J. Cent. South Univ. (2017) 24: 1968-1976
DOI: https://doi.org/10.1007/s11771-017-3605-7

Spatiotemporal distribution model for zinc electrowinning process and its parameter estimation
DENG Shi-jun(���˾�), YANG Chun-hua(������), LI Yong-gang(���¸�),
ZHU Hong-qiu(�����), WU Tie-bin(������)
School of Information Science and Engineering, Central South University, Changsha 410083, China
 Central South University Press and Springer-Verlag GmbH Germany 2017
Central South University Press and Springer-Verlag GmbH Germany 2017
Abstract: This paper focuses on the distributed parameter modeling of the zinc electrowinning process (ZEWP) to reveal the spatiotemporal distribution of concentration of zinc ions (CZI) and sulfuric acid (CSA) in the electrolyte. Considering the inverse diffusion of such ions in the electrolyte, the dynamic distribution of ions is described by the axial dispersion model. A parameter estimation strategy based on orthogonal approximation has been proposed to estimate the unknown parameters in the process model. Different industrial data sets are used to test the effectiveness of the spatiotemporal distribution model and the proposed parameter estimation approach. The results demonstrate that the analytical model can effectively capture the trends of the electrolysis reaction in time and thus has the potential to implement further optimization and control in the ZEWP.
Key words: zinc electrowinning; spatiotemporal distribution model; parameter estimation; orthogonal approximation
1 Introduction
In the metallurgical industry, the zinc electrowinning process (ZEWP) is a high-power-consuming process that accounts for 80% of the total electrical energy consumption of the hydrometallurgy process [1]. The recovery of zinc by electrolysis is a complicated reaction accomplished by the application of direct current through insoluble electrodes, causing decomposition of aqueous zinc sulfate electrolyte and deposition of metallic zinc onto the cathodes [2]. To guarantee the zinc deposition and achieve the desirable product indicators, high- performance control of the concentration of zinc ions (CZI) and the concentration of sulfuric acid (CSA) is essential. However, in practical production, the CZI and CSA can only be measured off-line. A large delay of measurement leads to difficulty in control, i.e., the concentrations cannot be controlled within the required ranges in an appropriate time. In this case, establishing an accurate process model is exceedingly important for implementing further optimization and control in the ZEWP [3].
Currently, many articles have reported on studies of the modeling methods for ZEWP to simulate the current efficiency or energy consumption. BARTON and SCOTT [4] built a dynamic, nonlinear model of an industrial pilot-plant scale zinc electrolysis cell that is based on fundamental thermodynamic and electrochemical relations and can be used to predict the current efficiency and the cell potential based on the configuration of the cell, the composition and temperature of the electrolyte, and the cathode current density. In a previous research [5], a simulation was proposed that improved the abilities of a conventional mass and energy balance approach by including predictions of the voltage, current efficiency and reaction kinetics. Sophisticated empirical relationships between the reaction conditions and the current efficiency have also been suggested. WANG et al [6] developed an empirical relationship that calculates an estimate of the current efficiency based on incremental fuzzy neural network in zinc electrolyte cell. BEHNAJADY et al [7] described a new approach for determination of effective parameters and optimization of zinc electrolyte hot purification process using statistical design of experiments. YANG et al [8] developed an optimal power-dispatching system depending on the varying prices of electricity to minimize the cost of power consumption in the electrochemical process of zinc. BARTON and SCOTT [9] built a mechanism model that reflects the conductivity of the electrolyte and the CZI and CSA. The good results of these prior studies inspired our investigation.
However, significant spatiotemporal distribution characteristics are found in the ZEWP. More precisely, the CZI and CSA are not distributed uniformly in electrolyte due to the special structure of electrolysis cell and the mass transfer between the plates. The modeling methods mentioned above cannot describe the time-space distribution of the electrolyte concentrations. Therefore, the spatiotemporal model should be established to provide accurate predicted values of the CZI and the CSA and promote optimization and control of the ZEWP.
2 Description of zinc electrowinning process
The flow sheet of a typical electrowinning process with an annual production capacity of 0.1 million tons is shown in Fig. 1.
Before electrowinning, the leaching solutions were preserved in the respective leaching solution tanks to allow the impurities to be deposited at bottoms of the tanks. Next, the leaching solution is mixed with the spent acid solution in the mixing cell. The mixture, also called the electrolyte, is passed to the electrolysis cell, where the zinc ions are deposited onto the cathode plate by the application of direct current. The electrolyte after the electrodeposition process is the spent acid solution, with lower CZI and higher CSA than the electrolyte. Most of the spent acid is pumped back to re-mix with the leaching solution, with the remainder preserved in the spent acid tanks. A series of cooling towers is also built to moderate the temperature of the electrolyte. The spent acid solution can be pumped through the cooling tower when the temperature of electrolyte is too high or into the mixing cell directly when the temperature is suitable.
Practically, the mass transfer and reaction kinetics of the electrolyte are more complicated than those of the leaching solution and the spent acid solution. First, the electrolyte is the only solution in which the electrochemical reaction occurs during the entire process of the ZEWP; second, as the electrolyte flows along the direction perpendicular to the plates, the CZI and CSA in the electrolyte gradually change along the flow direction and are homogeneous in the direction parallel to the plate. More importantly, the blockage of the plates will cause the inverse diffusion of Zn2+ and H2SO4. Therefore, the spatiotemporal dynamics of the electrolyte is of the highest concern during the process.
3 Framework of spatiotemporal model
The framework of the spatiotemporal model for the electrolyte is presented in Fig. 2. The framework is mainly composed of an axial dispersion model, a reaction rate model and a parameter estimation strategy. A detailed description for each part will be provided in the following sections.
3.1 Reaction rate model
In the ZEWP, the main chemical reaction is the zinc ion deposition onto metallic zinc under direct current, accompanied by the electrolysis of water; the reaction is given as follows [10]:
At the cathode:

 (1)
(1)
At the anode:
 (2)
(2)

Fig. 1 Flow sheet of zinc electrowinning process

Fig. 2 Framework of spatiotemporal distribution model of ZEWP
Based on the electrode kinetics, the deposition rate is proportional to the direct current. Accordingly, the following equation can be derived [11].

 (3)
(3)
where rZn is the deposition rate of zinc, rH.1 is the consumption rate of hydrogen ions at the cathode, rH.2 is the generation rate of hydrogen ions at the anode, S is the plate area, F is the Faraday constant and D is the current density. Based on the prior research, the current efficiency can be calculated using the following equation [12].
��= (4)
(4)
where the D, T, F, R, ZZn, and ZH are known parameters or can be measured; and 
 is the vector of kinetic parameters. A detailed description of the parameters in the kinetic model is given in Table 1 [13, 14].
is the vector of kinetic parameters. A detailed description of the parameters in the kinetic model is given in Table 1 [13, 14].
3.2 Axial dispersion model for electrolyte
As for the electrolyte, we focus on the mass transfer and reaction process shown in Fig. 3. Due to the blockage of plates, the electrolyte solution is not well mixed. More accurately, the concentrations of zinc ion and sulfate acid in electrolyte solution change gradually in horizontal axis direction. Therefore, we suppose that the electrolyte is well mixed along the longitudinal axis and that reverse diffusion of ions proceeds along the axis direction. The material balance analysis is conducted in a selected differential element dl of the axial direction within a unit dt.
Table 1 Parameters in the kinetic model

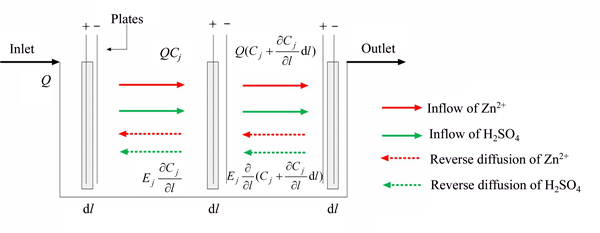
Fig. 3 Reaction unit in electrolysis cell
The material balance analysis is conducted in a selected differential element dl of the axial direction which is parallel to the flow of electrolyte within a unit dt. The left side of the equation represents the material quality flowing into the differential element. The right side of the equation consists of the material quality discharged from the differential element, the accumulating quality and the reaction consuming quality [15]. After the calculation of material, the two-dimension partial differential equations could be described as Eq. (6).
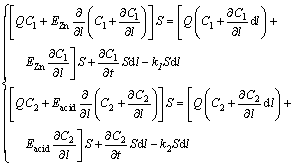 (6)
(6)
where Q is the flow rate of electrolyte, EZn and Eacid are the unknown axial diffusion coefficients of Zn2+ and H2SO4, respectively, [16] and C1 and C2 are the CZI and CSA of electrolyte, respectively. l is the axial position of the electrolysis cell. The inlet of the electrolysis cell is at l=0; the end of electrolysis cell is at l=L, (L=5.9 m is the length of the electrolysis cell). k1 and k2 are the reaction rates of zinc and sulfuric acid, respectively, which can be calculated as follows:
 (7)
(7)
Simplifying Eq. (6), the mass balance model can be described as follows:
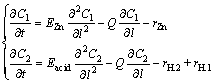 (8)
(8)
We can see from the partial differential Eq. (8) that the CZI and the CSA in the electrolysis cell are not only functions of time t but also functions of position l. This partial differential equation can be solved using a difference algorithm, the initial condition and boundary condition of which are given as follows [17]:
 (9)
(9)
4 Formulation and result of parameter estimation
The identification of such a spatiotemporal distribution system has become a paramount problem in modern control engineering and many other areas. Many researchers have studied the problem by using the Walsh function, block-pulse functions (BPFs), the Legendre function [18-21]. Among these functions, BPFs have been widely used in the identification of industrial process due to its reduced amount of calculations and minimal amount of pre-treatment [22].
4.1 Function approximated by two-dimension BPF
A set of two-dimension BPFs is defined as [23]

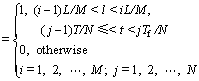 (10)
(10)
where Hi(l) and Hj(t) are the basis functions of the BPFs at the ith and jth sub-regions of l and t, respectively. L and Tf are the maximum values of l and t, respectively.
Therefore, the function u(l, t), which is absolutely integrable in the region �� can be approximated as



 (11)
(11)
where �� denotes transpose, U=(ui,j) is the coefficient matrix of u(l,t), and
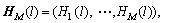
 (12)
(12)
The coefficients ui,j are the integral mean values of u(l,t) over the sub-regions:
��i,j
 (13)
(13)
The ui,j can be calculated by Eq. (14).
 (14)
(14)
Obviously, HM(l) and HN(t) have the following properties, which are useful for the next integrating step:
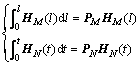 (15)
(15)
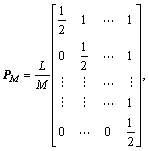
 (16)
(16)
4.2 Parameter identification based on orthogonal function for spatiotemporal distribution model
Given system (17) with initial condition and boundary conditions (18),
 (17)
(17)
 (18)
(18)
Using properties (15) and integrating Eq. (17) with respect to t from 0 to t and twice integrating with respect to l from 0 to l and then from L to l, we obtain the following:


 (19)
(19)
Transform Eq. (18) with Eq. (11) to obtain
 (20)
(20)
where  and
and  are the coefficients matrices of ��(l), g1(t), g2(t) and g3(t), respectively. Upon substituting Eq. (20) into Eq. (19) and integrating Eq.(17), we have
are the coefficients matrices of ��(l), g1(t), g2(t) and g3(t), respectively. Upon substituting Eq. (20) into Eq. (19) and integrating Eq.(17), we have
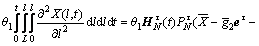
 (21)
(21)
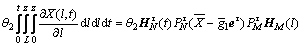 (22)
(22)
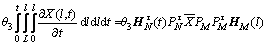 (23)
(23)
 (24)
(24)
where  Let
Let
 (25)
(25)
We change Eq. (19) to
 (26)
(26)
where Vec denotes the associated vector. Let
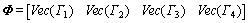 (27)
(27)
Then, Eq. (26) can be formulated as Eq. (28)
 (28)
(28)
Therefore, �� can be estimated as follows:
 (29)
(29)
4.3 Results of parameter identification
Based on the data obtained from a real-life ZEP of a zinc production factory in China, the parameters identification method is used to determine the parameters of Eq. (19). By comparing the distributed model of an electrolysis cell described by Eq. (19), we notice that
 (30)
(30)
where Q can be regarded as a constant during 0f. Therefore, the parameters that require estimation are EZn and Eacid. According to Ref. [24], when M=5 and N=4, a satisfying result can be achieved. The sample positions are shown in Fig. 4, and Tf=2 h for each group; the identification results are shown in Table 2.
The diffusion coefficient for Zn2+ was found to be 4.6��10-6 cm2/s by PAN et al [25], while YU et al [26] reported a value of 4.6��10-6 cm2/s. SUNG and LEE [27] reported the diffusion coefficient for sulfuric acid a value of (2.78-5.02)��10-6 cm2/s. These are similar with the results estimated by the proposed method.
5 Industrial application
Supported by these successive experiments, the proposed system model and parameter estimation strategy are applied to an actual zinc electrowinning process at the largest lead-zinc smelting factory in China. The prediction system based on the proposed model was developed using C language on the Wincc platform. SQL 2007 technology stored the original industrial data sampled by the DCS system. In the prediction system, the databases include two parts: the online measurement base and the offline measurement base [28]. The former base consists of the inlet flow rate of the electrolyte, the temperature of the electrolyte, the current density and the volume of the electrolyte, which are sampled by PLC once every 5 s. The latter off-line base consists of the concentration of zinc ions and sulfuric acid at each position of the electrolyte, which are measured and inputted once every half hour. The spatiotemporal distribution of the CZI and CSA concentrations of the electrolyte under different current densities are shown in Figs. 5 and 6. Along with the axial position of electrolysis cell, CZI clearly decreases, and CSA clearly increases gradually. By comparing Fig. 5 with Fig. 6, it is observed that the higher the current density is, the faster the CZI decreases and the faster the CSA increases.
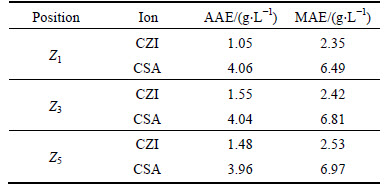
Another 50 sets of samples were selected to test the model performance, and the test results are given in Fig. 7 to Fig. 9. To accurately assess the running results of the predictive strategies, two indices are introduced in this work (as shown in Table 3): maximum absolute error (MAE) and average absolute error (AAE). The results show that by applying the axial dispersion model of the electrolysis cell, the proposed model tracks the trend of the concentrations of ions and also show the spatial and temporal distribution of the concentrations of ions in the electrolysis cell.

Fig. 4 Sampling positions
Table 2 Parameters estimation and their 95% confidence interval

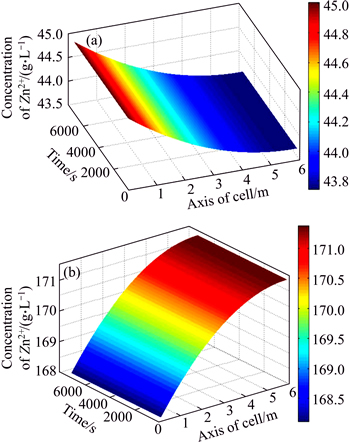
Fig. 5 Spatiotemporal distributions of CZI (a) and CSA (b) of electrolyte at low current density (D=260-300 A/m2)
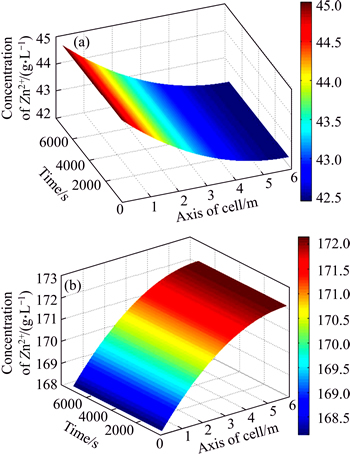
Fig. 6 Spatiotemporal distribution of CZI (a) and CSA (b) of electrolyte at high current density (D=400-450 A/m2)

Fig. 7 Prediction of CZI (a) and CSA (b) at Z1

Fig. 8 Prediction of CZI (a) and CSA (b)at Z3

Fig. 9 Prediction of CZI (a) and CSA (b) at Z5
Table 3 Performance of spatiotemporal distribution model
6 Conclusions
A spatiotemporal and parameter estimation model for the ZEWP based on orthogonal approximation was proposed. The system is described by a kinetic model combined with an axial dispersion model. To improve the model precision when applied to an actual process, a method to identify the unknown parameters in the spatiotemporal model using two dimension block-pulse functions was presented. The running results showed that the proposed model functions stably and reliably.
References
[1] ZHANG Bin, YANG Chun-hua, ZHU Hong-qiu, LI Yong-gang, GUI Wei-hua. Kinetic modeling and parameter estimation for competing reactions in copper removal process from zinc sulfate solution [J]. Industrial & Engineering Chemistry Research, 2013, 52(48): 17074-17086.
[2] SUN Bei, GUI Wei-hua, WU Tie-bin, WANG Ya-lin, YANG Chun-hua. An integrated prediction model of cobalt ion concentration based on oxidation�Creduction potential [J]. Hydrometallurgy, 2013, 140: 102-110.
[3] HERRERO D, ARIAS P L,  B, BARRIO V L, CAMBRA J F, REQUIES J. Hydrometallurgical process development for the production of a zinc sulphate liquor suitable for electrowinning [J]. Mineral Engineering, 2010, 23(6): 511-517.
B, BARRIO V L, CAMBRA J F, REQUIES J. Hydrometallurgical process development for the production of a zinc sulphate liquor suitable for electrowinning [J]. Mineral Engineering, 2010, 23(6): 511-517.
[4] BARTON G W, SCOTT A C. Scale-up effects in modelling a full-size zinc electrowinning cell [J]. Journal of Applied Electrochemistry, 1992, 22(8): 687-692.
[5] MAHON M, PENG S, ALFANTAZI A. Application and optimization studies of a zinc electrowinning process simulation [J]. Canadian Journal of Chemical Engineering, 2014, 92: 633-642.
[6] WANG Ya-lin, Gui Wei-hua, YANG Chun-hua, HUANG Tai-song. Intelligent modeling and optimization on time-sharing power dispatching system for electrolytic zinc process [J]. Transactions of Nonferrous Metals Society of China, 2000, 10(4): 561-565.
[7] BAHRAM B, JAVAD M. Statistical evaluation and optimization of zinc electrolyte hot purification process by Taguchi method [J]. Journal of Central South University, 2015, 22(6): 2066-2072.
[8] YANG Chun-hua, DECONINCK G, GUI Wei-hua, LI Yong-gang. An optimal power-dispatching system using neural networks for the electrochemical process of zinc depending on varying prices of electricity [J]. IEEE Transactions on Neural Network, 2002, 13(1): 229-236.
[9] BARTON G W, SCOTT A C. Industrial applications of a mathematical model for the zinc electrowinning process [J]. Journal of Applied Electrochemistry, 1994, 24(5): 377-383.
[10] MAHON M, WASIK L, ALFANTAZI A. Development and implementation of a zinc electrowinning process simulation [J]. Journal of Applied Electrochemistry, 2012, 159(8): D486-D492.
[11] ZHANG Qi-bo, HUA Yi-xin, DONG Tie-guang, ZHOU Dan-gui. Effects of temperature and current density on zinc electrodeposition from acidic sulfate electrolyte with H2SO4 as additive [J]. Journal of Applied Electrochemistry, 2009, 39(8): 1207-1216.
[12] GUI Wei-hua, YANG Chun-hua, CHEN Xiao-fang, WANG Ya-lin. Modeling and optimization problems and challenges arising in nonferrous metallurgical processes [J]. Acta Automatic Sinica, 2013, 39(3): 197-207.
[13] DENG Shi-jun, YANG Chun-hua, LI Yong-gang, ZHU Hong-qiu. On-line prediction model for concentrations of zinc ion and sulfuric acid in zinc electrowinning process [J]. Chinese Journal of Chemical Engineering, 2015, 66(7): 2588-2594.
[14] GUI Wei-hua, ZHANG Mei-ju, YANG Chun-hua, LI Yong-gang. Energy consumption optimization of zinc electrolysis process based on hybrid particle swarm algorithm [J]. Control Engineering of China, 2009, 16(6): 748-751.
[15] CHARTON S, DUHAMET J, BORDA G, ODE D. Axial dispersion in pulsed disk and doughnut columns: A unified law [J]. Chemical Engineering Science, 2012, 75(0): 468-477.
[16] LI Han-xiong, QI Chen-kun. Modeling of distributed parameter systems for applications-A synthesized review from time-space separation [J]. Journal of ProcessControl, 2010, 20(8): 891-901.
[17] BONIS I, XIE Wei-guo, THEODOROPOULOS C. Multiple model predictive control of dissipative pde systems [J]. IEEE Transactions on Control Systems Technology, 2014, 22(3): 1206-1214.
[18] XIANG Xiu-qiao, ZHOU Jiang-zhong, LI Mo, LUO Zhi-meng, LI Chao-shun. Improved algorithm about NSFOT [J]. Applied Mathematics & Computation, 2009, 215(3): 881-888.
[19] STANKOVIC A M, LEV-ARI H, PERISIC M M. Analysis and implementation of model-based linear estimation of dynamic phasors [J]. IEEE Transactions on Power System, 2004, 19(4): 1903-1910.
[20] ABBASBANDY S, KAZEM S, ALHUTHALI M S, ALSULAMI H H. Application of the operational matrix of fractional-order Legendre functions for solving the time-fractional convection�Cdiffusion equation [J]. Applied Mathematics & Computation, 2015, 266: 31-40.
[21] LEPIK U. Solving fractional integral equations by the Haar wavelet method [J]. Applied Mathematics & Computation, 2009, 214(2): 468-478.
[22] LI Yuan-lu, SUN Ning. Numerical solution of fractional differential equations using the generalized block pulse operational matrix [J]. Computers & Mathematics with Applications, 2011, 62(3): 1046-1054.
[23] MALEKNEJAD L, SOHRABI S, BARAJI B. Application of 2D-BPFs to nonlinear integral equations [J]. Communications in Nonlinear Science & Numerical Simulation, 2010, 15(3): 527-535.
[24] YU Shou-yi, CAO Yue-bin, ZHOU Xuan. Parameter identification for vertical quench furnace control system based on orthogonal function [J]. Control Engineering of China, 2009, 16(3): 251-253.
[25] PAN Jun-li, WEN Yue-hua, CHENG Jie, PAN Jun-qing, BAI Zhang-li, YANG Yu-sheng. Zinc deposition and dissolution in sulfuric acid onto a graphite-resin composite electrode as the negative electrode reactions in acidic zinc-based redox flow batteris [J]. Journal of Applied Electrochemistry, 2013, 43: 541-551.
[26] YU Jiang-xian, YANG Han-xi, AI Xin-ping, CHEN Yong-yan. Effect of anions on the zinc electrodeposition onto glassy-carbon electrode [J]. Russian Journal of Electrochemistry, 2002, 38(3): 321-325.
[27] SUNG BAE KIM, LEE Y. Diffusion of sulfuric acid within lignocellulosic biomass particles and its impact on dilute-acid pretreatment [J]. Bioresource Technology, 2002, 83(2): 165-171.
[28] LI Yong-gang, GUI Wei-hua, TEO KOK LAY, ZHU Hong-qiu, CHAI Qin-qin. Optimal control for zinc solution purification based on interacting CSTR models [J]. Journal of Process Control, 2012, 22(10): 1878-1889.
(Edited by HE Yun-bin)
Cite this article as: DENG Shi-jun, YANG Chun-hua, LI Yong-gang, ZHU Hong-qiu, WU Tie-bin. Spatiotemporal distribution model for zinc electrowinning process and its parameter estimation [J]. Journal of Central South University, 2017, 24(9): 1968�C1976. DOI:https://doi.org/10.1007/s11771-017-3605-7.
Foundation item: Project(61673400) supported by the National Natural Science Foundation of China; Project(2015cx007) supported by the Innovation-driven Plan in Central South University, China; Project(61321003) supported by the Foundation for Innovative Research Groups of the National Natural Science Foundation of China; Projects(61590921, 61590923) supported by the Major Program of the National Natural Science Foundation of China
Received date: 2016-03-10; Accepted date: 2016-07-14
Corresponding author: LI Yong-gang, PhD, Professor; Tel: +86-15973120578; E-mail: liyonggang@csu.edu.cn