600 ��C�ȴ���ʱAl-4Ni-2Mn�Ͻ���Al9(Mn,Ni)2������������Ϊ
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2018���10��
�������ߣ������� ������ ��ٻ
����ҳ�룺1913 - 1919
�ؼ��ʣ�Al-Ni-Mn�Ͻ���䣻�������ࣻ��������
Key words��Al-Ni-Mn alloys; phase transformation; quasicrystal approximant; slip phenomenon
ժ Ҫ������ɨ���������������������о�600 ��C�ȴ���ʱAl-4Ni-2Mn�Ͻ���Al9(Mn,Ni)2���������ò�ݱ估��ṹת�䡣������������ȴ��������У���ά״��Al9(Mn,Ni)2�����༸��ȫ��ת���������״����״���������ң������ȴ���ʱ����ӳ�����������Al9(Mn,Ni)2����������Oswald�컯�����⣬ͨ���о�֤ʵ�ȴ��������е�ȷ������Al9(Mn,Ni)2��O��������̡����������Ŀ�����ò������O��������Al9(Mn,Ni)2����ض��������κˣ�Ȼ����һ������ĸ�����������������У�����O���(010)[001]����ϵ�������ˣ���һЩO�������������˻��Ƽ��ߡ�
Abstract: The morphology evolution and phase transformation of Al9(Mn,Ni)2 eutectic phase in an Al-4Ni-2Mn alloy during heat treatment at 600 ��C were studied by scanning electron microscopy (SEM) and transmission electron microscopy (TEM). Results show that nearly all of the eutectic fibers change into prolate ellipsoid and spherical particles in the process of heat treatment, and Ostwald ripening phenomenon occurs in the eutectic region with the increase of the heat treatment time. Besides, a phase transformation from Al9(Mn,Ni)2 to O-phase is confirmed. The morphologies of the transformed particles indicate that the O-phase preferentially nucleates on the specific crystal plane of the Al9(Mn,Ni)2 eutectic phase and grows in a certain direction. During the phase transformation, the (010)[001] slip system in O-phase is activated, and the resultant slip traces appear on the surface of some O-phase particles.
Trans. Nonferrous Met. Soc. China 28(2018) 1913-1919
Wen-tao YU, Qi-tang HAO, Qian WANG
State Key Laboratory of Solidification Processing, School of Materials Science and Engineering, Northwestern Polytechnical University, Xi��an 710072, China
Received 23 September 2017; accepted 8 March 2018
Abstract: The morphology evolution and phase transformation of Al9(Mn,Ni)2 eutectic phase in an Al-4Ni-2Mn alloy during heat treatment at 600 ��C were studied by scanning electron microscopy (SEM) and transmission electron microscopy (TEM). Results show that nearly all of the eutectic fibers change into prolate ellipsoid and spherical particles in the process of heat treatment, and Ostwald ripening phenomenon occurs in the eutectic region with the increase of the heat treatment time. Besides, a phase transformation from Al9(Mn,Ni)2 to O-phase is confirmed. The morphologies of the transformed particles indicate that the O-phase preferentially nucleates on the specific crystal plane of the Al9(Mn,Ni)2 eutectic phase and grows in a certain direction. During the phase transformation, the (010)[001] slip system in O-phase is activated, and the resultant slip traces appear on the surface of some O-phase particles.
Key words: Al-Ni-Mn alloys; phase transformation; quasicrystal approximant; slip phenomenon
1 Introduction
During the last two decades, Al-based alloy systems including transition metals (TMs) have attracted increasing attention due to a multitude of binary and ternary intermetallics they often contain. Such as Al-Ni-Mn alloy system, studies have shown that several kinds of intermetallics can be formed in this alloy system when the material produced by different casting processes. Besides the binary intermetallics Al6Mn, Al4Mn, and Al3Ni, at least six ternary phases have been reported up to now [1-4], including three ternary thermodynamically stable intermetallics, the ��-phase (Al5Co2-type, hP26, P63/mmc; a=0.76632(16) nm, c=0.78296(15) nm), the ��-phase (��-Al14.4Cr3.4Nil.1-type, hP227, P63/m; a=1.7625(10) nm, c=1.2516(10) nm), and the O-phase (O-Al77Cr14Pd9-type, Pmmn, oP650; a=2.3316(16) nm, b=1.2424(15) nm, c=3.2648(14) nm), as well as three ternary metastable phases, the decagonal D3-phase with periodicity about 1.25 nm, the Al9(Mn,Ni)2-phase (Al9Co2-type, P1121/a, mP22; a=0.8585(16) nm, b=0.6269(9) nm, c=0.6205(11) nm, ��=95.34(10)��) and the O1-phase (base-centered orthorhombic, a��2.38 nm, b��1.24 nm, c��3.22 nm).
Although several intermetallics in Al�CNi�CMn alloy have been reported in recent years, little research has been conducted on the phase transformation of the known metastable phase. Taking Al9(Mn,Ni)2 phase as an example, this ternary compound has been firstly reported by BALANETSKYY et al [2]. However, the phase transformation of the Al9(Mn,Ni)2 phase is not clear. Recently, we observed that the Al9(Mn,Ni)2 eutectic phase can transform into a ternary hexagonal ��-phase after heat treatment at 350-550 ��C for a long time [5]. However, it should be noted that a new phase transformation appears in the Al-4Ni-2Mn alloy when the heat treatment temperature rises to 600 ��C. It is important to reveal this phase transformation process, because the research can not only enrich the understanding of Al9(Mn,Ni)2 phase but also lay a foundation for the further study of Al-4Ni-2Mn alloy. In this work, we will mainly discuss the phase transformation of Al9(Mn,Ni)2 based on SEM and TEM observation.
2 Experimental
Al-4Ni-2Mn (mass fraction, %, used throughout the paper unless noted) alloys were melted in a silicon carbide crucible in an electric resistance furnace by using high purity aluminum (99.99%), Al-10Ni and Al-10Mn master alloys. After refining with sodium-free refining agent, the melt was poured at 750 ��C into a steel mold preheated up to about 250 ��C. The composition was measured to be Al-3.67Ni-2.31Mn by using inductively coupled plasma optical emission spectrometry (ICP-OES). The heat treatment was performed in an electric resistance furnace with a temperature accuracy of ��2 ��C. The as-cast samples were heat treated at 600 ��C for 75, 312, and 432 h, respectively, and then quenched into water. The microstructures of eutectic phase were observed by ZEISS SUPRATM 55 scanning electron microscope. Phase transformation was identified using the selected area electron diffraction (SAED) and energy dispersive X-ray spectroscopy (EDX) in TecnaiTM G2 F30 transmission electron microscopy operated at 300 kV. Samples for SEM observation were prepared using standard metallographic techniques. Thin foils for TEM investigation were ground to a thickness of about 100 ��m and then punched into discs with a diameter of 3 mm. Finally, the discs were thinned using dimpling and ion milling.
3 Results and discussion
3.1 Morphological change during heat treatment
Figure 1 shows the eutectic microstructures in the specimen heat-treated at 600 ��C for 75, 312, and 432 h, respectively. In contrast to the morphology of the eutectic solidification microstructure in the as-cast Al-4Ni-2Mn alloy [6], an obvious change in the morphology of the Al9(Mn,Ni)2 eutectic phase can be observed during the heat treatment at 600 ��C. As can be seen, nearly all of the eutectic fibers have changed into prolate ellipsoid and spherical particles. With the increase of the heat treatment time, Ostwald ripening of the eutectic particles appears in the eutectic region. This coarsening phenomenon is particularly evident in the sample after heat treatment at 600 ��C for 432 h (Fig. 1(e)).
However, the detailed morphology evolution mechanism of the Al9(Mn,Ni)2 eutectic phase is beyond of the scope of the present investigation. In this study, we focus our attention on the structural phase transition during heat treatment at 600 ��C. Observations under higher magnification, i.e., the backscattered electron images shown in Figs. 1(b), (d) and (f), demonstrate that a phase transformation occurred during heat treatment at 600 ��C. As can be seen, the untransformed parts of the particles have a white contrast, while the transformation products have a darker contrast. Furthermore, the morphology characteristics of the partly transformed particles indicate that the new-phase particles are likely to nucleate at the interface between the Al matrix and Al9(Mn,Ni)2 eutectic phase, and then grow into the Al9(Mn,Ni)2 particles. During the growth of new-phase particles, a distinct phase boundary can be found on the partly transformed particles.

Fig. 1 Backscattered electron images of samples heat treated at 600 ��C for different time
3.2 Determination of phase transformation behavior
The phase transformation of Al9(Mn,Ni)2 eutectic phase has also been characterized by TEM. Figure 2(a) presents the morphology of the particle after phase transformation. The EDX result shows that the phase transformation product is a ternary intermetallic with a composition of Al79.70Mn13.51Ni6.78 (mole fraction, %), which is very close to the stoichiometric composition of the Al60Mn11Ni4 and C3,I-Al60Mn11Ni4 phases. However, it should be noted that even if the Al60Mn11Ni4 and C3,I-Al60Mn11Ni4 phases have the same nominal composition, the structure parameters of these two phases are not exactly the same. The Al60Mn11Ni4 phase is an orthorhombic intermetallic compound with the structure parameters: Bbmm, Al31Mn6Ni2-type, a=0.755 nm, b=1.25 nm and c=2.38 nm (the unit-cell parameters have been rearranged according to present systematization) [7]. The C3,I-Al60Mn11Ni4 phase is also an orthorhombic intermetallic compound, but it has different structure parameters: Pmmm, oP650, O-Al77Cr14Pd9-type; a=3.27 nm, b=1.24 nm and c= 2.40 nm [1,8]. In particular, orthorhombic Al60Mn11Ni4 and C3,I-Al60Mn11Ni4 phases belong to the (1/0, 2/1) and (3/2, 2/1) approximants of a decagonal quasicrystal (DQC), respectively. They have almost the same parameter c and the parameter a is rough in t3 (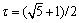 ) ratio [9]. Besides, it should be noted that the C3,I-Al60Mn11Ni4 phase is also termed as O-phase in Refs. [1,2], so we use O-phase to denote the C3,I-Al60Mn11Ni4 phase in this work. In order to identify the phase transformation product, typical SAED patterns have been taken from the transformed particle. As shown in Figs. 2(b) and (c), all of the SAED patterns can be consistently indexed based upon the O-phase with an orthorhombic structure. In addition, it can be found that the diffraction patterns shown here present strong spots in a hexagonal formation, which is a significant characteristic of the O-phase, as reported in Ref. [9]. Furthermore, based on the orthorhombic structure, the lattice parameters a, b and c of the phase transformation product can be easily determined to be a=3.33 nm, b=1.25 nm and c=2.42 nm from the SAED patterns, the results obtained in this study are in good agreement with that of O-phase reported by TENDELOO et al [8].
) ratio [9]. Besides, it should be noted that the C3,I-Al60Mn11Ni4 phase is also termed as O-phase in Refs. [1,2], so we use O-phase to denote the C3,I-Al60Mn11Ni4 phase in this work. In order to identify the phase transformation product, typical SAED patterns have been taken from the transformed particle. As shown in Figs. 2(b) and (c), all of the SAED patterns can be consistently indexed based upon the O-phase with an orthorhombic structure. In addition, it can be found that the diffraction patterns shown here present strong spots in a hexagonal formation, which is a significant characteristic of the O-phase, as reported in Ref. [9]. Furthermore, based on the orthorhombic structure, the lattice parameters a, b and c of the phase transformation product can be easily determined to be a=3.33 nm, b=1.25 nm and c=2.42 nm from the SAED patterns, the results obtained in this study are in good agreement with that of O-phase reported by TENDELOO et al [8].
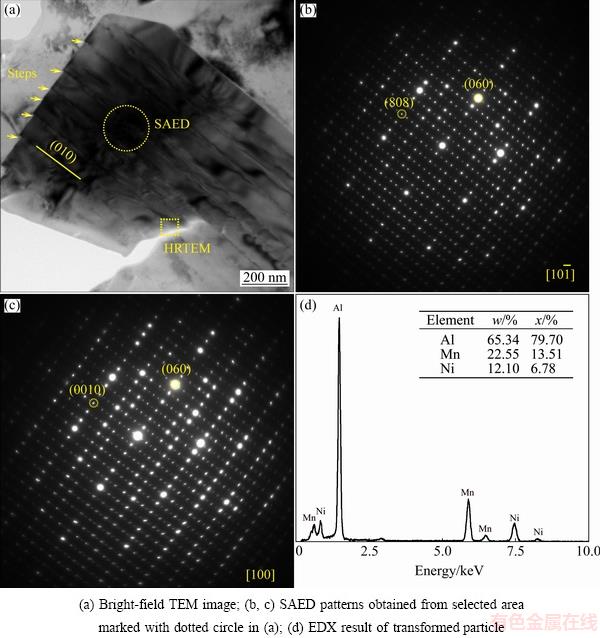
Fig. 2 TEM image of transformed product in specimen heat treated at 600 ��C for 312 h, SAED patterns and EDX result
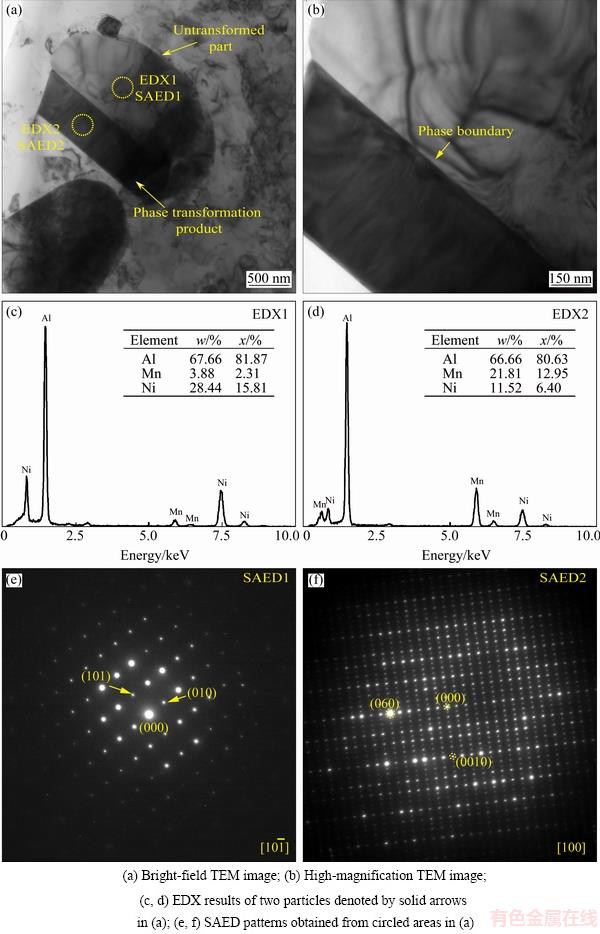
Fig. 3 TEM images of partly transformed eutectic particle in specimen heat treated at 600 ��C for 312 h, SAED patterns and EDX results
In order to further confirm that the phase transformation occurred in the specimen during the heat treatment at 600 ��C, many partly transformed particles were examined by TEM. One of the particles is shown in Fig. 3(a), bright-field TEM image shown in Fig. 3(b) is the high-magnification morphology of this particle. As can be seen, during the phase transformation, an unambiguous phase boundary formed between the parent phase and the phase transformation product. The two particles show different contrast even in the bright-field image. Figures 3(c) and (d) are the EDX results obtained from the two particles in Fig. 3(a) respectively, and corresponding SAED patterns are shown in Figs. 3(e) and (f). By SAED patterns together with the composition of the particles measured by EDX, the two particles have been identified to be the untransformed Al9(Mn,Ni)2 eutectic particle and transformation product (O-phase), respectively, as denoted in Fig. 3(a). These results confirm that the phase transformation from Al9(Mn,Ni)2 to O-phase occurred during the heat treatment at 600 ��C.
From the above analysis of phase transformation, it can be seen that the composition of transformation product is different from that of the parent phase. This means that the phase transformation observed in the present study is accompanied by redistribution of Ni and Mn atoms between the Al9(Mn,Ni)2 and O-phase. However, it should be noted that the Al9(Mn,Ni)2 phase is enriched with Ni atoms and the O-phase is enriched with Mn atoms, therefore, a large number of Mn atoms are required for the phase transformation to proceed. It is believed that the Mn solute atoms and Mn-containing particles in the Al matrix can supply Mn atoms for the phase transformation. But beyond that, the dissolution of the Al9(Mn,Ni)2 eutectic phase is considered to be an important contributing factor. Because the morphology evolution is always accompanied by the dissolution of eutectic phase [10]. In this case, a large amount of Ni and Mn atoms dissolve into the surrounding ��(Al), resulting in the enrichment of Ni and Mn atoms around the undissolved Al9(Mn,Ni)2 eutectic particles, which is beneficial for the nucleation of O-phase.
Furthermore, as can be seen from Fig. 1, it can be concluded that the nucleation of the O-phase particle most probably occurs at the boundary between the Al matrix and the Al9(Mn,Ni)2 eutectic particle, because nucleation at boundary zone is encouraged by several factors. First, the phase boundary has a high energy and the nucleation energy barrier can be easily overcome at the boundary. Second, the atomic diffusion is fast at the phase boundary, which can easily cause a relatively strong concentration fluctuation, and local solute concentration at boundary zone will satisfy the nucleation condition of the O-phase. In addition, the morphologies of the transformed particles shown in Fig. 1(f) also manifest that the O-phase preferentially nucleates on the specific crystal plane of the Al9(Mn,Ni)2 eutectic phase (i.e. the habit plane) and then grows in a certain direction in order to minimize the interface energy. As is reported in Ref. [6], Al9(Mn,Ni)2 has a low-symmetry monoclinic structure, which has few equivalent crystal planes. This means that the habit plane is probably a certain crystal plane of the Al9(Mn,Ni)2 eutectic phase. In this case, the phase transformation of Al9(Mn,Ni)2 during heat-treatment is likely to occur only at the specific interface between the Al9(Mn,Ni)2 and Al matrix. Moreover, Al9(Mn,Ni)2 eutectic phase in a eutectic cell has the same orientation, therefore, the transformed particles in the same eutectic cell will show similar phase transition behavior. This conclusion has been confirmed by the experimental results. As shown in Fig. 1(f), there are three transformed particles in the eutectic region. It is apparent that the phase transformation appears on the same side of the Al9(Mn,Ni)2 eutectic particles, and the interface between Al9(Mn,Ni)2 and O-phase nearly has the same orientation.
3.3 Slip phenomenon in new-phase particle
The slip traces were observed on the surface of the transformed particle, as shown in Fig. 2(a). In order to further investigate the slip phenomenon, high-resolution transmission electron microscopy (HRTEM) was used to characterize the O-phase particle. Figure 4(a) shows the high resolution TEM image of the selected rectangular region in Fig. 2(a). Figures 4(b) and (c) are the diffraction patterns obtained via Fast Fourier Transform (FFT) of the selected square (indicated as numbers 1 and 2) in Fig. 4(a), respectively. By comparing the two FFT patterns, it can be found that the FFT pattern obtained from the second area shows a characteristic of stacking fault displaying streak lines perpendicular to the (010) plane, which is believed to be the slip plane of O-phase. According to the classical theory of plastic deformation [11-14], different types of lattice have different slip systems (slip planes and slip directions). The dominant slip plane of orthorhombic system is (010) plane, slip direction is parallel to the [100] or [001] zone axis [15,16]. In the present study, it should be noted that the bright-field TEM image shown in Fig. 2(a) is taken along the [100] zone axis, and the corresponding SAED pattern (after calibration of magnetic declination) is shown in Fig. 2(c). According to the characteristics of slip steps and the SAED pattern, it can be concluded that the slip direction is parallel to the [001] zone axis of O-phase. Therefore, the results show that the (010)[001] slip system in O-phase was activated during the phase transformation.

Fig. 4 High resolution TEM images taken along  zone axis and FFT patterns
zone axis and FFT patterns
However, it should be noted that the existing experimental results are unable to fully reveal the primary cause of the slip phenomenon. It is believed that the activation of this slip system may be related to the elastic strain energy induced by the phase transformation. Although the microscopic structure of the interface between the Al9(Mn,Ni)2 eutectic phase and the O-phase is still not clear, the morphologies of the transformed particles show that the O-phase preferentially nucleates on specific crystal plane of the parent phase. Even though the habit-plane index cannot be determined in the present study, the existence of habit plane means that there exists a definite orientation relationship between the new phase and the parent phase, implying that the two-phase interface is coherent or semi-coherent. In this case, the lattice distortion near the phase interface is inevitable owing to the difference of lattice structure between the new phase and the parent phase, resulting in high elastic strain energy at the interface. Besides, the phase transformation is always accompanied by a volume change as result of the difference of specific volume between the parent phase and the new phase. Nevertheless, it is impossible for the new phase to be free to contract and expand under the constraint of surrounding parent phase. Thus, the elastic strain energy is stored during the phase transformation. The strain energy induced by phase transformation increases with the growth of the O-phase. Eventually, microscopic plastic deformation occurs when the phase transformation stress is higher than the elastic limit of the O-phase. This is why the slip traces are often observed on the surface of the O-phase particle.
Besides, as shown in Fig. 4(d), in one b=1.25 nm period, a stacking of six layers can be observed clearly along the [010] direction. This characteristic agrees well with the layer structural model of the O-phase constructed in Refs. [9,17].
4 Conclusions
1) After heat treatment of the Al-4Ni-2Mn alloy at 600 ��C for different time, nearly all of the Al9(Mn,Ni)2 eutectic fibers have changed into small particles with prolate ellipsoidal and spherical morphology. With the increase of the heat treatment time, Ostwald ripening phenomenon of the eutectic particles can be observed.
2) A phase transformation from Al9(Mn,Ni)2 to O-phase has been confirmed, for the first time, in Al-4Ni-2Mn alloy during the heat treatment at 600 ��C.
3) The slip traces observed on the surface of O-phase particle indicate that the (010)[001] slip system has been activated during the phase transformation.
Acknowledgments
We are grateful for the assistance from State Key Laboratory of Solidification Processing and Material Analysis and Testing Center of Shaanxi Province, China.
References
[1] BALANETSKYY S, MEISTERERNST G, FEUERBACHER M. The Al-rich region of the Al-Mn-Ni alloy system, Part I: Ternary phases at 750-950 ��C [J]. Journal of Alloys & Compounds, 2011, 509: 3787-3794.
[2] BALANETSKYY S, MEISTERERNST G, GRUSHKO B, FEUERBACHER M. The Al-rich region of the Al-Mn-Ni alloy system, Part II. Phase equilibria at 620-1000 ��C [J]. Journal of Alloys & Compounds, 2011, 509: 3795-3805.
[3] ZOLOTOREVSKY V S, BELOV N A, GLAZOFF M V. Casting aluminum alloys [M]. 1st ed. Oxford: Elsevier, 2007.
[4] FAN Y, HUANG K, MAKHLOUF M M. Precipitation strengthening in Al-Ni-Mn alloys [J]. Metallurgical & Materials Transactions A, 2015, 46: 5830-5841.
[5] YU Wen-tao, HAO Qi-tang. The phase transformation of Al9(Mn,Ni)2 eutectic phase in an Al-4Ni-2Mn alloy during heat treatment [J]. Materials Characterization, 2017, 129: 53-59.
[6] YU Wen-tao, HAO Qi-tang, Fan Li, LI Jie-hua. Eutectic solidification microstructure of an Al-4Ni-2Mn alloy [J]. Journal of Alloys & Compounds, 2016, 688: 798-803.
[7] ROBINSON K. The determination of the crystal structure of Ni4Mn11Al60 [J]. Acta Crystallographica, 1954, 7: 494-497.
[8] TENDELOO G V, LANDUYT J V, AMELINCKX S, RANGANATHAN S. Quasi-crystals and their crystalline homologues in the Al60Mn11Ni4 ternary alloy [J]. Journal of Microscopy, 2011, 149: 1-19.
[9] SUI H X, SUN K, KUOK H. A structural model of the orthorhombic C3,I-Al60Mn11Ni4 approximant [J]. Philosophical Magazine A, 1997, 75: 379-393.
[10] SHI Yun-jia, PAN Qing-lin, LI Meng-jia, LIU Zhi-ming, HUANG Zhi-qi. Microstructural evolution during homogenization of DC cast 7085 aluminum alloy [J]. Transactions of Nonferrous Metals Society of China, 2015, 11: 3560-3568.
[11] BUNGE H J, ESLING C. Texture development by plastic deformation [J]. Scripta Metallurgica, 1984, 18: 191-195.
[12] KUHLMANN-WILSDORF D. Theory of plastic deformation: Properties of low energy dislocation structures [J]. Materials Science & Engineering A, 1989, 113: 1-41.
[13] RODRIGUEZ P. Grain size dependence of the activation parameters for plastic deformation: Influence of crystal structure, slip system, and rate-controlling dislocation mechanism [J]. Metallurgical & Materials Transactions A, 2004, 35: 2697-2705.
[14] WEINBERGER C R, BOYCE B L, BATTAILE C C. Slip planes in bcc transition metals [J]. International Materials Reviews, 2013, 58: 296-314.
[15] YANG S J, NAM S W, HAGIWARA M. Abnormal acceleration of creep deformation rate above 700 ��C in the orthorhombic based Ti-22Al-27Nb alloy [J]. Journal of Alloys & Compounds, 2004, 368: 197-210.
[16] LI Guo-zhen, TANG Jia-zhen, XU Guo-shu. The structure and morphology of high temperature superconductive YBCO thin film [J]. Journal of Yunnan University, 1993, 15: 12-17. (in Chinese)
[17] DEMANGE V, WU J S, BRIEN V, MACHIZAUDF, DUBOIS JM. New approximant phases in Al-Cr-Fe [J]. Materials Science & Engineering A, 2000, 294-296: 79-81.
�����Σ������ã��� ٻ
������ҵ��ѧ ����ѧԺ ���̼��������ص�ʵ���ң����� 710072
ժ Ҫ������ɨ���������������������о�600 ��C�ȴ���ʱAl-4Ni-2Mn�Ͻ���Al9(Mn,Ni)2���������ò�ݱ估��ṹת�䡣������������ȴ��������У���ά״��Al9(Mn,Ni)2�����༸��ȫ��ת���������״����״���������ң������ȴ���ʱ����ӳ�����������Al9(Mn,Ni)2����������Oswald�컯�����⣬ͨ���о�֤ʵ�ȴ��������е�ȷ������Al9(Mn,Ni)2��O��������̡����������Ŀ�����ò������O��������Al9(Mn,Ni)2����ض��������κˣ�Ȼ����һ������ĸ�����������������У�����O���(010)[001]����ϵ�������ˣ���һЩO�������������˻��Ƽ��ߡ�
�ؼ��ʣ�Al-Ni-Mn�Ͻ���䣻�������ࣻ��������
(Edited by Xiang-qun LI)
Corresponding author: Qi-tang HAO; Tel: +86-29-88494276; E-mail: haoqitang@nwpu.edu.cn
DOI: 10.1016/S1003-6326(18)64836-8

