
Low temperature oxidation of Cr-alloyed MoSi2
E. Str?m1, Y. Cao2, Y. M. Yao2
1. Kanthal AB, Box 502, SE-734 27 Hallstahammar, Sweden;
2. Department of Materials and Manufacturing Technology, Chalmers University Technology,
SE-412 96 Gothenburg, Sweden
Received 15 July 2007; accepted 10 September 2007
Abstract: Cr-alloyed MoSi2 was compared with monolithic MoSi2 with respect to oxidation at 450 ℃ for 456 h. Phases formed on Cr-alloyed MoSi2 after exposure are Cr2(MoO4)3, MoO3, and cristobalite (SiO2) according to X-ray diffraction results. Monolithic MoSi2 forms MoO3 and mainly amorphous SiO2. X-ray photoelectron spectroscopy indicates that the main oxidation product on the outermost surface is SiO2 for all studied samples. The samples form a relatively loose oxide but the oxide adherence improves with increasing Cr content. It is indicated that Cr addition can benefit pesting control in MoSi2.
Key words: molybdenum silicides; pesting; oxidation; alloying
1 Introduction
Many intermetallics are under intensive investigation as high temperature materials. Of these, MoSi2 is perhaps the most interesting, because of its high melting point (2 030 ℃), excellent oxidation resistance up to 1 800 ℃ in air and low density (6.27 g/cm3). The oxidation resistance at high temperature is due to the formation of a viscous SiO2 scale that can be self-healing if damaged. However, at 400-600 ℃ MoSi2 suffers from catastrophic oxidation sometimes referred to as pesting[1].
There are several investigations that have dealt with the so-called pest oxidation in MoSi2-based materials. As summarised in Ref.[2], some say that pest is not an intrinsic property in MoSi2, as crack-free, hot isostatically pressed(HIPed) or single crystalline MoSi2 does not pest. However, some reports that hot pressed or single crystalline MoSi2 indeed suffers from pest oxidation[2]. In another study[3], silicon-rich MoSi2 alloys were able to form protective SiO2 even at 500 ℃ and showed no indication on disintegration. It has been reported that in MoSi2-composites with additions of Si3N4, pesting has been overcome[4]. In silicides based on the Mo5SiB2-phase it is argued that B additions increase the fluidity of the silica scale and encapsulate the silicide grains, thereby preventing oxidation induced by cracking. However, studies so far have not been extended to temperatures below 600 ℃[5].
Alloying additions to MoSi2 may alter its oxidation properties at low temperatures. Alloying studies so far have focused on additions of Ti, V, Cr, Zr, Nb, Ta, W, Re, and Al[6-7]. With a few exceptions, the alloying content has been on the order of 1% (molar fraction) corresponding to (Mo0.97M0.03)Si2, where M is the alloying element. YANAGIHARA et al[8] studied the pesting of Mo-Si-X (X=Al, Ta, Ti, Zr, and Y) to evaluate the third element effect. They reported that elements that have stronger affinity to oxygen than Si, e.g., Al, Ti, Zr, and Y, can prevent MoSi2 from pesting. A possible effect of the elements that have stronger affinity for oxygen than Si is that they decrease the oxygen flux toward the oxide-intermetallic interface and increase the plasticity of the amorphous oxide formed in cracks[2]. So far, there has been no study on the effect of Cr on pesting of MoSi2 reported in literature. Very promising results have been reported on Cr-rich silicide composites consisting of (Cr,Mo)3Si and (Cr,Mo)5Si3 that do not show any signs of pest even after 2 000 h at 500 ℃[9]. Therefore, in this work, polycrystalline, Cr-alloyed MoSi2 was compared with monolithic MoSi2 with respect to oxidation at 450 ℃.
2 Experimental
The samples were prepared by sintering powders of designed compositions, i.e., MoSi2, (Mo0.90Cr0.10)Si2 and (Mo0.85Cr0.15)Si2, in hydrogen at 1 615 ℃. Densities after sintering were 95.4%, 99.3% and 99.0 % of theoretical, respectively. Bulk chemical analysis was performed to determine the sample compositions as well as the C, O, and N contents. The results of the chemical analyses of the studied alloys are shown in Table 1. Nominal and measured compositions are listed in Table 2.
The microstructural characterisation and phase analysis of the alloys were carried out using scanning electron microscopy (SEM, LEO 1550 Gemini), energy dispersive spectrometry (EDS, Oxford) and X-ray diffraction (XRD, Bruker axs D8 ADVANCE diffractometer, Cr Kα radiation, 35 kV, 40 mA, secondary monochromator). Lattice parameter calculations of the Cr-alloyed samples were performed using monoclinic ZrO2 as an internal standard. Subsequent density calculations were based on chemical analysis data. Oxidation tests were performed for 456 h (three weeks) in ambient air at 450 ℃, using MoSi2 as reference material.
The samples subjected to oxidation were also characterized by X-ray photoelectron spectrometry(XPS) using a PHI5500 electron spectro- meter. The photoelectrons were generated with an Al Kα (1 486.6 eV) X-ray source. The acquisition conditions for the survey spectra (0-1 200 eV) were 93.9 eV pass energy, 45? take-off angle and 0.4 eV/step. Selected region spectra were recorded covering the Si2p, C1s and O 1s, Mo 3d and Cr 2p to estimate the surface concentration. The acquisition conditions were 23.5 eV pass energy, 45? take-off angle, 0.1 eV/step for the selected region spectra. The position of adventitious carbon (284.6 eV) was used to calibrate the peak shift.
3 Results and discussion
Table 2 shows the phase constitution, cell parameters and densities as calculated from XRD. It can be seen that the lattice parameters decrease with increasing Cr content, which indicates solid solution of Cr into the MoSi2 crystal.
According to the results of UMAKOSHI et al[11] MoSi2 can dissolve not more than 2.6% (molar fraction) Cr corresponding to the formula (Mo0.92Cr0.08)Si2, since their alloy with composition (Mo0.90Cr0.10)Si2 showed some C40 reflections in XRD. Some indications of C40 phase are also found in our work for this composition, i.e. 3.3% Cr. Interestingly, XRD indicates that Cr is completely dissolved into the C11b phase without forming hexagonal C40 phase for the alloy having 4.7% Cr additions, i.e., (Mo0.85Cr0.15)Si2, since no C40 reflections are found (see Fig.1). In SEM, Cr5Si3 grains containing high amounts of Mo are observed at grain junctions and grain boundaries in the matrix (see Fig.2). The Cr/Mo-ratios in Cr5Si3 as determined by EDS were 4.0 and 5.3 in Cr1 and Cr2, respectively, which explains their contrast being darker than that of the matrix phase.
Fig.3 shows the three studied samples after exposure at 450 ℃ in ambient air for 456 h. It can be noticed that there is a shift in colour with increasing Cr-addition. All samples have a loose oxide scale that is partly spalled during sample handling, though the adherence improves with Cr-alloying.
XRD patter indicates that the oxide products formed
Table 1 Chemical compositions of investigated samples (mass fraction, %)
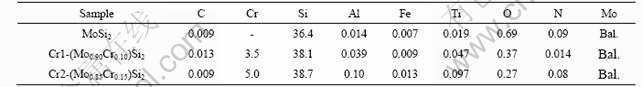
Table 2 Nominal and measured compositions of Cr-added samples (molar fraction, %)
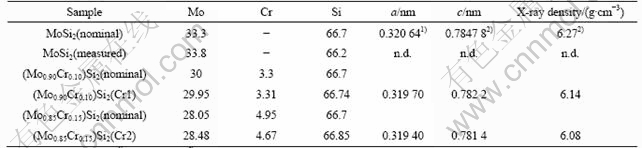
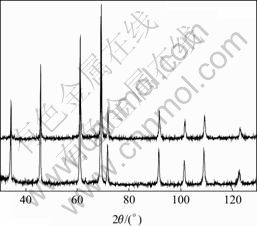
Fig.1 XRD patterns for (Mo0.90Cr0.10)Si2 (lower) and (Mo0.85Cr0.15)Si2 (upper) (Existence of C40 phase in (Mo0.90Cr0.10)Si2 at about 63? (2θ). All other reflections are from C11b phase)

Fig.2 Backscattered electron micrographs of sample Cr1 (a) and Cr2 (b), showing matrix phase (Mo,Cr)Si2 (bright contrast) and precipitates (indicated with arrows)
on MoSi2 are MoO3 and SiO2, though the latter seems to exist also in amorphous form due to the curvature of the background and the low acquisition rate in its diffractogram (see Fig.4). It is also indicated that the oxide is rather thick, since no reflections from the bulk are visible using normal Bragg-Brentano geometry. In contrast, reflections of the bulk are seen both for Cr1 and Cr2 despite that grazing incidence XRD is used (see Fig.5). Thus, the oxide formed on Cr1 and Cr2 is probably

Fig.3 Samples after exposure for 456 h in ambient air at 450 ℃ (From left to right: MoSi2, Cr1, and Cr2; sample diameter about 10 mm)
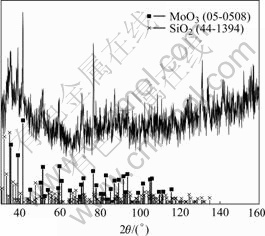
Fig.4 XRD patterns of MoSi2 after exposure at 450 ℃ in ambient air for 456 h
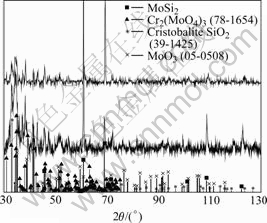
Fig.5 XRD patterns of Cr1 (lower) and Cr2 (upper) after exposure for 456 h in ambient air at 450 ℃
thinner than that formed on MoSi2. The phases that were identified by XRD on Cr1 and Cr2 after exposure were Cr2(MoO4)3, MoO3, and cristobalite (SiO2).
Fig.6 shows the XPS survey spectra of the samples MoSi2 and Cr1 after exposure at 450 ℃ for 456 h in ambient air. It is indicated that only small amounts of Mo and/or Cr exist on the top surface, while oxygen and silicon signals are dominant. It can be seen in the inserted Si and O peaks in Fig.6 that the Si 2p has its maximum at about 103.4 eV and the O 1s has its maximum at 532.5 eV, indicating the formation of SiO2. The main product on the top surface after exposure is thus SiO2.
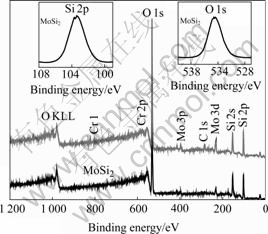
Fig.6 XPS spectra of exposed samples MoSi2 and Cr1 (Inserts are Si 2p (upper left) and O 1s (upper right) peaks)
It has been widely accepted that pesting of MoSi2 is a function of temperature, microstructure, alloying element, composition, and atmosphere. For alloyed C11b-MoSi2, STERGIOU and TSAKIROPOULOS reported that neither Ta (C40 structure stabilizer) nor W (C11b structure stabilizer) additions suppressed pesting, while Al (C40 stabilizer) added alloys with pure C40 structure did[12]. However, a study on the effect of Al concentration on pesting in MoSi2 showed that even as low as 5% (molar fraction) Al, i.e., single-phase C11b, eliminated pesting at 500 ℃[13]. So, the structure of MoSi2 is a less sensitive aspect for pesting than the alloying effect, even though the C40 structure might allow different solid solubilities than the C11b structure for the protective scale forming elements, e.g., Al and Cr. Therefore, the existence of small amounts of C40 phase detected by XRD in this study may play a secondary role on pesting.
Compared with monolithic MoSi2 in this study, it is confirmed that a thin and more protective oxide scale is formed on the surface of powder metallurgy produced (Mo,Cr)Si2 materials at 450 ℃. XRD and XPS analyses have shown that SiO2 is the main constituent in the surface oxides. The mechanism of pesting resistance in the materials is not clear at this stage. Whether protective Cr2O3 is formed beneath the outermost surface or not at the pesting temperature, which is the objective of the material design, needs further examination.
Usually, both monolithic and MoSi2-composites contain small amounts of Mo5Si3, resulting from excess Mo for manufacture reasons or oxidation of MoSi2 during sintering, due to the strict stoichiometry of MoSi2. However, it is pertinently noticed that Cr5Si3 grains with high Cr to Mo atomic ratio instead of Mo5Si3 phase is formed in the MoSi2 matrix in this study. It is understandable for such a high Cr-content in Mo5Si3, as Cr5Si3-Mo5Si3 is an infinitive solid-solute system according to AGEEV[14]. As a result, the excess Cr in the MoSi2 matrix is accommodated in the (Cr,Mo)5Si3 phase. It has been suggested that the existence of Mo5Si3 phase promotes accelerated oxidation due to high oxygen diffusion in Mo5Si3 phase[15]. Therefore, a mechanism of improved pesting resistance seen in this study for Cr-added MoSi2 can be due to the formation of highly Cr-containing Mo5Si3.
4 Conclusions
Cr-alloyed MoSi2 was compared with monolithic MoSi2 with respect to oxidation at 450 ℃, which is in the so-called pest temperature range for MoSi2, for 456 h. According to XRD, phases formed on Cr-alloyed MoSi2 after exposure are Cr2(MoO4)3, MoO3, and cristobalite (SiO2). Monolithic MoSi2 forms MoO3 and mainly amorphous SiO2 as indicated by XRD and XPS. As indicated by XPS, the main oxidation product on the very top surface is SiO2 for all three samples. All studied samples form a relatively loose, non-protective oxide but the oxide adherence improves with increasing Cr content. Also, the oxide thickness decreases with Cr-addition as indicated by the presence of bulk reflections in XRD for Cr-alloyed MoSi2 as opposed to its monolithic counterpart.
Acknowledgement
This study was partly sponsored by the Swedish Consortium for Materials Use in Energy Applications. Urban Jelvestam at Chalmers University of Technology is gratefully acknowledged for his help with the XPS experiments.
References
[1] GRABKE H J, MEIER G H. Accelerated oxidation, internal oxidation, intergranular oxidation, and pesting of intermetallic compounds [J]. Oxid Met, 1995, 44: 147-176.
[2] YANAGIHARA K, PRZYBYLSKI K, MARUYAMA T. The role of microstructure on pesting during oxidation of MoSi2 and Mo(Si,Al)2 at 773 K [J]. Oxid Met, 1997, 47: 277-293.
[3] McKAMEY C G, TORTORELLI P F, DeVAN J H, CARMICHAEL C A. A study of pest oxidation in polycrystalline MoSi2 [J]. J Mater Res, 1992, 10: 2747-2755.
[4] PETROVIC J J, VASUD?VAN A K. Key developments in high temperature structural materials [J]. Mater Sci Eng A, 1999, A261: 1-5.
[5] MEYER M K, THOM A J, AKINC M. Oxide scale formation and isothermal oxidation behavior of Mo-Si-B intermetallics at 600- 1 000 ℃ [J]. Intermetallics, 1999, 7: 153-162.
[6] HARADA Y, MURATA Y, MORINAGA M. Solid solution softening and hardening inalloyed MoSi2 [J]. Intermetallics, 1998, 6: 529-535.
[7] YI D, LAI Z, LI C, AKSELSEN O M, ULVENSOEN J H. Ternary alloying study of MoSi2 [J]. Metall Mater Trans A, 1998, 29: 119-129.
[8] YANAGIHARA K, MARUYAMA T, NAGATA K. Effect of third elements on the pesting suppression of Mo-Si-X intermetallics (X=Al, Ta, Ti, Zr and Y) [J]. Intermetallics, 1996, 4: S133-S139.
[9] RAJ S V. A preliminary assessment of the properties of a chromium silicide alloy for aerospace applications [J]. Mater Sci Eng A, 1995, 192/193: 583-589.
[10] HARADA Y, MORINAGA M, SASO D, TAKATA M, SAKATA M. Refinement in crystal structure of MoSi2 [J]. Intermetallics, 1998, 6: 523-527.
[11] UMAKOSHI Y, HIRANO T, SAKAGAMI T, YAMANE T. Yield stress and dislocation structure of MoSi2 and (Mo,Cr)Si2 single crystals [C]// High Temperature Aluminides and Intermetallics. TMS Warrendale, 1990: 111-132.
[12] STERGIOU A, TSAKIROPOULOS P. Oxidation studies on MoSi2-X (X=Al, Ta, W) alloys [C]// Structural Intermetallics 1997. TMS Warrendale, 1997: 869-875.
[13] ARVANITIS A, WHITING M J, TSAKIROPOULOS P. A study of the oxidation behaviour of Mo-Si-Al intermetallic alloys at low, intermediate and high temperatures [C]// Structural Intermetallics 2001. TMS Warrendale, 2001: 561-570.
[14] AGEEV N V. Diagrammy sostoianiia metallicheskikh system [M]. Moscow: Viniti Press, 1964: 182.
[15] MESCHTER P J. Low-temperature oxidation of molybderum disilicide [J]. Metall Trans A, 1992, 23: 1763-1772.
(Edited by LI Xiang-qun)
Foundation item: Project supported by the Swedish Consortium for Materials Use in Energy Applications
Corresponding author: E. Str?m; Tel: +46-220-21665; E-mail: erik.o.strom@kanthal.com