sn (99.9%) in argon atmosphere. The alloys were melted four times in a water-cooled copper crucible for homogenization and then suction cast into alloy sheet with dimensions of 1 mm?5 mm?15 mm. Then the ingots were treated at 1373 K, 1473 K and 1573 K for 2 h, respectively, and quenched in ice water. An optical microscope was used to examine the microstructure. D/max-IIA X-ray diffractometer was used to identify the phase structure of the alloys with Cu K�� radiation. A MVK-H3 microscopic hardness meter was used to measure the microhardness under 1.96 N for 15 s.
3 Results
3.1 Microstructure of Co38Ni34Al28-xSnx (x=0, 1, 2, 3) alloys
The microstructures of Co38Ni34Al28 alloy heated at 1473 K, and 1573 K respectively, are shown in Fig. 1. In the as-cast alloy, the primary phase is ��-phase which is surrounded by ��+�� eutectic phase structure. After being treated at 1473 K, ��-phase could be still observed with an average diameter less than 50 ��m, while the surrounding phase becomes only ��-phase. When the alloy was treated at 1573 K, the size of ��-phase is larger than 50 ��m and the amount of ��-phase is reduced. It seems that the higher treatment temperature helps ��-phase to dissolute into ��-phase. However, no martensite in this alloy was obtained after heat treatment at these heating temperatures.
In order to get martensite, Sn was introduced into Co38Ni34Al28-xSnx (x=1, 2, 3) alloys and their microstructure are shown in Fig. 2, Fig. 3 and Fig. 4.
From Fig. 2 it is known that the microstructure of as-cast Co38Ni34Al27Sn1 alloy is composed of body- centered B2 phase or called ��-phase and ��-phase. After the alloy was treated at 1373 K, the microstructure exhibits ��-phase with grain size of about 25 ��m. It can be also observed that ��-phase can surround ��-grains or appear in the middle of ��-grain. After being further treated at 1473 K, the ��-phase decreases but still surrounds or appears inside ��-grains. Finally, when the alloy was treated at 1573 K, it can also found that the content of ��-phase reduces due to its dissolution into ��-phase. By comparison of the microstructure between CoNiAl and CoNiAlSn alloys, a small portion of lamellar martensite could be formed after being treated at 1573 K due to introduction of 1% of Sn.

Fig. 1 Microstructures of Co38Ni34Al28 alloy: (a) As-cast; (b) Treated at 1473 K; (c) Treated at 1573 K

Fig. 2 Microstructures of Co38Ni34Al27Sn1 alloy: (a) As-cast; (b) Treated at 1373 K; (c) Treated at 1473 K; (d) Treated at 1573 K

Fig. 3 Microstructure of Co38Ni34Al26Sn2 alloy: (a) As-cast; (b) Treated at 1373 K; (c) Treated at 1473 K; (d) Treated at 1573 K
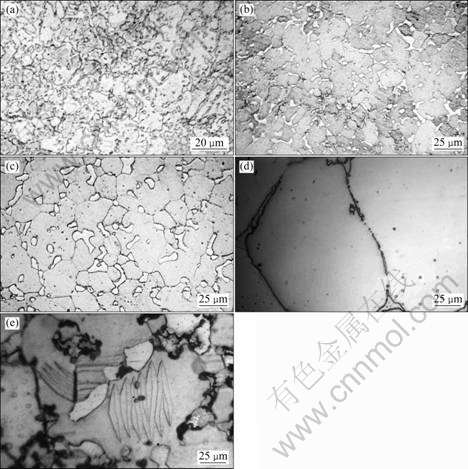
Fig. 4 Microstructures of Co38Ni34Al25Sn3 alloy: (a) As-cast; (b) Treated at 1373 K; (c) Treated at 1473 K; (d) Treated at 1573 K; (e) Treated at 1573 K with martensite
Figure 3 shows the microstructures of as-cast and treated Co38Ni34Al26Sn2 at 1373 K, 1473 K and 1573 K respectively. It is shown that higher treatment temperature will result in more and larger ��-phase and less ��-phase. Similarly, after being treated at 1573 K, the lamellar martensite structure can be clearly observed in Co38Ni34Al26Sn2 alloy. By comparison of the microstructure of the alloy with 1% and 2% Sn, it is found that the alloy with 2% Sn has more volume fraction of martensite after being treated at 1573 K.
Figure 4 shows the microstructures of as-cast and treated alloys at 1373 K, 1473 K and 1573 K, respectively, with 3% Sn. It can be seen that a higher treatment temperature induces more and larger ��-phase and less ��-phase. By comparison of microstructures in Fig. 3(d) and Fig. 4(e), it is observed that when alloy was treated at 1573 K, the lamellar martensite can be also formed, but the amount of martensite is reduced.
Figure 5 shows XRD patterns of Co38Ni34Al28-xSnx (x=1, 2, 3) alloys treated at 1573 K for 2 h. It can be confirmed that Co38Ni34Al27Sn1 alloy consists of martensite, B2 phase and ��-phase. However, for Co38Ni34Al25Sn2 and Co38Ni34Al25Sn3 alloys, XRD peaks of the martensite and B2 phase can be clearly observed. But the characteristic XRD peaks of ��-phase become very weak.
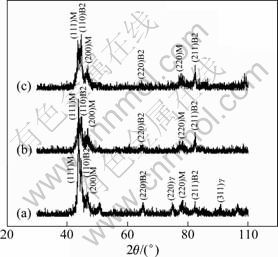
Fig. 5 XRD patterns of Co38Ni34Al28-xSnx alloys treated at 1573 K for 2 h: (a) Co38Ni34Al27Sn1; (b) Co38Ni34Al26Sn2; (c) Co38Ni34Al25Sn3
3.2 DSC curves of Co38Ni34Al28-xSnx (x=0, 1, 2, 3) alloys
From the DSC curves of Co38Ni34Al28-xSnx (x=0, 1, 2, 3) alloys treated at 1573 K for 2 h in Fig. 6, it is known that there are three endothermic peaks in DSC curves. In the heating process, reverse intermartensitic transformation first happens with a small endothermic peak. Very recent research [16] has attributed it to the intermartensitic transformation restrained by the residual stress. Then the second endothermic peak which falls sharply and rises slowly corresponds to reverse martensitic transformation. A final weaker endothermic peak corresponds to reverse pre-martensitic transformation. By comparison, we know that the reverse martensitic transformation temperature of Co38Ni34- Al28-xSnx (x=0, 1, 2, 3) alloys increases first and then decreases when x increases from 1 to 3.
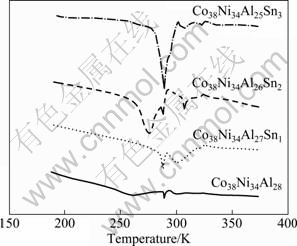
Fig. 6 DSC curves of Co38Ni34Al28-xSnx (x=0, 1, 2, 3) alloys treated at 1573 K
3.3 Microhardness of Co38Ni34Al28-xSnx (x=0, 1, 2, 3) alloys
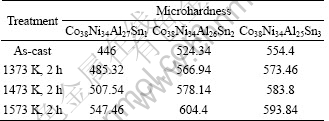
Table 1 shows microhardness of Co38Ni34Al28-xSnx (x=0, 1, 2, 3) alloys at different states. It is found that microhardness will increase as the treatment temperature increases. For example, the microhardness of Co38Ni34Al27Sn1 alloy increases from HV446.00 to HV485.32, HV507.54 and HV547.46 when treated at 1373, 1473 and 1573 K. Besides, at the same treatment temperature, more Sn substitution will improve the microhardness. For example, when treated at 1473 K, as x rises from 1 to 3, the microhardness of Co38Ni34Al28-xSnx increases from HV507.54, HV578.14 to HV583.80. However, there is one exception that the hardness of Co38Ni34Al26Sn2 alloy is higher than that of Co38Ni34Al25Sn3 alloy due to the higher portion of martensite in Co38Ni34Al26Sn2 alloy.
Table 1 Microhardness of CoNiAlSn alloys (HV0.2)
4 Discussion
Figure 7 shows the isothermal section of Co-Ni-Al diagram at 1373 K and 1573 K [17]. From the isothermal section, when treatment temperature is reduced, ��-phase with A1 structure (disordered-FCC) occurs in the region with a low aluminum content and the two-phase region of ��- and ��-phases widens. As the treatment temperature increases, the boundary between ��-phase region and two-phase region of ��- and ��-phases will shift to low aluminum area. The volume fraction of ��-phase will decrease as ��-phase region widens. Moreover, the increase of heat treatment temperature helps ��-phase to dissolve and the grains of ��-phase to become large in Co38Ni34Al28-xSnx alloys.
From Fig. 7, Co38Ni34Al28 alloy has �� (B2) structure at 1473 K and 1573 K. According to the experimental results, there exists more or less ��-phase in all the samples, which indicates that the ��-phase dissolves slowly into ��-phase. The addition of Sn causes the total content of Al to decrease, which may cause the ��+�� region to widen with the increase of Sn content. At the same time, Sn may also lead ��-phase to dissolve more quickly into ��-phase. That is why as Sn content increases, the volume fraction of ��-phase decreases in the Co38Ni34Al28-xSnx alloys.
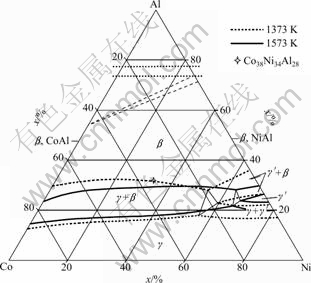
Fig. 7 Isothermal section diagram of Co-Ni-Al alloy at 1373 K and 1573 K
It is known that ��-phase with B2 (ordered-BCC) structure in the alloys can transform into ��-phase with L10 (ordered-FCC) structure above or below room temperature. Previous studies have shown that the martensitic transformation temperature in the ��+�� two-phase alloys rose up with the increase of treatment temperature. With the same alloy composition, the higher the treatment temperature, the less the volume fraction of ��-phase. Because of ��-phase solution, contents of Co and Ni in ��-phase increase as the treatment temperature increases, which will lead to the electron-to-atom-ratio (e/a) in ��-phase and the martensitic transformation temperature increasing. Therefore, the martensitic transformation occurs above room temperature.
As Sn content increases to 3%, the ��-phase grains grow up very quickly, especially at a higher treatment temperature. Therefore, the transformation process and martensitic transformation temperature will be affected and consequently the microstructure of Co38Ni34Al25Sn3 alloys is mainly composed of ��-phase after being treated at 1573 K.
Generally, the microhardness of alloys is associated with the alloy phase, alloying elements and their content, precipitates, etc. The microhardness of these alloys is mainly determined by the microhardness of the ordered ��-phase because the volume fraction of plastic ��-phase is small. The microhardness of these alloys increases with treatment temperature due to the dissolution of more ��-phase into ��-phase. After heat treatment, the ordered BCC ��-phase can transform into L10 thermoelastic martensite phase with a higher hardness. Therefore, the microhardness of the alloys will further increase. That is why the microhardness values of Co38Ni34Al27Sn1 and Co38Ni34Al26Sn2 alloys increase more obviously compared with Co38Ni34Al25Sn3 alloy after heat treatment at 1573 K.
5 Conclusions
1) The substitution of Sn for Al in Co38Ni34Al28-xSnx alloys causes the aluminum content to decrease and the content of ��-phase to decrease. The increase in heat treatment temperature helps ��-phase to dissolve into ��-phase and the ��-phase grain to grow up in Co38Ni34Al28-xSnx alloys.
2) Martensite was obtained in Co38Ni34Al28-xSnx (x=1, 2, 3) alloys by heat treatment at 1573 K for 2 h and then quenching in ice-water. The maximum martensite could be obtained in Co38Ni34Al26Sn2 alloy.
3) The microhardness of Co38Ni34Al28-xSnx alloys has a tendency of increasing with the increase in Sn content and heat treatment temperature. This effect will be more obvious when a part of ��-phase transforms into martensite phase.
4) Substitution of Sn for Al in Co38Ni34Al28-xSnx alloys causes their reverse martensitic transformation temperatures to increase at x=1 and x=2 and then to decrease at x=3.
References
[1] ENKOVARRA J, AYUELA A, ZAYAK A T, ZAYAK A T, ENTEL P, NORDSTROM L, DUBE M, JALKANEN J, IMPOLA J, NIEMINEN R M. Magnetically driven shape memory alloys [J]. Materials Science and Engineering A, 2004, 378(1-2): 52-60.
[2] WUTTIG M, LIU L, TSUCHIYA K, JAMES R D. Occurrence of ferromagnetic shape memory alloys [J]. Journal of Applied Physics, 2000, 87(9): 4707-4711.
[3] ULLAKKO K, HUANG J K, KANTNER C, OHANDLEY R C, KOKORIN V V. Large magnetic-field-induced strains in Ni2MnGa single crystals [J]. Applied Physics Letters, 1996, 69(13): 1966-1968.
[4] GEJIMA F, SUTOU Y, KAINUMA R, ISHIDA K. Magnetic transformation of Ni2AlMn Heusler-type shape memory alloys [J]. Metallurgical and Materials Transactions A, 1999, 30(10): 2721-2723.
[5] SUTOU Y, IMANO Y, KOEDA N, OMORI T, KAINUMA R, ISHIDA K, OIKAWA K. Magnetic and martensitic transformations of NiMnX(X=In, Sn, Sb) ferromagnetic shape memory alloys [J]. Applied Physics Letters, 2004, 85 (19): 4358-4360.
[6] OIKAWA K, OTA T, GEJIMA F, OHMORI T, KAINUMA R, ISHIDA K. Phase equilibria and phase transformations in new B2-type ferromagnetic shape memory alloys of Co-Ni-Ga and Co-Ni-Al systems [J]. Materials Transactions, 2001, 42(11): 2472-2475.
[7] OIKAWA K, OTA T, TANAKA Y, OMORI T, KAINUMA R, ISHIDA K. Magnetic and martensitic phase transitions in the Ni-Al-Fe shape memory alloy [J]. Trans Mater Res Soc Jpn, 2003, 28(2): 263-264.
[8] OIKAWA K, OTA T, SUTOU Y, OHMORI T, KAINUMA R, ISHIDA K. Magnetic and martensitic phase transformation in a Ni54Ga27Fe19 alloy [J]. Materials Transactions, 2002, 43(9): 2360-2362.
[9] OIKAWA K, OTA T, OHMORI T, MORITO H, FUJITA A, KAINUMA R, FUKAMICHI K, ISHIDA K. Magnetic and martensitic phase transitions in ferromagnetic Ni-Ga-Fe shape memory alloys [J]. Applied Physics Letters, 2002, 81(27): 5201-5203.
[10] OIKAWA K, KOEDA N, SUTOU Y, OMORI T, KAINUMA R, ISHIDA K. Martensitic transformation and magnetic properties of Cu-Ga-Mn �� alloys [J]. Materials Transactions, 2004, 45(8): 2780-2784.
[11] KOEDA N, SUTOU Y, OMORI T, OIKAWA K, KAINUMA R, ISHIDA K. Martensitic transformation of Cu-10at%Al-9at%Ga- 11at%Mn ferromagnetic �� alloy [J]. Scripta Materialia, 2005, 52(11): 1153-1156.
[12] TANAKA Y, OIKAWA K, SUTOU Y, OMORI T, KAINUMA R, ISHIDA K. Martensitic transition and superelasticity of Co-Ni-Al ferromagnetic shape memory alloys with ��+�� two-phase structure [J]. Materials Science and Engineering A, 2006, 438-440: 1054-1060.
[13] LIU J, LI J G. Magnetic force microscopy observations of Co-Ni-Ga and Co-Ni-Al alloys with two-phase structures [J]. Scripta Materialia, 2006, 55(9): 755-758.
[14] OIKAWA K, OMORI T, SUTOU Y, KAINUMA R, ISHIDA K. Development of the Co-Ni-Al ferromagnetic shape memory alloys [J]. Journal de Physique IV, 2003, 112: 1017-1020.
[15] GAO Z Y, LIU C, WU C, MA W J, ZHANG J, CAI W. Martensitic transformation and magnetocaloric properties of Sn doping Mn-Ni-Ga alloys [J]. Journal of Magnetism and Magnetic Materials, 2010, 322(17): 2488-2492.
[16] SEGU? C, CHERNENKO V A, PONS J, CESARI E, KHOVAILOC V, TAKAGI T. Low temperature-induced intermartensitic phase transformations in Ni�CMn�CGa single crystal [J]. Acta Materialia, 2005, 53(1): 111-120.
[17] SUTOU Y, KAINUMA R, ISHIDA K, TAYA M. Martensitic transformation behavior under magnetic field in Co-Ni-Al ferromagnetic shape memory alloys [J]. Smart Structures and Materials, 2003, 5053: 159-169.
����Sn���ȴ�����Co38Ni34Al28-xSnx������״����Ͻ�����֯����Ӳ�ȵ�Ӱ��
�ռѼ�1, 2��л��ޱ1����Ԫ��1
1. �㶫��ҵ��ѧ ���Ϻ���ԴѧԺ������ 510006��
2. �㶫����ְҵ����ѧԺ������ 510095
ժ Ҫ����Co38Ni34Al28�Ͻ���ϵ������Sn���о�Sn��������ͬ���ȴ����¶�(1373 K��1473 K��1573 K)�£�����2 h��Co38Ni34Al28-xSnx(x=1��2��3)�Ͻ�����֯��Ӳ�ȵ�Ӱ�졣�������������������Snʹ�Ͻ��Ц�����֯���٣���1573 K����2 h���������»�ò�����������֯����Sn���2%Alʱ��������֯����������֯�ı����ϸߡ�����Sn������������ȴ����¶ȵ����ߣ��Ͻ��Ӳ��Ҳ�����������⣬�Ͻ��������������¶���Sn����Ϊ1%��2%ʱ���ߣ���Sn����Ϊ3%ʱ�������͡�
�ؼ��ʣ�������״����Ͻ�Snȡ����Co-Ni-Al�Ͻ�����֯�����������
(Edited by YANG Hua)
Foundation item: Projects (50771037, 50371020) supported by the National Natural Science Foundation of China; Project (2011B090400485) supported by the Combination Project for Guangdong Province and the Ministry of Education, China
Corresponding author: SU Jia-jia; Tel: +86-13312868330; Fax: +86-20-38744009; E-mail: sugarsujiajia@163.com
DOI: 10.1016/S1003-6326(11)61443-X