
Electrochemical properties of high-power lithium ion batteries made from modified spinel LiMn2O4
LI Xiang-qun(����Ⱥ)1, WANG Zhi-xing(��־��)2, LIANG Ru-fu(���縣)2,
GUO Hua-jun(������)2, LI Xin-hai(���º�)2, CHEN Qi-yuan(����Ԫ)1
1. School of Chemistry and Chemical Engineering, Central South University, Changsha 410083, China;
2. School of Metallurgical Science and Engineering, Central South University, Changsha 410083, China
Received 20 December 2008; accepted 15 March 2009
Abstract: A prismatic 204056-type high��power lithium-ion battery was developed. Modified LiMn2O4 and carbonaceous mesophase sphere (CMS) were adopted as the cathode and anode, respectively. The effects of proportion of conductive carbon black in cathode and the rest time after discharge on the electrochemical properties of batteries were investigated. The electrochemical tests show that the proportion of conductive carbon black in cathodes affects the high rate capability and discharge voltage plateau distinctly. The battery with 3.0% of conductive carbon black in cathode shows excellent electrochemical performances when being charged/discharged within 2.5-4.2 V at room temperature. The discharge capacity at 20C rate is 94.4% of that at 1C rate, and the capacity retention ratio charged at 1C and discharged at 5C is 86.6% after 390 cycles at room temperature. The test result of impulse discharge at 50C for 5 s shows that the battery has outstanding high rate discharge performance when the battery is in the depth of charge of 90%, 75%, 60%, 45%, 30% and 15%. The battery also shows good charge performance. When the battery is charged at 0.5C, 1C, 2C and 4C, the ratios of capacity for constant current charge are 98.4%, 96.4%, 91.0% and 72.9% of the whole charge capacity, respectively. In addition, the rest time after discharge affects the cycle performance distinctly when the battery is discharged at high rate.
Key words: lithium ion batteries; LiMn2O4; electrochemical property; rate capability
1 Introduction
During the last decade, LiCoO2 has been extensively used as cathode material for cellular phones and notebook-type computers or other small size batteries due to its high energy density and perfect cycling performance. Recently, safety has become a major concern for lithium ion batteries. Due to the poor safety and high cost for LiCoO2[1-2], it is likely that LiCoO2 cathode material is not suitable for batteries used in power tool, miner��s lamps, electric vehicles(EVs) and hybrid electric vehicle(HEVs). Spinel LiMn2O4 has been extensively investigated as cathode for power tools, miner��s lamps, EVs, etc, for its high voltage, low cost, good safety and no toxicity[3-6]. Nevertheless, LiMn2O4 suffers from capacity fading, which limits its reversibility[7-9]. In order to improve the cycling performance, some researchers put forward modified methods of LiMn2O4 by doping with 3d metal ions (Cr, Fe, Ni, Co)[10-11], and coating with SrF2[12], AlPO4 [13], SiO2[14] or SnO2[15]. Nowadays, the demand for the high-power batteries for electrical toys and power tools is growing prominently. Therefore, it is significant to develop high-power batteries for their promising utilization.
In this work, the commercial modified LiMn2O4 cathode material was selected and the effect of the process conditions, such as the proportion of conductive carbon black in cathodes and the rest time after discharge, on the electrochemical properties of batteries was investigated.
2 Experimental
2.1 Methods
The particle morphologies of modified LiMn2O4 (From Hunan Shanshan Advanced Material Co. Ltd., China) and carbonaceous mesophase spheres (CMS, from Shanghai Shanshan Technology Co. Ltd., China) powders were observed using a scanning electron microscope (JOEL, JSM-5600LV).
2.2 Fabrication and electrochemical tests of 204056- type prismatic batteries
For cathode preparation, the modified LiMn2O4 powder was mixed with different proportions of conductive carbon black, 2% of squama graphite and 5% of polyvinylidene fluoride (PVDF) in N-methyl pyrrolidinone (NMP) in order to obtain uniform slurry. In succession, the slurry was coated onto aluminum current collector. For anode preparation, the CMS powder was mixed with 3.0% conductive carbon black and 6% bonding agent of LA133 (From Chengdu Indigo Power Sources Co. Ltd., China) in water. Then, the slurry was coated onto copper current collector. After coating, the cathode and the anode were dried in vacuum for 12 h at 130�� and 105 ��, respectively. The cathode and the anode were pressed with a bulk density of 2.55 g/cm3 and 1.25 g/cm3, respectively. Finally, the cathode and anode were assembled into 204056-type prismatic batteries using Celgard 2300 as separator and 1 mol/L LiPF6 in EC+DEC+DMC with 1?1?1 volume ratio as electrolyte. Before the electrolyte was injected, those semi finished batteries were dried at 80 �� for 48 h.
The batteries were charged/discharged between 2.5 and 4.2 V with 0.02C as cut-off current of charge at room temperature. The rate capability, charge performance and cycle performance were characterized.
2.3 AC impedance test of cathode with different proportions of conductive carbon black
The cathodes after pressing were assembled into CR2025 coin-type cells with the counter electrode of metallic lithium foil in a dry Ar-filled glove box. The separator and electrolyte were the same as the 204056-type prismatic batteries. The cells were charged and discharged for two cycles for activation at 0.2C current rate over a voltage range of 3.0-4.2 V vs Li/Li+ electrode at room temperature.
3 Results and discussion
3.1 Morphology analysis
Fig.1 shows the particle morphologies of modified LiMn2O4 and CMS powders using scanning electron microscope. It can be seen from Fig.1(a) that the primary particles of modified LiMn2O4 are around 0.4-2.0 ��m and have perfect shape of spinel. It can be seen from Fig.1(b) that the secondary particles of CMS are about 8-15 ��m, which are glomeration congregated tightly by primary particles.

Fig.1 SEM images of cathode and anode material samples: (a) Modified LiMn2O4; (b) CMS
3.2 Effect of proportion of conductive carbon black in cathode on electrochemical properties of batteries
Fig.2 shows the discharge rate capability of batteries with different proportions of conductive carbon black in the cathode. It can be seen from Fig.2 that the discharge capacity at high rate increases with the increase of conductive carbon black from 1.5% to 3.0%. While the proportion of conductive carbon black increases to 3.5%, the discharge capacity decreases slightly. This phenomenon can be explained from the two aspects of electronic conduction and ion conduction as follows. Obviously, the increase of the proportion of conductive carbon black contributes to the electronic conduction. So, the discharge capacity increases prominently at first. However, the voidage of the cathode that is pressed with fixed bulk density becomes small by increasing the proportion of conductive carbon black, because the density of conductive carbon black is lower than that of LiMn2O4 powder. Therefore, decreasing voidage of the cathode is difficult for ion conduction.
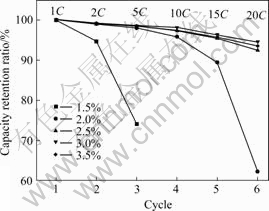
Fig.2 Discharge capacity ratios of batteries with various proportions of conductive carbon black in cathode at different current rates (Charge rate: 1C)
Fig.3 shows the EIS of cathode (tested at half-charged state). It can be seen from Fig.4 that the electrochemical impedance of the cathode decreases and then rises with the increase of the proportion of conductive carbon black in the cathode from 1.5% to 3.5%, which can interpret the phenomenon in Fig.2 further.

Fig.3 EIS of cathode (tested at half-charged state)
3.3 Electrochemical properties of batteries with 3.0% of conductive carbon black in cathode
Fig.4 shows the discharge curves of the battery with 3.0% of conductive carbon black in the cathode has excellent rate capacities. The discharge capacity at 20C rate is 94.4% of that at 1C rate. Table 1 lists the capacities and median voltages of discharge curves at various current rates. The rate capacity and voltage decline as the current rate of discharge rises.
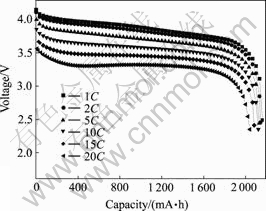
Fig.4 Discharge curves of batteries with 3.0% of conductive carbon black in cathode (Charge rate: 1C)
Table 1 Discharge capacities and median voltages at various current rates
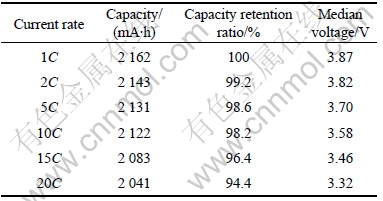
Fig.5 shows the cycling performance of batteries with 3.0% of conductive carbon black in cathode. It can be seen from Fig.5 that the batteries also have good cycling performance with capacity retention ratio of 86.6% when being charged at 1C and discharged at 5C after 390 cycles at room temperature. Fig.6 shows the impulse discharge curves at 50C for 5 s at the depth of charge of 90%, 75%, 60%, 45%, 30% and 15%, and discharge curves at 10C at other depths of charge. It can be seen from Fig.6 that the batteries have the outstanding high-current rate discharge performance. Fig.7 shows the charge curves of the battery with 3.0% of conductive carbon black in cathode at different rates. It can be seen from Fig.7 that when being charged at 0.5C, 1C, 2C and 4C, the capacities of constant current charge are 98.4%, 96.4%, 91.0% and 72.9% of the whole charge capacity, respectively.
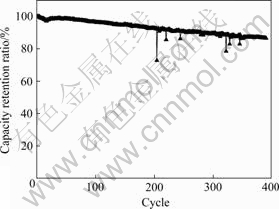
Fig.5 Cycling performance of batteries with 3.0% of conductive carbon black in cathode (Charge rate: 1C; Discharge rate: 5C)
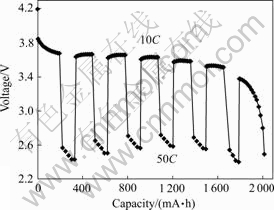
Fig.6 Impulse discharge of battery with 3.0% of conductive carbon black in cathode (Discharge at 50C for 5 s at depth of charge of 90%, 75%, 60%, 45%, 30% and 15%, and discharge at 10C in other depths of charge)
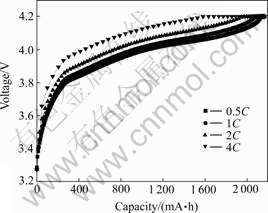
Fig.7 Charge curves of battery with 3.0% of conductive carbon black in cathode at different rates (Discharge rate: 1C)
3.4 Effect of rest time after discharge on cycling properties of batteries with 3.0% conductive carbon black in cathode
An interesting result is observed in the cycle curves at various discharge current rates ranging from 1C to 20C, as shown in Fig.8. Being charged at 1C and discharged at various current rates, the battery maintains a steady discharge capacity at the discharge rates from 1C to 10C. While discharge current rate is 15C or 20C, capacity rapidly declines at each discharge rate, especially at 20C. The battery shows good cycling performances again when the discharge current is reduced to 1C, which shows that the structure and properties decay little. Much effort is made to understand this phenomenon, and it is found out that the rest time after discharge at high current rate affects the capacity retention ratio obviously.

Fig.8 Cycling performance of battery with 3.0% of conductive carbon black in cathode discharged at various current rates (Charge rate: 1C)
Fig.9 shows the cycling performance of batteries with 3.0% of conductive carbon black in cathode tested with different rest time after discharge at 15C. It can be seen from Fig.9 that capacity retention ratio increases with the increase of the rest time after discharge. This phenomenon can be interpreted by electrolyte imbalance which results from discharging at high current rate. When the discharge current goes up, the electrolyte imbalance becomes greater. This will lead to increasing polarization of batteries, and can also interpret the phenomenon that the capacity declines rapidly when being discharged at 20C (Fig.8). It is obvious that the cycling performances tested at 15C and 20C become better with the increase of the rest time after discharge from 1 h to 4 h, as shown in Fig.9.
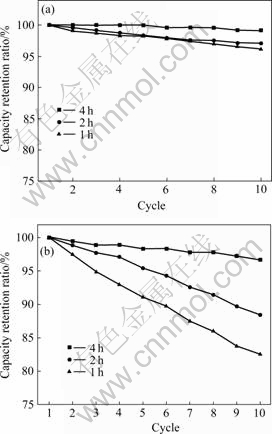
Fig.9 Cycling performance of batteries with 3.0% of conductive carbon black in cathode tested with various rest time after discharge at 15C(a) and 20C(b) (Charge rate: 1C)
4 Conclusions
1) A prismatic 204056-type high��power lithium- ion battery is developed by using modified LiMn2O4 as cathode and CMS as anode. When the cathode contains 3.0% of conductive carbon black, the electrochemical performances of batteries are the best.
2) The discharge capacity of battery at 20C is 94.4% of that at 1C, and the capacity retention ratio of battery charged at 1C and discharged at 5C is 86.6% after 390 cycles at room temperature.
3) The capacity retention ratio increases with the increase of rest time after discharge at high current rate. The capacity retention ratios of battery charged at 1C and discharged at 15C and 20C, respectively, both increase as the rest time after discharge increases from 1 h to 4 h.
References
[1] TODOROV Y M, NUMATA K. Effects of the Li:(Mn + Co + Ni) molar ratio on the electrochemical properties of LiMn{sub}(1/3)Co{sub}(1/3)Ni{sub}(1/3)O{sub}2 cathode material [J]. Electrochim Acta, 2004, 50(2/3): 495-499.
[2] SINGHAL R, DAS S R, TOMAR M S, OVIDEO O, NIETO S, MELGAREJO R E, KATIYAR R S. Synthesis and characterization of Nd doped LiMn2O4 cathode for Li-ion rechargeable batteries [J]. J Power Sources, 2007, 164(2): 857-861.
[3] SINGH D, KIM W S, CRACIUN V, HOFMANN H, SINGH R K. Microstructural and electrochemical properties of lithium manganese oxide thin films grown by pulsed laser deposition [J]. Appl Surf Sci, 2002, 197/198: 516-521.
[4] HAN J D, JIA Y Z, JIN S, JING Y, TILLARD M, BELIN C. Morphology and electrochemistry of spinel Li�CMn�CO optimized by composite technology [J]. Energy, 2006, 31(12): 2088-2093.
[5] YU L, QIU X, XI J, ZHU W, CHEN L. Enhanced high-potential and elevated-temperature cycling stability of LiMn2O4 cathode by TiO2 modification for Li-ion battery [J]. Electrochim Acta, 2006, 519(28): 6406-6411.
[6] SHIEH D T, HSIEH P H, YANG M H. Effect of mixed LiBOB and LiPF6 salts on electrochemical and thermal properties in LiMn2O4 batteries [J]. J Power Sources, 2007, 174(2): 663-667.
[7] SAHAN H, G?KTEPE H, PATAT S, ?LGEN A. The effect of LBO coating method on electrochemical performance of LiMn2O4 cathode material [J]. Solid State Ionics, 2008, 178(35/36): 1837-1842.
[8] TANIGUCHI I, FUKUDA N I, KONAROVA M. Synthesis of spherical LiMn2O4 microparticles by a combination of spray pyrolysis and drying method [J].Powder Technology, 2008, 181: 228-236.
[9] LIU D Q, LIU X Q, HE Z Z. The elevated temperature performance of LiMn2O4 coated with Li4Ti5O12 for lithium ion battery [J]. Materials Chemistry and Physics, 2007, 105(2/3): 362-366.
[10] WU Y P, RAHM E, HOLZE R. Effects of heteroatoms on electrochemical performance of electrode materials for lithium ion batteries [J]. Electrochim Acta, 2002, 47(21): 3491-3507.
[11] MOLENDA J, MARZEC J, ?WIERCZEK K, OJCZYK W, ZIEMNICKI M, WILK P, MOLENDA M, DROZDEK M, DZIEMBAJ R. The effect of 3d substitutions in the manganese sublattice on the charge transport mechanism and electrochemical properties of manganese spinel [J]. Solid State Ionics, 2004, 171(3/4): 215-217.
[12] LI Jiang-gang, HE Xiang-ming, ZHAO Ru-song. Electrochemical performance of SrF2-coated LiMn2O4 cathode material for Li-ion batteries [J]. Trans Nonferrous Met Soc China, 2007, 17(6): 1324-1327.
[13] LIU D Q, HE Z Z, LIU X Q. Increased cycling stability of AlPO4-coated LiMn2O4 for lithium ion batteries [J]. Materials Letters, 2007, 61(25): 4703-4706.
[14] BLYR A, SIGALA C, AMATUCCI G G, GUYOMARD D, CHABRES Y, TARASCON J M. Self-discharge of LiMn2O4/C Li-ion cells in their discharged state [J]. J Electrochem Soc, 1998, 145(1): 194-209.
[15] PARK S B, SHIN H C, LEE W G, CHO W I, JANG H. Improvement of capacity fading resistance of LiMn2O4 by amphoteric oxides [J]. J Power Sources, 2008, 180(1): 597-601.
Foundation item: Project(2007CB613607) supported by the National Basic Research Program of China
Corresponding author: WANG Zhi-xing; Tel/Fax: +86-731-88836633; E-mail: zxwang@mail.csu.edu.cn
DOI: 10.1016/S1003-6326(09)60058-3
(Edited by YANG Bing)