
Phase transformation in reductive roasting of
laterite ore with microwave heating
CHANG Yong-feng(畅永锋), ZHAI Xiu-jing(翟秀静), FU Yan(符 岩),
MA Lin-zhi(马林芝), LI Bin-chuan(李斌川), ZHANG Ting-an(张廷安)
School of Materials and Metallurgy, Northeastern University, Shenyang 110004, China
Received 20 November 2007; accepted 24 March 2008
Abstract: Selective reduction of laterite ores followed by acid leaching is a promising method to recover nickel and cobalt metal, leaving leaching residue as a suitable iron resource. The phase transformation in reduction process with microwave heating was investigated by XRD and the reduction degree of iron was analyzed by chemical method. The results show that the laterite samples mixed with active carbon couple well with microwave and the temperature can reach approximate 1 000 ℃ in 6.5 min. The reduction degree of iron is controlled by both the reductive agent content and the microwave heating time, and the reduction follows Fe2O3→Fe3O4→FeO→Fe sequence. Sulphuric acid leaching test reveals that the recoveries of nickel and iron increase with the iron reduction degree. By properly controlling the reduction degree of iron at 60% around, the nickel recovery can reach about 90% and iron recovery is less than 30%.
Key words: laterite; microwave; carbothermic reduction
1 Introduction
Laterite ores consist of the majority (65%) of the world’s nickel reserves. Although sulfides are still the primary source of nickel and cobalt, increasing underground costs and decreasing the grades of the sulfide ores attract more attention towards the exploitation of laterite ores[1]. One of the major economic factors, which limit the development of the laterite ores, is that the energy costs for processing these ores are 2-3 times higher than that for the sulfide ores[2]. Therefore, in the long term it will be necessary to develop new and more economical processing technologies.
A new technology, which is considered for application in extractive metallurgy, is microwave technology[3-7]. Microwave energy is a nonionizing electromagnetic radiation with frequencies in the range from 300 MHz to 300 GHz. Microwave heating is unique and offers a number of advantages over conventional heating, such as reduction in energy consumption and process duration, rapid and controllable heating, peculiar temperature distribution, and selectivity of energy deposition[8-10].
Selective reduction of laterite ores followed by acid leaching is a promising method to recover nickel and cobalt metal, leaving leaching residue as a suitable iron resource[11]. The iron in leach liquor can be precipitated as goethite, which is also a usable iron resource and has no contamination to the environment. In the present work, the microwave radiation was applied to the carbothermic reduction of limonite laterite ore. The phase transformation in the reduction process and the acid leaching of reduced calcine were investigated.
2 Experimental
2.1 Apparatus
Microwave heating was carried out at a frequency of 2.45 GHz at 800 W power level using WD800B Galanz microwave oven. X-ray diffractometry was utilized to analyze mineral compositions of laterite samples before and after reduction treatment.
2.2 Materials
The limonite laterite ore (<74 μm) from Philippines used in this work was dried at 90 ℃ for 12 h to remove the moist water. The chemical composition of the ore is given in Table 1. The analytically pure grade active carbon was used as reductive agent.
Table 1 Chemical composition of limonite laterite ore (mass fraction, %)

2.3 Carbothermic reduction with microwave heating
Reduction of iron oxides in a direct reduction system has been known to occur by gaseous phases, for example CO, rather than the solid carbon[12-13]. The reduction reactions are presented as Reaction (1). Fig.1 shows the equilibrium diagram of reduction. The Reaction (2) is the highly endothermic Boudouard reaction. The overall oxide reduction with carbon is shown in Reaction (3).
MO+CO=M+CO2 (1)
CO2+C=2CO (2)
MO+C=M+CO (3)

Fig.1 Equilibrium diagram of reduction with CO-CO2
According to Reactions (4)-(7), the reduction of iron oxide to magnetite (in the reaction region (Ⅰ) of Fig.1) needs 1.9% of carbon, adding the reduction of nickel and cobalt oxide to metallic state. About 5.2% of carbon is needed to completely reduce iron oxide to wustite state (in the reaction region (Ⅱ) of Fig.1). The third series of sample was added with 7.6% carbon, which was 50% carbon excessive to reduce iron to wustite.
3Fe2O3+C=2Fe3O4+CO (4)
Fe2O3+C=2FeO+CO (5)
NiO+C=Ni+CO (6)
CoO+C=Co+CO (7)
Sample mixed with active carbon was put in a covered ceramic crucible which was then heated by microwave for required time. After irradiation, a Chromel-Alumel thermocouple with diameter of 1.0 mm was rapidly inserted to the bottom of the sample to measure the temperature[14].
2.4 Leaching of reduced laterite with sulphuric acid
All the leaching processes were operated at room temperature, with the liquid-to-solid ratio of 20(v/w), sulphuric acid of 0.5 mol/L and stirring speed of 400 r/min for 1.0 h.
Iron content in the solution was determined by the titration method using K2Cr2O7. Nickel content was measured by the ICP-AES method. To evaluate the reduction degree of the laterite, the total iron, ferrous iron and metal iron content were analyzed with the standard chemical method[15].
3 Results and discussion
3.1 Microwave heating temperature
The results of microwave heating the laterite ores mixed with different carbon content can be seen in Fig.2. Although laterite ore has poor microwave absorbing ability at low temperature[14], when being mixed with active carbon, the temperature of samples rapidly reaches approximately 1 000 ℃ in 6.5 min. Then the temperature keeps stabilized in the next heating period until the end of 30 min.
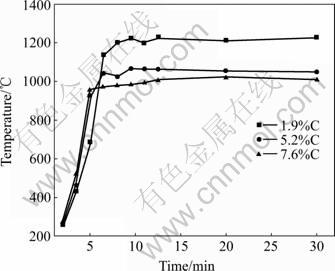
Fig.2 Temperature of laterite with different carbon contents heated by microwave
The fast heating behavior of the sample mixed with carbon may attribute to two factors. Firstly, carbon is an excellent microwave absorber. When it is mixed into other dielectric material, it can facilitate coupling with microwave. This has been proved by PICALES[14]. Secondly, magnetite is similar to the carbon, i.e. a hyperactive material in microwave. When the laterite sample is heated, the dominant mineral phase, goethite, converts to hematite, which is easy to be reduced to magnetite in weak reductive atmosphere. The fast appearance of magnetite phase, which can be seen in Figs.3-5, further enhances the heating behavior of sample.
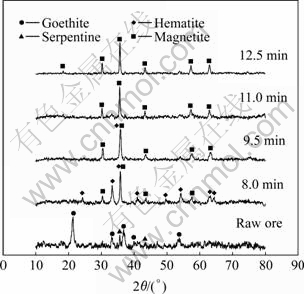
Fig.3 XRD patterns of laterite ores mixed with 1.9% carbon heated by microwave for different time

Fig.4 XRD patterns of laterite ores mixed with 5.2% carbon heated by microwave for different time
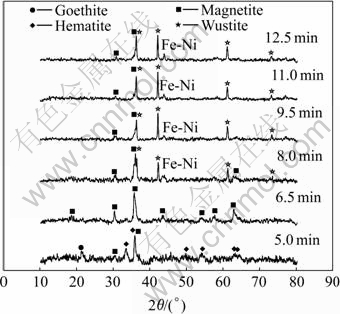
Fig.5 XRD patterns of laterite ores mixed with 7.6% carbon heated by microwave for different time
As far as the different stabilized temperature in Fig.2 is concerned, it may attribute to the different carbon content. Because the different carbon content, the iron oxides are reduced to different degree. In the XRD patterns of Fig.3, sample with 1.9% carbon has almost converted to Fe3O4, while in the XRD patterns of Fig.4 and Fig.5 the samples with higher content carbon have higher content of FeO phase after reduction. The Fe3O4 is a better microwave absorber than FeO, hence the higher the carbon content of the sample, the lower thestabilized temperature after long time microwave heating.
3.2 Phase transformation in reduction process
From the XRD patterns of Fig.3, it can be seen that the raw laterite ore mainly consists of goethite and minor serpentine phase. The reduction of Fe2O3 to Fe3O4 begins no later than 8.0 min and ends in 11.0 min for the sample containing 1.9% carbon. The hematite has the strongest peak at 2θ=33.04?, but the sub-strongest peak lies at 2θ=35.6?, which is close to the strongest peak of magnetite at 2θ=35.82?. So the two peaks are overlapped.
In the XRD patterns of Fig.4, it can be seen that laterite ores with 5.2% carbon have much larger reduction speed. At 6.5 min all the hematite peaks disappear, and at 9.5 min the wustite peaks are clearly visual. Similar to Fig.3, the strongest peak of magnetite at 2θ=35.82? overlaps with wustite at 2θ=36.22?. The strongest peak of wustite lies at 2θ= 42.06?.
Although the carbon is added in the stoichiometric ratio to reduce iron oxide to wustite, there is still minor magnetite phase after 12.5 min microwave heating. This may be because the reduction of iron oxide to wustite needs relatively higher CO partial pressure than magnetite. So the third series of sample added with 7.6% carbon, which is 50% carbon excessive to reduce iron oxide to wustite, is also tested. The XRD pattern is shown in Fig.5.
With the increase of carbon content, the Fe-Ni alloy peak appears in 8.0 min to 12.5 min in Fig.5, which is not found in Fig.3 and Fig.4. So if Fe-Ni alloy phase is wanted, the reduction reaction should lie in the region (Ⅲ) of Fig.1.
The reduction process of iron oxide was studied by many researchers. Above 570 ℃, the reduction follows the sequence of Fe2O3→Fe3O4→FeO→Fe[16]. This step-by-step reaction can be clearly seen in Figs.3-5.
One difficulty in microwave heating application is the temperature control. But in this work the ‘thermo-runaway’ phenomenon doesn’t appear. It can be seen that both the reduction agent content and the microwave irradiating time affect the reduction degree of laterite, so it is feasible to obtain the desired phase composition of product by adjusting the carbon content and microwave heating time.
3.3 Sulphuric acid leaching behavior of reduced laterite
It is commonly believed that the selective reduction of laterite should lie in the upper region of (Ⅰ) in Fig.1, that is, nickel and cobalt are reduced to metallic state, while iron only to magnetite. But under that condition the nickel recovery is relatively low. For the sample shown in Fig.3, the nickel recovery is less than 40% by sulphuric acid leaching. REDDY et al[17] also found in order to achieve more than 90% extraction of nickel, about 50% of iron should be reduced to the wustite phase. Since the oxide nickel in the ores is present mainly in solid solution, for complete reduction of nickel, the end product is iron-nickel alloy. So iron is inevitably partly reduced to wustite.
In this work, the reduced laterite ores were analyzed using the chemical method to determine the total iron, ferrous iron and iron metal content. The reductive degree (Dr) is defined as
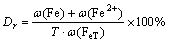 (8)
(8)
The reduction degree and the recovery of nickel and iron at different microwave heating time are shown in Fig.6 and Fig.7. The reduction degree increases with the microwave heating time. So does the recovery of nickel and iron. At 12.5 min, the maximal Ni recoveries of the laterite samples with 5.2% and 7.6% carbon are 92% and 97%, and the Fe recoveries are 39% and 55%, respectively. To enhance the selectivity of Ni recovery, the reduction degree should be controlled at 60% or so. In this condition, Ni recovery can reach about 90% and Fe recovery no more than 30%, as shown in Fig.6.
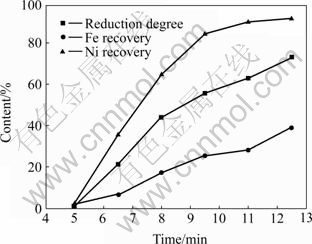
Fig.6 Effect of microwave heating time on extraction rate of nickel and iron and reduction degree of laterite mixed with 5.2% carbon
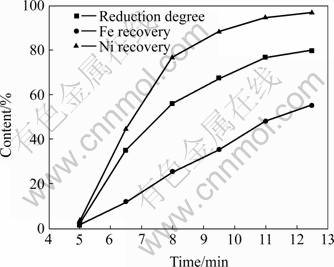
Fig.7 Effect of microwave heating time on extraction rate of nickel and iron and reduction degree of laterite mixed with 7.6% carbon
4 Conclusions
1) The laterite ores added with active carbon are heated rapidly under microwave radiation. The temperature of samples can reach approximately 1 000 ℃ in 6.5 min.
2) The reduction of iron oxide follows Fe2O3→Fe3O4→FeO→Fe sequence, and is a step-by-step reaction. Both the reduction agent content and the microwave heating time affect the reduction degree of laterite.
3) The recovery of nickel and iron increases with the reduction degree of laterite. To selectively leach nickel, the proper reduction degree is about 60%, with nickel recovery of 90% and iron recovery less than 30%.
References
[1] Dalvi A D, Bacon W G, Osborne R C. Past and future of nickel laterite projects [C]// International Laterite Nickel Symposium-2004. Charlotte, North Carolina, USA, 2004: 23-27.
[2] Thomas F T. Comparative costs of nickel sulphides and laterites [J]. Resources Policy, 1995, 21(3): 179-187.
[3] Amankwah R K, Pickles C A, Yen W T. Gold recovery by microwave augmented ashing of waste activated carbon [J]. Minerals Engineering, 2005, 18: 517-526.
[4] Harahsheh M A, Kingman S, Hankins N, SOMERFIELD C, BRADSHAW S, LOUW W. The influence of microwaves on the leaching kinetics of chalcopyrite [J]. Minerals Engineering, 2005, 18: 1259-1268.
[5] TONG Zhi-fang, BI Shi-wen, YU Hai-yan, WU Yu-sheng. Leaching kinetics of non-constant temperature process of calcium aluminate slag under microwave radiation [J]. The Chinese Journal of Nonferrous Metals, 2006, 16(2): 357-362. (in Chinese)
[6] USLU T, ATALAY U, AROL A I. Effect of microwave heating on magnetic separation of pyrite [J]. Colloids and Surfaces A: Physicochem Eng Aspects, 2003, 225: 161-167.
[7] OLUBAMBI P A, POTGIETER J H, HWANG J Y, NDLOVU S. Influence of microwave heating on the processing and dissolution behavior of low-grade complex sulfide ores [J]. Hydrometallurgy, 2007, 89: 127-135.
[8] THOSTENSON E T, CHOU T W. Microwave processing fundamentals and applications [J]. Composite: Part A, 1999, 30: 1055-1071.
[9] CLARK D E, FOLZ D C, WEST J K. Processing materials with microwave energy [J]. Materials Science and Engineering, 2000, A287: 153-158.
[10] BYKOV Y V, RYBAKOV K I, SENENOV V E. High-temperature microwave processing of materials [J]. Journal of Physics D: Applied Physics, 2001, 34: R55-R75.
[11] PURWANTO H, SHIMADA T, TAKAHASHI R, YAGI J I. Recovery of nickel from selectively reduced laterite ore by sulphuric acid leaching [J]. ISIJ International, 2003, 43(2): 181-186.
[12] STANDISH N, HUANG W. Microwave application in carbothermic reduction of iron ores [J]. ISIJ International, 1991, 31(3): 241-245.
[13] LIU Gui-su, STREZOV V, LUCAS J A, WIBBERLEY L J. Thermal investigations of direct iron ore reduction with coal [J]. Thermochimica Acta, 2004, 410: 133-140.
[14] PICALES C A. Microwave heating behaviour of nickeliferous limonitic laterite ores [J]. Minerals Engineering, 2004, 17: 775-784.
[15] Beijing Research Institute of Mining & Metallurgy. Chemical phase analysis [M]. Beijing: Metallurgical Industry Press, 1979: 241-245. (in Chinese)
[16] JOZWIAK W K, KACZMAREK E, MANIECKI T P, IGNACZAK W, MANIUKIEWICZ W. Reduction behavior of iron oxides in hydrogen and carbon monoxide atmospheres [J]. Applied Catalysis A: General, 2007, 326: 17-27.
[17] REDDY B R, MURTHY B V R, SWAMY Y V, RAY H S. Correlation of nickel extraction with iron reduction in oxidic nickel ore by a thermogravimetric method [J]. Thermochimica Acta, 1995, 264: 185-192.
Foundation item: Project(50774020) supported by the National Natural Science Foundation of China
Corresponding author: ZHAI Xiu-jing; Tel: +86-24-83687729; E-mail: zhaixj@smm.neu.edu.cn
(Edited by YANG Bing)