Synthesis and characterization of Cu-Cr-O nanocomposites
来源期刊:中南大学学报(英文版)2007年第3期
论文作者:李卫 程化
文章页码:291 - 295
Key words:copper chromite composite; nanomaterial; citric acid; solid-state propellant; catalyst; complexing
Abstract: Cu-Cr-O nanocomposites that can be used as additives for the catalytic combustion of AP(ammonium perchlorate)-based solid-state propellants were synthesized via a citric acid(CA) complexing approach. Techniques of TG-DTA, XRD as well as TEM were employed to characterize the thermal decomposition procedure, crystal phase, micro-structural morphologies and grain size of the as-synthesized materials respectively. The results show that well-crystallized Cu-Cr-O nanocomposites can be produced after the CA-Cu-Cr precursors are calcined at 500 ℃ for 3 h. Phase composition of the as-obtained Cu-Cr-O nanocomposites depends on the molar ratio of Cu to Cr in the starting reactants. Addition of the as-synthesized Cu-Cr-O nanocomposites as catalysts enhances the burning rate as well as lowers the pressure exponent of the AP-based solid-state propellants considerably. Noticeably, catalyst with a Cu/Cr molar ratio of 0.7 exhibits promising catalytic activity with high burning rate and low pressure exponent at all pressures, due to the effective phase interaction between the spinel CuCr2O4 and delafossite CuCrO2 contained in the as-synthesized Cu-Cr-O nanocomposites.
基金信息:the National High-Tech Research and Development Program of China
the Postdoctoral Foundation of Hunan University
J. Cent. South Univ. Technol. (2007)03-0291-05
DOI: 10.1007/s11771-007-0057-5
![]()
LI Wei(李 卫)1,2,3, CHENG Hua(程 化)2
(1. State Key Laboratory of Powder Metallurgy, Central South University, Changsha 410083, China;
2. School of Chemistry and Chemical Engineering, Central South University, Changsha 410083, China;
3. School of Materials Science and Engineering, Central South University, Changsha 410083, China)
Abstract: Cu-Cr-O nanocomposites that can be used as additives for the catalytic combustion of AP(ammonium perchlorate)-based solid-state propellants were synthesized via a citric acid(CA) complexing approach. Techniques of TG-DTA, XRD as well as TEM were employed to characterize the thermal decomposition procedure, crystal phase, micro-structural morphologies and grain size of the as-synthesized materials respectively. The results show that well-crystallized Cu-Cr-O nanocomposites can be produced after the CA-Cu-Cr precursors are calcined at 500 ℃ for 3 h. Phase composition of the as-obtained Cu-Cr-O nanocomposites depends on the molar ratio of Cu to Cr in the starting reactants. Addition of the as-synthesized Cu-Cr-O nanocomposites as catalysts enhances the burning rate as well as lowers the pressure exponent of the AP-based solid-state propellants considerably. Noticeably, catalyst with a Cu/Cr molar ratio of 0.7 exhibits promising catalytic activity with high burning rate and low pressure exponent at all pressures, due to the effective phase interaction between the spinel CuCr2O4 and delafossite CuCrO2 contained in the as-synthesized Cu-Cr-O nanocomposites.
Key words: copper chromite composite; nanomaterial; citric acid; solid-state propellant; catalyst; complexing
1 Introduction
Cu-Cr-O composite oxides are long recognized as a kind of versatile functional materials due to their wide commercial application as catalysts for chemical reactions of hydrogenation, dehydrogenation, oxidation, alkylation, and so on[1-4]. The catalyst is also known to be an active catalyst for pollution treatment, for example, in car exhaust purification, where it is used to achieve complete oxidation of carbon monoxide and hydro- carbons to carbon dioxide[5-8]. In addition to the aforementioned versatile commercial applications, the Cu-Cr-O composites in recent years are found great promising in application as burning rate catalysts (ballistic modifier) for solid propellants used in defense (ballistic missiles)[9-12] and space vehicles (rocket propellants)[13-16]. RAJEEV et al[13] prepared Cu-Cr-O composite oxides via thermal decomposition of copper ammonium chromate and found that the propellant burning rate is enhanced by the addition of Cu-Cr-O composite oxides. Copper chromite (CuCr2O4) obtained via both ceramic and coprecipitation methods has also been evaluated by KAWAMOTO et al[11] as burning rate catalyst for the combustion of hydroxyl-terminated- polybutadiene (HTPB)-based solid-state propellant and the results indicated that the highest burning rate can be obtained with samples that present a more defined crystalline shapes synthesized by the coprecipitation method. COMINO et al[8] showed that the Cu-Cr-O composites with a Cu/Cr molar ratio of 0.7 via a Pechini method displayed the highest catalytic activity. Hence, catalytic performance of the Cu-Cr-O composites is of much dependence on the crystal structure, phase composition as well as particle shape and size. It was therefore considered a better understanding of the correlations between microstructural parameters and combustion performance would be meaningful in formulating the propellant-catalyst compositions used in solid fuel propelled rockets.
In this study, Cu-Cr-O nanocomposites were synthesized via a citric acid(CA) complexing method (namely Pechini approach) and the characterizations of the as-synthesized Cu-Cr-O nanocomposites as additives for the catalytic combustion of AP(ammonium perchlorate)-based solid-state propellants were reported.
2 Experimental
2.1 Preparation of Cu-Cr-O nanocomposites
All the chemicals used in the experiments were of analytical purity, bought from Beijing Chemicals Reagent Factory, China, and used without further purification. In a typical synthesis procedure, 0.07 mol Cu(NO3)2 and 0.1 mol Cr(NO3)3 were dissolved in 100 mL deionized water to obtain a mixed metal nitrate aqueous solution. Then citric acid was added to this solution and the molar ratio of citric acid to the total metal ions was fixed to be 2:1. After stirring for 30 min, the solution was heated at 95 ℃ for several hours to evaporate the water solvent to produce dark brown transparent viscous gels. The gels were then dried at 160 ℃ for 2 h to obtain the foamy dark powders, which were denoted as precursor of CuCr2O4/CuO nanocomposites (CA-Cu-Cr). After grinding, the precursors were successively heated at 600 ℃ for 3 h to obtain the final black CuCr2O4/CuO nanocomposites. In an attempt to investigate the crystallinity and composition dependence of catalytic performances, several experiments were conducted by varying the calcination temperatures and the molar ratio of Cu to Cr.
2.2 Characterization of Cu-Cr-O nanocomposites
The crystal phase of the as-prepared products was identified by powder X-ray diffraction method (XRD, Bruker D8) using Cu Kα radiation (λ=0.154 18 nm) at a scan rate of 4 (?)/min. The microstructural morphology of the final products was characterized with a Hitachi H-800 transmission electron microscope (TEM) operated at 200 kV. The thermal decomposition behavior of the citrate gel precursors was examined by a thermo- gravimetric analyzer (TGA, Perkin Elmer, TAC 7/DX) in air. For TGA measurements, the samples were heated from room temperature to 1 000 ℃ at a heating rate of 5 ℃/min. α-Al2O3 was used as reference material and the samples were run in open platinum pans.
The samples of copper chromate synthesized were evaluated as burning rate catalyst in a typical composite propellant formation, containing 5% (mass fraction) catalyst. The propellant’s burning rate and pressure exponent were measured and compared. The burning rate was determined by performing burning tests in a Crawford bomb between 7 MPa and 18 MPa.
3 Results and discussion
3.1 Thermal analysis
Thermal decomposition procedure of the as- obtained CA-Cu-Cr precursors is studied by TG-DTA, as shown in Fig.1. Over the temperature range from ambient temperature to 400 ℃, there is a continuous mass loss (about 65%) in the TG curve. The variation of mass-loss is not so obvious when the temperature is further increased from 400 ℃ to 1 000 ℃. Three discrete regions of thermal decomposition can be found in the DTA curve at 100-200, 200-400, and 400-500 ℃. The first mass-loss corresponds to the elimination of water. The second mass-loss region can be ascribed to the pyrolysis of ![]() and organic phases (citric acid) to give an amorphous inorganic phase, which will be further discussed in detail by XRD in the following section. Subsequently elimination of the remaining organic materials (carbon and organic compounds) occurs in the temperature range from 400 to 500 ℃ and crystallized CuCr2O4/CuO inorganic phase is simultaneously formed.
and organic phases (citric acid) to give an amorphous inorganic phase, which will be further discussed in detail by XRD in the following section. Subsequently elimination of the remaining organic materials (carbon and organic compounds) occurs in the temperature range from 400 to 500 ℃ and crystallized CuCr2O4/CuO inorganic phase is simultaneously formed.

Fig.1 TG and DTA curves of as-synthesized CA-Cu-Cr precursors via Pechini method
(Inset is enlarged part of DTA curve from 400 to 500 ℃)
3.2 Crystal structure analysis
Fig.2 presents the variation of XRD patterns as a function of calcination temperature. It can be observed that the materials calcined at 400 ℃ (Figs.2(a) and (b)) have broad diffraction peaks that are ascribed to the CuO phase (JCPDS:801917) and no peaks referring to the desired spinel CuCr2O4 can be found. Based on the thermal analysis results, it is proposed that the organic compounds in the precursors begin to decompose and the inorganic phase Cu-Cr-O begins to form (but it is amorphous) at the temperature range from 300 to 500 ℃. When being calcined at 500 ℃, the main peaks associated with the spinel phase CuCr2O4 (JCPDS:880110) appear. At the same time, the diffraction of CuO becomes much less pronouncing, which suggests that the organic compounds have almost completely been decomposed and the well-crystallized inorganic phase CuCr2O4 is consequently formed. But the wide and weak diffraction peaks are indicative of a very fine particle size of the as-synthesized sample at 500 ℃ (Fig.2(b)). When the calcination temperature is further raised to 700 ℃ and 900 ℃ (Figs.2(c) and (d)), intensities of all diffraction peaks gradually increase and the peak width gradually becomes narrower, which reveals that the particle size increases and the crystallinity improves with temperature increasing while remaining its phase nature of spinel CuCr2O4.

Fig.2 XRD patterns of Cu-Cr-O nanocomposites after calcinated at different temperatures for 3 h
(Cu/Cr molar ratios of all samples are fixed at 0.5)
(a) 400 ℃; (b) 500 ℃; (c) 700 ℃; (d) 900 ℃
Fig.3 shows XRD patterns for the samples with various Cu/Cr molar ratios after being calcined at 700 ℃ for 3 h. It is clear that Cu-Cr-O composites with pure spinel CuCr2O4 can be obtained when the Cu/Cr molar ratio is 0.5 (Fig.3(b)). When the Cu/Cr molar ratio is lower than 0.5, for example, 0.3 as shown in Fig.3(a), the product is a mixture of CuCr2O4 and Cr2O3. And when it is increased to 0.55, the product is a mixture of CuCr2O4 and CuO (Fig.3(c)). However, the diffraction peaks referring to delafossite phase of CuCrO2 appear when the Cu/Cr molar ratio is further increased to 0.6 (Fig.3(d)). The relative diffraction intensity related to delafossite phase CuCrO2 becomes more and more pronouncing with the continuous increase of Cu/Cr molar ratio (Figs.3(e) and (f)) and the product consists of single phase delafossite CuCrO2 at a Cu/Cr molar ratio of 1.0 (Fig.3(g)). This phenomenon clearly demonstrates that phase composition of the final Cu-Cr-O composites can be turned just by changing the Cu/Cr molar ratio in the starting reactant, which is very important to take fully advantages of the merits of Cu-Cr-O nanocomposites as catalysts.
3.3 Morphologies
TEM images of the precursors and the samples after calcination are shown in Fig.4. It is clear from Fig.4(a) that CA-Cu-Cr precursors are amorphous, which is
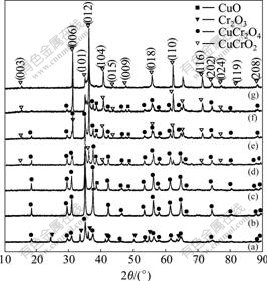
Fig.3 XRD patterns of as-synthesized Cu-Cr-O composite oxides with various Cu/Cr molar ratios
(Calcination temperatures for all samples are fixed at 700 ℃)
(a) 0.3; (b) 0.5; (c) 0.55; (d) 0.6; (e) 0.7; (f) 0.8; (g) 1.0
further confirmed by the selected area electron diffraction (SAED) pattern. TEM micrograph of the sample obtained by calcining at 500 ℃ for 3 h is displayed in Fig.4(b), which exhibits ultrafine nanoparticles with a mean grain size of 30 nm, but some small particles integrate into aggregates of 200 nm in diameter. SAED pattern in Fig.4(b) has two dim rings, which can be assigned to the two major planes of (211) and (202) for spinel CuCr2O4 and is an indication of fairly good crystallinity of the as-produced sample after being calcined at 500 ℃ for 3 h. When the calcination temperature reaches 700 ℃, the crystallinity of the as-obtained sample is improved and the average grain size is about 100 nm in Fig.4(c). SAED pattern in Fig.4(c) is composed of bright points that can be indexed as the crystal planes of spinel CuCr2O4, i.e. (104), (112), (211), (202), (321) and (411). This means that the sample obtained by heating at 700 ℃ is polycrystal in nature with good crystallinity. The microstructure analysis clearly shows the superiority of solution-based methods in synthesizing nanocomposites with controllable composition, shape and size.
3.4 Solid propellant properties
The pressure dependence on the burning rate is normally of interest for describing the combustion performance of solid state propellants. This function, in accordance with the Vieille’s law, can be expressed as follows:
v=βpα (1)
where v, β, p and α are denoted as burning rate, experimental constant, pressure, and pressure exponent,
respectively. In particular, high values of the burning rate, v, are favorable for a high power, and small values of the pressure exponent, α, are aimed for a stable burning at high pressure [16-19]. Table 1 lists a comparison of the pressure dependence on burning rates for the solid state propellants containing Cu-Cr-O nanocomposites catalysts obtained by Pechini method and for the propellant without using any catalyst. It can be concluded from Table 1 that after the addition of Cu-Cr-O composites as catalyst, burning rates of the propellants are greatly enhanced at all pressures, and at the same time the pressure exponents obviously reduce. Noticeably, the sample with a Cu/Cr molar ratio of 0.7 and calcined at 700 ℃ (sample 6#) exhibits a relatively high burning rate and the lowest pressure exponent, that is, the most stable combustion at all pressures. With a fixed Cu/Cr molar ratio of 0.5, when the calcination temperatures are raised from 500 ℃ to 900 ℃, the burning rate as well as the pressure exponent progressively decreases. When keeping the calcination temperature constant (at 700 ℃), the burning rate at low pressure gradually increases and then decreases with the increase of Cu/Cr molar ratio from 0.3 to 1.0. Although the pressure exponent shows a random relationship with the Cu/Cr molar ratio, the pressure exponent has an inclination to decrease corresponding to the increase of Cu/Cr molar ratio from 0.3 to 0.7 and then increases afterwards. As shown in Fig.2 and Fig.4, particles aggregate into bigger ones at the expense of small nanoparticles with the increase of calcination temperature, which results in a reduced surface area. As well known, catalysts with a high surface area exhibit a large amount of surface activity centers and can effectively react with the targeted catalyzed materials, leading to a faster combustion process. Therefore, catalyst obtained at 500 ℃ with a grain size of 30 nm shows the highest burning rate at all pressures (sample 1#). But the catalyst with higher surface area also contains larger surface free energy and is consequently

Fig.4 TEM images and corresponding SAED patterns of CA-Cu-Cr precursors (n(Cu)/n(Cr)=0.5) via Pechini method(a) and as-obtained samples after precursors are calcined at 500 ℃(b) and 700 ℃(c)
Table 1 Fitting burning equation of AP-based solid-state propellants under various pressures
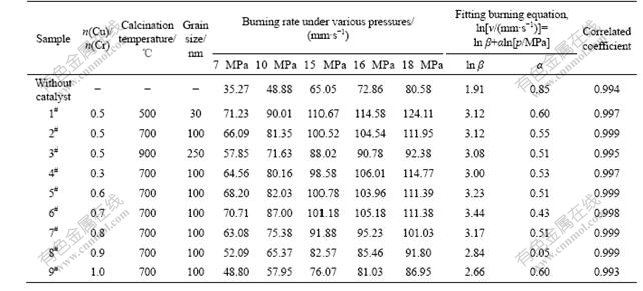
much sensitive to the pressure, therefore the pressure exponent is higher (sample 1# compared with samples 2# and 3#). Results from Fig.3 imply that samples with Cu/Cr molar ratio of 0.6-1.0 are composed of spinel CuCr2O4 and delafossite CuCrO2. It has been stated that in the catalysts consisting of CuCr2O4 and CuCrO2, phase-to-phase interaction occurs, resulting in the increase in surface area and catalytic activity[10]. And the sample with a Cu/Cr ratio of 0.7, in which the molar ratio of CuCr2O4 to CuCrO2 is much closer to unity, shows the most stable and the highest catalytic activity. It is considered to be the reason responsible for the high burning rate and low pressure-exponent of the sample 6# with a Cu/Cr molar ratio of 0.7.
4 Conclusions
1) Cu-Cr-O nanocomposites with controllable composition, uniform particle shape and size are synthesized by a citric acid(CA) complexing method.
2) Well-crystallized Cu-Cr-O nanocomposites can be obtained after the CA-Cu-Cr precursors are calcined at 500 ℃ for 3 h. Crystal structure of the as-obtained Cu-Cr-O nanocomposites varies with n(Cu)/n(Cr) in the starting reactants, that is, CuCr2O4-Cr2O3(n(Cu)/ n(Cr)< 0.5), CuCr2O4(n(Cu)/n(Cr)=0.5), CuCr2O4-CuO (0.5< n(Cu)/n(Cr)<0.6), CuCr2O4-CuCrO2(0.6
3) Addition of the as-synthesized Cu-Cr-O nanocomposites as catalysts obviously enhances the burning rate as well as lowers the pressure exponent of the AP-based solid-state propellants. Noticeably, catalyst with n(Cu)/n(Cr) of 0.7 exhibits the most promising catalytic activity, high burning rate and low pressure exponent (0.43) at all pressures, due to the effective phase interaction between spinel CuCr2O4 and delafossite CuCrO2 contained in the as-synthesized Cu-Cr-O nanocomposites.
References
[1] ADKINS H, CONNOR R. The catalytic hydrogenation of organic compounds over copper chromite[J]. J Am Chem Soc, 1931, 53(3): 1091-1095.
[2] CONNOR R, FOLKERS K, ADKINS H. The preparation of copper-chromium oxide catalysts for hydrogenation[J]. J Am Chem Soc, 1932, 54(3): 1138-1145.
[3] THOMAS W, RIENER W. An improved laboratory preparation of copper-chromium oxide catalyst[J]. J Am Chem Soc, 1949, 71(3): 1130-1130.
[4] ROY S, GHOSE J. Synthese and studies on some copper chromite spinel oxide composites[J]. Materials Research Bulletin, 1999, 34(7): 1179-1186.
[5] STEFANESCU M, SASCA V, MRACEC M. Investigation concerning the mechanism and kinetics of copper(II) chromite formation[J].Revue Roumaine de Chimie, 2000, 45(12): 1063-1068.
[6] KHASINA V, SIMENTSOVA I I, YUR'EVA. T M Moderate- temperature reduction of copper chromite by hydrogen and hydrogen desorption from the surface of reduced chromite[J]. Kinetics and Catalysis, 2000, 41(2): 282-286.
[7] PRASAD R. Highly active copper chromite catalyst produced by thermal decomposition of ammoniac copper oxalate chromate[J]. Materials Letters, 2005, 59(29/30): 3945-3949.
[8] COMINO G, GERVASINI A, RAGAINI V. Methane combustion over copper chromite catalysts[J]. Catalysis Letters, 1997, 48(1/2): 39-45.
[9] XANTHOPOULOU G, VEKINIS G. Investigation of catalytic oxidation of carbon monoxide over a Cu-Cr-oxide catalyst made by self-propagating high-temperature synthesis[J]. Applied Catalysis B: Environmental, 1998, 19(1): 37-44.
[10] ZHAO F, LI G, LI Y N, et al. Influence of phase-to-phase interaction between CuCrO2 and CuCr2O4 on their structure and catalytic activity[J].Chinese Journal of Catalysis, 2003, 24(1): 11-16.
[11] KAWAMOTO A M, PARDINI L C, REZENDE L C. Synthesis of copper chromite catalyst[J]. Aerospace Sci Technol, 2004, 8(7): 591- 598.
[12] JACOBS P W M, WHITEHEAD H M. Decomposition and combustion of ammonium perchlorate[J]. Chem Rev, 1969, 69(4): 551-590.
[13] RAJEEV R, DEVI K A, ABRAHAM A, et al. Thermal decomposition studies(Part 19): Kinetics and mechanism of thermal decomposition of copper ammonium chromate precursor to copper chromite catalyst and correlation of surface parameters of the catalyst with propellant burning rate[J]. Thermochimica Acta, 1995, 254(15): 235-247.
[14] PATRON L, POCOL V, CARP O, et al. New synthetic route in obtaining copper chromite(I): Hydrolysis of some soluble salts[J]. Mater Res Bull, 2001, 36(7/8): 1269-1276.
[15] STROUPE J D. An X-ray diffraction study of the copper chromites and of the “copper-chromium oxide” catalyst[J]. J Am Chem Soc, 1949, 71(2): 569-572.
[16] ARMSTRONG R W, BASCHUNG B, BOOTH D W, et al. Enhanced propellant combustion with nanoparticles[J]. Nano Letters, 2003, 3(2): 253-255.
[17] BASCHUNG B, GRUNE D, LICHT H H, et al. Combustion of Energy Materials[M]. KUO K K, DELUCA L T, ed. New York: Begell House, 2001.
[18] LI Wei. Synthesis and catalytic characteristics of Bi2O3/Cu-Cr-O core/shell structured nano-composites[J]. Journal of Central South University: Science and Technology: 2006, 37(6): 1019-1024. (in Chinese)
[19] LI Wei. Preparation and structure characteristics of nano-Bi2O3 powders with mixed crystal structure[J]. J Cent South Univ Technol, 2005, 12(3): 243-245.
(Edited by YANG Bing)
Foundation item: Project (2003AA305820) supported by the National High-Tech Research and Development Program of China; Project(2006) supported by the Postdoctoral Foundation of Central South University, China
Received date: 2006-12-29; Accepted date: 2007-03-12
Corresponding author: LI Wei, PhD; Tel:+86-731-8830768; E-mail: 973@mail.csu.edu.cn

