J. Cent. South Univ. Technol. (2007)05-0660-06
DOI: 10.1007/s11771-007-0127-8 
Preparation of p-menthane hydroperoxide from p-menthane in presence of metalloporphyrins
YANG Wei-jun(������), TAO Neng-ye(������), GUO Can-cheng(���ӳ�)
(College of Chemistry and Chemical Engineering, Hunan University, Changsha 410082, China)
Abstract The aerobic oxidation of p-menthane to p-menthane hydroperoxide (PMHP) in the presence of metalloporphyrins was investigated in an intermittent mode under an atmospheric pressure of air. Several important reaction parameters, such as the structure of metalloporphyrin, the air flow rate, and the temperature, were studied. The preliminary mechanism of the aerobic oxidation of p-menthane catalyzed by metalloporphyrins was also discussed. The results show that the reaction is greatly accelerated by the addition of metalloporphyrins at very low concentration, in terms of both the yield and formation rate of PMHP, and the high selectivity of PMHP is maintained during the reaction. Temperature of 120 �� and reaction time of around 5 h are the optimal conditions for the best result in the presence of 0.06 mmol/L monomanganeseporphyrins ((p-Cl)TPPMnCl). Furthermore, the yield of PMHP is increased remarkably when the reaction is carried out under programmed temperature compared with the constant temperature. When the reaction is catalyzed by 0.06 mmol/L((p-Cl)TPPMnCl) at the air flow rate of 600 mL/min and 120 �� for 4 h, and then the temperature is reduced to 110 ��, for another 4 h, the yield of PMHP reaches 24.3��, which is higher than that of the reaction at a constant temperature of 120 �� or 110 �� for 8 h.
Key words: metalloporphyrins; catalysis; oxidation; p-menthane
1 Introduction
p-menthane hydroperoxide (PMHP) is an excellent initiator of copolymerization of olefin. Compared with other hydroperoxides, PMHP is better for its performance, less dosage and more stability, which leads to the wide use in synthesis of styrene-butadiene rubber[1-2]. The main industrial process for PMHP production currently is carried out without any catalyst, which leads to the difficulty for the activation of the oxygen molecule. Consequently, there are some limitations associated with current industrial processes, such as long induction period, low reaction speed, numerous side-reactions and low selectivity of PMHP[2-4].
Metalloporphyrins, which resemble the important enzyme cytochrome P450 structurally, are among the most heavily investigated enzyme-mimic systems[5-7]. In recent years, researches have shown that metalloporphyrins can be utilized as catalysts in selective oxidation of hydrocarbons to produce commercial oxygenated compounds under medium pressure and at medium temperature[8-9]. A process, which is based on this catalytic, biomimetic oxidation, has been successfully developed in industry[10-11]. As green catalysts, metalloporphyrins possess many advantages, such as small amount, and wide use not only in homogeneous catalysis but also in heterogeneous catalysis when they are immobilized on solid supports. Though previous works have shown that hydrocarbons can be oxidized to produce alcohols, ketones and acids in the presence of metalloporphyrins[12-15], producing hydroperoxides directly from p-menthane with metalloporphyrin catalysts is still rare.
In this work, the aerobic oxidation of p-menthane to produce p-menthane hydroperoxide was investigated in the presence of metalloporphyrins at atmospheric pressure.
2 Experimental
2.1 Materials and reagents
All reagents and solvents used were analytical grade and obtained commercially. GC analysis showed that there was no impurity in p-menthane before use.
Various metalloporphyrins were synthesized according to Refs.[16-17] and purified via regular column chromatography. Their structures (Fig.1) were confirmed by MS, UV-Vis and 1H NMR spectra.

Fig.1 Structures of metalloporphyrins
(a) Monometalloporphyrin(R= Cl, H��M=Mn); (b) ��-oxo-bismetalloporphyrin(R1=Cl, H��M=Mn)
2.2 Device and analysis of products
The peroxidation of p-menthane was conducted in a 150 mL bubbling reactor shown in Fig.2.

Fig.2 Experimental device of peroxidation
1��Material inlet and product outlet; 2��Position for temperature controller; 3��Heater; 4��Support plate;
5��Buffer bottle; 6��Flowmeter; 7��Control valve;8��Air pump
In this work, the flow rate of air was maintained at 300-800 mL/min and the temperature was controlled below 130 ��. After the metalloporphyrins were dissolved in p-menthane, the liquid was filled into the reactor and heated to a desired temperature in 5 min. The air was then bubbled into the reactor and the reaction was started. At suitable time intervals, parts of the reactant mixture were sampled and analyzed.
The iodometric method was employed to analyze the content of PMHP in the reaction mixture[18]. The analytical process is as follows. A three-necked Pyrex kettle of 250 mL with a condenser was used. 20 mL isopropyl alcohol, 2 mL glacial acetic acid and 0.8-1.0 g samples were added into the three-necked bottle. The mixture was refluxed by heating, and 2 g KI was added into the bottle. The reflux was maintained for 5 min, then 5 mL water was put into the mixture. After the solution was cooled, the PMHP titration was carried out with 0.1 mol/L Na2S2O3 until the color of the solution was changed from orange to ivory-white. The PMHP content in reaction samples was calculated from the following equation:
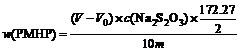 ��100% (1)
��100% (1)
where V is the volume of consumed Na2S2O3, V0 is the volume of consumed Na2S2O3 for blank sample, c(Na2S2O3) is the concentration of Na2S2O3, m is the mass of sample.
The structures of the hydroperoxides from the catalyzed reaction were identified by HPLC-MS. Byproducts were identified using GC�CMS and were quantified by GC using benzene as the internal standard substance. Before being injected into GC, PMHP in the reaction mixture samples was reduced to the corresponding alcohols.
3 Results and discussion
3.1 Reaction equation and products
In the presence of metalloporphyrins, the p-menthane oxidation process is shown as follows.

(2)
The tertiary carbon atom in p-menthane molecule is easily activated in the presence of air/oxygen under certain condition. The structures of the hydroperoxides from the catalyzed reaction are identified as the two forms of PMHP shown in Eqn.(2). The contents of the two forms of PMHP are nearly 50%, separately. The oxidation mixture containing the two monohydroperoxides is directly used as PMHP product in industry. There are also a few by-products in the reaction mixture( less than 5%), such as p-menthan-8-ol and p-menthan-1-ol, coming from the decomposition of PMHP under certain conditions. After analysis of the reaction samples, it is found that the components in the oxidation production are the same whether in the presence of metalloporphyrins or not.
3.2 Variation in PMHP yield with constant temperature
Due to the fact that the oxidation of p-menthane is relatively slow at low temperature and PMHP tends to decompose at higher temperature, in this work, the reaction was studied in the temperature range from 100 to 130 �� at the air flow rate of 600 mL/min and 0.06 mmol/L (p-Cl)TPPMnCl under atmospheric pressure. It is shown that the reaction rate is much lower at 100 �� or 110 �� than that at 120 �� or 130 �� (Fig.3). 18.3�� PMHP can be obtained within 5 h at 120 ��. Among the temperatures investigated, the initial reaction rate is the fastest at 130 ��, but the formed PMHP then quickly decomposes owing to the higher temperature, which leads to the lower PMHP yield during the reaction period. It seems that 120 �� is an optimum choice in the presence of 0.06 mmol/L (p-Cl)TPPMnCl.

Fig.3 Effect of temperature on p-menthane oxidation with (p-Cl)TPPMnCl
Reaction temperature/��: 1��100; 2��110; 3��120; 4��130
3.3 Variation in PMHP yield with programmed temperature
In the oxidation reaction, it is found that higher yield of PMHP can be obtained and higher selectivity can be maintained when changing the temperature regularly. Under the conditions of atmospheric pressure, 600 mL/min air, in the presence of 0.06 mmol/L (p-Cl)TPPMnCl or no catalyst, when the reaction temperature is kept at 120 �� for a period of time and then reduced the temperature to 110 �� and kept for another period of time, the maximal PMHP yield is higher than that at 120 �� constantly under the same conditions (Fig.4). Curve 1 in Fig.4 represents the reaction that is carried out at 120 �� firstly for 4 h, and then proceeds at 110 �� for another 4 h in the presence of (p-Cl)TPPMnCl, the PMHP yield is 24.3��. Curves 2 and 3 represent the reaction with catalyst or none respectively at 120 �� constantly. Curve 4 represents the reaction that is carried out at 120 �� for 6 h and then reduced to 110 �� without catalyst. After another 1 h reaction, the yield of PMHP is 16.9��, and after another 5 h reaction, the PMHP yield is 23.6��. It is shown in Fig.4 that the maximal PMHP yield at programmed temperature is higher than that at the constant temperature.

Fig.4 Effect of variational temperature on PMHP yield
1��Variational temperature with catalyst;
2��Constant temperature with catalyst;
3��Constant temperature without catalyst;
4��Variational temperature without catalyst
3.4 Variation in PMHP yield with catalysts
PMHP is thermal instable, which will be partly decomposed during the reaction under certain conditions. It is difficult to obtain high yield of PMHP in a relative short reaction time. A suitable catalyst can not only accelerate the reaction rate, but also produce high yield of PMHP. In this work, the catalytic activities of several kinds of simple metalloporphyrins, such as (p-Cl)TPPMnCl, O-[(p-Cl)TPPMn]2, O-[TPPMn]2 and TPPCo, were investigated in p-menthane aerobic oxidation (Fig.5). The reaction conditions are as follows: atmospheric pressure, 120 ��, the air flow rate of 600 mL/min and the metalloporphyrins�� concentration of 0.06 mmol/L (based on the content of metal ions). It is known from the results that all the metalloporphyrins used can accelerate the formation rate of PMHP remarkably, and the manganeseporphyrins, such as (p-Cl)TPPMnCl, O-[(p-Cl)TPPMn]2 and O-[TPPMn]2, can make the higher PMHP yield at the same time. When Mn(Ac)2 is used as catalyst, which is 4 times dosage than monomanganeseporphyrin on the content of Mn ions, the maximal yield of PMHP is nearly the same, but the reaction rate in early stage is obviously slower than that of manganeseporphyrins. The maximal yield of PMHP is 18.3�� after reaction for 5 h in the presence of (p-Cl)TPPMnCl, and that is 17.2�� after reaction for 8 h in presence of 4 times dosage of Mn(Ac)2 (Fig.5).

Fig.5 Effect of catalysts on PMHP yield
1��Without catalyst; 2��Mn(Ac)2; 3��O-[TPPMn]2; 4��O-[(p-Cl)TPPMnCl]; 5��(p-Cl) TPPMnCl; 6��TPPCo
3.5 Variation in PMHP yield with flow rate of air
The effect of air flow rate on the PMHP yield is shown in Fig.6. When varying air flow rate from 300 to 800 mL/min under atmospheric pressure, at 120 �� and using 0.06 mmol/L (p-Cl)TPPMnCl as catalyst, the oxidation rate of p-menthane to produce PMHP is accelerated with increasing flow rate of air. A huge increase of PMHP yield can be observed in the air flow rate range from 400 to 600 mL/min. When the flow rate of air is 400 mL/min, the maximal yield of PMHP is 15.6% within 6 h, and the maximal yield of PMHP is 18.3�� within 5 h at 600 mL/min. When the flow rate of air is further increased to 800 mL/min, the PMHP yield changes very little in the first 5 h, however, after that, the decomposition of PMHP is much faster than that of the reaction at 600 mL/min. This phenomenon can be explained as follows. Before reaching 600 mL/min, the oxygen dissolved in the liquid phase does not reach saturation level, thus the production of PMHP increases with the increase of the flow rate of air. Beyond the saturation point (600 mL/min), the increase of oxygen flow no longer increases the reaction rate since it does not alter the concentration of oxygen in p-menthane. Additionally, the higher flow rate of air can accelerate the decomposition of PMHP. Taking everything into consideration, it seems that the flow rate of 600 mL/min is the optimal choice.

Fig.6 Effect of flow rate of air on PMHP yield
Air flow rate/(mL?min-1): 1��300; 2��400; 3��600; 4��800
3.6 Variation in PMHP yield with concentration of metalloporphyrins
Table 1 lists the yield and selectivity of PMHP after reacting for 4 h with different catalyst concentrations under atmospheric pressure at 120 �� and 600 mL/min.
Table 1 Effect of (p-Cl)TPPMnCl concentration on yield and selectivity of PMHP

According to Table 1, under the same reaction condition, enhancing the catalyst concentration leads to increase in the yield of PMHP. When the concentration of (p-Cl)TPPMnCl is 0.04 mmol/L, the yield of PMHP is 11.6�� after 4 h, when the concentration of the catalyst is 0.06 mmol/L, the yield of PMHP increases to 15.8%, and when the concentration of the catalyst is 0.12 mmol/L, the yield of PMHP is only higher than that of the reaction with catalyst concentration of 0.06 mmol/L by 1.6��. However, enhancing the catalyst concentration leads to decrease in the selectivity of PMHP. It can be concluded that the metal ions from the self-oxidation of the metalloporphyrin catalysts in the reaction mixture may accelerate the decomposition of PMHP.
3.7 Preliminary mechanism of aerobic oxidation of p-menthane catalyzed by metalloporphyrins
PMHP is the target product in the reaction, and after examining the products by GC-MS, it is found that there are also a few by-products in the reaction mixture, such as p-menthan-8-ol and p-menthan-1-ol, and trace of p-menthan-5-ol can also be detected by GC-MS. According to the mechanism of hydrocarbons catalyzed by metalloporphyrins[19-20] and the aerobic auto- oxidation mechanism of hydrocarbons[21], a reasonable course of the reaction can be deduced as follows.
Step 1: Generation of p-menthane free radicals by (p-Cl)TPPMnCl.
 (3)
(3)
 (4)
(4)
 (5)
(5)
 (6)
(6)
Step 2: Formation of PMHP.

(7)

(8)
The side reaction is

(9)
4 Conclusions
1) An efficient and selective protocol for the oxidation of p-menthane to p-menthane hydroperoxide (PMHP) was developed using metalloporphyrins as catalysts with air under atmospheric pressure. The effects of different catalysts, the flow rate of air and temperature on the reaction were examined.
2) The reaction is greatly accelerated by the addition of metalloporphyrins, and the yield and formation rate of PMHP are also increased. Temperature of 120 �� and reaction time of around 5 h are the optimal conditions for the best result in the presence of 0.06 mmol/L (p-Cl)TPPMnCl. Higher concentration of (p-Cl)TPPMnCl, longer reaction time and higher reaction temperature are all improper to the production of PMHP from p-menthane.
3) The yield of PMHP is increased greatly in the presence of metalloporphyrins when the reaction is carried out under programmed temperature. Under the condition of 600 mL/min flow rate of air, in the presence of monomanganeseporphyrins ((p-Cl)TPPMnCl), when the reaction temperature is maintained at 120 �� for 4 h firstly, and then reduced to 110 ��, the maximal yield and formation rate of PMHP are higher than those at 120 �� or 110 �� constantly when keeping the same total reaction time.
References
[1] FISHER G S. Peroxides from turpentine II: Pinane hydroperoxide[J]. Journal of the American Chemical Society, 1953, 75(15): 3675-3678.
[2] STINSON J S. p-menthane hydroperoxide[J]. Journal of Organic Chemistry, 1959, 24(8): 1084-1088.
[3] LIU Xian-zhang, JIANG Tong-fu, HU xi-e, et al. Oxidation of p-menthane[J]. Journal of Chemical Industry of Forest Products, 1997, 31(1): 629-632. (in Chinese)
[4] CLARKE P D. Antiviral composition comprising p-menthane- 3,8-diol: European,1748769[P]. 2007.
[5] LANGE M, MANSUY D. N-substituted porphyrins formation from carbene iron-porphyrin complexes: A possible pathway for cytochrome P450 heme destruction[J]. Tetrahedron Letters, 1981, 22(27): 2561-2564.
[6] MANSUY D. A brief history of the contribution of metalloporphyrin models to cytochrome P450 chemistry and oxidation catalysis[J]. Comptes Rendus Chimie, 2007, 10: 1-12.
[7] ZAKHARIEVA O, TRAUTWEIN A X, VEEGER C. Porphyrin-Fe(III)-hydroperoxide and porphyrin-Fe(III)-peroxide anion as catalytic intermediates in cytochrome P450-catalyzed hydroxylation reactions: A molecular orbital study[J]. Biophysical Chemistry, 2000, 88(1/3): 11-34.
[8] GUO Can-cheng. Synthesis of ?-oxo-bisiron(III) porphyrin compounds and their catalysis for cyclohexane hydroxylation[J]. Journal of Catalysis, 1998, 178(1): 182-187.
[9] GUO Can-cheng, CHU Ming-fu, LIU Qiang, et al. Effective catalysis of simple metalloporphyrins for cyclohexane oxidation with air in the absence of additives and solvents[J]. Journal of Applied Catalysis A: General, 2003, 246(2): 303-309.
[10] YANG Wei-jun, GUO Can-cheng. Metalloporphyrins and their biomimetic catalytic oxidation for alkanes[J]. Chinese Journal of Applied Chemistry, 2004, 21(6): 541-545. (in Chinese)
[11] GUO Can-cheng, LIU Qiang, WANG Xu-tao, et al. Selective liquid-phase oxidation of toluene with air[J]. Journal of Applied Catalysis A: General, 2005, 282(1/2): 55-59.
[12] GUO Can-cheng, YANG Wei-jun, MAO Yan-li. Selectively aerobic oxidation of C=C and allylic C��H bonds in ��-pinene over simple metalloporphyrins[J]. Journal of Molecular Catalysis A: Chemical, 2005, 226(2): 279-284.
[13] GUO Can-cheng, HUANG Guan, ZHANG Xiao-bing, et al. Catalysis of chitosan-supported iron tetraphenylporphyrin for aerobic oxidation of cyclohexane in absence of reductants and solvents[J]. Journal of Applied Catalysis A: General, 2003, 247(2): 261-267.
[14] HUANG Guan, GUO Can-cheng, TANG Si-si. Catalysis of cyclohexane oxidation with air using various chitosan-supported metallotetraphenylporphyrin complexes[J]. Journal of Molecular Catalysis A: Chemical, 2007, 261(1/2): 125-130.
[15] YANG Wei-jun, GUO Can-cheng, MAO Yan-li, et al. Catalysis and substituent effects of monomanganese-porphyrins and monoiron-porphyrins for ��-pinene oxidation with air[J]. Chemical Journal of Chinese Universities, 2005, 26(9): 1695-1698. (in Chinese)
[16] ADLER A D, LONGO F R, FINARELLI J D, et al. A simplified synthesis for meso-tetraphenylporphine[J]. Journal of Organic Chemistry, 1967, 32(2): 476-481.
[17] ADLER A D, LONGO F R, KAMPAS F. On the preparation of metalloporphyrins[J]. Journal of Inorganic Nuclear Chemistry, 1970, 32(7): 2443-2447.
[18] WAGNER C D, SMITH R H, PETERS E D. Evaluation of modified iodometric method[J]. Analytical Chemistry, 1947, 19(12): 976-979.
[19] LYONS J E, ELLIS P E, MYERS H K. Halogenated metalloporphyrin complexes as catalysts for selective reactions of acyclic alkanes with molecular oxygen[J]. Journal of Catalysis, 1995, 155(1): 59-73.
[20] GRINSTAFF M W, HILL M G, LABINGER J A, et al. Mechanism of catalytic oxygenation of alkanes by halogenated iron porphyrins[J]. Science, 1994, 264 (5163): 1311-1313.
[21] MU Guang-zhao. Reaction of Free Radicals[M]. Beijing: Higher Education Press, 1985.(in Chinese)
Foundation item: Project (20606008) supported by the National Natural Science Foundation of China
Received date: 2007-03-26; Accepted date: 2007-04-28
Corresponding author: GUO Can-cheng, Professor; Tel: +86-731-8821603; E-mail: ccguo@hnu.cn
(Edited by CHEN Wei-ping)