��Ͻ������Ժ�������/��������(��ñ��)�Ѵ���������ı���
��Դ�ڿ����й���ɫ����ѧ��2015���4��
�������ߣ�Oktay CELEP Vedat SERBEST
����ҳ�룺1286 - 1297
Key words��refractory gold and silver ore; characterization; diagnostic leaching; mineral liberation analysis (MLA); alkaline pretreatment
ժ Ҫ��Ϊ��̽�����軯����֮ǰ����Ԥ�������գ���������Ϸ����Ժ���������/��������(��ñ��)�Ѵ�����/���������ϸ���������������������Ȼ��������(�ߴ�Ϊ6~24 ��m)���ڣ������ڷ���ʯ��ʯӢ�����еĺ�������������������С��������ȷ���(MLA)�����������������������ڣ�����������Ǧ����������/�������ʯӢ�С�����Ҫ��������(Ag2S)���������ڣ������ڿ�ʯ������Ǧ����軯����ʵ��������ڽ���ʱ��ﵽ24 h��ʯ(d80��50 ��m)�н�����Ľ����ʷֱ�ֻ��76%��23%����ʯ����Ͻ���ʵ�����ϸ�Ŀ�������������������Ҫ�������������������У��������/������ͻƼ������͵�����Ǧ������Щ�������������ڼ�����Һ�з��������������Щ�о������軯����֮ǰ����ʯ��KOH��Һ�н��м���Ԥ�������ܽ����ͽ�Ľ����ʷֱ���ߵ�87%��90%����Щ���ֱ������Խ�������Ϊһ���µ���Ϸ�������������������/���������Ľ�/�������Ѷȣ����ҿ���Ϊһ��Ԥ�������������������ʡ�
Abstract: A detailed characterization of an iron oxy/hydroxide (gossan type) bearing refractory gold/silver ore was performed with a new diagnostic approach for the development of a pretreatment process prior to cyanide leaching. Gold was observed to be present as native and electrum (6-24 ��m in size) and associated with limonite, goethite and lepidocrocite within calcite and quartz matrix. Mineral liberation analysis (MLA) showed that electrum is found as free grains and in association with beudantite, limonite/goethite and quartz. Silver was mainly present as acanthite (Ag2S) and electrum and as inclusions within beudantite phase in the ore. The cyanide leaching tests showed that the extractions of gold and silver from the ore (d80: 50 ��m) were limited to 76% and 23%, respectively, over a leaching period of 24 h. Diagnostic leaching tests coupled with the detailed mineralogical analysis of the ore suggest that the refractory gold and silver are mainly associated within iron oxide mineral phases such as limonite/goethite and jarosite-beudantite, which can be decomposed in alkaline solutions. Based on these characterizations, alkaline pretreatment of ore in potassium hydroxide solution was performed prior to cyanidation, which improved significantly the extraction of silver and gold up to 87% Ag and 90% Au. These findings suggest that alkaline leaching can be used as a new diagnostic approach to characterize the refractoriness of iron oxy/hydroxide bearing gold/silver ore and as a pretreatment method to overcome the refractoriness.
Trans. Nonferrous Met. Soc. China 25(2015) 1286-1297
Oktay CELEP, Vedat SERBEST
Hydromet B&PM Group, Division of Mineral & Coal Processing, Department of Mining Engineering, Karadeniz Technical University, Trabzon 61080, Turkey
Received 18 August 2014; accepted 10 December 2014
Abstract: A detailed characterization of an iron oxy/hydroxide (gossan type) bearing refractory gold/silver ore was performed with a new diagnostic approach for the development of a pretreatment process prior to cyanide leaching. Gold was observed to be present as native and electrum (6-24 ��m in size) and associated with limonite, goethite and lepidocrocite within calcite and quartz matrix. Mineral liberation analysis (MLA) showed that electrum is found as free grains and in association with beudantite, limonite/goethite and quartz. Silver was mainly present as acanthite (Ag2S) and electrum and as inclusions within beudantite phase in the ore. The cyanide leaching tests showed that the extractions of gold and silver from the ore (d80: 50 ��m) were limited to 76% and 23%, respectively, over a leaching period of 24 h. Diagnostic leaching tests coupled with the detailed mineralogical analysis of the ore suggest that the refractory gold and silver are mainly associated within iron oxide mineral phases such as limonite/goethite and jarosite-beudantite, which can be decomposed in alkaline solutions. Based on these characterizations, alkaline pretreatment of ore in potassium hydroxide solution was performed prior to cyanidation, which improved significantly the extraction of silver and gold up to 87% Ag and 90% Au. These findings suggest that alkaline leaching can be used as a new diagnostic approach to characterize the refractoriness of iron oxy/hydroxide bearing gold/silver ore and as a pretreatment method to overcome the refractoriness.
Key words: refractory gold and silver ore; characterization; diagnostic leaching; mineral liberation analysis (MLA); alkaline pretreatment
1 Introduction
Refractory gold and silver ores yield low gold and silver extractions in cyanide leaching [1]. A suitable pre-treatment process such as roasting [2], pressure oxidation [3], bio-oxidation [4], alkaline [5,6] or alkaline sulfide leaching [7] and, to a limited extent, ultrafine grinding [8-10], is often required to overcome refractoriness and render the encapsulated gold and silver accessible to the lixiviant action of cyanide and oxygen.
The gossan ores are intensely oxidized, weathered or decomposed rock, usually the upper and exposed part of an ore deposit or mineral vein. The gossan (or iron cap) ores mainly consist of quartz and iron oxides, limonite, goethite, and jarosites. It has been reported that the natural jarosite-type minerals such as beudantite [PbFe3(SO4)(AsO4)(OH)6], argentojarosite [AgFe3- (SO4)2(OH)6] and the silver-bearing plumbojarosite [(Ag,Pb)Fe3(SO4)2(OH)6] are the major silver (230 g/t Ag in average) carriers in the gossan ores from Rio Tinto (Spain) [11-14]. Gold and silver extractions from these types of ores are often limited to 70%-80% Au and 40% Ag since jarosite-type phases are not readily dissolved during the conventional cyanidation process [15].
The diagnostic leaching procedure developed by LORENZEN [16] is a characterization method to determine the reasons of refractory behavior of gold and silver ores. It involves cyanide leaching to extract unlocked gold or silver after each stage of acid treatment using a variety of mineral acids selected to decompose gold/silver-bearing target mineral(s) [17-19]. The decomposition of some mineral phases in different acid leaching stages may be represented by the following chemical reactions [20-22]:
CaCO3+2HCl��CaCl2+H2O+CO2 (1)
MS+H2SO4��MSO4+H2S [M=Zn, Cu, Pb, etc.] (2)
2FeS2+10HNO3��2Fe3++2H++4SO42-+10NO+4H2O (3)
SiO2+6HF��H2SiF6+2H2O (4)
LORENZEN and van DEVENTER [18] used diagnostic leaching to identify the refractory nature of gold ores (only 1.3%-2.4% leachable gold with direct cyanidation). It was found that approximately 95% of Au in ore was extracted by means of selective destruction of gold-bearing minerals by nitric and sulphuric acids within diagnostic leaching procedure. TEAGUE et al [23] studied diagnostic leaching of a refractory gold ore and a bulk flotation concentrates. HENLEY et al [24] studied the characterization of a refractory telluride ore by a two-stage diagnostic approach. CELEP et al [22] concluded that the refractoriness of a refractory gold ore was associated with the dissemination and encapsulation of the very fine gold particles largely within carbonates, oxides, sulfides and, to a small extent, silicates present in the ore matrix. Although the diagnostic leaching procedure includes different acid leaching stages and the subsequent cyanidation, it is inefficient to decompose jarosite type minerals. In this work, an alkaline leaching was adopted as a diagnostic leaching stage for decomposition of jarosite type minerals.
Detailed characterization such as mineral liberation size, mineral associations and modal mineralogical analysis is pre-requisite for plant design and mineral processing operations [25]. The distribution of precious metal, grain size and mode of occurrence (liberation, exposure, and mineral association) and their gangue components were quantitatively determined by means of automated scanning electron microscopic techniques (QEMSCAN/MLA). The mineral liberation analysis (MLA) system consists of a specially developed software package and a scanning electron microscopy (SEM) equipped with an energy dispersive spectrum (EDS) analyzer. The analysis was operated on particles mounted in 30 mm epoxy resin blocks to provide automated quantitative mineral liberation characterization [26]. The system utilized stable back-scattered electron (BSE) signals defined by its average atomic number to generate images of mineral grains from a scanning electron microscope (SEM). Mineral identification was verified by using of a single X-ray analysis at a point within the particle. Parameters such as liberation, mineral grain size, mineral locking and mineral associations were then measured by the system. In this system, X-ray mapping was also used when similar BSE brightness of minerals was identified [27]. There are seven different operating modes available for the MLA system: standard BSE liberation analysis (BSE), extended BSE liberation analysis (XBSE), sparse phase liberation analysis (SPL), particle X-ray mapping (PXMAP), selected particle X-ray mapping (SXMAP), X-ray modal analysis (XMOD), and rare phase search (RPS) [28].
In this work, the detailed characterization of a gold- and silver-bearing gossan type ore was performed to provide an insight into the refractory behaviour of the ore. The mineral liberation analysis was undertaken to determine the occurrence and liberation of gold and silver, and to identify refractory silver-bearing phases. A new, modified diagnostic approach was developed for gossan type ore so as to identify the refractions of silver-bearing minerals as well as gold-bearing phases. Based on the results of these characterizations, an alkaline pretreatment process was proposed and demonstrated on the ore sample.
2 Experimental
2.1 Materials
The ore sample (~150 kg) was obtained from  (Turkey) gold and silver deposit. It was crushed to <1 mm in a jaw crusher and then ground in a rod mill. The chemical composition of the ore sample (Table 1 and Table 2) was determined using inductively coupled plasma-emission spectroscopy (ICP-ES) after four kinds of acid digestion and X-ray fluoressans (XRF). The gold and silver contents of the ore were determined by lead collection fire assay fusion-gravimetric finish method. The ore sample was found to contain 8.4 g/t Au, 167.0 g/t Ag, 2.6% Pb, 4.8% Zn and 1.6% As mass fraction. Quartz, goethite, calcite and dolomite were identified as the major mineral phases by X-ray diffraction analysis (Fig. 1). The optical mineralogical analysis of the ore sample was also performed under an ore microscope (Leitz Wetzlar 1432) for which the polished sections of a number of hand-picked pieces were prepared from the as-received ore.
(Turkey) gold and silver deposit. It was crushed to <1 mm in a jaw crusher and then ground in a rod mill. The chemical composition of the ore sample (Table 1 and Table 2) was determined using inductively coupled plasma-emission spectroscopy (ICP-ES) after four kinds of acid digestion and X-ray fluoressans (XRF). The gold and silver contents of the ore were determined by lead collection fire assay fusion-gravimetric finish method. The ore sample was found to contain 8.4 g/t Au, 167.0 g/t Ag, 2.6% Pb, 4.8% Zn and 1.6% As mass fraction. Quartz, goethite, calcite and dolomite were identified as the major mineral phases by X-ray diffraction analysis (Fig. 1). The optical mineralogical analysis of the ore sample was also performed under an ore microscope (Leitz Wetzlar 1432) for which the polished sections of a number of hand-picked pieces were prepared from the as-received ore.
Table 1 Chemical analysis of ore sample (mass fraction, %)

Table 2 Element analysis of ore sample (mass fraction, %)

2.2 Methods
2.2.1 Sample preparation
The crushed ore sample was ground by a rod mill for 3-50 min at 50% pulp density. Ultra-fine grinding of the ore was carried out using 2 mm ceramic ball at 800 r/min stirring rate in a laboratory scale pin-type vertical stirred mill as described in details elsewhere [9]. The particle size analysis of the samples was carried out using the laser diffraction method with Malvern Mastersizer 2000.

Fig. 1 XRD pattern of ore sample showing major mineral phases
2.2.2 Mineralogical characterization
For the mineral liberation analysis (MLA), the samples were split using a quantachrome mini-riffler to obtain representative sub-samples, which were used to prepare a number of resin mounts. The polished mounts were examined for MLA using an FEI Quanta 600F scanning electron microscope (SEM). By a combination of image analysis employing atomic number contrast imaging (from BSE or BSE-signal intensity) and energy dispersive spectrometry (EDS) using two Bruker 5010 SDD detectors, minerals and other attributes were directly measured on the MLA system. BSE signal intensity is proportional to the mean atomic number of minerals. The field emission gun MLA was set at an accelerating voltage of 25 kV and a spot size of 5.5. XBSE measurements were performed for the characterization of modal mineralogy and associations. Sparse phase liberation (SPL) measurements were performed for gold and silver searches.
2.2.3 Diagnostic leaching tests
The cyanidation tests (lasted for 24 h) were carried out in a glass reactor. The sodium cyanide (1.5 g/L) was added to maintain the concentration of free NaCN at the predetermined level over the leaching period and the consumption of cyanide was recorded. Free CN- concentration was determined by titration with silver nitrate using p-dimethylaminobenzalrhodanine (0.02% (w/w) in acetone) as the indicator [10]. pH was controlled at ~10.5 by the addition of 1 mol/L NaOH. Pulp (25% (w/w) solids) was stirred at the speed of 700 r/min at room temperature.
Potassium hydroxide (KOH) reagent is the most effective alkaline reagent among reagents such as NH4OH, Na2CO3 and NaOH [20], which was used successfully in a pre-treatment method to obtain the improved gold and silver extractions from antimonial refractory gold and silver ore by cyanidation [6]. In this work, potassium hydroxide was used to ensure the most effectively decomposition as alkaline digestion stage at diagnostic leaching procedure. Diagnostic leaching tests were carried out by acid/alkaline leaching stages (KOH, HCl, H2SO4, HNO3 and HF) to destroy specific minerals, followed by cyanidation of the residue from each stage to determine the amounts of gold, silver and other metal. Diagnostic leaching procedure of the ore sample and the parameters are given in Fig. 2. The leaching tailings were digested by hot acid mixture (HCl, HNO3, HClO4 and HF). Analysis of gold and silver in the samples removed at predetermined intervals was carried out using atomic absorption spectrophotometer (AAS, Perkin Elmer AAnalyst 400). The leaching solutions and leaching residues at the end of each stage were also analyzed for Au, Ag, Fe, Pb, Zn and As to establish a mass balance and to determine the metal recovery.

Fig. 2 Diagnostic leaching procedure
2.2.4 Alkaline pre-treatment
Alkaline pretreatment experiments were conducted to investigate the effect of potassium hydroxide leaching (5 mol/L KOH). The sample (d80��50 ��m) was leached in a 1 L beaker submerged in a water bath to control the leaching temperature (80 ��C) within ��2 ��C. The beaker with 200 mL leaching solution (KOH) and ~70 g ore sample (solids 26% (w/v)) was continuously stirred for 3 h. At the end of leaching period (3 h), solid and liquid phases were separated by filtration and the dried residues were subjected to cyanidation for gold and silver extractions.
3 Results and discussion
3.1 Mineralogical characteristics of ore
3.1.1 Optical mineralogical examination
The optical mineralogical analysis indicated that the ore consisted of predominantly iron oxide minerals such as lepidocrocite, limonite, goethite, hematite; and pyrite and gangue minerals such as quartz, calcite, dolomite and goethite/limonite (Fig. 3). Gold particles were observed as 6-24 ��m in size to be associated with limonite, goethite, lepidocrocite minerals and within calcite and quartz matrix. In the previous study, it was found that ore consists of cerusite, anglesite, galena, pyromorphite, mimetite and plumbojarosite as lead minerals; smithsonite, hydrozincite, hemimorphite, adamite and sphalerite as zinc minerals; limonite, hematite, goethite, pyrite, magnetite and siderite as iron minerals [29].
3.1.2 Mineral liberation analysis (MLA) results
Modal mineralogy determined by the extended BSE liberation analysis (XBSE) measurement showed that the ore is mostly composed of limonite/goethite (50.62%), quartz (13.79%), calcite (5.62%), dolomite/ankerite (2.71%), hemimorphite (2.20%) and clay (7.00%) (Table 3). The limonite/goethite minerals contain variable amounts of Zn, Mn, Pb and As. The elemental composition of the selected minerals was determined by the semi-quantitative EDS analysis. Microprobe analysis is required for accurate determination of the elemental composition. Pb-bearing minerals found in trace amounts in the sample are galena and cerussite. The zinc minerals are smithsonite, which contain small amounts of Fe, hemimorphite, adamite and clay. The gangue minerals are comprised of quartz, feldspar, illite, kaolinite, calcite, mica/chlorite dolomite and ankerite. Gold and silver are native gold/electrum and acanthite (Fig. 4(a)-(f)). Electrum is found as free grains and in association with beudantite, limonite/goethite, mimetite and quartz (Fig. 4(e)). Silver is present as acanthite and electrum and in the form of beudantite phase in the ore [12]. Most of the acanthite is locked within particles containing limonite/goethite (Fig. 4(d)), barite (Fig. 4(f)), beudantite (Fig. 4(a)) (Table 4).

Fig. 3 Presence and association of gold particles
Mineral liberation analysis by free surface showed that 43% of gold/electrum and 11% of acanthite are present as free mineral in the ore. But, most of gold/ electrum and acanthite (47% and 63%, respectively) was associated with locked minerals (Fig. 5) which is responsible for the low gold and silver extractions during conventional cyanidation. The particle exposure gives an indication of the leachability of an ore at a specific particle size.
The classified images of MLA particle X-ray mapping (PXMAP) showing the associations of gold/ electrum and acanthite with each other mineral phases are presented in Fig. 6. Most of the locked gold/electrum is associated with beudantite or limonite/goethite minerals (Fig. 6(a)). Acanthite is mainly found as locked within quartz, beudantite cerussite, calcite and limonite/goethite iron oxide minerals in the ore (Fig. 6(b)).

Fig. 4 BSE images of ore samples
Table 3 Modal mineralogy as determined by extended BSE liberation analysis (XBSE) measurement

Table 4 EDS analysis of beudatite phase in ore (mass fraction, %)
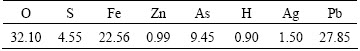

Fig. 5 Mineral liberation by free surface for gold/electrum and acanthite free
Native gold and electrum in the ore are mostly present as binary and ternary particles associated with limonite/goethite, quartz, beudantite and clay matrix (Fig. 4(c) and Table 5). Acanthite is mostly associated with cerussite, beudantite, limonite/goethite and quartz minerals (Table 6).
3.2 Cyanide leaching of ore
Effect of particle size and leaching time on gold and silver extractions are shown in Fig. 7. Most of gold and silver was extracted into solution over the cyanidation of 2 h. Gold extraction was improved from 76.5% to 87.8% when the particle size was decreased from 50.24 to 18.20 mm by conventional grinding. Similarly, silver extraction was improved from 23.4% to 43.4% when the particle size was decreased from 50.24 to 11.25 mm. By decreasing the particle size (d80: ~10 mm), the extractions of gold and silver were enhanced by 11.3% and 20.0%, respectively, as there was more contact between cyanide and the formed new surfaces (Fig. 8).
However, the decreasing in particle size (<10 mm) by stirred mill reduced the gold and silver extractions, especially gold extraction (26.5% reduction). Furthermore, the cyanide consumption increased from 2.3 to 4.7 kg/t (Fig. 8), which can be explained with the more liberation of cyanide consuming minerals. Thus, the dissolution of zinc during cyanidation can be referred as the reason of the increasing of cyanide consumption, except for Pb and Fe (not shown). The iron oxides or hydroxides such as lepidocrocite, limonite, goethite (FeOOH) and hematite (Fe2O3) are actually insoluble in alkaline cyanide solutions, which may coat gold particles and reduce sometimes significantly the leaching efficiency [21], which may express the reason of low gold extraction in the case of the ultrafine grinding.
Apart from gold and silver, many metals or minerals can be dissolved in dilute alkaline cyanide solutions [21]. They may cause a reduction in gold or silver extraction and the consuming of cyanide and oxygen due to the side reactions. The general reaction for a sulphide containing a divalent metal cation is given as follows:
2MS+2(x+1)CN-+O2+2H2O��2M(CN)x(x-2)-+2SCN-+4OH- (5)
3.3 Diagnostic leaching
Figure 9 illustrates the gold and silver extractions followed by standard cyanide leaching after acid/alkaline treatment stages in diagnostic leaching. Gold extraction (85%) represents the recoverable gold in cyanide leaching of the untreated ore (Fig. 9(a)). The remaining amount of the gold appeared to be refractory presumably due to inaccessibility of gold particles to the action of cyanide. Gold extraction was improved up to 100% at cyanidation after acid/alkaline treatments (KOH and H2SO4). The refractoriness of the ore for gold can be described as 8% Au associated with arsenates or jarasites such as beudantite can be decomposed in alkaline leaching and 6% Au with Cu-Zn sulphides and labile pyrite can be decomposed in acid leaching [16].

Fig. 6 MLA classified images showing gold/electrum (a) and acanthite (b) in ore sample
Table 5 Mineral associations for gold/electrum (mass fraction, %)

Table 6 Mineral associations for acanthite in ore (mass fraction, %)


Fig. 7 Effect of particle size on extraction of gold (a) and silver (b) from ore in cyanidation (1.5 g/L NaCN) over a period of 24 h

Fig. 8 Effect of particle size on final extraction of gold and silver, and consumption of cyanide over 24 h leaching period (1.5 g/L NaCN)
Silver extraction during cyanide leaching after each treatment stage in diagnostic leaching is given in Fig. 9(b). Silver extraction (~30%) obtained from direct cyanidation shows the recoverable amount of silver in cyanidation. The results of cyanidation after acid/alkaline (KOH, HCl, H2SO4 and HNO3) leaching treatment showed that ~60% of silver appeared to be related with beudantite type jarosites minerals which are decomposed in KOH alkaline leaching. The beudantite, plumbojarosite and potassium jarosite minerals are not decomposed during conventional cyanidation in gossan ores [13]. The remaining 7.3% of silver related with Fe-oxide minerals are soluble in HCl solution; and 3.5% of silver with sulphide minerals such as pyrite, galena and sphalerite are soluble in H2SO4 and HNO3 solutions [16,22].

Fig. 9 Extractions of gold (a) and silver (b) following each stage of acid/alkaline treatments in diagnostic leaching of ore
Figure 10 illustrates the extractions of iron, zinc, arsenic and lead in diagnostic leaching. The iron extraction (93%) arised from the decomposition of Fe-oxide minerals during HCl leaching (Fig. 10(a)). Iron oxide/hydroxide minerals can be dissolved in hydrochloric acid solutions by forming ferric ions [30]. 7.3% of Ag extraction was observed in cyanidation after hydrochloric acid leaching (Fig. 9(b)), which is probably the amount of silver associated with Fe-oxide minerals. The findings are confirmed with the results in Fig. 6. The remaining iron (6.6%) was dissolved at sulphuric acid leaching stage in which pyrite can be dissolved (Fig. 10(a)). Therefore, in presence of chloride ions, the chloroplumbate ion [(PbCl4)2-] is formed with the dissolution of lead sulfate (anglesite, PbSO4) [31].

Fig. 10 Extractions of iron (a), zinc (b), arsenic (c) and lead (d) following each stage of acid/alkaline treatments in diagnostic leaching of ore
The 32% lead extraction was observed in hydrochloric acid leaching probably with the dissolution of anglesite in the ore (Fig. 10(d)). The dissolution of some iron oxide minerals and anglesite (PbSO4) in hydrochloric acid solution is given as the following reactions [30,31]:
Fe2O3+6HCl��2FeC13+3H2O (6)
Fe(OH)3+3H+��Fe3++3H2O (7)
PbSO4+4Cl-��(PbCl4)2-+SO42- (8)
The oxides, hydroxides and carbonate minerals of zinc metal are easily solubilized to water-soluble compounds as sodium zincate (Na2ZnO2) in strong alkaline solutions (e.g. NaOH) [20]. It can be seen from Fig. 10(b) that 62.3% Zn was dissolved in KOH alkaline leaching. The zinc extraction is probably derived from the dissolution of the minerals such as smithsonite or hydrozincite in the ore [32]. CHEN et al [32] studied the recovery of Zn and its concomitant metals from refractory hemimorphite [Zn4(Si2O7)(OH)��H2O] zinc oxide ore by alkaline leaching in the presence of 5 mol/L sodium hydroxide, and 73% Zn can be extracted by NaOH alkaline leaching. The oxide of zinc and lead can be reacted as the following equations and produce dissoluble metals salts. Also, sodium hydroxide can dissolve PbS and ZnS minerals by forming water-soluble zincate (Na2ZnO2) and plumbate (Na2PbO2) (Fig. 10(d)). The chemical reactions are given below:
ZnO+2NaOH��Na2ZnO2+H2O (9)
ZnS+4NaOH��Na2ZnO2+Na2S+2H2O (10)
PbS+4NaOH��Na2PbO2+Na2S+2H2O (11)
Zn4Si2O7(OH)2��H2O+12NaOH��4Na2Zn(OH)4+2Na2SiO3 (12)
PbO+2NaOH+H2O��Na2Pb(OH)4 (13)
It can be seen from Figs. 10(b) and (d) that 25.6% Zn and 11% Pb were dissolved during cyanidation. Sphalerite commonly associated with gold ore reacts with cyanide and forms soluble zinc cyano complex [Zn(CN)42-] [21,30], which has a much lower stability constant than the gold cyano complex. Among different lead minerals, galena is the most common mineral associated with gold ore. The concentration of lead ions in cyanide solution is usually very low due to the presence of galena [33].
On the other hand, the beudantite, plumbojarosite or potassium jarosite solid phases remain unaltered in the conventional cyanidation process. These minerals are not decomposed by the cyanidation treatment in alkaline medium at room temperature and, consequently, the silver in these phases is inaccessible to the cyanide ions [11-13].
In alkaline decomposition of nature jarosite-type minerals such as beudantite, argentojarosite and plumbojarosite, the sulfate, sodium and potassium ions are removed, while iron, arsenic and lead remain in the solid [13]. The formation of soluble plumbite is insignificant [11]. The dissolution of arsenic in KOH alkaline medium (54.3% As) (Fig. 10(c)) probably resulted in the dissolution of arsenate minerals such as adamite or mimetite in the ore.
3.4 Cyanidation after alkaline pre-treatment
The cyanidation of as-received ore and after alkaline pretreatment using potassium hydroxide is shown in Fig. 11. While 23.4% Ag was extracted from the untreated ore, 87% Ag was dissolved in cyanidation followed alkaline leaching. Similarly, gold extraction was obtained to be improved from 76.6% to 90.2% in cyanidation by following the pretreatment (Fig. 11). The higher gold and silver extractions after alkaline pretreatment showed that high amount of silver is especially refractory in the beudantite type jarosite minerals that are apparently insoluble in cyanide solutions. The pretreatment in alkaline solution decomposed the jarosite-beudantite phases and improved the silver extraction up to 87%. The rest of silver probably remained in the residue enclosed in the Fe-oxide minerals.

Fig. 11 Effect of potassium hydroxide alkaline pretreatment
Some recent studies have already shown that alkaline leaching including sodium hydroxide or potassium hydroxide is an effective pretreatment method ahead of cyanide leaching for the extraction of gold and silver from antimonial refractory ores [5,6]. CELEP et al [34] also reported low gold and silver extractions (87% Au and 43% Ag) by cyanidation from a gossan- type ore containing various iron oxide and jarosite phases. Some studies showed that the alkaline decomposition in saturated Ca(OH)2, NaOH or alkaline sulphide solutions ensures the simultaneous removal of sulfate and Na+ or K+ from the lattice, resulting in the formation of amorphous hydroxy-arsenate of Fe, Pb and Ca or silver [11,13,15,35,36]. After alkaline decomposition, silver can be extracted from the decomposition residue by cyanide leaching [14]. The alkaline decomposition of beudantite, plumbojarosite and argentojarosite can be described by the following equations, respectively [13,36]:
PbFe3(SO4)(AsO4)(OH)6(s)+2OH-(aq)+1.5H2O��[3Fe(OH)3+Pb(OH)2+0.5As2O5]gel+SO42-(aq) (14)
PbFe6(SO4)4(OH)12+4Ca(OH)2+8H2O��6Fe(OH)3+Pb(OH)2+4CaSO4��2H2O (15)
AgFe3(SO4)2(OH)6(s)+4OH-(aq)��AgOH(s)+3Fe(OH)3(s)+2SO42-(aq) (16)
AgOH(s)+2CN-(aq)��Ag(CN)2-(aq)+OH-(aq) (17)
4 Conclusions
1) In the ore sample, gold was found to be 6-24 ��m in size and associated with limonite, goethite and lepidocrocite within calcite and quartz matrix. Silver was observed to be mainly present as acanthite (Ag2S) and electrum, and as inclusions within beudantite type jarosite phase.
2) The limited gold and silver extractions were obtained during cyanidation from the ore (d80: 50 ��m) (76% and 23%, respectively) over a leaching period of 24 h. Diagnostic leaching and detailed mineral liberation analysis (MLA) exhibited that the refractory gold and silver are mainly associated within iron oxide mineral phases such as limonite/goethite and jarosite-beudantite.
3) The potassium hydroxide alkaline pretreatment was found to improve the extractions of silver and gold (87% Ag and 90% Au) significantly. The alkaline leaching can be used as a new diagnostic approach to characterize the refractoriness of iron oxy/hydroxide (gossan type) bearing gold/silver ores and as a pretreatment method to overcome the refractoriness.
Acknowledgements
The authors would like to express their sincere thanks and appreciation to  Mining Company and Assoc. Prof.
Mining Company and Assoc. Prof.  Alp for kindly providing the ore samples, and to Assist. Prof. Mithat Vicil for the light micrographic images and Prof. Haci Deveci for proof reading of the article.
Alp for kindly providing the ore samples, and to Assist. Prof. Mithat Vicil for the light micrographic images and Prof. Haci Deveci for proof reading of the article.
References
[1] LA BROOY S R, LINGE H G, WALKER G S. Review of gold extraction from ores [J]. Minerals Engineering, 1994, 7(10): 1213-1241.
[2] DUNN J G, CHAMBERLAIN A C. The recovery of gold from refractory arsenopyrite concentrates by pyrolysis-oxidation [J]. Minerals Engineering, 1997, 10(9): 919-928.
[3] GUNYANGA F P, MAHLANGU T, ROMAN R J, MUNGOSHI J, MBEVE K. An acidic pressure oxidation pre-treatment of refractory gold concentrates from the Kwekwe roasting plant, Zimbabwe [J]. Minerals Engineering, 1999, 12(8): 863-875.
[4] IGLESIAS N, CARRANZA F. Refractory gold-bearing ore: A review of treatment methods and recent advances in biotechnological techniques [J]. Hydrometallurgy, 1994, 34(3): 383-395.
[5] CELEP O, ALP  ,
,  D, THIBAULT Y. Implementation of sodium hydroxide pretreatment for refractory antimonial gold and silver ores [J]. Hydrometallurgy, 2011, 108(1-2): 109-114.
D, THIBAULT Y. Implementation of sodium hydroxide pretreatment for refractory antimonial gold and silver ores [J]. Hydrometallurgy, 2011, 108(1-2): 109-114.
[6]  D, THIBAULT, Y. Influence of potassium hydroxide pretreatment on the extraction of gold and silver from a refractory ore [J]. Hydrometallurgy, 2014, 146: 64-71.
D, THIBAULT, Y. Influence of potassium hydroxide pretreatment on the extraction of gold and silver from a refractory ore [J]. Hydrometallurgy, 2014, 146: 64-71.
[7] CELEP O, ALP  , DEVECI H. Improved gold and silver extraction from a refractory antimony ore by pre-treatment with alkaline sulphide leach [J]. Hydrometallurgy, 2011, 105(3-4): 234-239.
, DEVECI H. Improved gold and silver extraction from a refractory antimony ore by pre-treatment with alkaline sulphide leach [J]. Hydrometallurgy, 2011, 105(3-4): 234-239.
[8] CORRANS I J, ANGOVE J E. Ultra fine milling for the recovery of refractory gold [J]. Minerals Engineering, 1991, 4(11): 763-776.
[9] CELEP O, ASLAN N, ALP  ,
,  G. Optimization of some parameters of stirred mill for ultra-fine grinding of refractory Au/Ag ores [J]. Powder Technology, 2011, 208: 121-127.
G. Optimization of some parameters of stirred mill for ultra-fine grinding of refractory Au/Ag ores [J]. Powder Technology, 2011, 208: 121-127.
[10] CELEP O, BAS A D, YAZICI E Y, ALP  , DEVECI H. Improvement of silver extraction by ultra-fine grinding prior to cyanide leaching of the plant tailings of a refractory silver ore [J]. Mineral Processing and Extractive Metallurgy Review, 2015, 36(4): 227-236.
, DEVECI H. Improvement of silver extraction by ultra-fine grinding prior to cyanide leaching of the plant tailings of a refractory silver ore [J]. Mineral Processing and Extractive Metallurgy Review, 2015, 36(4): 227-236.
[11]  C. Alkaline decomposition-cyanidation kinetics of argentian plumbojarosite [J]. Hydrometallurgy, 1994, 34: 279-291.
C. Alkaline decomposition-cyanidation kinetics of argentian plumbojarosite [J]. Hydrometallurgy, 1994, 34: 279-291.
[12]  C. Characterization and cyanidation of Rio Tinto gossan ores [J]. Canadian Metallurgical Quarterly, 1995, 34(2): 115-122.
C. Characterization and cyanidation of Rio Tinto gossan ores [J]. Canadian Metallurgical Quarterly, 1995, 34(2): 115-122.
[13] ROCA A,  J, ARRANZ M, CALERO J. Characterization and alkaline decomposition/cyanidation of beudantite-jarosite materials from Rio Tinto gossan ores [J]. Canadian Metallurgical Quarterly, 1999, 38(2): 93-103.
J, ARRANZ M, CALERO J. Characterization and alkaline decomposition/cyanidation of beudantite-jarosite materials from Rio Tinto gossan ores [J]. Canadian Metallurgical Quarterly, 1999, 38(2): 93-103.
[14] KASAINI H, KASONGO K, NAUDE N, KATABUA J. Enhanced leachability of gold and silver in cyanide media: Effect of alkaline pre-treatment of jarosite minerals [J]. Minerals Engineering, 2000, 21: 1075-1082.
[15]  L, CRUELLS M, ROCA A. Sulphidization-cyanidation of jarosite species: Applicability to the gossan ores of Rio Tinto [J]. Hydrometallurgy, 1996, 42: 35-49.
L, CRUELLS M, ROCA A. Sulphidization-cyanidation of jarosite species: Applicability to the gossan ores of Rio Tinto [J]. Hydrometallurgy, 1996, 42: 35-49.
[16] LORENZEN L. Some guidelienes to the desing of a diagnostic leaching experiment [J]. Minerals Engineering, 1995, 8(3): 247-256.
[17] LORENZEN L, TUMILTY J A. Diagnostic leaching as an analytical tool for evaluating the effect of reagents on the performance of a gold plant [J]. Minerals Engineering, 1992, 5(3): 503-512.
[18] LORENZEN L, van DEVENTER J S J. The identification of refractoriness in gold ores by the selective destruction of minerals [J]. Minerals Engineering, 1993, 5(3): 1013-1023.
[19] SABA M, MOHAMMADYOUSEF A, RASHCHI F, MOGHADDAM J. Diagnostic pre-treatment procedure for simultaneous cyanide leaching of gold and silver from a refractory gold/silver ore [J]. Minerals Engineering, 2011, 24: 1703-1709.
[20] GUPTA C K, MUKHERJEE T K. Hydrometallurgy in extraction processes, Volume I [M]. Florida, USA: CRC Press, 1990.
[21] MARSDEN J O, HOUSE C L. The chemistry of gold extraction [M]. Colorado, USA: Society for Mining Metallurgy and Exploration, 2006.
[22] CELEP O, ALP  , DEVECI H, VICIL M. Characterization of refractory behaviour of a complex gold/silver ore by diagnostic leaching [J]. Transactions of Nonferrous Metals Society of China, 2009, 19: 707-713.
, DEVECI H, VICIL M. Characterization of refractory behaviour of a complex gold/silver ore by diagnostic leaching [J]. Transactions of Nonferrous Metals Society of China, 2009, 19: 707-713.
[23] TEAGUE A J, SWAMINATHAN C, VAN DEVENTER J S J. The behaviour of gold bearing minerals during froth flotation as determined by diagnostic leaching [J]. Minerals Engineering, 1998, 11(6): 523-533.
[24] HENLEY K J, CLARKE N C, SAUTER P. Evaluation of a diagnostic leaching technique for gold in native gold and gold+silver tellurides [J]. Minerals Engineering, 2000, 14(1): 1-12.
[25] PETRUK W. Applied mineralogy in the mining industry [M]. Amsterdam, Holland: Elsevier Science BV, 2000.
[26] FANDRICH R, GU Y, BURROWS D, MOELLER K. Modern SEM-based mineral liberation analysis [J]. International Journal of Mineral Processing, 2007, 84: 310-320.
[27] GU Y. Automated scanning electron microscope based mineral liberation analysis: An introduction to JKMRC/FEI Mineral Liberation Analyser [J]. Journal of Minerals and Materials Characterisation and Engineering, 2003, 2(1): 33-41.
[28] GOODALL W R, SCALES P J. An overview of the advantages and disadvantages of the determination of gold mineralogy by automated mineralogy [J]. Minerals Engineering, 2007, 20(5): 506-517.
[29] ACARKAN N, BULUT G, 
 The effect of collector��s type on gold and silver flotation in a complex ore [J]. Separation Science and Technology, 2011, 46: 283-289.
The effect of collector��s type on gold and silver flotation in a complex ore [J]. Separation Science and Technology, 2011, 46: 283-289.
[30] HABASHI F. Principles of extractive metallurgy, Volume 2: Hydrometallurgy [M]. London, England: Gordon and Breach, Science Publishers, 1970.
[31] HABASHI F. A Textbook of hydrometallurgy [M]. Canada: M��tallurgie Extractive Qu��bec, 1999.
[32] CHEN A, ZHAO Z W, JIA X, LONG S, HUO G, CHEN X. Alkaline leaching Zn and its concomitant metals from refractory hemimorphite zinc oxide ore [J]. Hydrometallurgy, 2009, 97: 228-232.
[33] AGHAMIRIAN M. Reactivity of sulfide minerals and its effect on gold dissolution and its electrochemical behaviour in cyanide solution [D]. Ontario, Canada: Queen's University, 1997: 228.
[34] CELEP O, ALP  SERBEST V,
SERBEST V,  E. A preliminary research on gold and silver recovery from
E. A preliminary research on gold and silver recovery from 
 ore [C]//Proceedings of The XIII International Mineral Processing Symposium. Bodrum, Turkey, 2012: 605-612.
ore [C]//Proceedings of The XIII International Mineral Processing Symposium. Bodrum, Turkey, 2012: 605-612.
[35]  F, SALINAS E, CRUELLS M, ROCA A. Alkaline decomposition�Ccyanidation kinetics of argentian natrojarosite [J]. Hydrometallurgy, 1998, 49: 323-336.
F, SALINAS E, CRUELLS M, ROCA A. Alkaline decomposition�Ccyanidation kinetics of argentian natrojarosite [J]. Hydrometallurgy, 1998, 49: 323-336.
[36] ROCA A,  C. Alkaline decomposition-cyanidation kinetics of argentojarosite [J]. Hydrometallurgy, 1993, 33: 341-358.
C. Alkaline decomposition-cyanidation kinetics of argentojarosite [J]. Hydrometallurgy, 1993, 33: 341-358.
Oktay CELEP, Vedat SERBEST
Hydromet B&PM Group, Division of Mineral & Coal Processing, Department of Mining Engineering,
Karadeniz Technical University, Trabzon 61080, Turkey
ժ Ҫ��Ϊ��̽�����軯����֮ǰ����Ԥ�������գ���������Ϸ����Ժ���������/��������(��ñ��)�Ѵ�����/���������ϸ���������������������Ȼ��������(�ߴ�Ϊ6~24 ��m)���ڣ������ڷ���ʯ��ʯӢ�����еĺ�������������������С��������ȷ���(MLA)�����������������������ڣ�����������Ǧ����������/�������ʯӢ�С�����Ҫ��������(Ag2S)���������ڣ������ڿ�ʯ������Ǧ����軯����ʵ��������ڽ���ʱ��ﵽ24 h��ʯ(d80��50 ��m)�н�����Ľ����ʷֱ�ֻ��76%��23%����ʯ����Ͻ���ʵ�����ϸ�Ŀ�������������������Ҫ�������������������У��������/������ͻƼ������͵�����Ǧ������Щ�������������ڼ�����Һ�з��������������Щ�о������軯����֮ǰ����ʯ��KOH��Һ�н��м���Ԥ�������ܽ����ͽ�Ľ����ʷֱ���ߵ�87%��90%����Щ���ֱ������Խ�������Ϊһ���µ���Ϸ�������������������/���������Ľ�/�������Ѷȣ����ҿ���Ϊһ��Ԥ�������������������ʡ�
�ؼ��ʣ��Ѵ���������������軯�������������ȷ���(MLA)������Ԥ����
(Edited by Mu-lan QIN)
Foundation item: Project (8300) supported by the Research Foundation of Karadeniz Technical University, Turkey
Corresponding author: Oktay CELEP; Fax: +90-462-3257405; E-mail: ocelep@ktu.edu.tr
DOI: 10.1016/S1003-6326(15)63725-6

