J. Cent. South Univ. Technol. (2009) 16: 0066-0072
DOI: 10.1007/s11771-009-0011-9
Synthesis and property of temperature and pH sensitive xanthan–MA/PNIPAAm hydrogels
LONG Qing-de(隆清德), PAN Chun-yue(潘春跃), MENG Yan-hua(孟艳华),
ZHANG Bao-jin(张报进), XU Cheng-xi(许晨曦)
(School of Chemistry and Chemical Engineering, Central South University, Changsha 410083, China)
Abstract: A novel class of xanthan–maleic anhydride (Xan-MA)/poly(N-isopropylacrylamide) hybrid hydrogels was designed and synthesized by solution polymerization. The xanthan-based precursor (Xan-MA) was prepared by substituting the hydroxyl groups in Xan by MA. This Xan-MA precursor was then polymerized with a known temperature sensitive precursor (N-isopropylacrylamide, NIPAAm) to form hybrid hydrogels with a series range of composition ratio of Xan-MA to NIPAAm precursors. These smart hydrogels were characterized by Fourier transform infrared spectroscopy for structural determination, differential scanning calorimertry for thermal property. And maximum swelling ratio, swelling kinetics and temperature response kinetics were studied. The data obtained clearly show that these smart hydrogels are responsive to the external changes of temperature as well as pH value. The magnitudes of smart and hydrogel properties of these hybrid hydrogels depend on the feed composition ratio of the two precursors. With the increase of the content of Xan-MA the maximum swelling ratio, reswelling ratio and thermo-sensitivities increase, and the feed composition ratio of Xan-MA/NIPAAm increases the maximum swelling ratio augment from 13.88 to 23.21. From XMN0, XMN1, XMN3 to XMN5, the lower critical solution temperatures (LCSTs) are 33.02, 36.15, 40.28 and 41.92 ℃, respectively. By changing the composition ratio of these two precursors, the LCST of the hybrid hydrogels could also be adjusted to be or near the body temperature for the potential applications in bioengineering and biotechnology fields.
Key words: hydrogel; N-isopropylacrylamide; polysaccharide; temperature; pH
1 Introduction
Smart hydrogels that respond and change their shape and volume upon external stimuli, such as pH[1], temperature[2], photo field[3] and antigen[4], are very attractive materials for application in biomaterial science and technology. Recently, hydrogels, especially the temperature sensitive ones, have attracted extensive interest due to their potential and promising applications in many fields, such as protein-ligand recognition[5], on-off switches for modulated drug delivery[6] or artificial organs[7], and immobilization of enzyme[8]. poly(N-isopropylacrylamide) (PNIPAAm) hydrogel, a classical temperature intelligent hydrogel, is the most widely studied. It exhibits a lower critical solution temperature (LCST) of 33 ℃ in aqueous solution and shows an abrupt thermoreversible change in volume as external temperature changes[9-10]. An important parameter to consider in the design of these systems is the degree of swelling. Not only is the delivery of drugs accomplished by the swelling characteristics of hydrogels, but the release of drugs is also relative to the swelling behavior of hydrogels[11]. So much attention has been paid to study on the swelling behavior of hydrogels.
Xanthan (Xan), a microbial biopolymer produced by the Xanthomonas campestris, seems potentially useful for such purpose. It consists of D-glucose, D-mannose, D-glucuronic acid acetal linked pyruvic acid and O-acetyl; later, the currently accepted structure is identified. The acetate and pyruvate contents appear to vary due to the culture conditions and the post-fermentation processing as well. Some properties of the Xan depend on those contents[12-14]. In medical and pharmaceutical applications, Xan is used as a component in hydrogels[15-18]. Xan can form both physical and chemical gels. Typical physical gels formed simply from polysaccharide concentrated solutions are unstable; physical gelation process of Xan is usually reversible (sol-gel transition). By heating or dilution, Xan physical gels can easily be solubilized in aqueous solution, which limits their application to the drug controlled release carrier.
In order to obtain a stabile chemical hydrogel in drug delivery system, the Xan modification by esterification with unsaturated maleic anhydride was performed. Xan derivatives carrying reactive double bonds for subsequent grafting and crosslinking to form hydrogels were synthesized. Then temperature and pH sensitive parts were imported to the Xan-base polysaccharide with unsaturated binding functionalities. The Xan-MA/PNIPAAm hydrogels synthesized were characterized by Fourier transform infrared spectroscopy (FT-IR) and differential scanning calorimertry (DSC). The equilibrium swelling ratio, swelling kinetics, deswelling kinetics, and swelling ratio in buffer solutions of different pH values were also investigated.
2 Experimental
2.1 Materials
Xanthan (medium viscosity 1.0-1.2 Pa?s for 1% aqueous solution) was obtained from Deosen Co. (Shandong, China) and dried in a vacuum oven at 60 ℃ for 2 d. N-isopropylacrylamide (NIPAAm) was purchased from Tokyo Chemical Industry Co., Ltd. and recrystallized from hexane before used. Maleic acid, dimethyl formamide(DMF), N, N′-methylenebisacrylamide (BIS), ammonium persulfate(APS), N,N,N′,N′- tetramethyl-ethylenediamine (TEMED), acetone, ethanol were used without any purification.
2.2 Preparation of xanthan-based precursor (Xan-MA)
1.0 g Xan was introduced into 15 mL dimethyl formamide (DMF) at room temperature. The resulting suspension was stirred for 12 h so that the polysaccharide particles swelled well in solvent. MA (n(MA): n(monomole Xan)=11?1) was dissolved in other 10 mL DMF and then added dropwise to the system at 70 ℃. The reaction under magnetic stirring was conducted for 24 h. Then at a very slow rate, adding 30 mL acetone, the ropy product swelled and was agglomerated. After 120 mL acetone was added to precipitate xanthan-based precursor, and the mixture was separated by filtration, dissolved in water (50 mL) under stirring for 6 h, and precipitated in ethanol (200 mL). After 2 h, the upper layer clear solution was poured out. The remainder precipitate was filtered. The purification procedure was repeated two times. The product was dried at room temperature, first at normal pressure and then dried in a vacuum oven at room temperature for 24 h. The degree of substitution of Xan-MA precursor was 10.8% (Double bond per repeating unit was 1.18), which was estimated by HAMCERENCU et al[19].
2.3 Preparation of hydrogels
Different mass ratios of Xan-MA/NIPAAm and N,N′-methylenebisacrylamide(BIS) solutions were dissolved in distilled water. The feed compositions of these two precursors and other reactants are listed in Table 1, where XMN denotes Xan-MA/PNIPAAm hydroges. Nitrogen was bubbled through the monomer/solvent mixture at 15 ℃ for 20 min to remove oxygen dissolved in the reaction mixture. Then, 0.5 mL 2%(mass fraction) ammonium persulfate(APS) was incorporated. 40 ?L N,N,N′,N′-tetramethylethylenediamine (TEMED) was added as an accelerator, and then the solution was stirred for 3 min. The hydrogel samples were further purified with distilled water at room temperature for at least 48 h, and the distilled water was also replaced every several hours to let the purified hydrogels reach equilibrium state for the following characterization.
Table 1 Feed compositions of novel temperature and pH sensitive hydrogels
2.4 FT-IR analysis of Xan-MA/PNIPAAm hydrogels
The FT-IR analysis was performed by using Nicolet Fourier transform infrared spectroscopy (made by Thermo Nicolet), over the range of 500-4 000 cm-1. Prior to the measurement, the hydrogel samples were dried under vacuum till constant mass. The dried hydrogel was pressed into powder, mixed with 10 times as much as KBr powder and then compressed into a pellet for FT-IR characterization.
2.5 Thermo-behavior of XAN-MA/PNIPAAm hydrogels
The LCST property of the hydrogel samples was determined by using DSC (Q10 Modulated DSC, TA instruments, USA). All samples were immersed in distilled water at 48 ℃ for at least 2 d to reach a equilibrium state. About 10 mg swollen sample was placed inside a hermetic aluminum pan, and then sealed tightly by a hermitic aluminum lid. The thermal analyses were performed from -40 ℃ to 100 ℃ under nitrogen atmosphere at a flow rate of 50 mL/min and a heating rate of 10 ℃/min.
2.6 Equilibrium swelling ratio of Xan-MA/ PNIPAAm hydrogels
The room temperature swollen hydrogel samples were put into deionized water for 24 h at different temperatures to reach swelling balance. The classical gravimetric method was used to measure their swelling ratios. Swollen hydrogels were removed from test conditions, then gently wiped off the water on the hydrogel surfaces with moistened filter papers, and the mass was determined. And the equilibrium swelling ratio (Se) is defined as follows:
Se=(me-md)/md (1)
where me is the mass of hydrogel of equilibrium swollen hydrogel, and md is the dried mass of hydrogel.
2.7 Swelling kinetics of Xan-MA/PNIPAAm hydrogels at room temperature
The swelling kinetics of the dried hydrogels was measured gravimetrically at 22 ℃ over predetermined periods. The sample was taken out from 22 ℃ water at regular intervals. After wiping off the water on the hydrogel surfaces with moistened filter papers, the mass of hydrogel was recorded. The swelling ratio (S) is defined as follows:
S=(mt-md)/md (2)
where mt is the mass of the wet hydrogel at time t.
2.8 Deswelling kinetics of Xan-MA/PNIPAAm hydrogels
The deswelling kinetics of the hydrogels was measured by transferring swollen hydrogels of room temperature (after 24 h swelling) to the distilled water bath (48 ℃) for predetermined durations. This temperature (48 ℃) was selected because it is well above the LCST of the hydrogels, so that dramatic changes in volume or water content could be attained within a short intervals. The mass changes of hydrogels at different intervals after transferring to 48 ℃ were measured with the gravimetric method described above. The water retention (R) is defined as follows:
R=[(mt-md)/(me-md)]×100% (3)
2.9 pH sensitivity of Xan-MA/PNIPAAm hydrogels
Dried fresh hydrogels after synthesis were immersed in buffer solutions of different pH values for 24 h to reach the swelling equilibrium. The soaked hydrogels were removed, washed, the surface water was wiped by a wet filter paper, and weighed until stable mass was detected. Swelling ratio of hydrogel was measured as above.
3 Results and discussion
3.1 Synthesis of Xan-MA
The Xan modification by esterification with the unsaturated MA was performed, in view of establishing the optimal reaction conditions. The esterification proceeds predominantly to the primary hydroxyl groups (C6), although participation of some secondary ones was not wholly excluded. Preliminary trials of synthesizing Xan and maleates led to the conclusion that a considerable excess of esterification agent was necessary for attaining acceptable transformation degrees. In the experiments, a molar ratio of functionalization agent to Xan (11/1) was generally used corresponding to an equimolar ratio of reactive sites.
3.2 FT-IR spectra of Xan-MA/PNIPAAm hydrogels
The FT-IR absorption spectra of the Xan-MA/ PNIPAAm hydrogel samples are shown in Fig.1. From Fig.1, it can be seen that IR spectra of all samples are similar, although they differ slightly among each other due to different composition ratios of Xan-MA to NIPAAm precursor. The intensities of peak at about 1 722 cm-1 indicate the esterification with unsaturated maleic anhydride.

Fig.1 FT-IR spectra of Xan-MA/PNIPAAm hydrogels
Each spectrum shows a broad band in the range of 3 700-3 100 cm-1, which belongs to O—H (from Xan-MA) and N—H (from PNIPAAm) stretching vibrations. The typical amide I and II bands in PNIPAAm are evident at about 1 644 and about 1 534 cm-1. These findings indicate that both Xan-MA and PNIPAAm components exist in Xan-MA/PNIPAAm hydrogels.
3.3 LCST of Xan-MA/PNIPAAm hybrid hydrogels
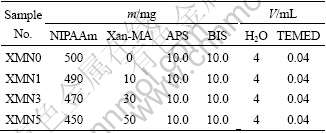
The LCSTs of the Xan-MA/PNIPAAm hydrogels determined from their DSC thermograms are shown in Fig.2. The data indicate that all XMNs have a higher LCST than the pure PNIPAAm hydrogel(XMN0) at 33 ℃[10].
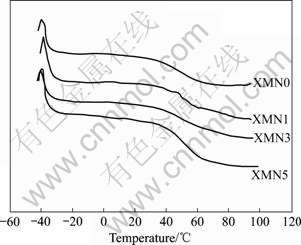
Fig.2 DSC thermograms of hybrid hydrogel as a function of mass ratio of Xan-MA to PNIPAAm
The increase in the LCSTs from 100% PNIPAAm, XMN1, XMN3 to XMN5 is believed to be attributed to the increased hydrophilic Xan-MA. The possible explanation may be that an incorporation of Xan-MA into PNIPAAm would increase the hydrophilicity of the whole hydrogel network due to the hydrophilic groups of Xan-MA; the hindrance of hydrophilic interactions to the shrinking of PNIPAAm results from intermolecular hydrogen bonding between hydrophilic groups of Xan-MA, PNIPAAm and water molecules. The more the Xan-MA content is, the stronger the interactions and inhibition are, leading to the higher LCST with smooth changes. This result is in agreement with the relationship derived from generally main mechanism for the phase separation of thermo-sensitive hydrogels that the hydrophilic moiety incorporated into PNIPAAm will enhance its LCST.
From the application point, a higher LCST of thermo-sensitive hydrogels is important since the LCST of the pure PNIPAAm hydrogel is lower than the body temperature. Thermo-sensitive hydrogels with LCSTs near the body temperature would permit us to use this intelligent property for the biomedical applications.
3.4 Equilibrium swelling ratio of Xan-MA/PNIPAAm hydrogels
The temperature-dependence of the swelling ratios of XMN was investigated. Fig.3 shows the typical temperature-dependent swelling ratio of the four hydrogels over the temperature range of 22-48 ℃ in deionized water. The equilibrium swelling ratios of those hybrid hydrogels exhibit similar temperature dependence. As the temperature increases, the swelling ratio decreases, with the most drastic decrease being observed at around the LCSTs of PNIPAAm series hydrogels. Generally, the main reason for this distinctive characteristic of the hydrogels can be attributed to the unique and rapid alternation of the hydrophilic and hydrophobic states. When the temperature is below the LCST, the hydrophilic groups of the hydrogels form hydrogen bonds with water molecules. These bonds act cooperatively to form a stable shell of hydration around the hydrophobic groups, resulting in greater water uptake and producing a larger swelling ratio. As the external temperature increases, the interactions among the hydrophobic groups rise and the entrapped water molecules from the hydrogel networks release. At 22 ℃, the swelling ratios of those hydrogels are about 13.88(for XMN0), 14.37(for XMN1), 18.71(for XMN3) and 23.21 (for XMN5), respectively. However, when the ambient temperature exceeds the LCST of the hydrogels, the swelling ratio decreases to about 2.83, 1.65, 1.06 and 1.51, respectively. The difference in swelling ratio may be attributed to the different contents of Xan-MA in the hydrogels. The interactions among the hydrophobic groups increase when the temperature increases, while the hydrophilic ability in the Xan-MA moiety can still lead to some water retention and this trend becomes stronger with the increase of Xan-MA content. So the LCST of the hydrogels gets higher. When the temperature exceeds the LCST, hydrogels collapse abruptly and the hydrophobic groups aggregate together. Thus, XMN3 and XMN5 with a higher Xan-MA content can maintain a larger swelling ratio below the LCST. The above results indicate that the thermo-sensitive properties of the hydrogels are improved mainly from PNIPAAm, and the copolymer composition can be used to control the swelling ratio of the NIPAAm-based hydrogels.

Fig.3 Equilibrium swelling ratio of Xan-MA/PNIPAAm hydrogels in deionized water
3.5 Swelling kinetics of Xan-MA/PNIPAAm hydrogels at room temperature
The swelling kinetics of Xan-MA/PNIPAAm hydrogels at room temperature was studied. Fig.4 shows the swelling ratios of XMN hydrogels as a function of the time of the dry hydrogels. As shown in Fig.4, with the increase of Xan-MA content in XMNs hydrogels, their reswelling rates from the dried state at room temperature are increased. This means the water penetrates into the three-dimensional structure hydrogel network faster if the network has higher Xan-MA component. This relationship may be attributed to a higher hydrophilicity of XMN hydrogels with a higher Xan-MA component, which exhibit a faster water uptake rate.

Fig.4 Swelling kinetic curves of Xan-MA/PNIPAAm hydrogels at 22 ℃
The swelling process is complicated and involves three successive steps: (1) the diffusion of water molecules into a polymer system; (2) the subsequent relaxation of hydrated polymer chains; (3) the expansion of polymer network into aqueous solution. The swelling ratio (S) will be proportional to the square root of time(t1/2) if the water diffusion is the rate-controlled step. However, S will be directly proportional to time, if the relaxation of polymer chains is the dominant step. Fig.5 shows the relationship between S and t1/2, which suggests that the swelling ratio of XMN hydrogels showing a line relationship swelling kinetic model and the slope of those lines becomes larger with the increase of Xan-MA’s content.
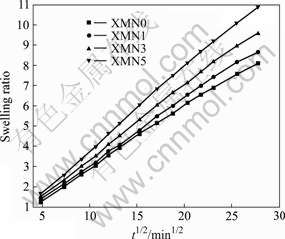
Fig.5 Swelling kinetic curves (S vs t1/2) of Xan-MA/PNIPAAm hydrogels at 22 ℃
3.6 Deswelling kinetics of Xan-MA/PNIPAAm hydrogels
Fig.6 shows the deswelling kinetic curves of Xan-MA/PNIPAAm hydrogels after the swollen hydrogel samples at 22 ℃ are quickly transferred to a hot water bath of 48 ℃ (>LCST). Fig.6 shows that the thermo-response capability depends on the mass ratio of Xan-MA to PNIPAAm precursors. Those hydrogels with higher Xan-MA exhibit stronger thermo-responsivity than pure PNIPAAm, irrespective of the dilution of the PNIPAAm component by Xan-MA.
Fig.6 Deswelling kinetic curves of Xan-MA/PNIPAAm hydrogels
A comparison of the response rates of XMN series (initial slope of the curves in Fig.6) with the pure PNIPAAm hydrogel indicates that XMN3 and XMN5 hybrid hydrogels show faster response rates; the response rate of XMN1, however, is only slightly faster than that of the pure PNIPAAm hydrogel, which exhibits no distinct difference between XMN1 and pure PNIPAAm hydrogels. The water retention almost gets to a constant mass for XMN series hydrogels when samples shrink for 18 h. At the end of the immersion period in hot water of 48 ℃ (1 500 min), pure PNIPAAm hydrogel and XMN1, XMN3, XMN5 samples shrink and lose water about 22.91%, 27.00%, 86.86%, and 94.28%, respectively. Among the XMN series, the thermo-response extent and rate of XMN1 hydrogel are the smallest and slowest, while those of XMN3 and XMN5 hydrogels are the largest and highest. From a theoretical point of view, an incorporation of non-thermo-responsive species such as Xan-MA into inherently thermo-responsive moiety such as PNIPAAm, the thermo-responsive property would be diluted and the level of dilution would depend on the amount of the non-thermo-responsive species incorporated. Based on this view, we should expect a reduction in the thermo-response extent and rate from pure PNIPAAm, XMN1 to XMN5 due to an increase in Xan-MA component. The data obtained in Fig.6, however, illustrate the opposite of that theoretical prediction.
It is well recognized that, during deswelling process of PNIPAAm hydrogel, the response rate of a thermo-responsive hydrogel also depends on the water-diffusing rate; and a faster response rate could be achieved, if entrapped water within a hydrogel could quickly release. Thus, in addition to the hydrophilic/ hydrophobic balance within the PNIPAAm segments, the characteristic of the dense skin layer formed during the collapse process of hydrogel network must also be considered as another possible factor that could influence the response rate[20]. Due to the poor water-permeability of this dense skin layer, the free water would accumulate underneath the hydrogel surface, which would lead to an increase in osmotic pressure inside the hydrogel. Some portions of the surface layer can be inflated to compensate the increasing osmotic pressure, i.e., bubble formation. Only after the breaking of these bubbles, could the free water release. While the incorporated hydrophilic chains in PNIPAAm may lead to a faster deswelling of PNIPAAm hydrogels due to the fact that the hydrophilic chains may act as releasing channels for water molecules, which would prevent or block the forming of the dense, thick skin layer during the shrinking process. Many bubble observed during thermo-induced shrinkage of pure PNIPAAm and XMN1 certainly support the theory of the existence of the dense skin layer on the XMN1 hydrogel surface. This may mainly attributed to the fact that the amount of Xan-MA incorporated in XMN1 may not be high enough to exert meaningful Xan-MA effect on the deswelling property. Other XMN hydrogels, however, show fewer bubbles on their surfaces in the same thermo-induced hydrogel shrinkage process. Their increased deswelling rate is contributed to an increase in hydrophilicity from higher incorporated Xan-MA component.
3.7 pH sensitivity of Xan-MA/PNIPAAm hydrogels
Fig.7 shows the swelling ratios of XMN hydrogels as a function of pH of the swelling media. All the XMN hybrid hydrogels exhibite a lower swelling ratio in an acidic medium, which increases with an increase in pH of the medium. In an acidic medium (i.e., pH 2.2), the swelling ratios of XMN1, XMN3, and XMN5 are 2.9, 3.0, and 3.6, respectively.
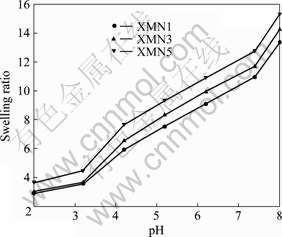
Fig.7 Swelling ratio of Xan-MA/PNIPAAm hydrogels in buffer solutions of different pH values
In an alkaline medium (pH 8.0), the swelling ratios augment greatly. This can be attributed to the free carboxylic acid groups in Xan-MA ionized, which will break hydrogen bonding and generate electrostatic repulsion among Xan-MA macromolecules. This repulsive force will push the network chain segments apart and attract more water into the hydrogel, which results in a higher swelling ratio.
4 Conclusions
(1) A novel class of hybrid hydrogel consisting of biocompatible polysaccharide (xanthan derivative, Xan-MA) and temperature sensitive PNIPAAm is designed and synthesized by solution polymerization. The resulting XMN hydrogels have the combined unique properties of their component precursors. Due to the presence of PNIPAAm in XMN series, they acquire thermo-responsive capability to Xan-MA.
(2) The properties of the XMN hydrogels, such as the lower critical solution temperature (LCST), swelling ratio, temperature responsive kinetics, swelling kinetics and the pH sensitivity are examined in terms of the composition ratio of Xan-MA to PNIPAAm precursors. The data obtained clearly demonstrate that the newly synthesized XMN hybrid hydrogel series have temperature as well as pH sensitivity.
(3) Most importantly, the properties of this class of new hydrogels are controllable. From XMN0, XMN1, XMN3 to XMN5, the LCSTs are 33.02, 36.15, 40.28 and 41.92 ℃, respectively. For instance, the phase transition temperature of these intelligent hydrogels can be modulated to be near the body temperature, which is critical for their biomedical applications.
References
[1] TORRES-LUGO M, PEPPAS N A. Molecular design and in vitro studies of novel pH-sensitive hydrogels for the oral delivery of calcitonin [J]. Macromolecules, 1999, 32(20): 6646-6651.
[2] HOFFMAN A S. Application of thermally reversible polymers and hydrogels in therapeutics and diagnostics [J]. Journal of Controlled Release, 1987, 6(1): 297-305.
[3] SUZUKI A, TANAKA T. Phase transition in polymer gels induced by visible-light [J]. Nature, 1990, 346: 345-347.
[4] MIYATA T, ASAMI N, URAGAMI T. A reversibly antigen- responsive hydrogel [J]. Nature, 1999, 399: 766-769.
[5] STAYTON P S, SHIMOBJI T, LONG C, CHILKOTI A, CHEN G, HARRIS J M, HOFFMAN A S. Control of protein-ligand recognition using a stimuli-responsive polymer [J]. Nature, 1995, 378: 472-474.
[6] PAN Yu-song, XIONG Dang-sheng, MA Ru-yin. Preparation and swelling behavior of polyvinyl alcohol physiological saline gel [J]. Journal of Central South University of Technology, 2006, 13(1): 27-31.
[7] OSADA Y, OKUZAKI H, HORI H. A polymer gel with electrically driven motility [J]. Nature, 1992, 355: 242-244.
[8] LIU F, TAO G L, ZHUO R X. Synthesis of thermal phase-separating reactive polymers and their applications in immobilized enzymes [J]. Polymer Journal, 1993, 25(6): 561-567.
[9] HIROKAWA Y, TANAKA T. Volume phase transition in a nonionic gel [J]. Journal of Chemical Physics, 1984, 81(12): 6379-6380.
[10] HOFFMAN A S. Environmentally sensitive polymers and hydrogels smart biomaterials [J]. MRS Bulletin, 1991, 16(9): 42-46.
[11] ZHANG X Z, YANG Y Y, CHUNG T S. The influence of cold treatment on properties of temperature-sensitive poly (N-isopropylacrylamide) hydrogels [J]. Journal of Colloid and Interface Science, 2002, 246: 105-111.
[12] HORTON D, MOLS O, WALASEK Z, WERNAU W C. Structural and biosynthetic studies on xanthan by 13C-N.M.R. spectroscopy[J]. Carbohydrate Research, 1985, 141(2): 340-346.
[13] HALL LD, YALPANI M. Determination of the primary structure of polysaccharides and their derivatives by solid-state carbon-13 N.M.R. spectroscopy [J]. Carbohydrate Research, 1981, 91(2): C1-C4.
[14] MILAS M, RINAUDO M. Conformational investigation on the bacterial polysaccharide xanthan [J]. Carbohydrate Research, 1979, 76(1): 189-96.
[15] WANG L F, SHEN S S, LU S C. Synthesis and characterization of chondroitin sulfate-methacrylate hydrogels [J]. Carbohydrate Polymers, 2003, 52(4): 389-396.
[16] MARTINEZ-RUVALCABA A, CHORNET E, RODRIGUE D. Viscoelastic properties of dispersed chitosan/xanthan hydrogels [J]. Carbohydrate Polymers, 2007, 67(4): 586-595.
[17] LIU Y Y, FAN X D. Preparation and characterization of a novel responsive hydrogel with β-cyclodextrin-based macromonomer [J]. Journal of Applied Polymer Science, 2003, 89(2): 361-367.
[18] IIJIMA M, SHINOZAKI M, HATAKEYAMA T, TAKAHASHI M, HATAKEYMA H. AFM studies on gelation mechanism of xanthan gum hydrogels [J]. Carbohydrate Polymers, 2007, 68(4): 701-707.
[19] HAMCERENCU M, DESBRIERES J, POPA M, KHOUKH A, RIESS G. New unsaturated derivatives of xanthan gum: Synthesis and characterization [J]. Polymer, 2007, 48(7): 1921-1929.
[20] KANEKO Y, YOSHIDA R, SAKAI K, SAKURAI Y, OKANO T. Temperature-responsive shrinking kinetics of poly (N-isopropylacrylamide) copolymer gels with hydrophilic and hydrophobic comonomers [J]. Journal of Membrane Science, 1995, 101: 13-22.
Received date: 2008?05?15; Accepted date: 2008?07?23
Corresponding author: PAN Chun-yue, Professor; Tel: +86?731?8836961; E-mail: panchunyue@sina.com
(Edited by YANG You-ping)