J. Cent. South Univ. Technol. (2009) 16: 0876-0880
DOI: 10.1007/s11771-009-0145-9
Hydrogen sorption properties of nanocrystalline Mg2FeH6-based
complex and catalytic effect of TiO2
LIU Yi(�� �D)1, 2, TANG Sheng-long(��ʢ��)1, FANG Yu-hu(���ڻ�)1, LIU Huai-fei(������)1,
CUI Jian-min(����)3, LI Song-lin(������)1
(1. State Key Laboratory of Powder Metallurgy, Central South University, Changsha 410083, China;
2. Shenzhen Jinzhou Precision Technology Corporation Ltd, Shenzhen 518116, China;
3. Powder Metallurgy Corporation Ltd, Laiwu Iron and Steel Group, Laiwu 271105, China)
Abstract: The diversities of hydrogen sorption properties of Mg2FeH6-based complexes with and without TiO2 were investigated. Mg2FeH6-based complexes with and without TiO2 were synthesized respectively by reactive mechanical alloying, and hydrogen sorption properties of the complexes were examined by Sieverts-type apparatus. The results show that the sample without TiO2 releases 4.43% (mass fraction) hydrogen in 1.5 ks at 653K under 0.1MPa H2 pressure and absorbs 90% of the total 4.43% (mass fraction) hydrogen absorbed in 85s at 623K under 4.0MPa H2 pressure. But for the sample with TiO2 addition under the same condition, it only needs 400s to release all of the stored hydrogen and 60s to absorb 90% of the total hydrogen absorbed. The activation energies for desorption process of the samples with and without TiO2 are determined to be 71.2 and 80.3kJ/(mol��K), respectively. The improvement in hydrogen sorption rate and and reduction in activation energy can be attributed to the addition of TiO2.
Key words: Mg-based hydrogen storage materials; reactive mechanical alloying; hydrogen sorption properties; kinetics; activation energy
1 Introduction
One of the key roadblocks to the widespread use of hydrogen as a renewable fuel on board of passenger vehicles is hydrogen storage[1]. The hydrogen storage alloy is the key material of dynamical battery and fuel cell system that are used in vehicles[2]. Mg and Mg-based alloys are a group of attractive materials for hydrogen storage due to its low density and cost, abundance in the earth and high hydrogen capacity. Dimagnesium iron hydride, Mg2FeH6, has one of the highest volumetric hydrogen density of 150 kg/m3, which is double of the volumetric hydrogen density of liquid hydrogen, and a gravimetric hydrogen density of 5.66% (mass fraction), which is far larger than that of conventional hydrogen storage materials such as LaNi5 or TiFe. So Mg2FeH6 has become a hot topic in recent years.
Mg2FeH6 is more difficult to synthesize than the conventional transition metal hydride, Mg2NiH4, mainly due to the absence of intermetallic compound of the type Mg2Fe in the Mg-Fe binary system[3-4]. The traditional way of preparing Mg2FeH6 is by sintering the raw powder at high temperature (723-793 K) under high hydrogen pressure (2-12 MPa)[5]. More recently, progress has been made by mechanical alloying. The aim is to obtain a maximum yield of Mg2FeH6 and to minimize the contents of Fe, MgH2, Mg and MgO. SAI et al[3] obtained a yield of 63% (mass fraction) Mg2FeH6 by ball milling 2Mg-Fe mixture under 1 MPa H2 pressure. Shang et al[4] synthesized Mg2FeH6 by milling 3MgH2-Fe mixture under Ar atmosphere for 60 h, leading to a yield of 79% (mass fraction) of Mg2FeH6 which co-existed with 16% (mass fraction) of MgO. Puszkiel et al[6] synthesized Mg2FeH6 utilizing mechanical milling of 2Mg-Fe mixture, followed by heating at 673 K under 6 MPa H2 pressure and the obtained yield of Mg2FeH6 was about 50%. In Refs.[7-8], effects of ball milling method and starting powder mole ratio of Mg to Fe on the synthesis and hydrogen storage properties of nanocrystalline Mg2FeH6 were investigated. So far, high sorption temperatures and slow kinetics are still the main drawbacks for practical application of Mg2FeH6. In recent years, many studies have shown that the addition of transition metal oxides such as V2O5, Cr2O3, Fe3O4, TiO2 and Fe2O3 in Mg-based hydrogen storage materials can obviously improve their hydrogen sorption properties[9-11]. WANG et al[12] found that TiO2 provided a diffusion pathway for hydrogen atoms. TiO2 improves the hydrogen sorption property of Mg-TiO2 mixture at low temperature (473 K). WANG et al[13] synthesized Mg-based hydrogen storage materials with good sorption kinetics by the addition of transition metal chloride or oxide in Mg-Ni powders. SHANG et al[4] and HERRICH et al[14] added Cu and Pt respectively to Mg2FeH6 and found that there was no catalytic effect on Mg2FeH6 hydrogen sorption properties for these additives. But little work has been done interiorly on the effect of additives on Mg2FeH6.
In this work, Mg2FeH6-TiO2 nanocrystalline hydrogen storage material was synthesized at mole ratio of Mg to Fe of 3?1 and 2.5% (mole fraction) TiO2 via reactive mechanical alloying. The structure and sorption kinetic properties of the Mg2FeH6-based complexes were examined by an X-ray diffractometry (XRD) and a hydrogen sorption apparatus, respectively. The effect of TiO2 additive was investigated.
2 Experimental
Elemental powders of Mg (purity��99%, 0.841 mm) and Fe (purity��99%) were mixed with mole ratio of 3?1, and 2.5% TiO2 was used as an additive. The sample handling was performed in a glove-box fulfilled with Ar (purity 99.99%). The powders were aggregately milled for 150 h under reactive H2 atmosphere. The samples with and without addition of TiO2 were designated as 3MFHTOL150 and 3MFH150, respectively. The details of the processing parameters are shown in Table 1.
Table 1 Processing parameters for reactive mechanical alloying of samples 3MFH150 and 3MFHTOL150

The phase compositions were tested by Rigaku D/max2550 diffractometer using Cu K�� radiation (��=0.154 18 nm). The grain size of phases was calculated using Scherrer equation. Hydrogen sorption properties of the milled powders were measured by a Sieverts-type apparatus. The activation energy for desorption process was estimated from Arrhenius plot of reaction rate constant K with temperature [15-20]:
K=K0exp[-Q/(RT)] (1)
where K is the reaction rate constant; K0 is the coefficient; Q is the activation energy; R is the gas constant; and T is the temperature. K is determined by fitting the desorption curves with the Johnson-Mehl-Avrami (JMA) equation:
��=1-exp[-(Kt)n] (2)
where n is the reaction exponent; and �� is the desorption fraction at time t.
3 Results and discussion
3.1 Synthesis of nanocrystalline Mg2FeH6-TiO2 complex
XRD patterns of powders with TiO2 as a function of reactive milling time are shown in Fig.1. At the initial step, Mg reacts with H2 to form MgH2, and then Mg2FeH6 is formed according to the following reaction[21-22]:
3MgH2+Fe Mg2FeH6+Mg (3)
Mg2FeH6+Mg (3)
Weak MgO diffraction peak appears after milling for 105 h, and the peaks of Mg2FeH6 and Fe on the XRD patterns are obviously broadened after milling for 150 h. There is no phase corresponding to TiO2 peaks observed, which is due to the small amount of TiO2 addition in the starting milling powder and the rapid reduction of TiO2 grain size.

Fig.1 XRD patterns of powders with TiO2 (3MFHTOL150) as function of reactive milling time
Table 2 certifies that there is an obvious reduction of grain size of each phase with increasing ball-milling
Table 2 Grain size (d) of each phase in milled powders after ball-milling for different time calculated from XRD patterns
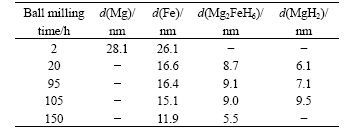
time. After milling for 2 h, the grain sizes of Mg and Fe are in the range of 20-30 nm, respectively. After milling for 20 h the grain sizes of MgH2 and Mg2FeH6 are 6.1 and 8.7 nm, respectively. The grain size of Mg2FeH6 becomes 5.5 nm after milling for 150 h. The nano- structure is favorable to the hydrogen sorption properties of the hydrogen storage material.
3.2 Hydrogen sorption properties of nanocrystalline Mg2FeH6-TiO2 and Mg2FeH6-based complex
The nanocrystalline Mg2FeH6-based complex synthesized by reactive mechanical alloying can desorb/absorb hydrogen directly without any activation, because the nanocrystalline Mg2FeH6-based complex has more crystal boundaries that can effectively increase the diffusion passageway for hydrogen atoms[23]. The hydrogen absorption and desorption curves of Mg2FeH6-based complex at different temperatures are shown in Fig.2.
Fig.2(a) shows that the as-synthesized Mg2FeH6- based complex possesses good hydrogen absorption performance. Only 85 s is needed for the accomplish- ment of 90% of hydrogen absorption at 623 K under 4.0 MPa H2 pressure. The synthesized powder can absorb 4.43% (mass fraction) hydrogen at 623 K under 4.0 MPa H2 pressure. It also takes only 90 s to accomplish 90% of hydrogen absorption and aggregately absorb 4.13%
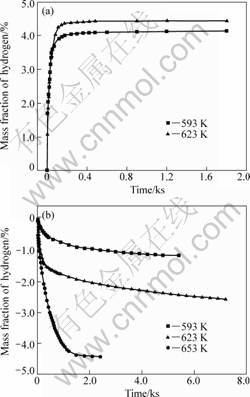
Fig.2 Hydrogen absorption��desorption curves of sample 3MFH150 from Sieverts-type apparatus at different temperatures: (a) Absorption curves (Initial H2 pressure 4.0 MPa); (b) Desorption curves (Desorption under 0.1 MPa H2 pressure)
hydrogen even at 593 K under 4.0 MPa H2 pressure.
The hydrogen desorption curves of the 3MFH150 sample are shown in Fig.2(b). Hydrogen desorption to 90% capacity of sample 3MFH150 can be accomplished in 1 ks and it completely releases 4.43% hydrogen at 653 K under 0.1 MPa H2 pressure in 1.5 ks. However, the hydrogen amount and desorption rate decrease remarkably with the decrease of desorption temperature. Sample 3MFH150 only releases 2.57% hydrogen at 623 K in 7.2 ks and 1.13% hydrogen at 593 K.
Hydrogen absorption curves of sample 3MFHTOL150 at different temperatures are shown in Fig.3(a). It can be seen that sample 3MFHTOL150 possesses good hydrogen absorption kinetics. More than 90% of hydrogen absorption is accomplished in 60 s at 653 K under 4.0 MPa H2 pressure, and finally hydrogen capacity reaches 3.12%. Compared with sample 3MFH150, it only takes 60 s to accomplish 90% of hydrogen absorption for sample 3MFHTOL150. Even though the temperature drops to 593 K, sample 3MFHTOL150 also displays higher hydrogen absorption rate, but the hydrogen capacity falls to 2.34%.
Sample 3MFHTOL150 also exhibits good hydrogen desorption kinetics (Fig.3(b)). The hydrogen desorption is accomplished in 400 s at 653 K and completely releases 2.58% hydrogen. The hydrogen desorption rate
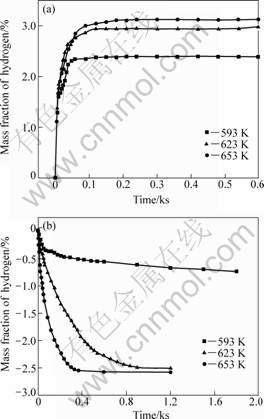
Fig.3 Hydrogen absorption��desorption curves of sample 3MFHTOL150 at different temperatures: (a) Absorption curves (Initial hydrogen pressure 4.0 MPa); (b) Desorption curves (Desorption under 0.1 MPa H2 pressure)
decreases with the decrease of temperature, but the amount of hydrogen desorption is less affected by the temperature when the temperature exceeds 623 K.
3.3 Hydrogen desorption kinetics of nanocrystalline Mg2FeH6-based complex and catalytical effect of TiO2
In order to further investigate the catalytical efficiency of TiO2 in Mg2FeH6-TiO2 complex, hydrogen desorption curves of samples 3MFH150 and 3MFHTOL150 were analyzed. The hydrogen desorption curves of samples 3MFH150 and 3MFHTOL150 at temperatures of 593, 623 and 653 K under 0.1 MPa H2 pressure are shown in Fig.4. By comparing the hydrogen

Fig.4 Hydrogen desorption curves of composite powders desorbed at 653 K (a), 623 K (b) and 593 K (c) under 0.1 MPa H2 pressure
desorption curves at different temperatures under 0.1 MPa H2 pressure, it is found that sample 3MFHTOL150 has higher hydrogen desorption rate that releases 90% of hydrogen capacity in 300 s at 653 K. This sample still keeps high hydrogen desorption rate even when the temperature falls to 623 K.
The reaction exponent n in Eq.(2) for all samples was taken as 3[20]. From the Arrhenius plot of K with temperature (see Fig.5, where R2 is the degree of fitting), the activation energies of desorption for samples 3MFH150 and 3MFHTOL150 are calculated to be 80.3 and 71.2 kJ/(mol��K), respectively. Czujko et al[20] estimated the activation energies of desorption for Mg+10% X (X=V, Y, Zr) submicrocrystalline composites from similar plot. By taking the data into Eqs.(1) and (2), the hydrogen desorption equations of samples 3MFH150 and 3MFHTOL150 are given respectively as follows:
��=1-exp{-[11 669texp(-9 652/T)]3} (4)
��=1-exp{-[3 707texp(-8 562/T)]3} (5)
The addition of TiO2 obviously improves hydrogen absorption/desorption rate. Several explanations are available: (1) TiO2 can accelerate the decomposition of molecular hydrogen and its c-axis is a passageway for fast diffusion of hydrogen atom; (2) TiO2 nano-particles
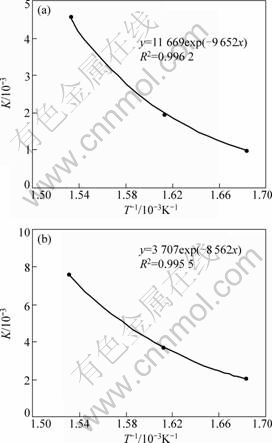
Fig.5 Arrhenius plot curves for samples 3MFH150 (a) and 3MFHTOL150 (b)
distributed on the surface of matrix speed up hydro- genation rate at the beginning, and those distributed in matrix supply a freeway for hydrogen atom diffusing into matrix[11]; and (3) TiO2 can destroy the layer of oxygenation.
4 Conclusions
(1) The as-synthesized sample 3MFH150 releases 4.43% hydrogen in 1.5 ks at 653 K under 0.1 MPa H2 pressure and absorbs 90% of total 4.43% hydrogen stored in 85 s at 623 K under 4.0 MPa H2 pressure. It also indicates that the hydrogen storage material Mg2FeH6-based complex synthesized by reactive mechanical alloying can release/absorb hydrogen directly without any activation.
(2) At 653 K under 0.1 MPa H2 pressure, it only needs 400 s to release all of hydrogen for sample 3MFHTOL150. At 623 K under 4.0 MPa H2 pressure, it only takes 60 s to absorb 90% of hydrogen stored. The addition of TiO2 improves hydrogen absorption/ desorption rate of Mg2FeH6-based complex.
(3) The activation energies for desorption process of samples 3MFH150 and 3MFHTOL150 are 80.3 and 71.2 kJ/(mol��K), respectively. The addition of TiO2 helps to the decrease of the activation energy for Mg2FeH6 desorption process, which gives the kinetical explanation for the higher desorption rate of Mg2FeH6-TiO2 complex.
References
[1] Ritter J A, Ebner A D, Wang J, ZIDAN R. Implementing a hydrogen economy[J]. Materials Today, 2003, 6(9): 19-23.
[2] LIU Kai-yu, HUANG Bai-yun, ZHANG Ping-min, HE Yue-hui, ZHOU Ke-chao, SU Geng. Application of thermal electrochemical equation to metal-hydride half-cell system[J]. Journal of Central South University of Technology, 2006, 13(2): 146-150.
[3] Sai R S S, Davidson D J, Bobet J L, Srivastava O N. Investigations on the synthesis, structural and microstructural characterizations of Mg-based K2PtCl6 type (Mg2FeH6) hydrogen storage material prepared by mechanical alloying[J]. Journal of Alloys and Compounds, 2002, 333: 282-290.
[4] Shang C X, Bououdina M, Gao Z X. Direct mechanical synthesis and characterization of Mg2Fe(Cu)H6[J]. Journal of Alloys and Compounds, 2003, 351/356: 626-629.
[5] Didisheim J J, Zolliker P, Yvon K, FISCHER P, SCHEFER J, GUBELMANN M, WILLIAMS A F. Magnesium iron hydride, Mg2FeH6, containing octahedral FeH64- anions[J]. Inorganic Chemistry, 1984, 23(13): 1953-1957.
[6] PUSZKIEL J A, LAROCHETTE P A A, GENNARI F C. Thermodynamic and kinetic studies of Mg-Fe-H after mechanical milling followed by sintering[J]. Journal of Alloys and Compounds 2008, 463: 134-142.
[7] LI S L, LIU Y, VARIN R A, LIU H F, CUI J M, CHEN S Q. Effect of ball milling methods on synthesis and desorption properties of nanocrystalline Mg2FeH6 hydrogen storage materials[J]. The Chinese Journal of Nonferrous Metals, 2008, 18(1): 42-47. (in Chinese).
[8] LI Song-lin, LIU Yi, CUI Jian-min, YANG Wen-zhi, LI Hao-peng, HE Yi-lun. Synthesis and hydrogen desorption properties of Mg2FeH6 hydrogen storage material by reactive mechanical alloying[J]. Journal of Central South University: Science and Technology, 2008, 39(1): 1-6. (in Chinese).
[9] Jin S A, Shim J H, Ahn J P, CHO Y W, YI K W. Improvement in hydrogen sorption kinetics of MgH2 with Nb hydride catalyst[J]. Acta Materialia, 2007, 55(15): 5073-5079.
[10] Hanada N, Ichikawa T, Hino S, FUJII H. Remarkable improvement of hydrogen sorption kinetics in magnesium catalyzed with Nb2O5[J]. Journal of Alloys and Compounds, 2006, 420: 46-49.
[11] Barkhordarian G, Klassen T, Bormann R. Effect of Nb2O5 content on hydrogen reaction kinetics of Mg[J]. Journal of Alloys and Compounds, 2004, 364: 242-246.
[12] Wang P, Wang A M, Zhang H F, DING B Z, HU Z Q. Hydrogenation characteristics of Mg-TiO2 (rutile) composite[J]. Journal of Alloys and Compounds, 2000, 313: 218-223.
[13] WANG Er-de, LEI Zheng-long, YU Zhen-xing. A review on the development of Mg-based hydrogen storage materials[J]. Powder Metallurgy Technology, 2003, 21(1): 31-36. (in Chinese)
[14] Herrich M, Ismail N, Lyubina J, HANDSTEIN A, PRARR A, GUTFLEISCH O. Synthesis and decomposition of Mg2FeH6 prepared by reactive milling[J]. Materials Science and Engineering B, 2004, 108(1/2): 28-32.
[15] Graetz J, Reilly J J. Kinetically stabilized hydrogen storage materials[J]. Scripta Materialia, 2007, 56(10): 835-839.
[16] YU Zhen-xing, WANG Er-de, ZHANG Wen-cong, FANG Wen-bin, SUN Hong-fei, LIANG Ji. Shell and shrinking core kinetics model of Mg-based hydrogen storage alloys[J]. Trans Nonferrous Met Soc China, 2005, 15(1): 178-182.
[17] HU Lian-xi, WANG Er-de. Hydrogen generation via hydrolysis of nanocrystalline MgH2 and MgH2-based composites[J]. Trans Nonferrous Met Soc China, 2005, 15(5): 965-970.
[18] Hout J, Pelletier J F, Lurio L B, SUTTON M, SCHULZ R. Investigation of dehydrogenation mechanism of MgH2-Nb nanocomposites[J]. Journal of Alloys and Compounds, 2003, 348: 319-324.
[19] Hout J, Liang G, Boily S, VAN NESTE A, SCHULZ R. Structural study and hydrogen sorption kinetics of ball-milled magnesium hydride[J]. Journal of Alloys and Compounds, 1999, 293/295: 495-500.
[20] Czujko T, Varin R A, Chiu C, WRONSKI Z. Investigation of the hydrogen desorption properties of Mg+10%X (X=V, Y, Zr) submicrocrystalline composites[J]. Journal of Alloys and Compounds, 2006, 414: 240-247.
[21] ZHOU D W, LI S L, VARIN R A, PENG P, LIU J S, YANG F. Mechanical alloying and electronic simulations of 2Mg-Fe mixture powders for hydrogen storage[J]. Materials Science and Engineering A, 2006, 427(1/2): 306-315.
[22] Gennari F C, Castro F J, Gamboa J J A. Synthesis of Mg2FeH6 by reactive mechanical alloying: Formation and decomposition properties[J]. Journal of Alloys and Compounds, 2002, 339: 261-267.
[23] PENG Neng, LIANG Zhen-feng, XIAO Fang-ming, WANG Ying, LU Qi-yun, TANG Ren-heng. Study on nanocrystalline rare earth Mg-based system hydrogen storage alloys with AB3-type[J]. Materials Research and Application, 2007, 1(1): 23-36. (in Chinese).
(Edited by CHEN Wei-ping)
Foundation item: Project (50574105) supported by the National Natural Science Foundation of China; Project (NCET-06-0683) supported by the Program for the New Century Excellent Talents in University; Project (08-030239) supported by the Program for 121 Excellent Talents in Hunan Province; Project (07MX21) supported by Mittal Student Innovation Foundation of Central South University
Received date: 2009-01-17; Accepted date: 2009-04-10
Corresponding author: LI Song-lin, Professor; Tel: +86-731-88830614; E-mail: lisl@mail.csu.edu.cn