���ʳ���ǿ����Ǧ��-�һ���ԭ��ظ�ѡ��ϵ
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2020���4��
�������ߣ�Can GUNGOREN Yasin BAKTARHAN Ismail DEMIR Safak Gokhan OZKAN
����ҳ�룺1102 - 1110
�ؼ��ʣ���Ǧ���һ���ԭ��أ���ѡ������������-����ճ��ʱ��
Key words��galena; potassium ethyl xanthate; flotation; ultrasound; bubble-particle attachment time
ժ Ҫ���о����һ���ԭ���(KEX)���ڵ����������ó�����߷�Ǧ��ɸ��ԵĿ����ԡ�Ϊ�ˣ������滯ѧ�о��⣬��������ѡʵ�飬����������ͬ��������ˮƽ�͵���ʱ��ʱ��Zeta���ơ��Ӵ��Ǻ�����-����ճ��ʱ�䡣�о��������������������Ϊ30 W������ʱ��Ϊ2 minʱ��ѡ���������Ϊ77.5%�����⣬ʹ�ó������ø�����Zeta���ơ�����ĽӴ��Ǻ��̵�����-����ճ��ʱ�䣬������������ʹ��Ǧ�����ˮ�����ӡ�
Abstract: The objective of this study is to investigate the improvement possibilities of the floatability of galena with ultrasonic application in the presence of potassium ethyl xanthate (KEX). For this purpose, micro-flotation experiments were carried out in addition to surface chemistry studies including zeta potential, contact angle, and bubble-particle attachment time measurements at various ultrasonic power levels and conditioning time. The results showed that, the maximum micro-flotation recovery of 77.5% was obtained with 30 W ultrasound power and 2 min conditioning time. In addition, more negative zeta potential values were obtained with ultrasound as well as higher contact angle and lower bubble-particle attachment time, which indicated the increased hydrophobicity of galena with ultrasound.
Trans. Nonferrous Met. Soc. China 30(2020) 1102-1110
Can GUNGOREN1, Yasin BAKTARHAN1, Ismail DEMIR1, Safak Gokhan OZKAN1,2
1. Department of Mining Engineering, Engineering Faculty, Istanbul University-Cerrahpasa, Buyukcekmece, Istanbul 34500, Turkey;
2. Department of Robotics and Intelligent Systems, The Institute of the Graduate Studies in Science and Engineering, Turkish-German University, Beykoz, Istanbul 34820, Turkey
Received 12 July 2019; accepted 16 February 2020
Abstract: The objective of this study is to investigate the improvement possibilities of the floatability of galena with ultrasonic application in the presence of potassium ethyl xanthate (KEX). For this purpose, micro-flotation experiments were carried out in addition to surface chemistry studies including zeta potential, contact angle, and bubble-particle attachment time measurements at various ultrasonic power levels and conditioning time. The results showed that, the maximum micro-flotation recovery of 77.5% was obtained with 30 W ultrasound power and 2 min conditioning time. In addition, more negative zeta potential values were obtained with ultrasound as well as higher contact angle and lower bubble-particle attachment time, which indicated the increased hydrophobicity of galena with ultrasound.
Key words: galena; potassium ethyl xanthate; flotation; ultrasound; bubble-particle attachment time
1 Introduction
Lead (Pb) is an important metal for electronics, chemistry, metallurgy, medical, nuclear military, and mineral processing industries [1]. The main commercial mineral of lead is galena (PbS). Galena is generally recovered by flotation which is an effective method used to separate valuable minerals from gangue minerals by taking advantage of differences between the wettability of their surfaces [2]. The surface of galena is hydrophilic and has high wettability. Xanthates are the most common collectors used in galena flotation [3,4].
There are several studies carried out to enhance the galena flotation. SONG et al [5] flocculated the galena fines with potassium amyl xanthate (KAX) and performed floc flotation. Their results showed that the floatability of galena is strongly related to the floc size and is increased from 40% to 100% under the optimum conditions. They also reported that the flotation recovery can be greatly improved with a small addition of kerosene with decreasing potassium ethyl xanthate (KEX) consumption. MCFADZEAN et al [6,7] used some collector mixtures in the flotation of galena. AWATEY et al [8] studied the effect of particle size on the flotation of galena and reported that the recovery of flotation is strongly influenced by the overall particle size distribution. ANG et al [9] investigated the detachment of galena particles from air bubbles in the presence of two different frother types. SUN et al [10] used diethyl dithiocarbamate (DDTC) as a collector in galena single mineral flotation and showed that the interaction between galena and DDTC is an electrochemical process. LUO et al [11] reported that the increase of solid- in-pulp concentration has the most significant effect on the recovery and selective separation of galena mineral. The density functional theory (DFT) studies carried out by LONG et al [12] indicated that the presence of water molecules improves the reactivity of the atoms on galena surface during the reaction with xanthate molecules.
Ultrasound can be used successfully to improve the flotation of minerals benefitting from the extreme conditions such as acoustic streaming, cavitation, and micro-jets in company with high temperature (5000 K) and high pressure (1.013��108 Pa) [13]. However, there are very few studies on galena flotation with ultrasound. CELIK [14] used ultrasound as a pre-treatment in galena flotation and reported that the positive effects of ultrasound on galena flotation can be related to the surface cleaning, and micro-bubble generating effects, which lead to an enhanced bubble-particle attachment.
In literatures, ultrasound has been used for improving the beneficiation of several minerals in the recent studies [15] including quartz [13], colemanite, coal [16], ilmenite [17], phosphate [18], copper [19], pyrite [20], zinc [21], graphite [22], graphene [23], and feldspar [24] successfully; however, the studies about the effect of ultrasound on the floatability of galena are quite limited [14,25,26]. The motive behind this study is to fill the gap in this area.
Furthermore, agitation vessels are not necessarily very efficient liquid-solid contactors, owing to the uneven dissipation of energy throughout the pulp in the vessels [25]. Ultrasonic application during conditioning can be a useful alternative for solving this problem. Therefore, the main objective of this study is to reveal the effect of ultrasonic application on the flotation of galena in terms of surface chemistry studies using an ultrasonic probe. In line with this purpose, in this study, ultrasound was applied during the conditioning process of galena with KEX and micro-flotation studies were carried out. In addition, the effects of ultrasound on the surface properties and the wettability of galena were observed with zeta potential, contact angle, and bubble-particle attachment time measurements.
2 Experimental
2.1 Materials
The galena samples with high purity were obtained from a Pb-Zn processing plant in Turkey. The d50 and d80 sizes of the samples were 70 and 160 ��m, respectively. The chemical composition of the sample, which is seen in Table 1, indicated that the sample contained 68.282 wt.% Pb.
Table 1 Chemical composition of galena sample (wt.%)

The results of the chemical analyses were obtained from the mine site which was described by the standard method in ISO 12743 (Chapter 9) [27]. In addition, in order to use the contact angle measurements, a high purity galena sample (>90% galena) (~15 cm) was obtained from the feed of the same mineral processing plant. The sample was prepared using cutting equipment (Struers Discoplan, Denmark) and polished manually using silica carbide powder.
In the flotation experiments, potassium ethyl xanthate (KEX) was used as a collector and pine oil was employed as a frother. These reagents were obtained from Kimsan, Turkey. In addition, the pH adjustment was carried out using HCl and NaOH which were purchased from Merck, Germany. All of the solutions were prepared with deionized (DI) water (resistivity: 18.2 M����cm) (Millipore Milli-Q, Merck, Germany), freshly.
In the ultrasonic experiments, the ultrasonic probe (Bandelin HD 3200, Germany) which can be adjusted by 30-150 W ultrasonic power, was used as source of ultrasound.
2.2 Micro-flotation experiments
For the micro-flotation experiments, 2 g of galena sample under 300 ��m particle size was conditioned with the desired dosage of KEX, and at a constant dosage of pine oil (250 mL/L), 1% solid ratio and 500 r/min stirring speed using a magnetic stirrer for 10 min. Then, the suspension was transferred to a glass micro-flotation cell (30 mm �� 210 mm) with 200 mL volume and frit with 10-16 ��m pore size. Firstly, the micro-flotation experiments were performed at various KEX dosages (50, 75, 150, 300, and 600 g/t) without ultrasound in order to determine the optimum KEX dosage for the further micro-flotation experiments. All micro-flotation experiments were performed at room temperature (296.15 K), natural pH (9.0-9.5), and 50 cm3/min N2 flow rate for 2 min. The flotation products were dewatered with a black ribbon filter paper and dried at 378.15 K in a drying oven. The micro-flotation recoveries were calculated using the product weights.
In the ultrasonic experiments, firstly, the effect of the ultrasonic power on galena flotation was investigated. In this context, ultrasound was applied in the conditioning process at various ultrasonic powers (30, 90, and 150 W) during magnetic stirring and a constant conditioning time of 10 min. Then, the effect of the conditioning time on the micro-flotation recovery was investigated with decreasing the conditioning time to 5 and 2 min, separately. During the conditioning process with ultrasound, the temperature and pH of the suspension were also measured as a function of time in order to observe the changes in the conditioning environment.
2.3 Zeta potential measurements
The zeta potential measurements of galena were carried out as a function of pH by electrophoresis method by using a zetameter (Brookhaven Zetaplus, USA). The pH values were adjusted using 0.1 mol/L of HCl or NaOH (Merck, Germany). In order to carry out the zeta potential measurements, the galena samples were ground under 38 ��m using an automated agate mortar (Retsch, Germany). 1 g galena sample in 100 mL of DI water or KEX + pine oil solution at the desired dosage was stirred at 500 r/min with a magnetic stirrer for 10 min without and with ultrasound. The suspensions were waited for 5 min to allow larger particles to settle down. Then, a small amount of suspension was taken from the top of the suspension and transferred to the measurement cell [28]. 20 measurements were conducted at each pH value and the average values were calculated.
2.4 Contact angle measurements
Prior to contact angle measurements, the galena sample with a polished surface was conditioned with KEX by immersing it into the desired KEX solution with and without ultrasound at 30 W, and then dried at room temperature. The contact angle was measured with the sessile drop method using a goniometer (Attension Theta Lite, Biolin Scientific, Finland). For this purpose, the galena sample was placed with its polished surface upwards and a DI water droplet was placed onto the polished surface using a glass micro-syringe. Then, the contact angle was measured automatically by the goniometer software with observing the droplet shape with the help of a CCD camera [29].
2.5 Bubble-particle attachment time measurement
The bubble-particle attachment time measurements were carried out using a BCT-100 bubble-particle attachment timer (Bratton Engineering and Technical Associates, LLC, USA) with the measurement setup given in Fig. 1.
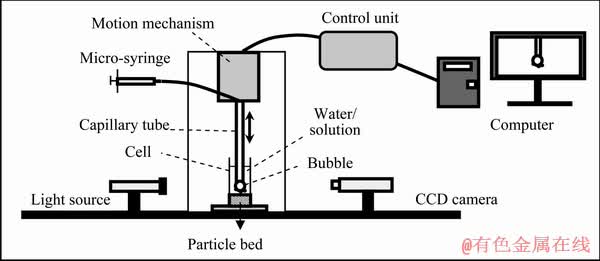
Fig. 1 Bubble-particle time measurement setup
In the bubble-particle attachment time measurements, firstly, the same galena-KEX conditioning procedure in the micro-flotation experiments was carried out for 2, 5, and 10 min with and without ultrasound, respectively. Then, approximately 5 mL of the suspension was transferred to the measurement cell beneath the capillary tube of the attachment timer. A galena particle bed was formed at the bottom of the measurement cell. The measurement cell was elevated with the stand of the attachment timer and the glass capillary tube of the attachment timer was located in the measurement cell. A bubble of about 3 mm in diameter was formed at the end of the capillary tube using a micro-syringe, and then the distance between the bubble and the particle bed was adjusted properly. The bubble was kept in contact with the particle bed for a pre-determined contact time (1, 10, 100, and 1000 ms). Then, the attachment of the particles to the bubbles was observed through a CCD camera linked to a monitor. 20 measurements were performed at different locations of the particle bed and the observations were recorded. The bubble-particle attachment time will be determined if 50% of the observations result in attachment [30,31].
3 Results and discussion
3.1 Micro-flotation
The results of the micro-flotation experiments carried out at various KEX dosages without ultrasound are given in Fig. 2. As seen in Fig. 2, the micro-flotation recovery was 29.80% in the absence of KEX because of the entrainment of the hydrophilic particles. It increased with increasing KEX dosage sharply and reached a plateau at 62.8% at 200 g/t KEX. These results are in accordance with the study of VUCINIC et al [32]. In order to observe the effects of ultrasound, 75 g/t KEX dosage which gave a micro-flotation recovery at an intermediate level (54.55%) was chosen to use in further ultrasonic experiments.
The results of the micro-flotation experiments at conditioning time of 10 min are shown in Fig. 3 as a function of ultrasonic power. The results in Fig. 3 indicated that the use of ultrasound at 30 W for 10 min did not significantly affect the floatability of galena. However, the micro-flotation recoveries tend to decrease at higher ultrasonic powers.
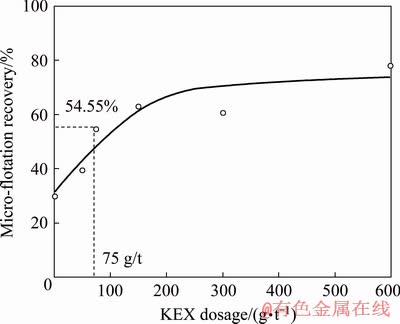
Fig. 2 Micro-flotation recovery of galena with respect to KEX dosage
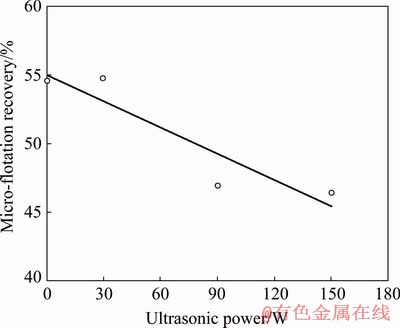
Fig. 3 Micro-flotation recovery of galena as function of ultrasonic power at conditioning time of 10 min
There are several studies in the literature that mentioned the importance of the ultrasonic power [33-35] and the detrimental effect of high power ultrasound in flotation [30,36].
The pH and temperature of the conditioning suspension were also measured during the ultrasonic conditioning process and the results are shown in Figs. 4(a) and (b), respectively.
As known from the literature, the flotation of galena was affected by the pH of the medium [37,38]. Galena-KEX flotation could be carried out effectively between pH 2.5 and 10.5 [39]; but above pH 11, galena was depressed due to the formation of lead hydroxyl species, such as Pb(OH)3-, which were retained on the surface [40]. The pH of the medium can change during the ultrasonic application [33]. Meanwhile, water dissociated under ultrasonic treatment and H2, O2, H2O2 and HO2 generated, as well as H�� and OH�� radicals. Furthermore, the ultrasonic application could cause some other ions (e.g. Ca2+ and Mg2+) to be adsorbed onto the mineral surface. These ions could become hydroxyl complexes and precipitate on the mineral surfaces related to the pH of the medium, therefore, ultrasonic treatment could also affect the adsorption of the collectors [33,41]. The pH measurements of this study indicated that the pH of the conditioning suspension fluctuated between 8 and 9 at all ultrasonic powers, as seen in Fig. 4(a).
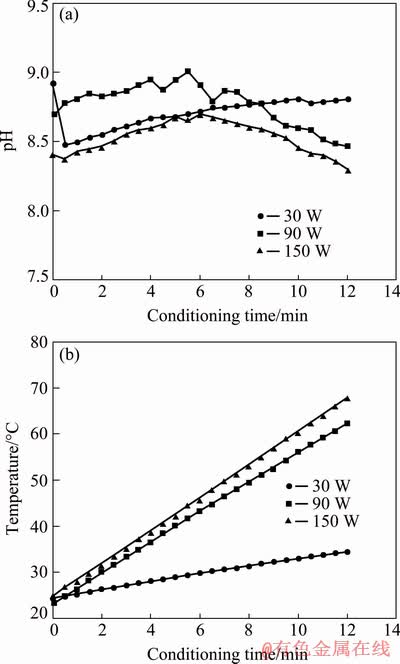
Fig. 4 pH (a) and temperature (b) of conditioning suspension with conditioning time at different ultrasonic powers
The temperature of the conditioning medium also affects the flotation success by changing the reagent adsorption behaviors [13]. The temperature of the medium can also alter the trade-off between the increase in cavitation bubbles and the reduction in the intensity of bubble collapse [25].
Figure 4(b) showed that the temperature of the suspension increased linearly with conditioning time. The maximum temperatures reached at 10 min were 34, 62, and 78 ��C at 30, 90, and 150 W, respectively.
The time of the ultrasonic application is another important factor for the flotation recovery and concentrate grade. ALDRICH and FENG [25] reported that sulfide flotation recovery and rate could be enhanced with ultrasound in the xanthate conditioning process as well as concentrate grade compared with other mechanical conditioning related to the increased hydrophobicity of the sulfides. However, they also reported that prolonging ultrasonic conditioning time resulted in decreased recovery. GHADYANI et al [42] and GUNGOREN et al [30] stated that prolonging conditioning time with ultrasound influenced the particle-collector adsorption negatively as well.
In this context, it is thought that high power and prolonged use of the ultrasound can be detrimental for the conditioning process by creating turbulent flow conditions in the conditioning medium and/or increasing its temperature. Therefore, further studies were carried out at the minimum ultrasonic power (30 W) and the conditioning time was reduced down to 5 and 2 min. The results of the micro-flotation experiments after 2, 5, and 10 min conditioning with and without ultrasound are given in Fig. 5.
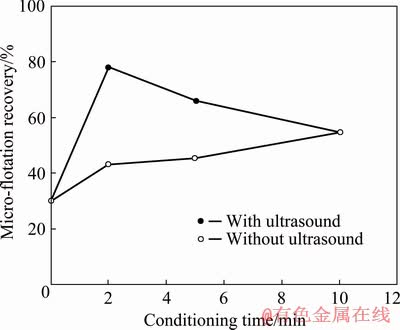
Fig. 5 Micro-flotation recovery with respect to conditioning time in the presence of 75 g/t KEX, without and with 30 W ultrasound
As seen in Fig. 5, the micro-flotation recovery was 29.80% in the absence of KEX and was in accordance with the result in Ref. [14], and it increased with conditioning time.
In the case of the 30 W ultrasonic application, the micro-flotation recovery increased sharply at conditioning time from 0 to 2 min; however, it decreased with further increasing conditioning time.
3.2 Zeta potential
The zeta potential results of galena are seen in Fig. 6 in the absence and presence of KEX and without and with ultrasound.
Figure 6(a) showed that the zeta potential of galena was negative at all pH values, which was in accordance with the result in Ref. [38]. The results of the zeta potential measurements also corrected the results of MA et al [43]. It was seen that the zeta potential became more negative at alkaline pH values. It is also clear in Fig. 6(b) that the zeta potential of galena became more negative with ultrasound in the presence of 75 g/t KEX.
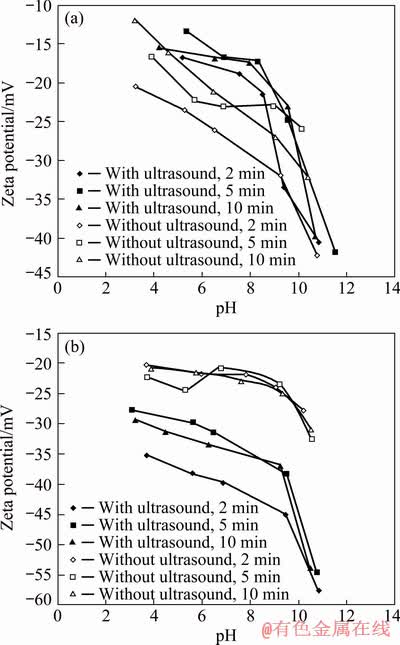
Fig. 6 Zeta potentials of galena in the absence (a) and in the presence of 75 g/t KEX (b), without and with 30 W ultrasound
Xanthate ions were electrochemically active and adsorbed onto galena via a one-electron transfer chemisorption reaction. Galena could be easily floated with xanthate due to both chemisorption of xanthate ions and formation of lead xanthate on the galena surfaces [44].
The result of the zeta potential measurements was an indicator of an increase in the adsorption of anionic KEX molecules which were chemically adsorbed on the negative galena surfaces. Moreover, it was also seen in Fig. 6(b) that 2 min conditioning time gave the most negative zeta potential values, supporting the micro-flotation and contact angle results.
The zeta potential of galena conditioned with 75 g/t KEX at natural pH (9.0-9.5) with respect to conditioning time without and with ultrasound is seen in Fig. 7. It was clearly seen in Fig. 7 that more negative zeta potentials were obtained with ultrasound.
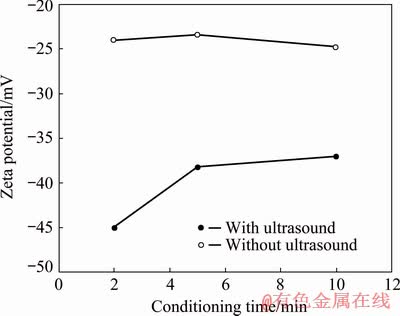
Fig. 7 Zeta potential of galena conditioned with 75 g/t KEX at natural pH 9.0-9.5 with respect to conditioning time without and with ultrasound
3.3 Contact angle
Figure 8 shows the results of the contact angle measurements at 75 g/t KEX with respect to conditioning time.
As seen in Fig. 8, the contact angle of galena increased from 43�� to 47�� and 50�� at 2, 5, and 10 min conditioning time, respectively without ultrasound. On the contrary, the contact angle was 55�� at 2 min, it decreased to 49�� and 41�� at 5 and 10 min, respectively with ultrasound. It is clear that in the case of KEX adsorption, the contact angle increased with conditioning time without ultrasound, and an increase in the conditioning time had a negative influence on the contact angle with ultrasound.
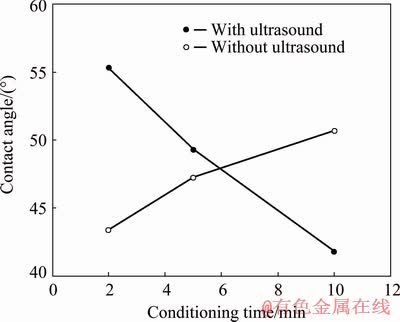
Fig. 8 Contact angle of galena with conditioning time without and with ultrasound
3.4 Bubble-particle attachment time
Apart from contact angle measurements, bubble-particle attachment time was a strong indicator of the hydrophobicity. The bubble- particle attachment efficiency for galena without and with ultrasound is given in Figs. 9(a) and (b), respectively. It was seen in Fig. 9(a) that the attachment efficiency increased with conditioning time without ultrasound in keeping with micro- flotation, zeta potential, and contact angle measurements. On the other hand, higher bubble- particle attachment efficiency was obtained at less conditioning time with ultrasound such that the highest attachment efficiency was obtained at conditioning time of 2 min.
Bubble-particle attachment time as a function of conditioning time is given in Fig. 10. As seen in Fig. 10, the bubble-particle attachment time was 510 ms at conditioning time of 2 min and decreased to 40 and 25 ms at conditioning time of 5 and 10 min, respectively without ultrasound. On the other hand, attachment time of 6 ms was obtained for galena at conditioning time of 2 min with ultrasound, which increased with increasing conditioning time.
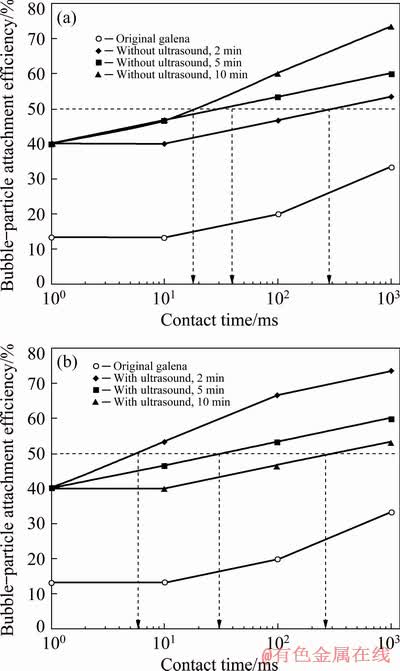
Fig. 9 Bubble-particle attachment efficiency for galena without (a) and with (b) ultrasound
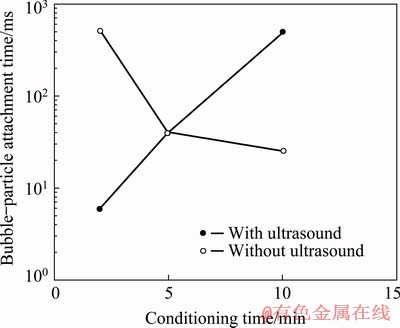
Fig. 10 Bubble-particle attachment time of galena as function of conditioning time
The increase in the hydrophobicity of galena could be attributed to the adsorption of the collector molecules more effectively to the fresh galena surfaces created by the surface cleaning effect of ultrasound, which was reported by many researchers, previously [19,45]. Furthermore, low power ultrasound could affect the organization of the adsorbed collector molecules and neaten them, which caused more equal distribution of the hydro- phobic regions on the mineral surface and therefore enhanced the bubble-particle attachment [26,30]. On the other hand, using ultrasound at high powers could cause some detrimental effects on the floatability [46].
4 Conclusions
(1) The limited use of ultrasound increased the micro-flotation recovery of galena. The power and application time of ultrasound have a crucial effect on the floatability of galena.
(2) More negative zeta potential values are obtained with ultrasound, which supports the increased adsorption of KEX molecules.
(3) Higher contact angle and lower bubble-particle attachment time are obtained under the optimum ultrasonic conditions (30 W power and 2 min conditioning time).
(4) The increase in the micro-flotation recovery is related to the increased hydrophobicity of galena by using ultrasound in the conditioning process with KEX. The increase in the hydro- phobicity of galena with low power ultrasound can be attributed to the increase in the fresh galena surfaces and thus the efficiency of the collector adsorption.
Acknowledgments
This work was supported by the Research Fund of Istanbul University under grant FAB-2017- 25658.
References
[1] FANG S, XU L, WU H, SHU K, XU Y, ZHANG Z, CHI R, SUN W. Comparative studies of flotation and adsorption of Pb(II)/benzohydroxamic acid collector complexes on ilmenite and titanaugite [J]. Powder Technology, 2019, 345: 35-42.
[2] FENG B, ZHANG W, GUO Y, WANG T, LUO G, WANG H, HE G. The flotation separation of galena and pyrite using serpentine as depressant [J]. Powder Technology, 2019, 342: 486-490.
[3] FLORES-ALVAREZ, J M, ELIZONDO-ALVAREZ M A, DAVILA-PULIDO G I, GUERRERO-FLORES A D, URIBE-SALAS A. Electrochemical behavior of galena in the presence of calcium and sulfate ions [J]. Minerals Engineering, 2017, 111: 158-166.
[4] LAN L H, CHEN J H, LI Y Q, LAN P, YANG Z, AI G Y. Microthermokinetic study of xanthate adsorption on impurity-doped galena [J]. Transactions of Nonferrous Metals Society of China, 2016, 26: 272-281.
[5] SONG S, LOPEZ-VALDIVIESO A, REYES-BAHENA J L, LARA-VALENZUELA C. Floc flotation of galena and sphalerite fines [J]. Minerals Engineering, 2001, 14: 87-98.
[6] MCFADZEAN B, CASTELYN D G, O��CONNOR C T. The effect of mixed thiol collectors on the flotation of galena [J]. Minerals Engineering, 2012, 36-38: 211-218.
[7] MCFADZEAN B, MHLANGA S S, O��CONNOR C T. The effect of thiol collector mixtures on the flotation of pyrite and galena [J]. Minerals Engineering, 2013, 50-51: 121-129.
[8] AWATEY B, SKINNER W, ZANIN M. Effect of particle size distribution on recovery of coarse chalcopyrite and galena in Denver flotation cell [J]. Canadian Metallurgical Quarterly, 2013, 52: 465-472.
[9] ANG Z, BOURNIVAL G, ATA S. Influence of frothers on the detachment of galena particles from bubbles [J]. International Journal of Mineral Processing, 2013, 121: 59-64.
[10] SUN W, SUN C, LIU R Q, CAO X F, TAO H B. Electrochemical behavior of galena and jamesonite flotation in high alkaline pulp [J]. Transactions of Nonferrous Metals Society of China, 2016, 26: 551-556.
[11] LUO X, FENG B, WONG C, MIAO J, MA B, ZHOU H. The critical importance of pulp concentration on the flotation of galena from a low grade lead�Czinc ore [J]. Journal of Materials Research and Technology, 2016, 5: 131-135.
[12] LONG X, CHEN Y, CHEN J, XU Z, LIU Q, DU Z. The effect of water molecules on the thiol collector interaction on the galena (PbS) and sphalerite (ZnS) surfaces: A DFT study [J]. Applied Surface Science, 2016, 389: 103-111.
[13] GUNGOREN C, OZDEMIR O, OZKAN S G. Effects of temperature during ultrasonic conditioning in quartz-Amine flotation [J]. Physicochemical Problems of Mineral Processing, 2017, 53: 687-698.
[14] CELIK M S. Effect of ultrasonic treatment on the floatability of coal and galena [J]. Separation Science and Technology, 1989, 24: 1159-1166.
[15] CHEN Y, TRUONG V N T, BU X, XIE G. A review of effects and applications of ultrasound in mineral flotation [J]. Ultrason Sonochem, 2020, 60: 1-12.
[16] MAO Y, BU X, PENG Y, TIAN F, XIE G. Effects of simultaneous ultrasonic treatment on the separation selectivity and flotation kinetics of high-ash lignite [J]. Fuel, 2020, 259: 1-10.
[17] FANG S, XU L, WU H, XU Y, WANG Z, SHU K, HU Y. Influence of surface dissolution on sodium oleate adsorption on ilmenite and its gangue minerals by ultrasonic treatment [J]. Applied Surface Science, 2020, 500: 1-12.
[18] HASSANI F, NOAPARAST M, SHAFAEI TONKABONI S Z. A study on the effect of ultrasound irradiation as pretreatment method on flotation of sedimentary phosphate rock with carbonate�Csilicate gangue [J]. Iranian Journal of Science and Technology, Transactions A: Science, 2019: 2787-2798.
[19] VIDELA A R, MORALES R, SAINT-JEAN T, GAETE L, VARGAS Y, MILLER J D. Ultrasound treatment on tailings to enhance copper flotation recovery [J]. Minerals Engineering, 2016, 99: 89-95.
[20] OZUN S, VAZIRI HASSAS B, MILLER J D. Collectorless flotation of oxidized pyrite [J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 2019, 561: 349-356.
[21] BAGHERI B, VAZIFEH MEHRABANI J, FARROKHPAY S. Recovery of sphalerite from a high zinc grade tailing [J]. J Hazard Mater, 2019, 381: 1-12.
[22] BARMA S D, BASKEY P K, RAO D S, SAHU S N. Ultrasonic-assisted flotation for enhancing the recovery of flaky graphite from low-grade graphite ore [J]. Ultrason Sonochem, 2019, 56: 386-396.
[23] MCCOY T M, PARKS H C W, TABOR R F. Highly efficient recovery of graphene oxide by froth flotation using a common surfactant [J]. Carbon, 2018, 135: 164-170.
[24] MALAYOGLU U, OZKAN S G. Effects of ultrasound on desliming prior to feldspar flotation [J]. Minerals, 2019, 9: 784.
[25] ALDRICH C, FENG D. Technical note-Effect of ultrasonic preconditioning of pulp on the flotation of sulphide ores [J]. Minerals Engineering, 1999, 12: 701-707.
[26] GUNGOREN C, SOW M A, BAKTARHAN Y, DEMIR I, OZKAN S G, OZDEMIR O. An investigation of flotation behaviour of ultrasound treated galena[C]// Proceedings of International Symposium on Multidisciplinary Academics Studies (IMASES 2018). Istanbul, Turkey, 2018: 93-102.
[27] ISO 12743. Copper, lead, zinc and nickel concentrates- Sampling procedures for determination of metal and moisture content [S]. 2018.
[28] WANG X, OZDEMIR O, HAMPTON M A, NGUYEN A V, DO D D. The effect of zeolite treatment by acids on sodium adsorption ratio of coal seam gas water [J]. Water Res, 2012, 46: 5247-5254.
[29] OZDEMIR O, TARAN E, HAMPTON M A, KARAKASHEV S I, NGUYEN A V. Surface chemistry aspects of coal flotation in bore water [J]. International Journal of Mineral Processing, 2009, 92: 177-183.
[30] GUNGOREN C, OZDEMIR O, WANG X, OZKAN S G, MILLER J D. Effect of ultrasound on bubble-particle interaction in quartz-amine flotation system [J]. Ultrason Sonochem, 2019, 52: 446-454.
[31] OZDEMIR O, KARAGUZEL C, NGUYEN A V, CELIK M S, MILLER J D. Contact angle and bubble attachment studies in the flotation of trona and other soluble carbonate salts [J]. Minerals Engineering, 2009, 22: 168-175.
[32] VUCINIC D R, LAZIC P M, ROSIC A A. Ethyl xanthate adsorption and adsorption kinetics on lead-modified galena and sphalerite under flotation conditions [J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 2006, 279: 96-104.
[33] SHU K, XU L, WU H, FANG S, WANG Z, XU Y, ZHANG Z. Effects of ultrasonic pre-treatment on the flotation of ilmenite and collector adsorption [J]. Minerals Engineering, 2019, 137: 124-132.
[34] CAO Q, CHENG J, FENG Q, WEN S, LUO B. Surface cleaning and oxidative effects of ultrasonication on the flotation of oxidized pyrite [J]. Powder Technology, 2017, 311: 390-397.
[35] SOW M A. The effect of energy applied on the surface of a sulfied mineral on mineral surface properties [D]. Istanbul: Istanbul University, 2017. (in Turkish)
[36] CHEN Y, BU X, TRUONG V N T, PENG Y, XIE G. Study on the effects of pre-conditioning time on the floatability of molybdenite from the perspective of cavitation threshold [J]. Minerals Engineering, 2019, 141: 1-6.
[37] ELIZONDO-ALVAREZ M A, DAVILA-PULIDO G I, BELLO-TEODORO S, URIBE-SALAS A. Role of pH on the adsorption of xanthate and dithiophosphinate onto galena [J]. Canadian Metallurgical Quarterly, 2019, 58: 107-115.
[38] WANG D, JIAO F, QIN W, WANG X. Effect of surface oxidation on the flotation separation of chalcopyrite and galena using sodium humate as depressant [J]. Separation Science and Technology, 2017, 53: 961-972.
[39] FUERSTENAU M C, CHANDER S, WOODS R. Sulfide mineral flotation[C]// Froth Flotation: A Century of Innovation. USA: Society for Mining, Metallurgy and Exploration, Inc., 2007: 425-464.
[40] RALSTON J. The chemistry of galena flotation: Principles & practice [J]. Minerals Engineering, 1994, 7: 715-735.
[41] NGUYEN T D, AL TAHTAMOUNI T M, HUONG P T, THANG P Q. Selective flotation separation of ABS/PC from ESR plastic wastes mixtures assisted by ultrasonic catalyst/H2O2 [J]. Journal of Environmental Chemical Engineering, 2019, 7: 1-5.
[42] GHADYANI A, NOAPARAST M, TONKABONI S Z S. A study on the effects of ultrasonic irradiation as pretreatment method on high-ash coal flotation and kinetics [J]. International Journal of Coal Preparation and Utilization, 2018, 38: 374-391.
[43] MA Y W, HAN Y X, ZHU Y M, LI Y J, LIU H. Flotation behaviors and mechanisms of chalcopyrite and galena after cyanide treatment [J]. Transactions of Nonferrous Metals Society of China, 2016, 26: 3245-3252.
[44] LOPEZ VALDIVIESO A, SANCHEZ LOPEZ A A, SONG S, GARCIA MARTINEZ H A, LICON ALMADA S. Dextrin as a regulator for the selective flotation of chalcopyrite, galena and pyrite [J]. Canadian Metallurgical Quarterly, 2013, 46: 301-309.
[45] CELIK M S, HANCER M, MILLER J D. Flotation chemistry of boron minerals [J]. J Colloid Interf Sci, 2002, 256: 121-131.
[46] GURPINAR G, SONMEZ E, BOZKURT V. Effect of ultrasonic treatment on flotation of calcite, barite and quartz [J]. Mineral Processing and Extractive Metallurgy, 2013, 113: 91-95.
Can GUNGOREN1, Yasin BAKTARHAN1, Ismail DEMIR1, Safak Gokhan OZKAN1,2
1. Department of Mining Engineering, Engineering Faculty, Istanbul University-Cerrahpasa, Buyukcekmece, Istanbul 34500, Turkey;
2. Department of Robotics and Intelligent Systems, The Institute of the Graduate Studies in Science and Engineering, Turkish-German University, Beykoz, Istanbul 34820, Turkey
ժ Ҫ���о����һ���ԭ���(KEX)���ڵ����������ó�����߷�Ǧ��ɸ��ԵĿ����ԡ�Ϊ�ˣ������滯ѧ�о��⣬��������ѡʵ�飬����������ͬ��������ˮƽ�͵���ʱ��ʱ��Zeta���ơ��Ӵ��Ǻ�����-����ճ��ʱ�䡣�о��������������������Ϊ30 W������ʱ��Ϊ2 minʱ��ѡ���������Ϊ77.5%�����⣬ʹ�ó������ø�����Zeta���ơ�����ĽӴ��Ǻ��̵�����-����ճ��ʱ�䣬������������ʹ��Ǧ�����ˮ�����ӡ�
�ؼ��ʣ���Ǧ���һ���ԭ��أ���ѡ������������-����ճ��ʱ��
(Edited by Wei-ping CHEN)
Corresponding author: Can GUNGOREN; E-mail: can@istanbul.edu.tr
DOI: 10.1016/S1003-6326(20)65281-5

