Uniform AlF3 thin layer to improve rate capability ofLiNi1/3Co1/3 Mn1/3O2 material for Li-ion batteries
来源期刊:中国有色金属学报(英文版)2010年第5期
论文作者:王海燕 唐爱东 黄可龙 刘素琴
文章页码:803 - 808
Key words:Li-ion battery; LiNi1/3Co1/3Mn1/3O2; coating; uniform thin AlF3 layer; rate capability
Abstract: LiNi1/3Co1/3Mn1/3O2 was coated with uniform nano-sized AlF3 layer by chemical precipitation method to improve its rate capability. The samples were characterized by X-ray diffractometry (XRD), transmission electron microscopy (TEM), energy dispersive spectroscopy (EDS), charge-discharge cycling, cyclic voltammetry (CV), and electrochemical impedance spectroscopy (EIS). Uniform coated layer with a thickness of about 3 nm was observed on the surface of LiNi1/3Co1/3Mn1/3O2 particle by TEM. At 0.5C and 2C rates, 1.5% (mass fraction) AlF3-coated LiNi1/3Co1/3Mn1/3O2/Li in 2.8-4.3 V versus Li/Li+ after 80 cycles showed less than 3% of capacity fading, while those of the bare one were 16.5% and 45.9%, respectively. At 5C rate, the capacity retention of the coated sample after 50 cycles maintained 91.4% of the initial discharge capacity, while that of the bare one decreased to 52.6%. EIS result showed that a little change of charge transfer resistance of the coated sample resulting from uniform thin AlF3 layer was proposed as the main reason why its rate capability was improved obviously. CV result further indicated a greater reversibility for the electrode processes and better electrochemical performance of AlF3-coated layer.
基金信息:supported by the National Natural Science Foundation of China
supported by Graduate Degree Thesis Innovation Foundation of Central South University, China

WANG Hai-yan(王海燕), TANG Ai-dong(唐爱东), HUANG Ke-long(黄可龙), LIU Su-qin(刘素琴)
School of Chemistry and Chemical Engineering, Central South University, Changsha 410083, China
Received 6 May 2009; accepted 23 July 2009
Abstract: LiNi1/3Co1/3Mn1/3O2 was coated with uniform nano-sized AlF3 layer by chemical precipitation method to improve its rate capability. The samples were characterized by X-ray diffractometry (XRD), transmission electron microscopy (TEM), energy dispersive spectroscopy (EDS), charge-discharge cycling, cyclic voltammetry (CV), and electrochemical impedance spectroscopy (EIS). Uniform coated layer with a thickness of about 3 nm was observed on the surface of LiNi1/3Co1/3Mn1/3O2 particle by TEM. At 0.5C and 2C rates, 1.5% (mass fraction) AlF3-coated LiNi1/3Co1/3Mn1/3O2/Li in 2.8-4.3 V versus Li/Li+ after 80 cycles showed less than 3% of capacity fading, while those of the bare one were 16.5% and 45.9%, respectively. At 5C rate, the capacity retention of the coated sample after 50 cycles maintained 91.4% of the initial discharge capacity, while that of the bare one decreased to 52.6%. EIS result showed that a little change of charge transfer resistance of the coated sample resulting from uniform thin AlF3 layer was proposed as the main reason why its rate capability was improved obviously. CV result further indicated a greater reversibility for the electrode processes and better electrochemical performance of AlF3-coated layer.
Key words: Li-ion battery; LiNi1/3Co1/3Mn1/3O2; coating; uniform thin AlF3 layer; rate capability
1 Introduction
Layered LiNi1/3Co1/3Mn1/3O2, reported firstly by OHZUKU and MAKIMURA[1], has been extensively studied as a promising cathode material for Li-ion battery. Due to its high discharge capacity, stable cycling performance, relatively low cost and high safety[2-4], LiNi1/3Co1/3Mn1/3O2 is even considered to be one of the best cathode materials for power source system of hybrid electric vehicle (HEV) or electric vehicle(EV)[5]. However, it shows poorer rate capability, compared with LiCoO2, which limits its further application[6-7].
It’s well known that surface modification on cathode materials with electrochemically inactive metal oxides such as Al2O3[8-10], ZrO2[11], MgO[12], ZnO[13] and Y2O3[14] could significantly improve their electrochemical performance. Among these materials, Al2O3 was widely used to coat LiCoO2, LiMn2O4, LiNiO2 or their composite metal compounds. Using time-of-flight secondary ion mass spectrometry (ToF-SIMS), MYUNG et al[15] found that the Al2O3 coating layer on Li[Li0.05Ni0.4Co0.15Mn0.4]O2 acted as HF scavenger, which could reduce HF generation, resulting in less decomposition of the cathode by forming AlF3 on the surface of Al2O3 coating layer. Based on these results, AlF3-coated LiCoO2 and LiNi1/3Co1/3Mn1/3O2 with significant improvement of high voltage cycling behavior were prepared by SUN et al[16-17]. Recently, AlF3 layer with the thickness of about 10 nm was employed to modify LiNi1/3Co1/3Mn1/3O2 by PARK et al[18] and obvious improvement of electrochemical properties in high voltage region was achieved. Considering that asymmetrical and thick coated-layer will be not propitious for further improvement of electrochemical properties, in this work, accordingly, LiNi1/3Co1/3Mn1/3O2 was coated by uniform and thin AlF3 layer with chemistry precipitation method, in order to ameliorate its rate capability. Electrochemical impedance spectroscopy (EIS) and cyclic voltammetry (CV) tests were also carried out to explain the reason for the improvement of electrochemical performance.
2 Experimental
LiNi1/3Co1/3Mn1/3O2 powder chosen for this work was provided by Dongguan Amperex Electronics Technology Co., Ltd (ATL). To prepare AlF3-coated LiNi1/3Co1/3Mn1/3O2, aluminum nitrate and hydrogen ammonium fluoride were separately dissolved in distilled water. After LiNi1/3Co1/3Mn1/3O2 powder was immersed into the aluminum nitrate solution, a certain excessive amount of hydrogen ammonium fluoride was slowly added to the solution with the speed of 1.5 mL/min. At the same time, the solution was heated to 45 ℃ and constantly stirred by magnetic force stirrer in order to disperse the powder adequately. The amount of AlF3 in theory in the solution corresponded to 1.5% (mass fraction) of the LiNi1/3Co1/3Mn1/3O2 powder. After the solution containing the cathode material was filtered and then washed by distilled water, the powder was dried at 110 ℃ for 3 h and then was calcined at 600 ℃ for 4 h to obtain AlF3-coated LiNi1/3Co1/3Mn1/3O2 under argon atmosphere.
The crystalline structures of the bare and coated LiNi1/3Co1/3Mn1/3O2 were identified with Rigaku X-ray diffractometer using Cu Kα radiation in the 2θ range of 10?-80? with a step of 0.02?. Transmission electron microscopy was operated by JEOL 200CX instrument and energy dispersive spectroscopy was carried out on Oxford INCA energy system.
Electrochemical tests were operated using CR2032 coin-type cells. This cell consisted of a cathode and a lithium metal anode, separated by a porous polyethylene film (Celgard 2500). The positive electrode was constituted from 85% active material, 10% carbon black as conducting additive, and 5% polytetrafluoroethylene (PTFE) as binder. It was pressed onto a 100 mm2 stainless steel mesh used as the current collector at 300 kPa and then dried at 120 ℃ for 8 h in a vacuum oven in order to remove moisture. A commercial 1 mol/L lithium-hexafluoro phosphate (LiPF6) solution in a mixture of ethylene-carbonate (EC), dimethyl carbonate (DMC) and propylene carbonate (PC) at a volume ratio of 1?1?1 was used as the electrolyte. The cells were cycled galvanostatically in the voltage range of 2.8-4.3 V versus Li/Li+ at a desired current density with a Land tester system.
AC impedance measurements were performed using a Zahner Elektrik IM6 impedance analyzer over the frequency range from 500 kHz to 10 mHz with an amplitude of 5 mV. Tested cell was charged to 3.95 V versus Li/Li+ and the voltage was kept stable until the current decreased to less than 50 uA. Cyclic voltammetry was also performed using a Zahner Elektrik IM6 analyzer at a scan rate of 1 mV/s. The scan voltage was from 3.0 to 4.5 V versus Li/Li+. Differential scanning calorimetry (DSC/TG) was carried out with a Model STA449C differential scanning calorimeter at a ramp rate of 10 ℃/min. The temperature ranged from 45 to 600 ℃.
3 Results and discussion
3.1 Physical characteristics of coated and uncoated samples
Fig.1 shows the XRD patterns of the bare, 1.5% and 3.0% AlF3-coated LiNi1/3Co1/3Mn1/3O2. All three samples had α-NaFeO2 layered structure and belonged to ![]() space group. In comparison with the bare LiNi1/3Co1/3Mn1/3O2, no diffractions of impurity in both 1.5% and 3.0% AlF3-coated sample were observed, which showed that the AlF3-coated layer had no influence on the structure of LiNi1/3Co1/3Mn1/3O2. The absence of diffraction peaks corresponding to AlF3 may be due to very low concentration of AlF3 on the surface of LiNi1/3Co1/3Mn1/3O2 particle. The lattice parameters of the bare, 1.5% and 3.0% AlF3-coated LiNi1/3Co1/3Mn1/3O2 were calculated as a=2.863 ?, c=14.242 ?; a=2.863 ?, c=14.245 ?; a=0.2864 ?, c=14.261 ?; respectively. There was little change in a lattice parameter, while a little extending in c axis direction existed with the increase of the concentration of coated AlF3, which suggested that the phase of AlF3-coated layer nearly close to the surface of LiNi1/3Co1/3Mn1/3O2 could form the solid solution under 600 ℃ for 4 h[19].
space group. In comparison with the bare LiNi1/3Co1/3Mn1/3O2, no diffractions of impurity in both 1.5% and 3.0% AlF3-coated sample were observed, which showed that the AlF3-coated layer had no influence on the structure of LiNi1/3Co1/3Mn1/3O2. The absence of diffraction peaks corresponding to AlF3 may be due to very low concentration of AlF3 on the surface of LiNi1/3Co1/3Mn1/3O2 particle. The lattice parameters of the bare, 1.5% and 3.0% AlF3-coated LiNi1/3Co1/3Mn1/3O2 were calculated as a=2.863 ?, c=14.242 ?; a=2.863 ?, c=14.245 ?; a=0.2864 ?, c=14.261 ?; respectively. There was little change in a lattice parameter, while a little extending in c axis direction existed with the increase of the concentration of coated AlF3, which suggested that the phase of AlF3-coated layer nearly close to the surface of LiNi1/3Co1/3Mn1/3O2 could form the solid solution under 600 ℃ for 4 h[19].

Fig.1 X-ray diffraction patterns of bare and AlF3-coated LiNi1/3Co1/3Mn1/3O2
The surface morphology of the 1.5% AlF3-coated LiNi1/3Co1/3Mn1/3O2 powder was observed by transmission electron microscopy (TEM). In Fig.2(a), a thin and uniform AlF3 layer with the thickness of about 3 nm was shown on the particle surface. To the best of our knowledge, the coated layer was much thinner than those reported[8-9, 17]. In our opinion, the thin coated layer would make the lithium ion transfer well and decrease the polarization of the electrode.
Energy dispersive spectroscopy (EDS) result of the AlF3-coated LiNi1/3Co1/3Mn1/3O2 powder is recorded in

Fig.2 TEM morphology (a) and EDS result (b) of 1.5% AlF3-coated LiNi1/3Co1/3Mn1/3O2 powder
Fig.2(b). The existence of peaks corresponding to F and Al indicated probably the presence of AlF3 coating layer on the surface of LiNi1/3Co1/3Mn1/3O2 particle[17].
3.2 Electrochemical performance
Fig.3 indicates the cyclic performance of the bare, 1.5% and 3.0% AlF3-coated LiNi1/3Co1/3Mn1/3O2/Li at 0.5C rate (1C rate was defined as 150 mA・h/g in this work) between 2.8 and 4.3 V versus Li/Li+. The bare LiNi1/3Co1/3Mn1/3O2/Li had an initial discharge capacity of 139.6 mA・h/g and exhibited good electrochemical performance at the first 40 cycles. However, capacity fading ratio increased to 13.5% after 80 cycles. On the other hand, although 1.5% AlF3-coated LiNi1/3Co1/3Mn1/3O2/Li showed lower discharge capacity (132.3 mA・h/g), the capacity after 80 cycles was still 128.2 mA・h/g, only with nearly 3% capacity fading. As for 3.0% AlF3-coated LiNi1/3Co1/3Mn1/3O2/Li, it demonstrated good cyclic performance, with 2.4% capacity fading after 50 cycles. However, its discharge capacity was less than that of the 1.5% AlF3-coated sample by about 6 mA・h/g. So, 1.5% AlF3-coated LiNi1/3Co1/3Mn1/3O2 was chosen to take further test.
Fig.4(a) depicts cyclic stability of the bare and 1.5% AlF3-coated LiNi1/3Co1/3Mn1/3O2/Li, operated at 2C rate between 2.8 and 4.3 V versus Li/Li+ and the discharge curves after 80 cycles are also given in Fig.4(b). As obviously observed, the AlF3-coated LiNi1/3Co1/3Mn1/3O2 had only about 2.4% capacity fade after 80 cycles, while the bare sample suffered from about 46% fading of the initial discharge capacity. For the coated and bare cells, the discharge capacity of 119.7 mA・h/g and 63.6 mA・h/g was observed at the 80th cycle, respectively. Such comparison was also shown by the 80th discharge curves in Fig.4(b). Clearly, AlF3-coated LiNi1/3Co1/3Mn1/3O2
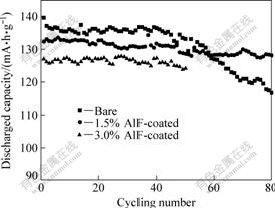
Fig.3 Cyclic performances of bare, 1.5% and 3.0% AlF3-coated LiNi1/3Co1/3Mn1/3O2/Li at 0.5C rate

Fig.4 Cyclic performance (a) and discharge curves after 80 cycles (b) of bare and 1.5% AlF3-coated LiNi1/3Co1/3Mn1/3O2/Li at 2C rate
indicated much better electrochemical performance at such current density.
Fig.5(a) shows the discharge capacity versus cycle number of the bare and AlF3-coated LiNi1/3Co1/3Mn1/3O2/Li cells operated at 5C rate between 2.8 and 4.3 V versus Li/Li+ and the discharge curves of both samples at different cycles (1st, 10th, 30th, 50th) are given in Fig.5(b) and Fig.5(c), respectively. As can be seen, discharge curve of the bare sample became stepper with the cycle number increasing, while that of the coated one changed a little, indicating that the AlF3-coated LiNi1/3Co1/3Mn1/3O2 electrode had much

Fig.5 Cyclic performance (a) and discharge curves of bare (b) and AlF3-coated (c) LiNi1/3Co1/3Mn1/3O2/Li at different cycles operated at 5C rate
better charge-discharge platform retention than the bare one. From the Fig.5(a), the bare electrode suffered from a severe capacity fading, showing only 52.6% of initial discharge capacity (44.6 mA・h/g) after 50 cycles was retained. As for AlF3-coated LiNi1/3Co1/3Mn1/3O2, 91.4% of the initial discharge (99.2 mA・h/g) was maintained. It is very interesting that the initial discharge capacity of the coated electrode at 2C rate or 5C rate was larger than that of the bare one, although it is well known that the discharged capacity often decreased to some extent after being coated[12], just like that at 0.5C rate. Such phenomenon could be explained as follows. The amount of unavailable active material (lithium ion) because of polarization at high current density was reduced effectively, which would enhance the capacity relatively. That is to say, the specific capacity fading of the coated sample resulting from polarization was less than that of the bare one. Apparently, uniform AlF3 nano-sized layer in this work was a well coated material to improve rate capability for cathode material. Most researchers attributed the performance improvement to the formation of a barrier isolated electrolyte from electrode, which would lower the activity of the second reaction[10, 12]. Besides, SUN et al[20] clarified that the excellent cyclability of ZnO-coated LiNi0.5Mn1.5O4 in the 5 V region was due to the reduced HF content and thus suppressed Mn dissolution. MYUNG et al[15] considered that the Al2O3 coating layer on Li[Li0.05Ni0.4Co0.5Mn0.4]O2 acted as a HF scavenger, which would reduce HF generation, resulting in less decomposition of the cathode particle by forming AlF3 on the surface of Al2O3 coating layer. It is known the strain and defect resulting from the fast Li+ extraction and intercalation in the cathode material were considered as the important cause of capacity fading when the battery was charged at high current density[21], which would increase the surface activity of cathode material largely and then result in electrolyte decomposition. In this work, compared with thick layer, uniform and thin AlF3 layer could not only suppress the electrolyte decomposition and hinder the second reaction in the material surface effectively, but also be better for the improvement of rate capability, since it was much easy for the Li+ extraction and intercalation when the electrode was charged at high current density. The conclusion above could be approved by the compared results of electrochemical impedance and CV test.
In order to gain further understanding of the role of the AlF3-coated layer, Nyquist plots of the bare and AlF3-coated LiNi1/3Co1/3Mn1/3O2/Li at 3.95 V versus Li/Li+ after different cycles (1 and 50) at 5C rate between 2.8 and 4.3 V versus Li/Li+ are recorded in Fig.6. In general, each impedance spectrum of the bare and the AlF3-coated cells consisted of two overlapped

Fig.6 Nyquist plots of bare (a) and AlF3-coated (b) LiNi1/3Co1/3Mn1/3O2/Li at 3.95 V versus Li/Li+ after different cycles at 5C rate
semicircles at the high frequency side and a well defined semicircle at the low frequency followed by a slop line at the very low frequency range. It has been confirmed that the high frequency semicircle was due to the interface parameters such as the porous nature of the electrode, the surface film contribution, and/or the bulk of the material while the low frequency semicircle was considered as the contribution of the charge-transfer process[22]. As for the slop line, it was due to Warburg resistance which reflected Li ion diffusion in the solid-state electrodes. AURBACH et al[23] reported that capacity fading of LiCoO2 on cycling and storage at elevated temperature was attributed to the increase of surface film resistance rather than the bulk degradation. SUN et al[16] considered that the variation in charge transfer resistance(Rct) was a key to capacity fading for bare and Al2O3-coated electrode. SHAJU et al[24] found the proportional increase in the charge transfer resistance of the LiNi1/3Co1/3Mn1/3O2 electrode with cycling number. In Fig.6, it is shown that the total resistance of the bare electrode was much larger than that of AlF3-coated one. Especially, charge transfer resistance of the bare electrode drastically increased during cycling, while that of the coated electrode changed a little. From the results above, we concluded that the improvement of rate capability of the AlF3-coated cathode material was attributed to the uniform and thin AlF3 layer, which lowered the activity of interface between active material and electrolyte, and also considerably suppressed impedance growth, especially the charge transfer resistance.
Cyclic voltammetry curves of the bare and AlF3-coated LiNi1/3Co1/3Mn1/3O2/Li at the first 3 cycles, operated at 1 mV/s in the potential range from 3.0 to 4.5 V versus Li/Li+ are compared in Fig.7. Both studied cells were those after 50 cycles at 5C rate. It was reported that LiNi1/3Co1/3Mn1/3O2/Li under small current often showed two couple peaks at about 3.9 V and 4.5 V, respectively. One was assigned to the Ni2+/Ni3+ or Ni2+/Ni4+ redox couple and the other was due to Co3+/Co4+ redox couple[1, 25]. However, the polarization of the bare and coated cells after cycling made the second redox couple nearly disappear and the interval of oxidation and reduction peaks became large. From Fig.7, the oxidation and reduction peaks of the AlF3-coated sample were centered at 4.3 and 3.4 V, respectively, but those of the bare one even located at 4.5 V and 3.1 V, respectively, which indicated the polarization of the bare one after such charge-discharge test was very larger than that of the coated one. The reduced interval of each redox couple and little change of peak area further demonstrated a greater reversibility for the electrode processes upon AlF3-coated layer. The EIS and CV test results approved that the uniform and thin AlF3 layer could reduce the polarization of the battery and improve the electrochemical performance when cycling at high rate.
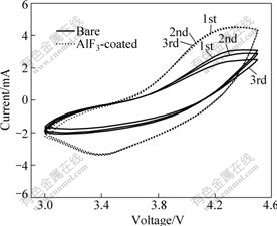
Fig.7 Cyclic voltammetry curves of bare and 1.5% AlF3-coated LiNi1/3Co1/3Mn1/3O2/Li cells with scan rate of 1 mV/s after 50 cycles at 5C rate
4 Conclusions
1) Uniform and thin AlF3 layer with a thickness of about 3 nm was coated on the surface of LiNi1/3Co1/3Mn1/3O2 successfully by chemical precipitation method.
2) At 0.5C rate and 2C rate, the 1.5% (mass fraction) AlF3-coated LiNi1/3Co1/3Mn1/3O2/Li in 2.8-4.3 V versus Li/Li+ after 80 cycles showed less than 3% capacity fading, while the bare electrode exhibited 16.5% and 45.9% capacity loss, respectively. At 5C rate, capacity retention of the coated electrode after 50 cycles maintained 91.4%, while that of the bare one decreased to 52.6%.
3) EIS result showed that a little change of charge transfer resistance of the coated electrode was the main reason why the rate capability was improved obviously. CV tests also indicated that a great reversibility for the electrode processed upon AlF3-coated layer.
4) All the test results approved that the uniform and thin AlF3 layer could reduce the polarization of the battery evidently and improve the electrochemical performance when cycling at high rate.
References
[1] OHZUKU T, MAKIMURA Y. Layered lithium insertion material of LiCo1/3Ni1/3Mn1/3O2 for lithium-ion batteries [J]. Chem Lett, 2001, 30: 642-645.
[2] SHAJU K M, SUBBA G V, CHOWDARI B. Performance of layered Li(Ni1/3Co1/3Mn1/3)O2 as cathode for Li-ion batteries [J]. Electrochim Acta, 2002, 48: 145-151.
[3] TANG A D, HUANG K L. Electrochemical properties and structural characterization of layered LixNi0.35Co0.3Mn0.35O2+δ cathode materials [J]. Mater Sci Eng B, 2005, 122: 115-120.
[4] KANG S H, KIM J, STOLL M E, ABRAHAM D, SUN Y K, AMINE K. Layered Li(Ni0.5-xMn0.5-xM′2x)O2 (M′=Co, Al, Ti; x=0, 0.025) cathode materials for Li-ion rechargeable batteries [J]. J Power Sources, 2002, 112: 41-48.
[5] BELHAROUAK I, SUN Y K, LIU J, AMINE K. Li(Ni1/3Co1/3Mn1/3)O2 as a suitable cathode for high power applications [J]. J Power Sources, 2003, 123: 247-252.
[6] LUO X F, WANG X Y, LIAO L, GAMBOA S, SEBASTIAN P J. Synthesis and characterization of high tap-density layered Li[Ni1/3Co1/3Mn1/3]O2 cathode material via hydroxide co-precipitation [J]. J Power Sources, 2006, 158: 654-658.
[7] WANG Hai-yan, TANG Ai-dong, HUANG Ke-long, JING Tao, ZHAO Wei. Preparation and rate capability study of LiNi1/3Co1/3Mn1/3O2 as cathode material [J]. Chinese Journal of Inorganic Chemistry, 2008, 24: 593-599. (in Chinese)
[8] GEORGE T K, KAO H M, MURALIDHARAN P, KUMAR Y D. Electrochemical and solid-state NMR studies on LiCoO2 coated with Al2O3 derived from carboxylate-alumoxane [J]. J Power Sources, 2006, 163: 135-143.
[9] LIU L J, WANG Z X, LI H, CHEN L Q, HUANG X J. Al2O3-coated LiCoO2 as cathode material for lithium ion batteries [J]. Solid State Ionics, 2002, 152/153: 341-346.
[10] LEE S W, KIM K S, MOON H S. Electrochemical characteristics of Al2O3-coated lithium manganese spinel as a cathode material for a lithium secondary battery [J]. J Power Sources, 2004, 126: 150-155.
[11] HU S K, CHENG G H, CHENG M Y, HWANG B J, SANTHANAM R. Cycle life improvement of ZrO2-coated spherical LiNi1/3Co1/3Mn1/3O2 cathode material for lithium ion batteries [J]. Power Sources, 2009, 188: 564-569.
[12] KWEON H J, KIM S J, PARK D G. Modification of LixNi1-yCoyO2 by applying a surface of MgO [J]. J Power Sources, 2000, 88: 255-261.
[13] FANG T, DUH J G, SHEEN S R. LiCoO2 cathode material coated with nano-crystallized ZnO for Li-ion batteries [J]. Thin Solid Films, 2004, 469/470: 361-365.
[14] WU F, WANG M, SU Y F, CHEN S. Surface modification of LiNi1/3Co1/3Mn1/3O2 with Y2O3 for lithium-ion battery [J]. J Power Sources, 2009, 189: 743-747.
[15] MYUNG S T, IZUMI K, KOMABA S, SUN Y K, YASHIRO H, KUMAGAI N. Role of alumina coating on Li-Ni-Co-Mn-O particles as positive electrode material for lithium ion batteries [J]. Chem Mater, 2005, 17: 3695-3704.
[16] SUN Y K, HAN J M, MYUNG S T, LEE S W, AMINE K. Significant improvement of high voltage cycling behavior AlF3-coated LiCoO2 cathode [J]. Electrochem Commun, 2006, 8: 821-826.
[17] SUN Y K, CHO S W, LEE S W, YOON C S, AMINE K. AlF3-coating to improve high voltage cycling performance of LiNi1/3Co1/3Mn1/3O2 cathode materials for lithium secondary batteries [J]. J Electrochem Soc, 2007, 154: A168-A172.
[18] PARK B C, KIM H B, MYUNG S T, AMINE K, BELHAROUAK I, LEE S M, SUN Y K.Improvement of structural and electrochemical properties of AlF3-coated LiNi1/3Co1/3Mn1/3O2 cathode materials on high voltage region [J]. J Power Sources, 2008, 178: 826-831.
[19] FEY G T, MURALIDHARAM P, LU C Z, CHO Y D. Surface modification of LiNi0.8Co0.2O2 with La2O3 for lithium ion batteries [J]. Solid State Ionics, 2005, 176: 2759-2767.
[20] SUN Y K, LEE Y S, YOSHIO M, AMINE K. Synthesis and electrochemical properties of ZnO-coated LiNi0.5Mn1.5O4 spinel as 5 V cathode material for lithium secondary batteries [J]. Electrochemical and Solid-State Letters, 2002, 5(5): A99-A102.
[21] CHO J, KIM Y J, PARK B. Novel LiCoO2 cathode material with Al2O3 coating for a Li-Ion cell [J]. Chem Mater, 2000, 12: 3788-3791.
[22] KANG Y J, KIM J H, LEE S W, SUN Y K. The effect of Al(OH)3 coating on the Li[Li0.2Ni0.2Mn0.6]O2 cathode material for lithium secondary battery [J]. Electrochim Acta, 2005, 50: 4784-4791.
[23] AURBACH D, MARKOVSKY B, RODKIN A, LEVI E, COHEN Y S, KIM H J, SCJMIDT M. On the capacity fading of LiCoO2 intercalation electrodes: The effect of cycling, storage, temperature, and surface film forming additives [J]. Electrochim Acta, 2002, 47: 4291-4306.
[24] SHAJU K M, SUBBA R G V, CHOWDARI B V R. Influence of Li-ion kinetics in the cathodic performance of layered LiNi1/3Co1/3Mn1/3O2 [J]. J Electrochem Soc, 2004, 151: A1324- A1332.
[25] KOYAMA Y, TANAKA I, ADACHI H, MAKIMURA Y, OHZUKU T. Crystal and electronic structures of superstructural Li1-x[Co1/3Ni1/3Mn1/3]O2 (0≤x≤1) [J]. J Power Sources, 2003, 119/121: 644-648.
(Edited by YANG Bing)
Foundation item: Project(50542004) supported by the National Natural Science Foundation of China; Project(1960-71131100017) supported by Graduate Degree Thesis Innovation Foundation of Central South University, China
Corresponding author: HUANG Ke-long; Tel: +86-731-88879616; E-mail: huangkelong@yahoo.com.cn
DOI: 10.1016/S1003-6326(09)60217-X

