
A thermodynamic approach to assess glass-forming ability of bulk metallic glasses
JI Xiu-lin (纪秀林)1, PAN Ye(潘 冶)2
1. Engineering Research Center of Dredging Technology of Ministry of Education, Hohai University,
Changzhou 213022, China;
2. School of Materials Science and Engineering, Southeast University, Nanjing 211189, China
Received 28 September 2008; accepted 6 March 2009
Abstract: According to the Gibbs free energy difference between liquid and crystal, a thermodynamic glass-forming ability(GFA) parameter related to characteristic temperatures, onset crystallization temperature(Tx) and liquidus temperature(Tl), was proposed for evaluating the GFA of bulk metallic glasses(BMGs). The new parameter defined as ω=Tl(Tl+Tx)/(Tx(Tl-Tx)) has good correlation with the critical section thickness(Zc) of Ca-Mg-Cu BMGs. Being verified by the glasses data, including oxide glasses, which were used to validate the former GFA parameters, ω is one of the most reliable and applicable GFA parameters among Trg (=Tg/Tl), γ(=Tx/(Tl+Tg)), α (=Tx/Tl), δ (=Tx/(Tl-Tg), and so on. Finally, predicting GFA of Cu-Ag-Zr-Ti and Cu-Zr-Ti-Al BMGs using ω was compared with the experimental results.
Key words: glass-forming ability; bulk metallic glass; Gibbs free energy
1 Introduction
Whether the vitrification of an alloy is easy or difficult, glass-forming ability(GFA) is vital to develop new bulk metallic glasses(BMGs). Scientific efforts for quantification of GFA have been started when the first Au-Si metallic glass was reported[1]. TURNBULL[2] identified reduced glass transition temperature(Trg), which is the ratio of glass transition temperature(Tg) to liquidus temperature(Tl), as a GFA gauge. INOUE et al [3] proposed the supercooled liquid region ?Tx (=Tx-Tg, where Tx is the onset crystallization temperature of the glass) to measure GFA. Since 2002, LU and LIU[4-6] have published a serial of papers to show the strongest correlation of the GFA parameter γ (=Tx/(Tl+Tg)) with the GFA of various glass formers among the parameters suggested so far. Therefore, γ has been frequently used to predict the GFA of alloys today.
However, the predominant situation of γ is challenged in recent two years by other GFA indicators, α(=Tx/Tl)[7], δ(=Tx/(Tl-Tg)[8],  (=Trg(?Tx/Tg)0.143)[9], Trx(=Tx/Ts, where Ts is onset temperature of solidification)[10], and so on. Origins of those GFA parameters mentioned above obtained from time―temperature―transformation(TTT) diagram analysis[5, 7-10] or kinetics analysis, such as viscosity[11], fragility[9] and homogenous nucleation and growth[4, 8]. Lately, LU et al[1] restated that γ was still the best GFA indicator in terms of reliability and applicability.
(=Trg(?Tx/Tg)0.143)[9], Trx(=Tx/Ts, where Ts is onset temperature of solidification)[10], and so on. Origins of those GFA parameters mentioned above obtained from time―temperature―transformation(TTT) diagram analysis[5, 7-10] or kinetics analysis, such as viscosity[11], fragility[9] and homogenous nucleation and growth[4, 8]. Lately, LU et al[1] restated that γ was still the best GFA indicator in terms of reliability and applicability.
In this work, based on thermodynamic analysis, mainly Gibbs free energy difference between liquid and crystal, a new GFA parameter was proposed and the new glass criterion was validated with lots of experimental data to assess the GFA of BMGs.
2 Origins of new parameter
Gibbs free energy difference(?Gl-s) between liquid and crystal means the driving force of crystallization. In a supercooled alloying liquid, the less ?Gl-s means the more stable and the better GFA of the liquid. Thus, ?Gl-s plays an important role in appraising the GFA of BMGs and the GFA parameter maybe has a solid interrelationship with ?Gl-s. In this way, a new GFA indicator was developed by this approach.
Scientific efforts for ?Gl-s estimation of the super- cooled alloying liquid have started for a long time and many approximate expressions of ?Gl-s have been derived [12-14]. The expression developed by THOMPSON and SPAEPEN[12] is one of the most important and reliable:
 (1)
(1)
where Tm is the melting temperature; ?Hm is the enthalpy of fusion; and T is the temperature of the supercooled liquid.
As discussed above, GFA of metallic glasses is associated with ?Gl-s. So, their relationship can be expressed as follows:
GFA∝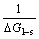 ∝
∝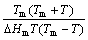 (2)
(2)
At slower cooling rates, the glass freezes at lower temperature. But the glass produced at higher cooling rates undergoes structural relaxation readily at lower temperature[15]. So, Tg detected from DSC (differential scanning calorimetry) is different from the actual freezing temperature of an metallic glass. But the expression of ?Gl-s is derived from continuous cooling process. Therefore, it is not surprising that the GFA parameter directly using Eq.(2) has no good correlation with GFA.
For glass transition of an alloying system, two equilibrium states, amorphous and liquid states, should be emphasized. From the viewpoint of heating process, the glass is still in amorphous state before system temperature exceeds Tx and the alloying system is not in a real liquid state before it reaches Tl. That is to say, Tx and Tl are thermal stability gauges of the glass and the liquid, respectively[7]. Thus, Tl but Tm characterizes the temperature of a liquid state and Tx but Tg characterizes the temperature of an amorphous state. Therefore, T and Tm in Eq.(2) are substituted by Tx and Tl, respectively, meaning that GFA is in inverse proportion to the Gibbs free energy difference of the alloying system from Tl to Tx. So, the following expression can be obtained from Eq.(2):
GFA∝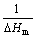 ?
?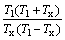 (3)
(3)
If there is no inner relationship between ?Hm and Tl (or Tx), the above expression can be simplified as
GFA∝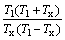 (4)
(4)
Hence, the new parameter ω for inferring the relative GFA among BMGs is defined as
 (5)
(5)
To reveal the validity of ω as a GFA parameter, Eq.(5) is deduced as:

(6)
Compared with Tl, the difference between Tx and Tg is very little, usually less than 5% Tl for the characteristic temperatures of BMGs. Therefore, the GFA parameters, Trg=Tg/Tl[2], γ=Tx/(Tg+Tl)[4-6] and α=Tx/Tl[7] are close, respectively, to Tx/Tl, Tx/(Tx+Tl) and Tx/Tl. All the GFA parameters, Trg, γ and α can be treated as functions of Tx/Tl. On the other hand, Eq.(6) indicates that ω is also the function of Tx/Tl. And the positive correlation between ω and α is illustrated in Fig.1. So, ω not only has the accordant trends of GFA description with α, but also expands the difference of values between descriptive parameters. And it may be more effective than other GFA parameters.

Fig.1 Function relationship between GFA parameters ω and α
3 Results and discussion
3.1 Correlation between new parameters and GFA
The most direct GFA indicator is the critical cooling rate(Rc) and the critical section thickness(Zc). Unfortunately, it is very difficult to measure Rc experimentally and Zc is rough. Table 1 shows the glass transition temperature(Tg), onset crystallization temperature(Tx), liquidus temperature(Tl) and Zc of Ca-Mg-Cu BMGs. The data in Table 1 were obtained from Ref.[16]. And the calculated GFA indicators (γ and ω) are also listed in Table 1. The relationships between ω, γ and Zc are shown in Fig.2. In order to reveal how closely the GFA indicators ( γ and ω) correspond to the actual experimental data of Zc, the statistical correlation parameter, R2, was also computed. The higher the R2, the more reliable the GFA parameter. As shown in Fig.2, R2 of ω is 0.70, but the corresponding R2 of γ is 0.64, indicating the new GFA parameter ω is more reliable than γ for Ca-Mg-Cu BMGs.
Table 1 Tg, Tx, Tl, Tm, Zc, ?Hm and calculated γ and ω for Ca-Mg-Cu BMGs

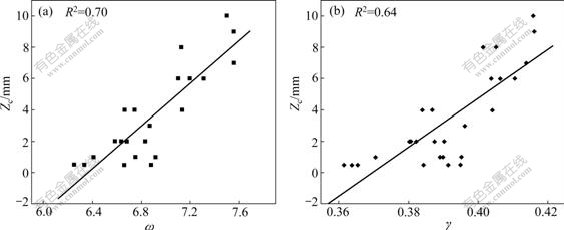
Fig.2 Correlation between Zc and ω (a), γ (b) for Ca-Mg-Cu BMGs
3.2 Comparison of ω and other GFA parameters
To verify the reliability of the new GFA parameter and to compare it with others, more BMGs data with their characteristic temperatures (Tg, Tx and Tl) and Zc, together with more calculated GFA parameters are listed in Table 2. The data in Table 2 were obtained from Table 1 in Ref.[4] and Table 1 in Ref.[8], which are used to interpret γ and α. The relationships between GFA indicators (Trg, γ, α, δ,  and ω) and the actual experimental data of Zc, are shown in Fig.3. By comparing R2 of every GFA indicator shown in Figs.3(a)-(f), R2 of ω is obviously higher than others, and the order of GFA indicators from the best to the worst is ω, δ, α,
and ω) and the actual experimental data of Zc, are shown in Fig.3. By comparing R2 of every GFA indicator shown in Figs.3(a)-(f), R2 of ω is obviously higher than others, and the order of GFA indicators from the best to the worst is ω, δ, α, 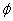 , γ and Trg. This result suggests the ω is the best GFA parameter. The relationship between the GFA and ω for these BMGs can be expressed as
, γ and Trg. This result suggests the ω is the best GFA parameter. The relationship between the GFA and ω for these BMGs can be expressed as
Zc=-77.98+11.93ω (7)
Table 2 Tg, Tx, Tl, Zc and calculated Trg, γ, α, δ,  and ω for reported BMGs
and ω for reported BMGs

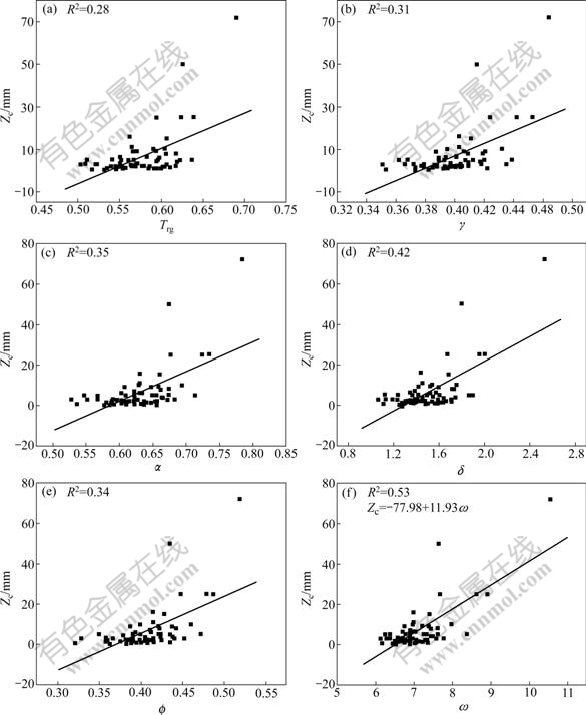
Fig.3 Correlations between Zc and Trg (a), γ (b), α (c), δ (d),  (e) and ω (f) for BMGs
(e) and ω (f) for BMGs
Besides Trg, γ, α, δ and  , other GFA parameters, such as ?Trg (=(Tx-Tg)/(Tl-Tg))[17], ?Tx, Trgx (=(TgTx)/ (TlTm))[18] and Kgl (=(Tx-Tg)/(Tm-Tx))[19] are also compared with ω. R2 of ?Trg and ?Tx calculated with data in Table 2 are 0.27 and 0.13, respectively, which are clearly lower than 0.53 of ω. From the viewpoint of emphasis on the two states, the relationships between GFA and those GFA parameters which import other characteristic temperatures beyond the amorphous and the liquid state will be injured. For example, Tg/Tl is a better representation of GFA than Tg/Tm[20]. R2 of Trgx and Kgl calculated with data in Table 1 are 0.53 and 0.42, respectively, which are obviously lower than 0.64 of γ and 0.70 of ω. On the other hand, except α and ω, all other GFA parameters mentioned above need the knowledge of Tg or Ts. However, Tg is not usually available for some glasses[7] and Ts needs to be measured during cooling of liquid, which increases the operational complexity. Based on above comparison, it can be concluded that ω is one of the most reliable and applicable GFA parameters.
, other GFA parameters, such as ?Trg (=(Tx-Tg)/(Tl-Tg))[17], ?Tx, Trgx (=(TgTx)/ (TlTm))[18] and Kgl (=(Tx-Tg)/(Tm-Tx))[19] are also compared with ω. R2 of ?Trg and ?Tx calculated with data in Table 2 are 0.27 and 0.13, respectively, which are clearly lower than 0.53 of ω. From the viewpoint of emphasis on the two states, the relationships between GFA and those GFA parameters which import other characteristic temperatures beyond the amorphous and the liquid state will be injured. For example, Tg/Tl is a better representation of GFA than Tg/Tm[20]. R2 of Trgx and Kgl calculated with data in Table 1 are 0.53 and 0.42, respectively, which are obviously lower than 0.64 of γ and 0.70 of ω. On the other hand, except α and ω, all other GFA parameters mentioned above need the knowledge of Tg or Ts. However, Tg is not usually available for some glasses[7] and Ts needs to be measured during cooling of liquid, which increases the operational complexity. Based on above comparison, it can be concluded that ω is one of the most reliable and applicable GFA parameters.
3.3 Comparison of ω in glasses including oxides
To verify the applicability of the new GFA parameter, Tg, Tx, Tl and Rc of some BMGs and oxide glasses are listed in Table 3, together with the calculated GFA parameters. The data of BMGs and oxide glasses in Table 3 were obtained from Table 3 in Ref.[4] and Table 2 in Ref.[6], respectively, which are applied to interpreting γ. According to the data in Table 3, six plots of the critical cooling rate as the functions of the parameters, Trg, γ, α, δ,  and ω, respectively, are shown in Fig.4. The statistical factor R2 for these regressions are high among which R2 of ω is the highest. This suggests a reliable relationship between the GFA and ω for these BMGs and oxide glasses. And this relationship is expressed in an approximation formula:
and ω, respectively, are shown in Fig.4. The statistical factor R2 for these regressions are high among which R2 of ω is the highest. This suggests a reliable relationship between the GFA and ω for these BMGs and oxide glasses. And this relationship is expressed in an approximation formula:
lg Rc=6.72-0.80ω (8)
Table 3 Tg, Tx, Tl, Rc and calculated Trg, γ, α, δ, and ω for reported BMGs and oxide glasses
and ω for reported BMGs and oxide glasses

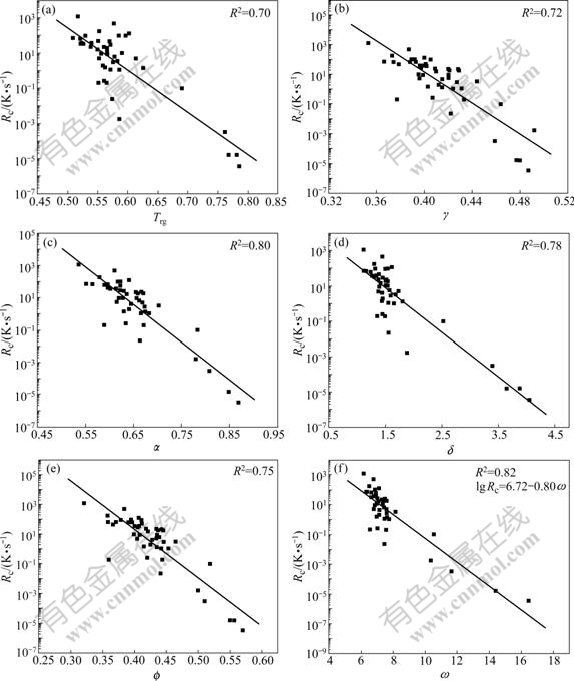
Fig.4 Correlations between Rc and Trg (a), γ (b), α (c), δ (d),  (e) and ω (f) for glasses including BMGs and oxides
(e) and ω (f) for glasses including BMGs and oxides
3.4 Applications of ω in bulk metallic glasses
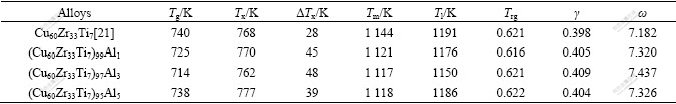
As an applicable GFA parameter, ω should indicate the actual GFA of BMGs. A centimeter-diameter Cu-based BMG had been developed[21]. Their experimental data and the calculated values are listed in Table 4. Values of ω and the calculated values of Zc are computed with Eq.(5) and Eq.(7), respectively. The relationship between the experimental values of Zc (ZcExp) and the calculated values of Zc (ZcCal) is shown in Fig.5. Although the values of ZcExp are not usually equal to the values of ZcCal for every alloying composition, they present a solid linear relationship with 0.92 of R2. Therefore, with the knowledge of Tx and Tl, the critical section thickness and the critical cooling rate of the alloy can be estimated approximately using Eqs.(7) and (8), respectively. Of course, these two expressions would be refined by more data of glasses to predict GFA of BMGs.
Table 4 Tg, Tx, Tl, actual critical section thickness ZcExp and calculated ω and ZcCal of reported Cu-Ag-Zr-Ti alloys


Fig.5 Actual critical section thickness ZcExp vs calculated critical section thickness ZcCal for Cu-based BMGs
Moreover, GFA of Cu-Zr-Ti-Al alloys [22] is described by ω parameter. The characteristic temperatures of the alloys are listed together with GFA parameters in Table 5. The results of scanning electron microscopy(SEM) and X-ray diffractometry(XRD) indicated the GFA of Cu-Zr-Ti-Al alloys are enhanced firstly and then depressed with minor additions of Al. Compared with the GFA parameters in Table 5, ΔTx, γ and ω are accordant to these experimental results, but except Trg, showing that ω is one of the best applicable and reliable GFA parameters.
Table 5 Characteristic temperatures and GFA parameters of prepared Cu-Zr-Ti-Al alloys
4 Conclusions
1) Based on the analysis of Gibbs free energy between liquid and crystal, a new GFA parameter, ω=Tl(Tl+Tx)/[Tx(Tl-Tx)], is suggested for bulk metallic glasses.
2) Together with other GFA parameters proposed formerly, including Trg, γ, α, δ and  , ω was verified with alloying and oxide glasses. Results indicate the new GFA parameter ω is the most reliable and applicable approach to assess the GFA of various glasses.
, ω was verified with alloying and oxide glasses. Results indicate the new GFA parameter ω is the most reliable and applicable approach to assess the GFA of various glasses.
3) ω is applied to estimating the GFA of Cu-Ag-Zr-Ti and Cu-Zr-Ti-Al BMGs and the predicted data are consistent with the experimental results.
Acknowledgments
The authors would like to thank Dr. CAI An-hui (Institute of Powder Metallurgy, Central South University, China) for helpful discussions.
References
[1] Lu Z P, Bei H, Liu C T. Recent progress in quantifying glass-forming ability of bulk metallic glasses [J]. Intermetallics, 2007, 15: 618-624.
[2] Turnbull D. Under what conditions can a glass be formed [J]. Contemp Phys, 1969, 10(5): 473-488.
[3] Inoue A, Zhang T, Masumoto T. Reductilization of embrittled La-Al-Ni amorphous alloys by viscous flow deformation in a supercooled liquid region [J]. J Non-Cryst Solids, 1993, 156/158: 598-602.
[4] Lu Z P, Liu C T. A new glass-forming ability criterion for bulk metallic glasses [J]. Acta Mater, 2002, 50: 3501-3512.
[5] Lu Z P, Liu C T. Glass formation criterion for various glass-forming systems [J]. Phys Rev Lett, 2003, 91: 115505 1-4.
[6] Lu Z P, Liu C T. A new approach to understanding and measuring glass formation in bulk amorphous materials [J]. Intermetallics, 2004, 12: 1035-1043.
[7] Mondal K, Murty B S. On the parameters to assess the glass forming ability of liquids [J]. J Non-Cryst Solids, 2005, 351: 1366-1371.
[8] Chen Q J, Shen J, Zhang D L, Fan H B, Sun J F, McCartney D G. A new criterion for evaluating the glass-forming ability of bulk metallic glasses [J]. Mater Sci Eng A, 2006, 433: 155-160.
[9] Fan G J, Choo H, Liaw P K. A new criterion for the glass-forming ability of liquids [J]. J Non-Cryst Solids, 2007, 353: 102-107.
[10] Kim J H, Park J S, Lim H K, Kim W T, Kim D H. Heating and cooling rate dependence of the parameters representing the glass forming ability in bulk metallic glasses [J]. J Non-Cryst Solids, 2005, 351: 1433-1440.
[11] Takeuchi A, Inoue A. Calculations of dominant factors of glass-forming ability for metallic glasses from viscosity [J]. Mater Sci Eng A, 2004, 375/377: 449-454.
[12] Thompson C V, Spaepen F. On the approximation of the free energy change of crystallization [J]. Acta Metall, 1979, 27(12): 1855-1859.
[13] Lad K N, Raval K G, Pratap A. Estimation of Gibbs free energy difference in bulk metallic glass forming alloys [J]. J Non-Cryst Solids, 2004, 334/335: 259-262.
[14] Mondal K, Chatterjee U K, Murty B S. Gibbs free energy of crystallization of glass forming liquids [J]. Appl Phys Lett, 2003, 83: 671-673.
[15] Chen H S. Metallic glasses [J]. Chinese J Phys, 1990, 28(5): 407-425.
[16] Senkov O N, Scott J M, Miracle D B. Composition range and glass forming ability of ternary Ca-Mg-Cu bulk metallic glasses [J]. J Alloy Compd, 2006, 424: 394-399.
[17] Xiao X S, Fang S S, Wang G M, Hua Q, Dong Y D. Influence of beryllium on thermal stability and glass-forming ability of Zr-Al-Ni-Cu bulk amorphous alloys [J]. J Alloy Compd, 2004, 376: 145-148.
[18] Senkov O N, Scott J M. Glass forming ability and thermal stability of ternary Ca-Mg-Zn bulk metallic glasses [J]. J Non-Cryst Solids, 2005, 351: 3087-3094.
[19] Cabral Jr A A, Fredericci C, Zanotto E D. A test of the Hruby parameter to estimate glass-forming ability [J]. J Non-Cryst Solids, 1997, 219: 182-186.
[20] Lu Z P, Tan H, Li Y, Ng S C. The correlation between reduced glass transition temperature and glass forming ability of bulk metallic glasses [J]. Scripta Mater, 2000, 42(7): 667-673.
[21] Dai C L, Guo H, Shen Y, Li Y, Ma E, Xu J. A new centimeter-diameter Cu-based bulk metallic glass [J]. Scripta Mater, 2006, 54: 1403-1408.
[22] Ji X L, Pan Y, Cai A H. The effect of Al on glass forming ability of Cu-based bulk metallic glasses [J]. J Cent South Univ Technol, 2007, 14(S2): 20-23.
Corresponding author: JI Xiu-lin; Tel: +86-519-85191969; E-mail: xiulinji@gmail.com
DOI: 10.1016/S1003-6326(08)60438-0
(Edited by YANG Hua)