
ARTICLE
J. Cent. South Univ. (2019) 26: 422-429
DOI: https://doi.org/10.1007/s11771-019-4014-x
Regional-scale investigation of salt ions distribution characteristics in bauxite residue: A case study in a disposal area
XUE Sheng-guo(Ѧ����)1, WANG Qiong-li(������)1, TIAN Tao(����)1,YE Yu-zhen(Ҷ����)1, ZHANG Yi-fan(��һ��)1, ZHU Feng(���)1, 2
1. School of Metallurgy and Environment, Central South University, Changsha 410083, China;
2. South China Institute of Environmental Sciences, Ministry of Environment Protection,Guangzhou 510655, China
 Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2019
Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2019
Abstract: Revegetation on bauxite residue disposal areas is the most promising strategy to reduce its potential ecological risk during stacking or disposing. Migration of salt ions in bauxite residue is one of the major issues to stimulate soil formation to support plant growth. 21 residue samples were collected and the related parameters including exchangeable cations, soluble ions, total salt, pH, electrical conductivity (EC) and exchangeable sodium percentage (ESP) were selected to evaluate alkalization and salinization of bauxite residue. High levels of ions, cation exchange capacity (TOC), total salt, exchangeable sodium percentage (ESP) and cation exchange capacity (CEC) in bauxite residue were detected with greater coefficient of variation (CV), which indicated that distribution characteristics of salt ions varied significantly. The percentage of sulfate-chloride-soda type in the residues accounted for 71.43%. The mean value of pH was 10.10, whilst mean value of ESP was 52.05%. It indicated that the residues in this case study belonged to sulfate-chloride-soda saline and alkaline soil. The research results could provide theoretical basis for soil formation in bauxite residue.
Key words: bauxite residue; bauxite residue disposal area; salt; saline-alkali soil; soil formation in bauxite residue
Cite this article as: XUE Sheng-guo, WANG Qiong-li, TIAN Tao, YE Yu-zhen, ZHANG Yi-fan, ZHU Feng. Regional-scale investigation of salt ions distribution characteristics in bauxite residue: A case study in a disposal area [J]. Journal of Central South University, 2019, 26(2): 422�C429. DOI: https://doi.org/10.1007/s11771-019-4014-x.
1 Introduction
Bauxite residue, an alkaline solid waste, is produced by the Bayer process in which bauxite is dissolved in hot NaOH [1]. The estimated global inventory of bauxite residue has reached to 4.6 billion tons, increasing by approximately 0.2 billion tons annually [2]. Large amounts of insoluble alkaline substances including sodalite, cancrinite, calcite, tricalcium aluminate, and perovskite cause bauxite residue a high value of pH (10.5�C12.8), and high Na+ concentration in solution phases leads to a high value of EC (electrical conductivity, 1.4�C28.4 mS/cm) [3, 4]. Due to the high alkalinity and salinity of bauxite residue, the utilization ratio is less than 5% [5]. A large amount of bauxite residue requires to be disposed in the disposal areas which may cause potential environmental risks [6, 7]. Management strategies are now moving towards in situ remediation in the disposal areas to reduce environmental risks and land reclamation [8, 9]. Further perspectives should be focused on the transformation process from a residue waste to a novel soil-like material to reconstruct a sustainable ecosystem in bauxite residue disposal areas [10].
Due to the typical physical and chemical properties of bauxite residue, a large number of experiments have been carried out on the mechanism of soil formation [11]. ZHU et al [12] demonstrated that natural weathering processes including climate and stacking time changed the fractal features of residue aggregates and enhanced soil formation process of bauxite residue. Gypsum and organic waste have been regarded as effective ameliorants to promote sustained rehabilitation on the disposal areas [13, 14]. ANDREW [15] illustrated that gypsum and organic matter addition lowered the alkalinity and salinity of bauxite residue and thus produced a novel substrate which was more suitable for biological colonization. ZHU et al [16] found that gypsum and vermicompost could reduce alkalinity via the transformation of alkaline phases and improve stable aggregate formation in bauxite residue.
Aggregates, the fundamental unit of soil, are formed through the combination of clay particles with organic and inorganic materials [17]. Aggregate stability, which is used to predict soil erosion and surface runoff, is affected by many factors [18]. Salt ions, especially for cations, are the major factors to determine the flocculation or dispersion process in aggregate particles. The related researches demonstrated that polyvalent metal ions including Ca2+, Mg2+, Al3+, and Fe3+ could enhance soil structure through cationic bridging between clay minerals and organic matter [19, 20]. The high concentration of Na+ resulted in dispersion of aggregates [21]. Furthermore, high alkalinity and salinity in soil restricted plant growth. Soil with an ESP (exchangeable sodium percentage) >15% and a pH>8.5 is generally classified as sodic, whilst soil with an EC of 400 mS/cm and a pH<8.5 is classified as saline [1]. In soil science, bauxite residue is both saline and sodic with an extremely high pH value. Salt management is therefore necessary in the disposal area. The current measures including gypsum addition could replace sodium to calcium from cation exchange sits. However, current rehabilitation strategies only focused on alkaline regulation of bauxite residue and its relationship between alkalinity and soil formation process, while little evidences exhibited the variation of salt ions in bauxite residues. Understanding salt ions distribution of bauxite residue was crucial to control residue desalinization, enhance aggregate formation and vegetation establishment.
A typical bauxite residue disposal area was selected as a case study to investigate the distribution characteristics of salt. The objectives of this study were: 1) to quantify distribution characteristics of salt ions in bauxite residue; 2) to identify major salt composition in bauxite residue by statistical method and 3) to evaluate degree of salinization and alkalization of bauxite residue.
2 Materials and methods
2.1 Site description and sampling
The study area locates in Northeast of Henan Province which is one of the major alumina production areas in China. The climate of the study area belongs to the warm temperate zone monsoon climate, with a mean daily temperature of 14.2�C14.8 ��C and an annual precipitation of 600�C700 mm.
There were two disposal areas. The disposal area (A) was taken into use in May 2001, and terminated in August 2015. The designed capacity of the disposal area was 16��106 m3 and the height of the dam was about 30 m. The top area (C) was capped with mineral waste residues during processing process to eliminate the potential immigration of alkaline granular and air pollution. The disposal area (B) was older and the residues had been stacked for more than 25 years. The height of dam was about 50 m. Under natural weathering processes, vegetation encroachment occurred in this area.
In October 2017, a total of 21 surface residue samples were collected using a sample shovel. The sampling depth was 20 cm. Among them, 16 of the samples were collected in the disposal area (A) and the rest were collected from the disposal area (B).For each sampling point, five samples were collected from the same point and mixed together, then the final samples were taken out using quartering method and stored in plastic bags. Each residue sample weighted approximately 2.0 kg.
2.2 Data source and processing
All of the samples were taken back to the laboratory and air dried at 40 ��C for two weeks, then sieved to <2 mm to remove gravel prior to chemical and physical analyses. The pH and EC (electrical conductivity) of the residue samples were measured in water (ratio to solid ratio water of 1:5). The supernatant was extracted by deionized water to determine soluble Ca2+, Mg2+, K+ and Na+ using inductively coupled plasma atomic emission spectroscopy [22]. The CO32�C and HCO3�C were assessed by the double indicator-neutralization titration method. SO42�C and Cl�C were determined by the barium sulfate turbidity and the silver nitrate titration, respectively [23]. Cation exchange capacity (CEC) was determined by barium chloride�Csulfuric acid titration method. Total salt was calculated by the mass percentage of water-soluble salt ions in bauxite residue. Total organic carbon (TOC) was oxidized by the potassium dichromate and determined using the ultraviolet spectrophotometry [24]. Exchangeable bases (Ca2+, Mg2+, K+, Na+) were extracted with 1 mol/L ammonium acetate (pH=7) and analyzed using inductively coupled plasma atomic emission spectroscopy. Exchangeable sodium percentage (ESP) was calculated using the following equation [16]:

Raw bauxite residue sample and residue solids (0.04�C0.05 mm) were carried out by X-ray powder diffraction (XRD). X-ray diffraction patterns ran under 2�� scan range from 10�� to 80�� at a step size of 1��/min.
2.3 Statistical analysis
The statistical analysis of the data was performed via the IBM SPSS version 20.0, Microsoft Office Excel 2007 and Origin 9.0. Descriptive statistics was used for the analysis of salinity in the residue samples. The measured data were individually determined using one-way ANOVA (analysis of variance) and homogeneity of variance tests.
3 Results and discussion
3.1 Salt composition in bauxite residue
The salt ions concentration in bauxite residue are presented in Figure 1 The mean values of soluble Ca2+, Mg2+, K+, Na+, CO32�C, HCO3�C, Cl�C and SO42�C were 86.48, 6.364, 487.5, 3420, 22969, 5243, 819.8 and 597.0 mg/kg, respectively. The contents of soluble cations were accounted as Na+>K+>Ca2+>Mg2+, and the contents of soluble anions were accounted as CO32�C>HCO3�C>Cl�C>SO42�C. It indicated that Na+, K+, CO32�C and HCO3�C were the main salt ions in bauxite residue. Among the exchangeable cations, the content of exchangeable K+ was higher than other cations. Furthermore, the contents of exchangeable Na+ and K+ were higher than those of water-soluble Na+ and K+, whilst reverse changes occurred in exchangeable Ca2+ and Mg2+. The variation of CO32�C, HCO3�C, SO42�C, Na+ and Ca2+ was mainly derived from the dissolution of various minerals in bauxite residue [25]. High total salt was one of the major limitations for plant growth on bauxite residue [11]. The mean value of pH was 10.10, whilst mean EC of residues achieved to 1.37 mS/cm. pH and ESP were the main indicators to determine the alkalization of the soil. The higher pH and ESP value indicated a stronger alkalization degree.
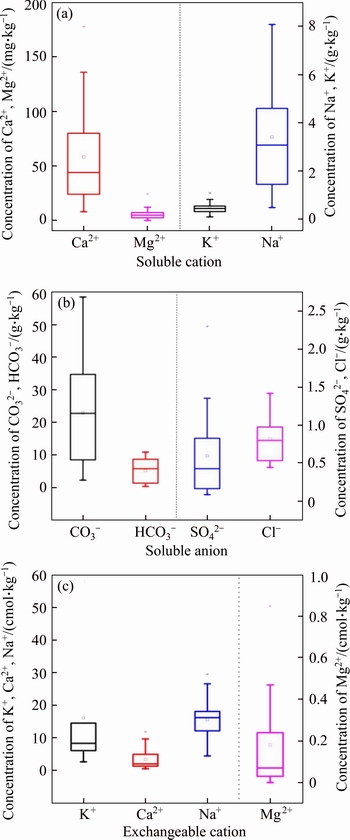
Figure 1 Salt ions concentration (a), salinity (b), and alkalinity (c) of bauxite residue samples in disposal area
Descriptive statistical analysis is a basic analysis of variables, which can explain the characteristics of variables from the perspective of traditional statistics. The descriptive statistical analysis of salt ions in bauxite residue could generalize the general situation of bauxite residue salt in the disposal area [26]. The coefficient of variation (CV) was the ratio of standard deviation to mean value, which could measure the relative dispersion degree of each test value in a set of data. CV could be classified as the weak variability (CV<0.1), the medium variability (0.1��CV��1.0) and the strong variability (CV>1.0). The coefficient of variation of ions, TOC, total salt, ESP and CEC were 0.636, 0.572, 0.423 and 0.188, respectively, and the CV of ions was strong, which indicated that the spatial distribution characteristics of these indicators were strong in variability. In addition, the CV of pH and exchangeable Ca2+ in bauxite residue had a higher variation (Table 1 and Table 2).
3.2 Mineralogical composition in bauxite residue
XRD analysis of bauxite residue revealed the compositions of chemical minerals in bauxite residue (Figure 2). The peak intensity of different XRD images could reflect the relative content of the crystal image to a certain extent. The higher value of the corresponding peak indicated a higher concentration of mineral phase. The major mineral phases included cancrinite (Na8Al6Si6O24(CO3)��(H2O)2), andradite (Ca3(Fe0.87Al0.13)2(SiO4)1.65��(OH)5.4), hematite (Fe2O3), quartz (SiO2), tri-calcium aluminate (TCA, Ca3Al2(OH)12), katoite(Ca3Al2(SiO4)3�Cx(OH)4x) and Al hydroxide (AlO(OH)). The peaks of cancrinite and andradite were the highest in all the alkaline minerals, which indicated that cancrinite and andradite were major alkaline minerals. The mineral characteristics of cancrinite and andradite in bauxite residue had positive effects on its high alkalinity. The possible equations on alkaline conversion were as follows [22]:
Ca3(Fe0.87Al0.13)2(SiO4)1.65(OH)5.4+H2O��3Ca2++(Fe0.87Al0.13)(OH)3+H4SiO4+6OH�C (1)
Na8Al6Si6O24(CO3)(H2O)2+22H2O��6Al(OH)3+6H4SiO4+6OH�C+CO32�C+8Na+ (2)
3.3 Correlation analysis of salt ions in bauxite residue
The correlation analysis of all salt ions could provide scientific basis for salt regulation of soil formation in bauxite residue. The pH value was significantly positively correlated to the CO32�C and HCO3�C, and the effect of CO32�C and HCO3�C on pH of bauxite residue were more significant than Cl�C and SO42�C (Table 3). Ca2+ and Mg2+ could replace exchangeable Na+ in soil colloids and reduce the alkalinity of bauxite residue [22]. pH was negatively correlated with Ca2+ and Mg2+, and positively correlated with Na+. There was a significant correlation between pH and Ca2+ as Ca2+ had a great effect on pH. TOC was an important indicator to evaluate soil quality and played an important role in soil chemical, physical, and biological properties [27]. TOC was positively correlated with Ca2+ and negatively correlated with other ions. Furthermore, ESP was positively correlated with Na+, HCO3�C and CO32�C, and negatively correlated with Ca2+. PAL et al [28] found that calcium could restrain the rise of pH and ESP value, which was consistent with the result in this study.
Table 1 Coefficient of variation (CV) of salt ions in bauxite residue
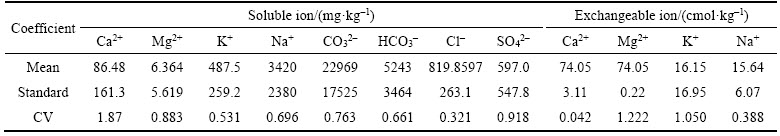
Table 2 Coefficient of variation (CV) of alkaline and saline indicators in bauxite residue


Figure 2 X-ray diffraction spectra of four bauxite residue samples
It was difficult to find independent and representative variables which could lead to further understanding on studies [29]. In this study, the relationship between water-soluble salt ions in bauxite residue was clustered and classified into three groups according to the shortest distance method (Figure 3). The results of the cluster analysis could reflect the variation rule of water- soluble salt ions. The first category was CO32�C, HCO3�C, K+ and Na+, whilst Ca2+, SO42�C and Mg2+ were in another category and Cl�C was divided into a separate category. Based on the results of cluster analysis, Na+, Cl�C and Mg2+ could reflect variation characteristics of water-soluble salt ions in bauxite residue.
3.4 Degree of salinization and alkalization in bauxite residue
The salt contents of <1 g/kg, 1�C2 g/kg, 2�C4 g/kg, 4�C6 g/kg and >6 g/kg in soils represented to the non-salinization, mild salinization, moderate salinization, severe salinization and saline soil, respectively [30]. According to the obtained data, all of bauxite residue belonged to salinization. 66.67% of bauxite residue belonged to saline soil, whilst the proportions of severe salinization, moderate salinization, mild salinization were 9.52%, 19.05%, 4.76%, respectively (Figure 4).
The classification of the soil salinization type was mainly determined by the ratio of HCO3�C, SO42�C and Cl�C. HCO3�C dominated sodas, SO42�C dominated sulfates and Cl�C dominated chlorides in soils (Table 4). Through the analysis of salt type, the main salt ions and saline-alkali soil type could be determined. Various soluble salt ions had different degrees of damage to crops. Chloride salts were the most harmful, whilst sulfate salts were relatively light [31]. In this study, there were three salt types including sulfate-chloride-soda, sulfate- soda-chloride and soda-sulfate-chloride in bauxite residue (Figure 4). HCO3�C and CO32�C accounted for the highest proportions in all saline anions and the distribution frequency of sulfate- chloride-soda was the highest among the three types in the case study.
Table 3 Descriptive statistics eigenvalues of soil salinity elements


Figure 3 Dendrogram of clustering analysis based on correlation coefficients using furthest neighbour linkage method
Table 4 Classification of soda-containing soil type based on ionic scale
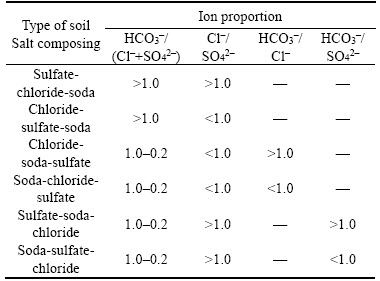
According to the related standard from American, ESP>15% and pH>8.5 were used as indicators to classify alkaline soil, which was used in this study to determine the possible soil type of bauxite residue [1]. According to Australian standard, ESP>15% and pH>9.5 were used as indicators to classify alkaline soil [32]. In China, the standard of alkaline soil depended on different soil types. LI et al [33] considered that soils with an ESP>40% and a pH>8.5 were generally classified as desert alkaline soil of China. According to American standard, all samples belonged to alkaline soil type. According to Australian standard, 90% of the residue samples belonged to alkali soil. According to Chinese standard, all bauxite residue samples belonged to alkaline soil as all samples had pH values greater than 8.5 (Figure 5). As the definition and standard of alkaline soil were not uniform, the alkaline soil type required to be further determined based on a large amount of bauxite residue samples. Due to the results from Figure 5, the soil type of the residue samples mainly belonged to alkaline soil.

Figure 4 Salt composition (a) and salinization degree (b) of bauxite residue
4 Conclusions
Na+, HCO3�C and CO32�C were the major salt ions with high coefficient of variation in bauxite residue. Na+, HCO3�C and CO32�C were positively correlated to pH and ESP. Through the clustering analysis, major water-soluble salt ions were divided into three categories to reflect variation of salt ions distribution in bauxite residue. In this case study, the salinization type of bauxite residue included sulfate-chloride-soda, sulfate-soda-chloride and soda-sulfate-chloride. The distribution frequency of sulfate-chloride-soda type was the highest among the three types, which indicated that salinized degree of bauxite residue in the disposal area belonged to salinization. The proportion of saline soil in bauxite residue was 66.67%, while the proportions of severe salinization, moderate salinization, mild salinization were 9.52%, 19.05%, 4.76%, respectively, which illustrated that the salt contents of bauxite residue were in states of dynamic change. According to the related standards about alkaline soil, the soil type of bauxite residue in this case study mainly belonged to alkaline soil.
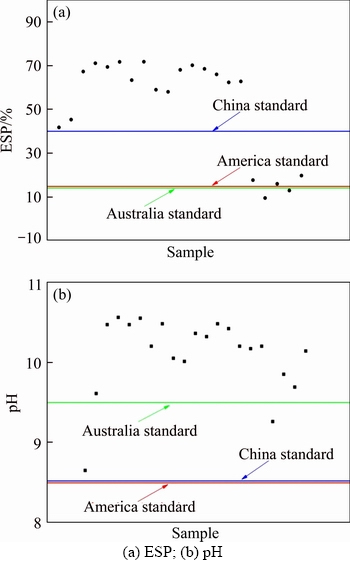
Figure 5 Alkalinization in bauxite residue samples following different standards:
References
[1] JONES B E H, HAYNES R J. Bauxite processing residue: A critical review of its formation, properties, storage, and revegetation [J]. Critical Reviews in Environmental Science & Technology, 2011, 41(3): 271�C315. DOI: 10.1080/ 10643380902800000.
[2] XUE Sheng-guo, KONG Xiang-feng, ZHU Feng, HARTLEY W, LI Xiao-fei, LI Yi-wei. Proposal for management and alkalinity transformation of bauxite residue in China [J]. Environmental Science & Pollution Research, 2016, 23(13): 12822�C12834. DOI:10.1007/s11356-016- 6478-7.
[3] ZHU Feng, LIAO Jia-xin, XUE Sheng-guo, HARTLEY W, ZOU Qi, WU Hao. Evaluation of aggregate microstructures following natural regeneration in bauxite residue as characterized by synchrotron-based X-ray micro-computed tomography [J]. Science of the Total Environment, 2016, 573(24): 155�C163. DOI: 10.1016/j.scitotenv.2016.08.108.
[4] GRAFE M, POWER G, KLAUBER C. Bauxite residue issues: III. Alkalinity and associated chemistry [J]. Hydrometallurgy, 2011, 108(1, 2): 60�C79. DOI: 10.1016/ j.hydromet.2011.02.004.
[5] LIU Wan-chao, CHEN Xiang-qing, LI Wang-xing, YU Yan-fen, YAN Kun. Environmental assessment, management and utilization of red mud in China [J]. Journal of Cleaner Production, 2014, 84: 606�C610. DOI: 10.1016/ j.jclepro.2014.06.080.
[6] KONG Xiang-feng, GUO Ying, XUE Sheng-guo, HARTLEY W, WU Chuan, YE Yu-zhen, CHENG Qin-yu. Natural evolution of alkaline characteristics in bauxite residue [J]. Journal of Cleaner Production, 2017, 143: 224�C230. DOI: 10.1016/j.jclepro.2016.12.125.
[7] GELENCSER A, KOVATS N, TUROCZI B, ROSTASI, HOFFER A, IMRE K, NYIRO K I, CSAKBERENYI M D, TOTH, CZITROVSZKY A. The Red Mud Accident in Ajka (Hungary): Characterization and potential health effects of fugitive dust [J]. Environmental Science & Technology, 2011, 45(4): 1608�C1615. DOI: 10.1021/es104005r.
[8] ZHU Feng, XUE Sheng-guo, HARTLEY W, HUANG Ling, WU Chuan, LI Xiao-fei. Novel predictors of soil genesis following natural weathering processes of bauxite residues [J]. Environmental Science & Pollution Research, 2016, 23(3): 2856�C2863. DOI: 10.1007/s11356-015-5537-9.
[9] SANTINI T C, BANNING N C. Alkaline tailings as novel soil forming substrates: Reframing perspectives on mining and refining wastes [J]. Hydrometallurgy, 2016, 164: 38�C47. DOI: 10.1016/j.hydromet.2016.04.011.
[10] KONG Xiang-Feng, TIAN Tao, XUE Sheng-guo, HARTLEY W, HUANG Long-bin, WU Chuan, LI Chu-xuan. Development of alkaline electrochemical characteristics demonstrates soil formation in bauxite residue undergoing natural rehabilitation [J]. Land Degradation and Development, 2018, 29(1): 58�C67. DOI: 10.1002/ldr.2836.
[11] ZHU Feng, LI Xiao-fei, XUE Sheng-guo, HARTLEY W, WU Chuan, HAN Fu-song. Natural plant colonization improves the physical condition of bauxite residue over time [J]. Environmental Science and Pollution Research, 2016, 23(22): 22897�C22905. DOI: 10.1007/s11356-016-7508-1.
[12] ZHU Feng, CHENG Qing-yu, XUE Sheng-guo, LI Chu-xuan, HARTLEY W, WU Chuan, TIAN Tao. Influence of natural regeneration on fractal features of residue microaggregates in bauxite residue disposal areas [J]. Land Degradation & Development, 2018, 29(1): 138�C149. DOI: 10.1002/ ldr.2848.
[13] BANNING N C, SAWADA Y, PHILLIPS I R, MURPHY D V. Amendment of bauxite residue sand can alleviate constraints to plant establishment and nutrient cycling capacity in a water-limited environment [J]. Ecological Engineering, 2014, 62(1): 179�C187. DOI: 10.1016/ j.ecoleng.2013.10.034.
[14] XEU Sheng-guo, YE Yu-zhen, ZHU Feng, WANG Qiong-li, JIANG Jun, HARTLEY W. Changes in distribution and microstructure of bauxite residue aggregates following amendments addition [J]. Journal of Environmental Sciences, 2019, 78: 276�C286. DOI: 10.1016/j.jes.2018.10.010.
[15] ANDREW W B, DOUGLAS I S, RONAN C, SIMON P R, PAUL N H, WILLIAM M M, IAN T B. Sustained bauxite residue rehabilitation with gypsum and organic matter 16 years after initial treatment [J]. Environmental Science & Technology, 2018, 52(1): 152�C161. DOI: 10.1021/acs.est. 7b03568.
[16] ZHU Feng, HOU Jing-tao, XEU Sheng-guo, WU Chuan, WANG Qiong-li, HARTLEY W. Vermicompost and gypsum amendments improve aggregate formation in bauxite residue [J]. Land Degradation & Development, 2017, 28(7): 2109�C2120. DOI: 10.1002/ldr.2737.
[17] ZHAO Jin-song, CHEN Shan, HU Rong-gui, LI Ya-yu. Aggregate stability and size distribution of red soils under different land uses integrally regulated by soil organic matter, and iron and aluminum oxides [J]. Soil & Tillage Research, 2017, 167: 73�C79. DOI: 10.1016/j.still.2016.11.007.
[18] SHI P, ARTER C, LIU Xing-yu, KELLER M, SCHULIN R. Soil aggregate stability and size-selective sediment transport with surface runoff as affected by organic residue amendment [J]. Science of the Total Environment, 2017, 607�C608: 95. DOI: 10.1016/j.scitotenv.2017.07.008.
[19] WU Xin-liang, CAI Chong-fa, WANG Jun-guang. Spatial variations of aggregate stability in relation to sesquioxides for zonal soils, South-central China [J]. Soil & Tillage Research, 2016, 157: 11�C22. DOI: 10.1016/j.still.2015. 11.005.
[20] XUE Sheng-guo, WU Yu-jun, LI Yi-wei, KONG Xiang-feng, ZHU Feng, HARTLEY W, LI Xiao-fei. Industrial wastes applications for alkalinity regulation in bauxite residue: A comprehensive review [J]. Journal of Central South University, 2019, 26(2): 268�C288.
[21] LEVY J G, TORRENTO R J. Clay dispersion and macroaggregate stability as affected by exchangeable potassium and sodium [J]. Soil science, 1995, 160(5): 352�C358. DOI: 10.1097/00010694-199511000-00004.
[22] XUE Sheng-guo, LI Meng, JIANG Jun, MILLAR G J, LI Chu-xuan, KONG Xiang-feng. Phosphogypsum stabilization of bauxite residue: Conversion of its alkaline characteristics [J]. Journal of Environmental Sciences, 2019, 77: 1�C10. DOI: 10.1016/j.jes.2018.05.016.
[23] LI Yi-wei, JIANG Jun, XUE Sheng-guo, MILLAR G, KONG, Xiang-feng, LI Xiao-fei, LI Meng, LI Chu-xuan. Effect of ammonium chloride on leaching behavior of alkaline anion and sodium ion in bauxite residue [J]. Transactions of Nonferrous Metals Society of China, 2018, 28(10): 2125�C2134. DOI: 10.1016/S1003-6326(18)64857-5.
[24] JONES B E H, HAYNES R J, PHILLIPS I R. Influence of organic waste and residue mud additions on chemical, physical and microbial properties of bauxite residue sand [J]. Environmental Science and Pollution Research, 2011, 18(2): 199�C211. DOI: 10.1007/s11356-010-0364-5.
[25] MARTIN A M, FORTE G, OSTAP S, SEE J. The mineralogy of bauxite for producing smelter-grade alumina [J]. Mineralogy Overview, 2001, 12(1253): 36�C40. DOI: 10.1007/s11837-001-0011-1.
[26] KILIC K, KILIC S. Spatial variability of salinity and alkalinity of a field having salination risk in semi-arid climate in northern Turkey [J]. Environmental Monitoring and Assessment, 2007, 127(1�C3): 55�C65. DOI: 10.1007/ s10661-006-9258-x.
[27] REEVES D W. The role of soil organic matter in maintaining soil quality in continuous cropping systems [J]. Soil & Tillage Research, 1997, 43(1): 131�C167. DOI: 10.1016/s0167-1987(97)00038-x.
[28] PAL D K, BHATTACHARYYA T, RAY S K, CHANDRAN P, SRIVASTAVA P, DURGE S L, BHUSE S R. Significance of soil modifiers (Ca-zeolites and gypsum) in naturally degraded Vertisols of the Peninsular India in redefining the sodic soils [J]. Geoderma, 2006, 136(1, 2): 210�C228. DOI: 10.1016/j.geoderma.2006.03.020.
[29] MEI J P, CHEN L. Fuzzy clustering with weighted medoids for relational data [J]. Pattern Recognition, 2010, 43(5): 1964�C1974. DOI: 10.1016/j.patcog.2009.12.00.
[30] LU Ru-kun. Soil agrochemical analysis methods [M]. Nanjing: China Agricultural Science and Technology Press, 1999. (in Chinese)
[31] MOR R P, MANCHANDA H R. Influence of phosphorus on the tolerance of table pea to chloride and sulfate salinity in a sandy soil [J]. Arid Soil Research and Rehabilitation, 1992, 6: 41�C52. DOI: 10.1080/15324989209381295.
[32] GUPTA R K, ABROL I P. Salt affected soils: Their reclamation and management for crop production [J]. Advances in Soil Science, 1990, 11: 224�C288. DOI: 10.1007/ 978-1-4612-3322-0-7.
[33] LI Shu-gang, YU Qi-li, WANG Zhou-qiong. Investigation on hydrolytic alkalinity of desert alkaline soil [J]. Acta Pedologica Sinica, 1982, 19(3): 311�C314. (in Chinese)
(Edited by YANG Hua)
���ĵ���
����ѳ��ηֲַ������μ�������
ժҪ���η�Ǩ��ת���dz������������õ���ҪӰ������֮һ��ͨ���Զѳ������μ�����IJⶨ�������ѳ������ηֲַ������μ������ԡ�����������������κ�������Ҫ�η����Ӻ����������������ӱȡ������ӽ������ı���ϵ���ϴ���ѳ��ηֲַ����������Բ��죻�������κ�����ֵΪ9.61 g/kg������������������������������-�Ȼ���-�մ���Ϊ����ռ�ȴﵽ71.43%������pH��ֵΪ10.10���ɽ��������ӱȾ�ֵΪ52.05%�����м��������������о����Ϊ�����μ���ؼ������������ṩ���������ݡ�
�ؼ��ʣ����ࣻ����ѳ����η֣��μ���������������
Foundation item: Project(41701587) supported by the National Natural Science Foundation of China; Project(PM-zx703-201803-057) supported by Basic Scientific Research Business of Central Institutes of Environmental Protection, China
Received date: 2018-10-30; Accepted date: 2018-12-11
Corresponding author: ZHU Feng, PhD, Associate Professor; Tel: +86-15573189415; E-mail: zhufeng1990@163.com; ORCID: 0000- 0002-8468-183X