非晶态催化剂电催化水分解制氢进展
来源期刊:中国有色金属学报2021年第4期
论文作者:余林键 熊昆 陈佳 张海东 向阳 许景钫
文章页码:1007 - 1019
关键词:非晶态催化剂;表/界面;电解水;析氢反应;析氧反应
Key words:amorphous catalyst; surface/interface; water electrolysis; hydrogen evolution reaction; oxygen evolution reaction
摘 要:非晶态催化剂作为一类新型催化材料,具有短程有序长程无序的结构特点,在相同质量下拥有更大的比表面积和结构缺陷,使其在催化反应中具有更多配位不饱和活性位,同时,它不易发生偏析和异相,能保持均匀性和各向同性,可以根据电催化条件自我调节并提供反应所需体积和有效比表面,从而在电解水阴极析氢反应(HER)和阳极析氧反应(OER)中具有优异的催化活性。基于此,结合本课题组在电解水方面的研究工作,综述了近几年非晶态电解水催化剂的最新研究进展,重点介绍了几类非晶态催化剂的可控合成、电催化水分解性能调控策略、表面电子结构与催化活性和稳定性之间的构效关系,并对非晶态的电解水催化剂活性中心的调控面临的挑战和发展进行了展望。
Abstract: Amorphous catalyst as a new type of catalytic material has the characteristics of short-range order and long-range disorder. Compared with the crystalline catalyst, the amorphous catalyst has larger specific surface area and structural defects under the same mass, which makes it more active sites for the catalytic reaction. In addition, the excellent flexibility and flexibility of the amorphous catalyst can induce the active sites to form suitable morphology for promoting the reaction rate due to its uniformity and isotropy, which can adjust their catalytic conditions and provide volume and capacity according to the surface electron transfer, promoting the electrocatalytic activities of hydrogen evolution reaction (HER) and oxygen evolution reaction (OER) in water splitting. Herein, the latest progress in several major types of the amorphous catalysts for electrocatalytic water splitting in recent years was reviewed, especially in the controllable synthesis of several kinds of amorphous catalysts, the control strategy on electrocatalytic performance for water decomposition, and the structure-activity relationship between surface electronic structure and catalytic activity and stability. Moreover, the prospects for the challenges and research direction of the adaptation relationship and modulation of the active site of the catalysts were proposed to shed light on further development of electrolysis water.
DOI: 10.11817/j.ysxb.1004.0609.2021-35988
余林键,熊 昆,陈 佳,张海东,向 阳,许景钫
(重庆工商大学 环境与资源学院 废油资源化技术与装备教育部工程研究中心,重庆 400067)
摘 要:非晶态催化剂作为一类新型催化材料,具有短程有序长程无序的结构特点,在相同质量下拥有更大的比表面积和结构缺陷,使其在催化反应中具有更多配位不饱和活性位,同时,它不易发生偏析和异相,能保持均匀性和各向同性,可以根据电催化条件自我调节并提供反应所需体积和有效比表面,从而在电解水阴极析氢反应(HER)和阳极析氧反应(OER)中具有优异的催化活性。基于此,结合本课题组在电解水方面的研究工作,综述了近几年非晶态电解水催化剂的最新研究进展,重点介绍了几类非晶态催化剂的可控合成、电催化水分解性能调控策略、表面电子结构与催化活性和稳定性之间的构效关系,并对非晶态的电解水催化剂活性中心的调控面临的挑战和发展进行了展望。
关键词:非晶态催化剂;表/界面;电解水;析氢反应;析氧反应
文章编号:1004-0609(2021)-04-1007-12 中图分类号:TQ151.1;O643.36 文献标志码:A
引文格式:余林键, 熊 昆, 陈 佳, 等. 非晶态催化剂电催化水分解制氢进展[J]. 中国有色金属学报, 2021, 31(4): 1007-1018. DOI: 10.11817/j.ysxb.1004.0609.2021-35988
YU Lin-jian, XIONG Kun, CHEN Jia, et al. Progress on hydrogen production from electrocatalytic water splitting on amorphous catalysts[J]. The Chinese Journal of Nonferrous Metals, 2021, 31(4): 1007-1018. DOI: 10.11817/j.ysxb.1004.0609.2021-35988
近年来,随着全球环境污染问题和能源短缺问题加剧,人们对清洁可再生能源的需求变得越来越紧迫[1-3]。其中,氢能由于能量高、产物清洁等优点受到了广泛关注,被认为是替代化石能源、解决能源和环境危机的最佳能源之一。因此,关于氢能的开发与利用已成为世界能源技术变革的重要研究方向,围绕“氢能经济”的制氢、储氢和氢燃料电池研究也受到高度关注[4-7]。在制氢方面,与化石能源裂解制氢相比,电解水制氢具有零碳排放且氢气纯度高的特点,然而,该技术的主要瓶颈是催化剂催化HER和OER所需的过电位高[8-10],尤其是阳极OER涉及更为复杂的四电子过程,严重限制了电解水制氢的整体效率[11]。研究者通过合金化、表面修饰或杂化改变催化剂的化学组成和电子结构,提高催化剂的本征活性和活性位密度,使电解水催化性能得到明显提升。
自从SMITH等[12]发表了第一篇关于非晶态催化剂的制备及应用并阐述了非晶态催化剂的优点及前景后,国内外开始关注非晶态材料在各个领域的应用。其中,非晶态催化剂在电解水中表现出优异的电催化性能,是近年来引起高度关注的一类新兴催化剂[13]。
因此,本文将结合本课题组在电解水方面的研究工作,对近几年应用于电解水的非晶态催化材料的最新研究进行总结,重点介绍非晶态过渡金属硫化物、磷化物、氧化物和氢氧化物催化剂等的合成、催化性能提高策略、表面几何结构和电子结构与催化活性和稳定性之间的构效关系,并对非晶态电解水催化剂活性中心的调变面临的挑战和发展方向进行了展望。
1 非晶态催化剂的特性
非晶态催化剂由于具有两个显著的特点,即短程有序和长程无序,导致在原子水平上以有序结构的状态随机堆积,从而在三维空间上呈现拓扑无序排列的形式[14-15],因此,与晶体型催化剂相比,非晶态催化剂在相同质量下包含更大的比表面积和结构缺陷,使其在催化反应中具有丰富的活性位[16-17]。这种非平衡的亚稳态结构增强了非晶态催化剂的灵活性和柔韧性,不仅容易使催化活性位点形成有助于催化反应的形态,提高反应速率,还增强了非晶态催化剂的抗腐蚀性和热力学稳定性[18-19]。
2 非晶态催化剂的研究进展
虽然电解水性能最好的催化剂仍然是铂族类贵金属,但已有研究表明[20],将贵金属制备成非晶态催化剂,其催化性能可进一步提升,如利用光驱动分解底物制备非晶态IrOx用于电解水OER。与晶体型IrOx相比,非晶态IrOx的Tafel斜率低至34 mV/dec,电流密度达到1 mA/cm2和10 mA/cm2时所需过电位可减少50 mV,仅为190 mV和220 mV,并在反应中保持良好的稳定性。SALIMI等[21]通过电化学法在金属Ir表面原位形成了非晶态IrOx。该催化剂具有较大的电化学比表面,催化OER的活性远高于晶态IrOx。TSUJI等[22]利用电沉积和射频磁控溅射在FTO基板上制备了RuO2薄膜并调节退火温度形成晶态和非晶态结构。由于非晶态RuO2的独特排列结构增加了催化反应的表面与空间结构,促使反应活性位增加,因此其起始电位明显降低。然而,这些贵金属在地壳中的储量较少、价格昂贵,难以满足大规模商业应用[23]。近年来,关于非晶态过渡金属电催化剂的研究受到了广泛关注,一些催化剂的催化性能甚至可以媲美贵金属催化剂[24]。下面将主要针对这些非晶态催化剂进行介绍和讨论。
2.1 非晶态过渡金属硫化物
非晶态硫化物富含硫原子并相互作用形成了二硫键,从而具有大量的不饱和配位硫原子,因此电解水HER活性优异[25]。为了增加该活性位密度,SUN等[26]利用电沉积法在FTO电极上获得CoS2,然后在氩气条件下300 ℃退火形成非晶态Co-S/FTO。与其他在酸性条件下具有较好HER活性的晶体型催化剂相比,该催化剂在中性条件下也能显示出优异的HER活性。由于高温退火处理可以使晶体内部原子来不及在结晶温度时规则排列,从而形成混乱的无序的非晶态结构;反之,给予非晶态材料一定温度,非晶态中混乱排列的原子可以在结晶温度下重新有序的排列成为晶体[27]。基于此,KORNIENKO等[28]通过调控电沉积参数,在氩气条件下退火制备了非晶态CoSx催化剂并用于HER研究。EDS表明该催化剂的Co/S为1.4,当电流密度达到10 mA/cm2时,仅需83 mV的过电位。通过结合拉曼和XPS发现,其良好的活性主要来源于形成的非晶态CoSx的团簇富含硫原子缺陷结构,从而容易被质子所包围,导致接受的S原子倾向于在质子的解离中向上输送H2,因此HER活性显著提高。在此基础上,GUO等[29]调整了催化剂的表面几何结构,将咪唑类金属框架材料进行原位硫化,合成了非晶态中空CoS2纳米盒。他们通过测试电化学比表面及双电容发现,该催化剂的活性中心密度显著提高,在碱性介质中仅需290 mV即可达到10 mA/cm2的电流密度,并可连续稳定运行40 h不发生明显的衰减。LIU等[30]用水热法合成了非晶态CoSx薄膜,并将其沉积在泡沫铜(CF)上合成了新型Cu@CoSx/CF纳米复合材料。该催化剂在碱性溶液中对HER和OER均显示出优异的催化活性和稳定性,并且在碱性电解槽中的活性可与Pt/C-IrO2耦合电极相媲美。CAI等[31]则在非晶态CoSx晶格中引入同族元素O,获得了A-CoS4.6O0.6PNC催化剂,从而进一步调节硫化物表面的电子结构。通过DFT计算表明,引入元素O后,显著提高了O*的吸附量,从而使活性得到显著提高。该催化剂除了具有良好的HER性能外,在电解水OER中还显示出类似于RuO2的催化特性,因此有望作为一类双功能催化剂用于全电解水反应中。
对于晶态硫化钼而言,只有边缘位置具有较高的反应活性,因此活性位密度低[32-34],难以实现规模化应用。近年来,研究者发现,通过改变硫化钼的晶态,实现非晶化并调整表面几何结构和电子结构,可以显著提高其HER催化活性。YU等[35]通过乙酸乙酯诱导形成非晶态MoSx纳米团簇,并将其改造为3D结构,从而得到了更多的不饱和S原子,从而显著提高了产氢速率。SONG等[36]则在Ni3S2/NF纳米片表面包覆修饰非晶态MoS2,HRTEM结果表明,MoS2纳米层呈现非晶态(见图1),MoS2垂直于Ni3S2紧密连接;LSV及Tafel测试显示,经过Ni3S2/NF纳米片包覆修饰的非晶态MoS2活性远高于晶态MoS2。其主要原因是选用的晶态Ni3S2增加导电性的同时,还通过构造非晶态MoS2增加催化剂的活性比表面,暴露更多活性位,因此在HER和OER中均展现出优异的催化作用。由于硫化钨具有硫化钼的相似电子结构,因此WU等[37]在设计催化剂时,在非晶态硫化钨的基础上引入过渡金属原子Ni和Co,分别形成了非晶态NiWS催化剂和非晶态CoWS催化剂。其中NiWS活性最佳,Tafel斜率为55 mV/dec,在250 mV的过电位下电流密度为8.6 mA/cm2。这是由于引入的Ni和Co改变了硫化钨的表面电子结构,使硫化钨催化剂表面的活性位点增多,电催化活性得到显著提高。
2.2 非晶态过渡金属磷化物
2005年,RODRIGUEZ等[38]首次提出Ni2P催化HER的作用机理类似于氢化酶,当质子受体位点和氢化物受体位点共存于Ni2P的表面时,其作用机理与Ni/Fe氢化酶的催化机理近似。当活性氢吸附于Ni活性位时,由于P原子的强电负性,部分电子从Ni转移到相邻的P原子,使得带负电的P可以捕获带正电的活性氢,从而辅助吸附H在Ni2P表面析出。在该过程中,金属中心和磷共同促进了析氢反应的进行,这一重要理论促进了金属磷化物催化HER的研究[39-41]。

图1 非晶态MoS2的合成示意图和MoS2的SEM像及SAED谱[36]
Fig. 1 Synthesis diagram(a) of amorphous MoS2, SEM(b) and SAED(c) images of MoS2[36]
基于此,研究者开始构建多元过渡金属磷化物[42],并将其拓展到OER研究中,形成优异的双功能催化剂[43-45]。LI等[44]利用水热法在纳米磷化镍基底表面制备了非晶态NiCoPi双功能电催化剂,在碱性电解液中仅需73 mV过电位即可达到10 mA/cm2的电流密度,在OER中也只需要234 mV的过电位。其展示出优异催化性能的主要原因在于纳米磷化镍基底不仅提高了催化剂的稳定性,还增大了催化剂比表面积并构建了电极与活性中心NiCoPi的通道,使电解水反应速率加快。ZHANG等[45]利用水热法联合固相磷化,得到非晶态CoFePx,通过调控NaH2PO2用量可以将催化剂厚度从350 nm减少到50 nm,使其更容易暴露活性位点,增强电化学活性。进一步研究发现非晶态结构可以使金属―磷化学键和部分氧缺陷位点磷化,从而调节催化剂表面电子结构并稳定,进而防止内部CoFeP的腐蚀,并促进电子从内部传输到外部的非晶态壳上,促进了催化剂和电解质之间的接触和催化剂的电荷转移。因此在电流密度达到10 mA/cm2时,HER过电位仅为80 mV,OER也仅为242 mV。WANG等[46]通过控制水热法条件的基础上联用高温处理,制备了催化性能更加优异的非晶态纳米球Co-Fe-P催化剂。电流密度为10 mA/cm2时,OER过电位约为217 mV,Tafel斜率为40 mV/dec,不仅优于商用IrO2,而且超过大多数磷化物。然而,与水热法相比,ZHOU等[47]则认为,利用电沉积制备无定型磷化物更加简便有效,对催化剂的界面调控也更容易,通过调控电解液浓度、pH值、电流密度和沉积时间等电沉积参数,可以在镍网上制得一系列非晶态过渡金属磷化物。其中,合成的三元非晶态NiFeCoP/NM粒子大小均匀,孔结构有序,在电流密度为10 mA/cm2时,催化OER所需过电位为277 mV,Tafel斜率36.4 mV/dec;催化HER所需过电位为33 mV,Tafel斜率为71.1 mV/dec。他们认为,在NiFeCoP/NM的电镀过程中,氢气泡模板形成的独特微孔结构增大了催化体积和表面,丰富了界面,并且非晶态催化剂拥有的丰富缺陷结构增加了活性中心和多金属原子在磷化物中形成的协同效应共同作用,使该催化剂对于H*和O*的吸附量得到提高,增强了电化学活性。最近,本课题组[48]通过在有序多孔Ni表面上经电化学P化制备了非晶态Ni-P/p-Ni催化剂。该催化剂经过非晶化后拥有了更加丰富的界面区域,使HER活性显著增强,且Ni-P/p-Ni表面Ni2+容易进一步被氧化为Ni(OH)2。而Ni(OH)2加速了H―OH键的断裂,其HER活性相比于晶态p-Ni得到了显著的提升(见图2)。
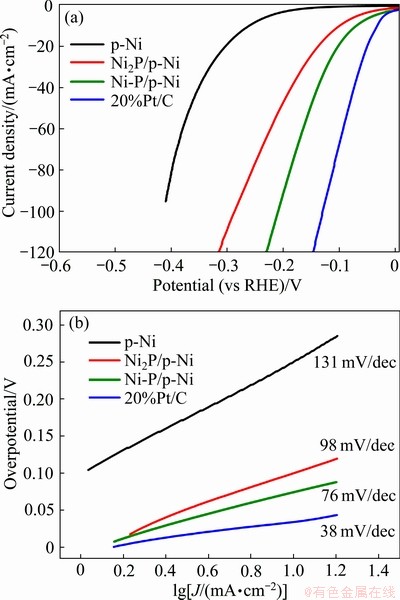
图2 不同电极的极化曲线和相应的Tafel曲线[48]
Fig. 2 Polarization curves(a) of different electrodes and corresponding Tafel curves(b)[48]
2.3 非晶态过渡金属氧化物
一般而言,具有p型半导体的过渡金属氧化物在OER阳极电位下容易产生表面空穴积累,空间电荷层中的电位降可以忽略不计,而对于n型半导体则没有表面空穴的积累,界面上的空间电荷层导致电荷载流子的附加势垒,增加电势,因此,p型半导体更适合作为OER催化剂[42, 49-50]。CoOx是研究最多的p型非晶态氧化物催化剂,SWITZER等[51]利用电沉积法制备了晶态和非晶态Co3O4,并用Tafel分析法比较了晶态和非晶态Co3O4的OER催化活性。实验表明,在碱性条件下晶体型Co3O4的Tafel斜率为49 mV/dec,交换电流密度为2.0×10-10 A/cm2,而非晶型Co3O4的Tafel斜率降低为36 mV/dec,交换电流密度为5.4×10-12 A/cm2,由此可见,非晶型Co3O4的析氧活性明显优于晶体型。
CHEN等[52]使用介质阻挡放电等离子体合成了高度分散的晶体CoOx团簇(LS-Co),并在800 ℃下氩气退火形成非晶态CoOx(LS-CoOx)。通过一系列电化学性能测试表明,非晶化后的CoOx拥有更大的电化学有效表面,LS-CoOx的电容比晶体型高2.45倍;在碱性条件下,非晶态LS-CoOx的Tafel斜率为47 mV/dec,晶体型为87 mV/dec;在电化学阻抗测试中,LS-CoOx的Rct为45 Ω,明显低于晶体型的115 Ω。他们认为,该催化剂活性提高的主要原因是非晶态结构在相同质量下具有更大的比表面积和结构缺陷。
然而,CoOx的稳定性不高,特别是在高电流密度下,稳定性明显下降。为了进一步提高其催化性能,CHEN等[53]采用光化学金属有机沉积(PMOD)合成了含Mo、Co、Fe的非晶态混合金属氧化物OER催化剂,将高价态Mo引入CoOx、FeOx后,Co50Mo50Ox和Fe50Mo50Ox的Tafel斜率下降明显且稳定性以及电化学活性得到显著提升,作者通过动力学实验探究了引入不同元素后催化机制的变化,如图3所示,图3(a)中不含Co时显示了一个起始电位>1.75 V的红色区域,然而当三种元素按比例混合在一起时,则出现了起始电位约为1.5 V的蓝色岛状区域;同样地,图3(b)中不含Co时显示了一个Tafel斜率>70 mV/dec的红色区域,当三种元素按比例混合后,Tafel斜率约为30 mV/dec并与图3(a)的深蓝色区域重合。由此可见,通过多元素掺杂后,不仅有效降低了催化剂的起始电位,还可以改变电解水反应机制。
因此,在催化剂的合成过程中,引入过渡金属形成多元过渡金属OER催化剂,可以有效地调节催化剂的电子结构,尤其是引入的Co、Fe、Mn、Ni等具有多种化合价的过渡金属,容易形成MOOH活性中心,同时,在引入其它元素形成协同效应时,混合价态的存在也更利于调控各元素之间与催化剂活性中心的适配关系,从而有效提高电解水反应速率。
2.4 非晶态过渡金属氢氧化物
氢氧化物作为一类具有较大比表面,亲水性能良好的层状材料,对H2O中H―OH键的断裂具有较好的催化活性,但由于受到其电导率和吸附H脱附等因素的限制,在碱性条件下催化HER活性较差,所以多用于阳极OER研究[54-57]。SHAO等[58]介绍了一种新型高效的非晶态催化剂合成方法,在室温条件下,利用5% H2O2氧化FeN和Co(NO3)2,仅需5 min即可在大孔泡沫铁上原位生成非晶态Co-Fe-OH纳米片阵列。在电解水OER中,该催化剂具有较低的Tafel斜率,当电流密度达到10 mA/cm2和100 mA/cm2时,其过电位分别仅为208 mV和298 mV,法拉第效率达到93%。经过稳定性测试后的Co-Fe-OH的微观形貌图依然保持原有结构。ZOU等[59]利用离子交换法修饰改性Ni3S2纳米片,在其表面生成非晶态Ni-Fe-OH薄膜,形成Ni-Fe-OH@Ni3S2/NF分层核壳结构。该催化剂中的非晶态Ni-Fe-OH薄膜作为OER的主活性中心,而Ni3S2纳米片承担增强电荷转移速率和进一步增大活性表面积的作用,因此在电催化OER中具有优异的催化性能。
虽然氢氧化物常用于OER研究,但通过改性后也可促进阴极HER活性,XU等[60]将用电沉积的方法将NiS和Ni2P嵌入非晶态Ni(OH)2纳米片形成双功能复合催化剂,如图4所示,直接沉积的Ni-P易团聚,形成纳米球,比表面较小,引入S后则形成了具有较大比表面积的花瓣状结构,同时,S有助于改变Ni-P晶体取向,使Ni-P晶面从(111)转变为(201),极大程度上丰富了界面区域,提升了电化学活性,使其在碱性条件下电流密度达到10 mA/cm2时,HER仅需要120 mV的过电位,OER为219 mV。
YUAN等[61]受到表面工程形态学中杂原子掺杂和缺陷调控的启发,利用电沉积技术将非晶态Ni(OH)2修饰在贵金属Pt表面(见图5)。由于晶体结构Pt与非晶态Ni耦合协同,导致Ni(OH)2-Pt界面存在大量的晶格缺陷,本征活性进一步提升,电子迁移速率加快,反应动力学提高。当电流密度为10、50和100 mA/cm2时,Ni(OH)2-Pt的过电位分别只有 38、114和203 mV,显著优于商用Pt/C和Pt片。此外,该催化剂在甲醇燃料电池研究中也表现出了优异的甲醇氧化活性和稳定性。他们认为,除了Ni(OH)2本身的优异水解离能力外,Pt与Ni物种形成的协同效应以及改性后所导致的非晶态表面缺陷结构增加是该催化剂性能优异的主要因素。

图3 含Mo、Fe和Co的非晶态金属氧化物动力学参数的等高线图[53]
Fig. 3 Contour plots of kinetics parameters for amorphous metal oxides containing Mo, Fe, and Co[53]

图4 电沉积Ni-P(a)和Ni-P-S(b)的SEM像以及电沉积Ni-P(c)和Ni-P-S(d)的HR-TEM像[60]
Fig. 4 SEM images of electrodeposited Ni-P(a) and Ni-S-P (b), and HR-TEM images of electrodeposited Ni-P (c) and Ni-S-P (d)[60]
2.5 其他非晶态催化剂
除了上述常用于电解水的非晶态催化剂外,还有少数磷酸盐、硼酸盐、氮化物(TMNs)等非晶态催化剂也被用于电解水[62-67]。NOCERA等[62]利用电沉积制备了可催化OER的非晶态金属磷酸盐和硼酸盐催化剂,其合成非晶态金属磷酸盐和硼酸盐催化剂的方法和理论对后续科研工作者提供了巨大的帮助。SURYAWANSHI等[64]在3D多孔NF上制备了非晶态Co-Fe-Bi催化剂,如图6所示,Co-Fe-Bi均匀的覆盖于NF表面,选区电子衍射证实了非晶型的存在,EDS表明Co、Fe、Bi分布均匀,其ECSA和OER活性远高于其它晶体型催化剂。在此基础上,LIU等[65]进行了系统优化,分别在NF和玻碳(GC)电极上制备了非晶态CoFeBO催化剂,并详细讨论了两种不同基底合成的催化剂在OER催化性能的差异以及不同Fe掺杂量对OER催化活性的影响。以GC为基底,Fe含量为0.3时,合成的非晶态CoFeBO催化OER活性最佳,且显著优于标准商业RuO2。经过28 h的稳定性测试显示,该催化剂的电位无明显波动。这是由于非晶态材料拥有丰富的配位不饱和结构,以及B的存在加强了Co、Fe间的热力学稳定效应,因此催化活性和稳定性显著增强。

图5 非晶态Ni(OH)2修饰贵金属Pt示意图[61]
Fig. 5 Schematic diagram of precious metal Pt modified by amorphous Ni(OH)2[61]

图6 Co-Fe-Bi的SEM和HR-TEM像、Co-Fe-Bi的EDS谱和Co-Fe-Bi的相对ECSA及LSV曲线图[64]
Fig. 6 SEM and HR-TEM images of Co-Fe-Bi((a)-(c)), EDS images of Co-Fe-Bi((d1)-(d4)), ECSA(e) and LSV(f) curves of Co-Fe-Bi[64]
3 结论
综上所述,非晶态催化剂凭借其独特的性质在电催化水分解反应中取得了显著成效,其中,非晶态硫化物和磷化物对氢气析出具有更优异的活性,因此被广泛用作阴极析氢反应;非晶态氧化物催化剂由于本身的P型电子结构,使其在碱性条件下用于OER反应时具有较低的过电位;氢氧化物作为一类亲水层状材料,具有较大的比表面积,在电解水反应时易使H2O中H―OH断裂,但由于受到其电导率和吸附H脱附等因素的限制,在碱性条件下催化HER活性较差,所以也多用于阳极OER研究。近年来,许多科研工作者不再局限于研究单一的催化HER和OER的非晶态催化剂,而是通过优化催化剂的合成方法并通过调控表面几何结构和电子结构,从而制备高效、稳定的双功能催化剂。
虽然关于双功能非晶态催化剂的设计合成已取得一定进步,但仍有许多方面需要探究完善:1) 当前关于非晶态催化剂的活性位结构仍存在一定的争议,开展非晶态催化剂的反应机理研究,通过现代原位表征谱学技术结合理论分析深入探究各催化剂催化HER和OER机理非常有必要;2) 精确控制双功能非晶态催化剂表面组分,通过多元素掺杂和复配,调控其结构形貌,优化电子结构及表面几何结构,确定最佳适配关系,以此获得具有多活性位的稳定非晶态催化剂;3) 完善和改进现有制备非晶态催化剂的方法,同时探索合成过程中不同制备方法的组合和交叉结合,形成多级结构的活性中心,从而使非晶态催化剂在全电解水催化性能中具有更丰富的可调性。可以相信,随着对非晶态催化剂活性位结构的深入理解,人们可以精确控制活性位并有效增加活性位密度,从而推进非晶态催化剂催化水分解制氢的实际应用。
REFERENCES
[1] ACAR C, BICER Y, DEMIR M E, et al. Transition to a new era with light-based hydrogen production for a carbon-free society: An overview[J]. International Journal of Hydrogen Energy, 2019, 44(47): 25347-25364.
[2] HAN K H, GAO J, QI J H. The study of sulphur retention characteristics of biomass briquettes during combustion[J]. Energy, 2019, 186: 115788.
[3] 熊 昆, 高 媛, 周桂林. 电解水析氢非铂催化剂的设计与发展[J]. 中国有色金属学报, 2017, 27(6): 1289-1301.
XIONG Kong, GAO Yuan, ZHOU Gui-lin. Design and development of non-Pt catalysts in water electrolysis for hydrogen production[J]. The Chinese Journal of Nonferrous Metals, 2017, 27(6): 1289-1301.
[4] LI J J, ZHOU J F, ZHAO X Y, et al. Electrochemical hydrogen storage properties of non-equilibrium Ti2-xMgxNi alloys[J]. Transactions of Nonferrous Metals Society of China, 2015, 25(11): 3729-3735.
[5] 王彦欣, 黄林军, 唐建国, 等. 石墨烯/银纳米复合膜对非晶态Mg-Ni-La贮氢合金的表面改性及机理[J]. 中国有色金属学报, 2018, 28(3): 509-517.
WANG Yan-xin, HUANG Lin-jun, TANG Jian-guo, et al. Surface modification and mechanism research of amorphous Mg-Ni-La hydrogen storage alloy with graphene/Ag nanocomposite[J]. The Chinese Journal of Nonferrous Metals, 2018, 28(3): 509-517.
[6] LIU K H, ZHONG H X, LI S J, et al. Advanced catalysts for sustainable hydrogen generation and storage via hydrogen evolution and carbon dioxide/nitrogen reduction reactions[J]. Progress in Materials Science, 2018, 92: 64-111.
[7] ZHANG K, BAO W R, CHANG L P, et al. A review of recent researches on Bunsen reaction for hydrogen production via S-I water and H2S splitting cycles[J]. Journal of Energy Chemistry, 2019, 33: 46-58.
[8] ZHU W J, ZHU G X, YAO C G, et al. Porous amorphous FeCo alloys as pre-catalysts for promoting the oxygen evolution reaction[J]. Journal of Alloys and Compounds, 2020, 828: 154465.
[9] XIONG K, HUANG L P, GAO Y, et al. Formation of a thin-layer of nickel hydroxide on nickel phosphide nanopillars for hydrogen evolution[J]. Electrochemistry Communications, 2018, 92: 9-13.
[10] 郭亚肖, 商昌帅, 李 敬, 等. 电催化析氢、析氧及氧还原的研究进展[J]. 中国科学: 化学, 2018, 48(8): 926-940.
GUO Ya-xiao, SHANG Chang-shuai, LI Jing, et al. Recent development of hydrogen evolution, oxygen evolution and oxygen reduction reaction[J]. Scientia Sinica Chimica, 2018, 48(8): 926-940.
[11] CAI W Z, CHEN R, YANG H B, et al. Amorphous versus crystalline in water oxidation catalysis: A case study of NiFe alloy[J]. Nano Letters, 2020, 20(6): 4278-4285.
[12] SMITH G V, BROWER W E, MATYJASZCZYK M S, et al. Metallic glasses: New catalyst systems[J]. Studies in Surface Science and Catalysis, 1981, 7(Part A): 355-363.
[13] ANANTHARAJ S, NODA S. Amorphous catalysts and electrochemical water splitting: An untold story of harmony[J]. Small, 2020, 16(2): 1905779.
[14] ZONG B N, MU X H, ZHANG X X, et al. Research, development, and application of amorphous nickel alloy catalysts prepared by melt-quenching[J]. Chinese Journal of Catalysis, 2013, 34(5): 828-837.
[15] 张家华, 蒋丽红, 伍水生, 等. 非晶态镍硼/石墨烯复合材料的制备及其蒎烯催化加氢活性[J]. 化工学报, 2016, 67(6): 2363-2370.
ZHANG Jia-hua, JIANG Li-hong, WU Shui-sheng, et al. Preparation of amorphous Ni-B/graphene composites for catalytic hydrogenation of pinene[J]. Journal of Chemical Industry and Engineering (China), 2016, 67(6): 2363-2370.
[16] JIANG R, CUI Z, XU W, et al. Highly efficient amorphous np-PdFePC catalyst for hydrogen evolution reaction[J]. Electrochimica Acta, 2019, 328: 135082.
[17] HUI T L, MIAO C G, FENG J T, et al. Atmosphere induced amorphous and permeable carbon layer encapsulating PtGa catalyst for selective cinnamaldehyde hydrogenation[J]. Journal of Catalysis, 2020, 389: 229-240.
[18] 魏 磊, 马麦霞, 王东升, 等. 非晶态NiB纳米催化剂的制备及其对对硝基苯酚加氢的催化活性[J]. 中国有色金属学报, 2019, 29(5): 1118-1124.
WEI Lei, MA Mai-xia, WANG Dong-sheng, et al. Preparation of amorphous NiB nanocatalysts and its catalytic activity for hydrogenation of 4-nitrophenol[J]. The Chinese Journal of Nonferrous Metals, 2019, 29(5): 1118-1124.
[19] 黄林军, 王彦欣, 唐建国. 非晶态Mg-Ni-La贮氢合金电化学吸释氢过程中的贮氢机制[J]. 中国有色金属学报, 2014, 24(12): 3059-3065.
WANG Lin-jun, WANG Yan-xin, TANG Jian-guo. Hydrogen-storage mechanism of amorphous Mg-Ni-La hydrogen-storage alloys in electrochemical absorbtion/ desorbtion process[J]. The Chinese Journal of Nonferrous Metals, 2014, 24(12): 3059-3065.
[20] SMITH R D L, SPORINOVA B, FAGAN R D, et al. Facile photochemical preparation of amorphous iridium oxide films for water oxidation catalysis[J]. Chemistry of Materials, 2014, 26(4): 1654-1659.
[21] SALIMI P, NAJAFPOUR M M. A simple method for synthesizing highly active amorphous iridium oxide for water-oxidation under acidic conditions[J]. Chemistry―A European Journal, 2020. https://doi.org/10.1002/chem. 202000955.
[22] TSUJI E, IMANISHI A, FUKUI K, et al. Electrocatalytic activity of amorphous RuO2 electrode for oxygen evolution in an aqueous solution[J]. Electrochimica Acta, 2011, 56(5): 2009-2016.
[23] SUN W, MA C L, TIAN X L, et al. Amorphous lanthanum-Iridium solid solution with open structure for efficient water splitting[J]. Journal of Materials Chemistry A, 2020, 8: 12518-12525.
[24] DONG Z H, LIN F, YAO Y H, et al. Crystalline Ni(OH)2/amorphous NiMoOx mixed-catalyst with Pt-like performance for hydrogen production[J]. Advanced Energy Materials, 2019, 9(46): 1902703.
[25] MORALESGUIO C G, HU X. Amorphous molybdenum sulfides as hydrogen evolution catalysts[J]. Accounts of Chemical Research, 2014, 47(8): 2671-2681.
[26] SUN Y, LIU C, GRAUER D C, et al. Electrodeposited cobalt-sufide catalyst for electrochemical and photoelectrochemlcal hydrogen generation from water[J]. Journal of the American Chemical Society, 2013, 135(47): 17699-17702.
[27] Minouei H, Akbari G H, Enayati M H, et al. Non-isothermal nano-crystallization kinetics in amorphous Ni55Nb35Si10 alloy[J]. Transactions of Nonferrous Metals Society of China, 2019, 29(2): 358-364.
[28] KORNIENKO N, RESASCO J, BECKNELL N, et al. Operando spectroscopic analysis of an amorphous cobalt sulfide hydrogen evolution electrocatalyst[J]. Journal of the American Chemical Society, 2015, 137(23): 7448-7455.
[29] GUO X, LIANG G. Designed formation of CoS2 nanoboxes with enhanced oxygen evolution reaction electrocatalytic properties[J]. International Journal of Hydrogen Energy, 2019, 44(59): 32020-31028.
[30] LIU Y P, LI Q J, SI R, et al. Coupling sub-nanometric copper clusters with quasi-amorphous cobalt sulfide yields efficient and robust electrocatalysts for water splitting reaction[J]. Advanced Materials, 2017, 29(13): 1606200.
[31] CAI P W, HUANG J H, CHEN J X, et al. Oxygen-containing amorphous cobalt sulfide porous nanocubes as high-activity electrocatalysts for the oxygen evolution reaction in an alkaline/neutral medium[J]. Angewandte Chemie― International Edition, 2017, 129(17): 4936-4939.
[32] KONG D, CHA J J, WANG H T, et al. First-row transition metal dichalcogenide catalysts for hydrogen evolution reaction[J]. Energy & Environmental Science, 2013, 6: 3553-3558.
[33] KIBSGAARD J, CHEN Z, REINECKE B N, et al. Engineering the surface structure of MoS2 to preferentially expose active edge sites for electrocatalysis[J]. Nature Materials, 2012, 11: 963-969.
[34] 陶 锴, 徐一锋, 刘继宁, 等. 二硫化钼基电极材料空间限域法制备研究进展[J]. 硅酸盐学报, 2020, 48(7): 1081-1089.
TAO Kai, XU Yi-feng, LIU Ji-ning, et al. Research progress on space-confined synthesis of molybdenum disulfide[J]. Journal of the Chinese Ceramic Society, 2020, 48(7): 1081-1089.
[35] YU H, YUAN R, GAO D. Ethyl acetate-induced formation of amorphous MoSx nanoclusters for improved H2-evolution activity of TiO2 photocatalyst[J]. Chemical Engineering Journal, 2019, 375: 121934.
[36] SONG S, WANG Y, LI W, et al. Amorphous MoS2 coated Ni3S2 nanosheets as bifunctional electrocatalysts for high-efficiency overall water splitting[J]. Electrochimica Acta, 2020, 332: 135454.
[37] YANG L, WU X L, ZHU X S, et al. Amorphous nickel/cobalt tungsten sulfide electrocatalysts for high-efficiency hydrogen evolution reaction[J]. Applied Surface Science, 2015, 341: 149-156.
[38] LIU P, RODRIGUEZ J A. Catalysts for hydrogen evolution from the [NiFe] hydrogenase to the Ni2P(001) surface: The importance of ensemble effect[J]. Journal of the American Chemical Society, 2005, 127(42): 14871-14878.
[39] POPCZUN E J, MCKONE J R, READ C G, et al. Nanostructured nickel phosphide as an electrocatalyst for the hydrogen evolution reaction[J]. Journal of the American Chemical Society, 2013, 135(25): 9267-9270.
[40] MCENANEY J M, CROMPTON J C, CALLEJAS J F, et al. Amorphous molybdenum phosphide nanoparticles for electrocatalytic hydrogen evolution[J]. Chemistry of Materials, 2014, 26(16): 4826-4831.
[41] MOON J S, JANG J H, KIM E G, et al. The nature of active sites of Ni2P electrocatalyst for hydrogen evolution reaction[J]. Journal of Catalysis, 2015, 326: 92-99.
[42] XU W, WANG H X. Earth-abundant amorphous catalysts for electrolysis of water[J]. Chinese Journal of Catalysis, 2017, 38(6): 991-1005.
[43] 黄丽萍, 周 琳, 高 媛, 等. 双功能过渡金属化合物的构建与应用[J]. 功能材料, 2018, 49(11): 11005-11013.
HUANG Li-ping, ZHOU Lin, GAO Yuan, et al. Construction and application of bifunctional transition metal compounds[J]. Journal of Functional Materials, 2018, 49(11): 11005-11013.
[44] LI C, MEI X H, LAM F L Y, et al. Hybridizing amorphous nickel cobalt phosphate and nickel phosphide as an efficient bifunctional nanocatalyst towards overall water splitting[J]. Catalysis Today, 2020, 358(1): 215-220.
[45] ZHANG W Q, LI Y, ZHOU L X, et al. Ultrathin amorphous CoFeP nanosheets derived from CoFe LDHs by partial phosphating as excellent bifunctional catalysts for overall water splitting[J]. Electrochimica Acta, 2019, 323: 134595.
[46] WANG T Y, WANG C, JIN Y, et al. Amorphous Co-Fe-P nanospheres for efficient water oxidation[J]. Journal of Materials Chemistry A, 2017, 5(48): 25378-25384.
[47] ZHOU M, SUN Q Q, SHEN Y Q, et al. Fabrication of 3D microporous amorphous metallic phosphides for high- efficiency hydrogen evolution reaction[J]. Electrochimica Acta, 2019, 306: 651-659.
[48] XIONG K, GAO Y, CHEN J, et al. Ordered porous Ni in situ decorated by thin-layer amorphous nickel-phosphorus via mild electrochemical-phosphorization for enhancing the hydrogen evolution performance[J]. Chemical Communications, 2020, 56(4): 611-614.
[49] LI N, XIA W Y, WANG J, et al. Manganese oxides supported on hydrogenated TiO2 nanowire arrays catalysts for electrochemical oxygen evolution reaction in water electrolysis[J]. Journal of Materials Chemistry A, 2015, 3: 21308-21313.
[50] ZHAO C T, YU C, HUANG H W, et al. Co ion-intercalation amorphous and ultrathin microstructure for high-rate oxygen evolution[J]. Energy Storage Materials, 2018, 10: 291-296.
[51] KOZA J A, HE Z, MILLER A S, et al. Electrodeposition of crystalline Co3O4―A catalyst for the oxygen evolution reaction[J]. Chemistry of Materials, 2012, 24(18): 3567-3573.
[52] CHEN D, DONG C L, ZOU Y Q, et al. In situ evolution of highly dispersed amorphous CoOx clusters for oxygen evolution reaction[J]. Nanoscale, 2017, 9(33): 11969-11975.
[53] CHEN C W, CHIANG C Y. Molybdenum-containing amorphous metal oxide catalysts for oxygen evolution reaction[J]. International Journal of Hydrogen Energy, 2017, 42(50): 29773-29780.
[54] YOON S, YUN J Y, LIM J H, et al. Enhanced electrocatalytic properties of electrodeposited amorphous cobalt-nickel hydroxide nanosheets on nickel foam by the formation of nickel nanocones for the oxygen evolution reaction[J]. Journal of Alloys and Compounds, 2017, 693: 964-969.
[55] LI H B, YU M H, WANG F X, et al. Amorphous nickel hydroxide nanospheres with ultrahigh capacitance and energy density as electrochemical pseudocapacitor materials[J]. Nature Communications, 2013, 4: 1894.
[56] GAO Y Q, LI H B, YANG G W. Amorphous Co(OH)2 nanosheet electrocatalyst and the physical mechanism for its high activity and long-term cycle stability[J]. Journal of Applied Physics, 2016, 119(3): 034902.
[57] GAO Y Q, LI H B, YANG G W. Amorphous nickel hydroxide nanosheets with ultrahigh activity and super-long-term cycle stability as advanced water oxidation catalysts[J]. Crystal Growth & Design, 2015, 15(9): 4475-4483.
[58] SHAO B, PANG W, TAN X Q, et al. Rapid growth of amorphous cobalt-iron oxyhydroxide nanosheet arrays onto iron foam: Highly efficient and low-cost catalysts for oxygen evolution[J]. Journal of Electroanalytical Chemistry, 2020, 856: 113621.
[59] ZOU X, LIU Y P, LI G D, et al. Ultrafast formation of amorphous bimetallic hydroxide films on 3D conductive sulfide nanoarrays for large-current-density oxygen evolution electrocatalysis[J]. Advanced Materials, 2017, 29(22): 1700404.
[60] XU Q L, GAO W L, WANG M, et al. Electrodeposition of NiS/Ni2P nanoparticles embedded in amorphous Ni(OH)2 nanosheets as an efficient and durable dual-functional electrocatalyst for overall water splitting[J]. International Journal of Hydrogen Energy, 2020, 45(4): 2546-2556.
[61] YUAN G, WEN B Y, HU Y J, et al. Aqueous substitution synthesis of platinum modified amorphous nickel hydroxide on nickel foam composite electrode for efficient and stable hydrogen evolution[J]. International Journal of Hydrogen Energy, 2019, 44(28): 14258-14265.
[62] KANAN M W, NOCERA D G. In situ formation of an oxygen-evolving catalyst in neutral water containing phosphate and Co2+[J]. Science, 2008, 321(5892): 1072-1075.
[63] GUPTA S, PATEL N, FERNANDES R, et al. Co-Ni-B nanocatalyst for efficient hydrogen evolution reaction in wide pH range[J]. Applied Catalysis B: Environmental, 2016, 192: 126-133.
[64] SURYAWANSHI U P, SURYAWANSHI M P, GHORPADE U V, et al. An earth-abundant, amorphous cobalt-iron-borate (Co-Fe-Bi) prepared on Ni foam as highly efficient and durable electrocatalysts for oxygen evolution[J]. Applied Surface Science, 2019, 495: 143462.
[65] LIU G, HE D Y, YAO R, et al. Amorphous CoFeBO nanoparticles as highly active electrocatalysts for efficient water oxidation reaction[J]. International Journal of Hydrogen Energy, 2018, 43(12): 6138-6149.
[66] THEERTHAGIRI J, LEE S J, MURTHY A P, et al. Fundamental aspects and recent advances in transition metal nitrides as electrocatalysts for hydrogen evolution reaction: A review[J]. Current Opinion in Solid State & Materials Science, 2020, 24(1): 100805.
[67] ZOU H Y, LI G, DUAN L L, et al. In situ coupled amorphous cobalt nitride with nitrogen-doped graphene aerogel as a trifunctional electrocatalyst towards Zn-air battery deriven full water splitting[J]. Applied Catalysis B: Environmental, 2019, 259: 118100.
YU Lin-jian, XIONG Kun, CHEN Jia, ZHANG Hai-dong, XIANG Yang, XU Jing-fang
(Engineering Research Center for Waste Oil Recovery Technology and Equipment, Ministry of Education, College of Environment and Resources, Chongqing Technology and Business University, Chongqing 400067, China)
Abstract: Amorphous catalyst as a new type of catalytic material has the characteristics of short-range order and long-range disorder. Compared with the crystalline catalyst, the amorphous catalyst has larger specific surface area and structural defects under the same mass, which makes it more active sites for the catalytic reaction. In addition, the excellent flexibility and flexibility of the amorphous catalyst can induce the active sites to form suitable morphology for promoting the reaction rate due to its uniformity and isotropy, which can adjust their catalytic conditions and provide volume and capacity according to the surface electron transfer, promoting the electrocatalytic activities of hydrogen evolution reaction (HER) and oxygen evolution reaction (OER) in water splitting. Herein, the latest progress in several major types of the amorphous catalysts for electrocatalytic water splitting in recent years was reviewed, especially in the controllable synthesis of several kinds of amorphous catalysts, the control strategy on electrocatalytic performance for water decomposition, and the structure-activity relationship between surface electronic structure and catalytic activity and stability. Moreover, the prospects for the challenges and research direction of the adaptation relationship and modulation of the active site of the catalysts were proposed to shed light on further development of electrolysis water.
Key words: amorphous catalyst; surface/interface; water electrolysis; hydrogen evolution reaction; oxygen evolution reaction
Foundation item: Projects(22078032, 21606028) supported by the National Natural Science Foundation of China; Project(cstc2020jcyj-msxmX0345) supported by the Natural Science Foundation of Chongqing, China; Projects(KJQN201800819, KJZD-K201900805, yds183007) supported by the Science and Technology Research Program of Chongqing Municipal Education Commission, China; Project(CXQT19023) supported by the Innovation Group of Chongqing Municipal Education Commission, China
Received date: 2020-08-04; Accepted date: 2020-11-23
Corresponding author: XIONG Kun; Tel: +86-23-62768317; E-mail: kunxiong@ctbu.edu.cn
(编辑 何学锋)
基金项目:国家自然科学基金资助项目(22078032,21606028);重庆市自然科学基金资助项目(cstc2020jcyj-msxmX0345);重庆市教委资助项目(KJQN201800819,KJZD-K201900805,yds183007);重庆市高校创新群体资助项目(CXQT19023)
收稿日期:2020-08-04;修订日期:2020-11-23
通信作者:熊 昆,副研究员,博士;电话:023-62768317;E-mail:kunxiong@ctbu.edu.cn

