
Hydriding/dehydriding properties of Mg-Ni-based ternary alloys synthesized by mechanical grinding
CHEN Yu-an(����)1, 2, YANG Li-ling(������)1, LIN Jia-jing(�ּξ�)1,
CHENG Ji(�� ��)1, PAN Fu-sheng(�˸���)1, 2
1. College of Materials Science and Engineering, Chongqing University, Chongqing 400044, China;
2. National Engineering Research Center for Magnesium Alloys, Chongqing University, Chongqing 400044, China
Received 23 September 2009; accepted 30 January 2010
Abstract: The Mg-Ni-based ternary alloys Mg2-xTixNi(x=0, 0.2, 0.4) and Mg2Ni1-xZrx(x=0, 0.2, 0.4) were successfully synthesized by mechanical grinding. The phases in the alloys and the hydriding/dehydriding properties of the alloys were investigated. Mg2Ni and Mg are the main hydrogen absorption phases in the alloys by XRD analysis. Hydriding kinetics curves of the alloys indicate that the hydrogen absorption rate increases after partial substitution of Ti for Mg and Zr for Ni. According to the measurement of pressure-concentration-isotherms and Van��t Hoff equation, the relationship between ln p(H2) and 1 000/T was established. It is found that while increasing the content of correspondingly substituted elements at the same temperature, the equilibrium pressure of dehydriding increases, the enthalpy change and the stability of the alloy hydride decrease.
Key words: Mg-Ni-based hydrogen storage alloy; mechanical grinding; p-C-T measurement; hydriding properties; enthalpy change
1 Introduction
It is well-known that Mg and Mg-Ni-based alloys are considered as one of the most potentially important hydrogen storage materials owing to several advantages: light weight, rich natural resources, and high hydrogen storage capacity together with being friendly to the environment. However, it usually takes a long time and a high temperature to absorb/desorb hydrogen. The slow desorption kinetics hinders its application[1-3]. Mg2Ni is one of the most representative materials with the hydrogen storage capacity of 3.6% (mass fraction) and much smooth desorption isotherm, which can be definitely regarded as an ideal hydrogen storage alloy[4-7]. Elemental substitution, chemical plating and surface treatment are always done by researchers in order to improve the properties and provide a theoretical instruction for the practical application of hydrogen storage materials.
In this work, the ternary alloys in which Mg was partially substituted by Ti and Ni by Zr were synthesized by mechanical grinding. The phases in the alloys and the hydriding/dehydriding properties of the alloys were investigated. According to the measurement of pressure-concentration-isotherms (p-C-T) of the prepared samples at different temperatures and Van��t Hoff equation, it is easy to obtain linear relationship between hydrogen pressure (ln p(H2)) and temperature (1 000/T) for each alloy.
2 Experimental
The alloys of Mg2-xTixNi(x=0, 0.2, 0.4) and Mg2Ni1-xZrx(x=0, 0.2, 0.4) were synthesized by mechanical grinding, with the equipment of planetary ball mill (Fritsch Pulverisette P-5). Powders of magnesium, nickel and titanium/zirconium with 99% in purity were commercially gotten and mixed in a certain molar ratio of Mg2-xTixNi(x=0, 0.2, 0.4) and Mg2Ni1-xZrx(x=0, 0.2, 0.4). Grinding was done with a ball-to-powder mass ratio of 20:1, and the speed was 600 r/min for 30 h at room temperature under argon atmosphere (99.999% in purity).
The phase structures of different samples prepared were analyzed by X-ray diffraction (XRD) on a RigakuD/max-2500PC X-ray diffractometer with the experimental condition: Cu K�� radiation, graphite monochromator, anode voltage of 40 kV, the anode current of 150 mA and scanning speed of 4.000 (?)/min.
The absorption/desorption properties of different samples were tested on p-C-T measurement apparatus which was produced by Sichuan University, China. All those parameters were investigated at reaction temperatures of 473 K, 573 K, 673 K respectively and hydrogen pressure (99.999% in purity) of 2.0-3.5 MPa. Together with Van��t Hoff equation and the equilibrium pressure of dehydrogenation obtained, it is clear to establish a relationship among ln p(H2), 1 000/T and the enthalpy change (��H) of each alloy. It is also easy to calculate the equilibrium pressure of dehydriding in a permitted temperature.
3 Results and discussion
3.1 XRD analysis of ternary alloys
3.1.1 XRD analysis of Mg2-xTixNi(x=0, 0.2, 0.4) alloys
The X-ray diffraction patterns of Mg2-xTixNi(x=0, 0.2, 0.4) prepared by mechanical grinding are shown in Fig.1. It can be seen from Fig.1 that the main phase is Mg2Ni which has been clearly identified as an efficient hydrogen adsorption phase and the intensity of Mg2Ni is increased by the addition of titanium. The sharp peaks of Ni become flat and the phases of NixTi(x=0.5, 1, 2, 3) also exist in the alloys, which illuminates that there is a solid solution with an increasing of the lattice volume of Ni, making the lattice distortion in order to absorb more hydrogen. Besides, Ni peak moves to a low angle with the increase of Ti content with the strongest peak of Ni from 44.416? to 44.351?, compared with the similar investigation reported by LI et al[8]. In addition, there is residual element of Ti in the alloys.
3.1.2 XRD analysis of Mg2Ni1-xZrx(x=0, 0.2, 0.4) alloys
The X-ray diffraction patterns of Mg2Ni1-xZrx(x=0, 0.2, 0.4) prepared by mechanical grinding are shown in Fig.2. It can be clearly seen that, the main phases are Mg2Ni and Mg. And the element of Zr and Ni can easily

Fig.1 XRD patterns of alloys prepared by mechanical grinding: (a) Mg2Ni; (b) Mg1.8Ti0.2Ni; (c) Mg1.6Ti0.4Ni
form some new substances by grinding, such as Ni11Zr9 and Ni7Zr2. According to Ref.[9], these compounds of Ni-Zr can also absorb and desorb hydrogen reversibly and take on active site for desorption. A large number of crystal lattice defects exist because of the addition of Zr, which provides a passway for hydrogen atoms to diffuse. Besides, the broad peak shows the existence of amorphous alloys through the addition of Zr, which is helpful to hydriding[10] .

Fig.2 XRD patterns of alloys prepared by mechanical grinding: (a) Mg2Ni; (b) Mg2Ni0.8Zr0.2; (c) Mg2Ni0.6Zr0.4
3.2 Hydriding/dehydriding properties of ternary alloys
3.2.1 Hydriding kinetics of ternary alloys
The hydriding kinetic characterises of the ternary alloys at different temperatures are clearly shown in Fig.3. This indicates that the hydrogen absorption rate increases after being substituted for Ti and Zr at 473 K and 573 K, respectively. Basically, although the maximum of hydriding capacity decreases for those ternary alloys, they could get close to the value at about 60 s or 120 s, and Mg2Ni needs about 300 s at least. But, the hydrogen absorption capacity of those ternary alloys increases compared with Mg2Ni at 673 K. The hydriding absorption capacities of Mg2Ni0.8Zr0.2 and Mg2Ni0.6Zr0.4 reach 3.48% and 2.92%, whereas that of Mg2Ni could only achieve 2.60%. The detailed data are shown in Table 1.
There would be active sites of atoms, reducing the distance of diffused hydrogen atoms and increasing the fresh surface atoms after adding the elements of Ti and Zr. Thus, it could result in the expansion of the lattice volume and form more defects along the interface of particles, in order to provide a way for hydrogen atoms to easily diffuse and improve the performance of ternary alloys. This conclusion was the similar with KWON et al[11] and LU et al[12].

Fig.3 Kinetics curves of Mg2-xTixNi(x=0, 0.2, 0.4) and Mg2Ni1-xZrx(x=0, 0.2, 0.4) at different temperatures: (a), (d) 473 K; (b), (e) 573 K; (c), (f) 673 K
3.2.2 Dehydriding dynamic of ternary alloys
The comparisons of desorbing p-C-T curves for different alloys at the same temperature are detailedly shown in Fig.4. Clearly, the dehydriding properties of Mg2-xTixNi(x=0.2, 0.4) could desorb little hydrogen, while Mg2Ni1-xZrx(x=0.2, 0.4) had an evident indication of dehydriding, compared with Mg2Ni at 473 K and 573 K respectively. All the dehydriding results are considerable with clear and broad platform at the temperature of 673 K.
It is also indicated that the equilibrium pressure of dehydriding would be increased, when increasing the content of correspondingly substituted elements at the same temperature. For example, the equilibrium pressure of Mg2Ni is 0.040 8 MPa, is about 0.041 2 MPa for Mg1.8Ti0.2Ni and 0.066 1 MPa for Mg1.6Ti0.4Ni; and the equilibrium pressures of Mg2Ni1-xZrx(x=0.2, 0.4) are about 0.065 2 and 0.065 5 MPa at 673 K, respectively.
Table 1 Comparison of hydriding/dehydriding data for ternary complex hydride Mg-based alloys
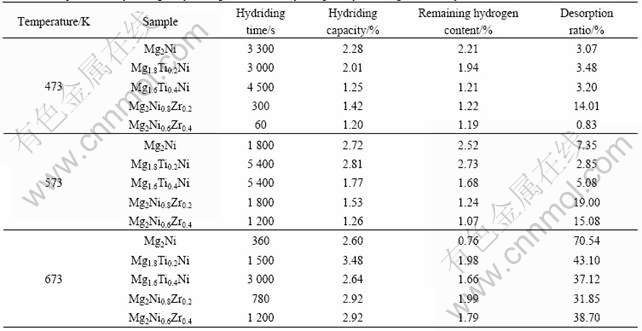
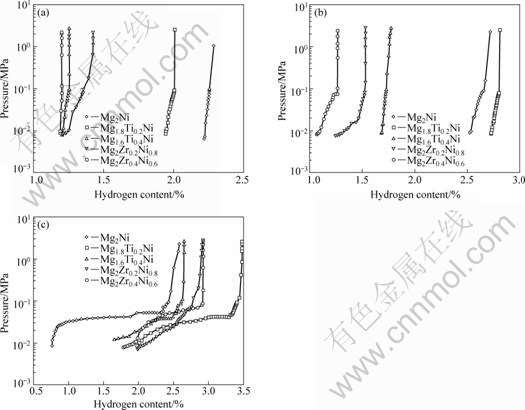
Fig.4 Comparisons of desorbing p-C-T curves for different alloys at same temperature: (a) 473 K; (b) 573 K; (c) 673 K
The detailed desorbing p-C-T curves of each alloy at different temperatures are shown in Fig.5. We could see that the relationship between dehydrogen pressure and temperature for the same alloy is just the same as the calculation of enthalpy change later.
3.3 Effects on enthalpy change of dehydrogenation
According to Van��t Hoff equation and correlational data of dehydrogenation p-C-T curves of the prepared samples at different temperatures in Fig.4 and Fig.5, the relationship between ln p(H2) and 1 000/T was established. The enthalpy change (��H) and the entropy change (��S) for different alloys can be gotten from the rate of slope and intercept of the linear equations, as shown in Table 2.
The linear relationships of hydrogen pressure and
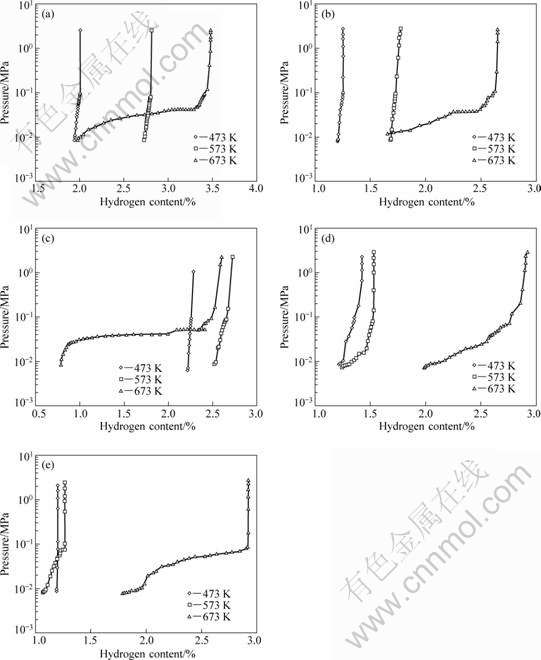
Fig.5 Desorbing p-C-T curves of each alloy at different temperatures: (a) Mg1.8Ti0.2Ni ; (b) Mg1.6Ti0.4Ni ; (c) Mg2Ni; (d) Mg2Ni1.8Zr0.2 ; (e) Mg2Ni1.6Zr0.4
Table 2 Comparison of dehydrogen data for ternary complex hydride
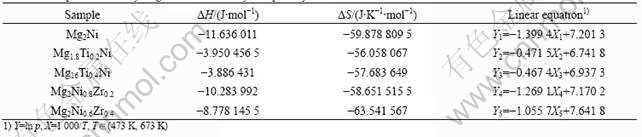
temperature are shown in Fig.6 and Fig.7 respectively, and the temperature ranges from 473 to 673 K. They illuminate that the dehydrogenation plateau is elevated by increasing the amount of substituting elements at the same temperature, which is equivalent to the phenomenon that the dehydrogenation temperature of alloys would slow down under the same equilibrium pressure while raising the replacing elements.
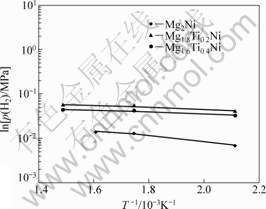
Fig.6 Linear relationship between hydrogen pressure and temperature for Mg2-xTixNi (x=0, 0.2, 0.4) alloys
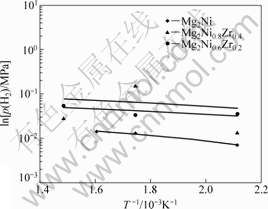
Fig.7 Linear relationship between hydrogen pressure and temperature for Mg2Ni1-xZrx(x=0, 0.2, 0.4) alloys
KANDAVEL and RAMAPRABHU[13] concluded that: the volume of Mg phase was decreased in the unit cell due to the formation of solid solution after adding the element of Ti, which causes an increase in strain energy to accommodate the interstitial hydrogen. Thus, the equilibrium pressure of hydrogen was increased, too.
The comparison of dehydrogen data for ternary complex hydride are given in Table 2, including ��H, ��S and the linear equations of different alloys. The results indicate that ��H decreases while increasing the content of substitutive elements, which makes clear the instability of hydrides, and is helpful to desorbing hydrogen at a low temperature. Besides, the new phases are formed because of the addition of substituted elements, which decrease the ��H while desorbing hydrogen. Thus, the instability of metal hydrides is increased. These results are in accordance with the conclusions that reported by TANG et al[14] and VAJO et al[15].
4 Conclusions
1) Although the hydrogen storage capacity of Mg2-xTixNi(x=0, 0.2, 0.4) and Mg2Ni1-xZrx(x=0, 0.2, 0.4) alloys is much lower than that of Mg2Ni, the absorption kinetics of them are better.
2) The ��H decreases while increasing the content of substitutive elements, which makes clear the instability of hydrides and is helpful to desorbing hydrogen at a relatively low temperature.
References
[1] HU Zi-long. Hydrogen storage [M]. Beijing: Chemical Industry Press, 2002: 12-44. (in Chinese)
[2] BONGDANOVIC B, SPLIETHOFF B. Active MgH2-Mg-systems for hydrogen storage [J]. Hydrogen Energy, 1987, 12(12): 863-873.
[3] FUJII H, ICHIKAWA T. Recent development on hydrogen storage materials composed of light elements [J]. Physica B, 2006, 383: 45-48.
[4] SONG M Y, PARK H R. Pressure-composition isotherms in the Mg2Ni-H2 system [J]. Journal of Alloys and Compounds, 1998, 270(1): 164-167.
[5] ZHANG Yang-huan, ZHAO Dong-liang, DONG Xiao-ping, QI Yan, GUO Shi-hai, WANG Xin-lin. Effects of rapid quenching on structure and electrochemical characteristics of La0.5Ce0.2Mg0.3Co0.4Ni2.6-xMnx (x=0-0.4) electrode alloys [J]. Trans Nonfereous Met Soc China, 2009, 19: 364-371.
[6] LIU Dong-ming, WEI Tao, LI Li-quan. Preparation and property of magnesium based hydrogen storage alloy hydride Mg2NiH4 [J]. Materials Science & Technology, 2007,15(3): 362-365. (in Chinese)
[7] KODERA Y, YAMASAKI N, YAMAMOTO T, KAWASAKI T, OHYANAGI M, MUNIR Z A. Hydrogen storage Mg2Ni alloy produced by induction field activated combustion synthesis [J]. Journal of alloys and Compounds, 2007, 446/447: 138-141.
[8] LI Ze-min, CHEN Xiu-juan, LI Jian-wei. Mechanical alloying for Ti-Mg system [J]. Power Metallurgy Industry, 2008, 18(3): 14-17. (in Chinese)
[9] ZHANG Yun-shi, YANG Hua-bin, YUAN Hua-tang, YANG En-dong, ZHOU Zuo-xiang, SONG De-ying. Dehydriding properties of ternary Mg2Ni1-xZrx hydrides synthesized by ball milling and annealing [J]. Journal of Alloys and Compounds, 1998, 269: 278-283.
[10] WANG Yi-cun, ZHANG Wen-feng, ZHU Yun-feng, LI Li-quan. Effect of ball-milling with Ni on hydriding combustion synthesizing Mg2NiH4 [J]. Battery Bimonthly, 2008, 138(6): 343-345. (in Chinese)
[11] KWON S, BAEK S, MUMM D R, HONG S H, SONG M. Enhancement of the hydrogen storage characteristics of Mg by reactive mechanical grinding with Ni, Fe and Ti [J]. International Journal of Hydrogen Energy, 2008, 33: 4586-4592.
[12] LU Guo-jian, ZHOU Shi-xue, MA Huan-ying, TANG Qi, ZHOU Zhuang-fei. Preparation of magnesium/carbon nanocomposites for hydrogen storage by reaction milling [J]. Journal of Functional Materials, 2007, 38(7): 1128-1131. (in Chinese)
[13] KANDAVEL M, RAMAPRABHU S. Correlation between hydrogen storage properties and amount of alloy particles in Mg-based composites [J]. Journal of Alloys and Compounds, 2007, 438: 285-292.
[14] TANG Gong-li, LIU Xiao-peng, JIANG Li-jun, WANG Shu-mao, LI Zhi-nian, LI Hua-ling. Dehydrogenation characteristic of Zr1-xMxCo(M=Hf, Sc) alloy [J]. Trans Nonferrous Met Soc China, 2007, 17: 949-953.
[15] VAJO J J, SALGUERO T T, GROSS A F, SKEITH S L, OLSON G L. Thermodynamic destabilization and reaction kinetics in light metal hydride systems [J]. Journal of Alloys and Compounds, 2007, 446/447: 409-414.
(Edited by YANG You-ping)
Foundation item: Project(2006BB4197) supported by Chongqing Natural Science Foundation, China
Corresponding author: CHEN Yu-an; Tel: +86-13996300268; Fax: +86-23-65127306; E-mail: chenyuan@cqu.edu.cn