J. Cent. South Univ. Technol. (2011) 18: 1068-1073
DOI: 10.1007/s11771-011-0805-4
Degradation of chlorothalonil by ultrasonic irradiation: Kinetics and impact factors
GUO Hong-guang(�����), GAO Nai-yun(������), LI Lei(����),
JIN Xuan-xiong(������), CHEN Zhe(��)
State Key Laboratory of Pollution Control and Resources Reuse, Tongji University, Shanghai 200092, China
? Central South University Press and Springer-Verlag Berlin Heidelberg 2011
Abstract: Factors on degradation of chlorothalonil (CLT) in water by high frequency ultrasonic irradiation were investigated. The effects of initial concentration of chlorothalonil, dosages of tertiary butyl alcohol, humic acid and initial pH value on degradation of chlorothalonil, as well as the reaction mechanism were studied. The results reveal that chlorothalonil could be effectively degradated by ultrasonic irradiation. The reaction constant value kapp decreased from 0.014 1 to 0.010 2 min-1 with the initial concentration increasing from 50 to 400 ��g/L during 180 min irradiation. Tertiary butyl alcohol had negative effect on chlorothalonil degradation, while lower concentration of humic acid promoted the sonolysis, and kapp declined with the further concentration increasing. The kapp varied little when the pH value ranged from 3.10 to 10.28. It may be concluded that mechanical and pyrolysis process played main roles on the degradation of chlorothalonil in ultrasonic irradiation rather than ?OH attack. The electrical energy per order (EEo) values for sonolysis degradation of CLT were also calculated to evaluate the cost of the process.
Key words: ultrasonic irradiation; chlorothalonil; kinetics; impact factors
1 Introduction
Chlorothalonil(2, 4, 5, 6-tetrachloroisophthalonitrile, CLT) is a high-efficient non-systemic organ chlorine pesticide used as an insecticide for vegetable, peanut, rice and wheat, and is also used on turf [1]. Like other pesticides, environmentally persistent and long-term residual effect is caused by the abuse of CLT which has obvious cumulative toxicity to fish and invertebrates like mollusks, shrimp, and marine organisms with acute toxicity levels of 10-80 ?g/L in aquatic systems and considerable concentrations of CLT have been found in waters [2-3]. Besides, CLT is also an estrogen, which has interference on human and animal endocrine systems by influencing reproductions, causing feminization, gland deterioration, and vitality degeneration [4]. It is apparent that alternative treatments for control of aqueous CLT require investigation.
CLT elimination from water has been studied by many processes and several studies have been published on the degradation of CLT. PE?UELA et al [5] has reported that chlorothalonil was degraded in deionized water with half-lives of 101.17 and 36.86 h when exposed to sunlight and simulated sunlight, respectively. Water/sediment systems have been used to investigate partitioning behavior and the degradation of CLT, indicating that CLT was primarily degraded by photodegradation rather than adsorption to sediment in the early stages with removal rate of 87%-88% and 3%-6%, respectively [6]. The efficiency of a dark Fenton system (H2O2/Fe2+) to remove CLT from water was investigated by GUTI?RREZ [7]. Nearly complete degradation of CLT (>98%) was observed in less than 10 min, and the Gram-negative bacteria was supposed to be responsible for the degradation and transformation of CLT to 4-hydroxy-2, 5, 6-trichloroisophthalonitrile according to MOTONAGA��s study [8]. Results of studying the transformation of a CLT solution using zero valent iron (ZVI) revealed that incomplete slow dechlorination merely occurred with ZVI. However, complete rapid dechlorination reactions took place with modified zero valent iron (MZVI), especially Fe/Pd by using UV and MS analyses [9].
Recently, ultrasonic irradiation as a potential advanced oxidation process has received increasing attentions for the degradation of organic pollutants commonly found in water, such as pesticide, dye, EDCs and pharmaceuticals [10-13]. Ultrasonic irradiation process is an alternative method to traditional water, based on hotspot theory as a result of continuous formation and collapse of cavitation bubbles in a microsecond time scale [10], which has advantages in operation, high efficiency, and without introducing other chemicals and byproducts pollution [14-16]. To our best knowledge, few studies have been published on the degradation of CLT by ultrasonic irradiation. This research aims at studying the degradation of CLT by ultrasonic irradiation in aqueous solutions, and the effects of initial concentration of CLT, tert-butyl alcohol, humic acid and pH value were investigated. Additionally, the electrical energy per order (EEo) values for sonolysis degradation of CLT were introduced and calculated.
2 Experimental
2.1 Chemicals
Chlorothalonil (99% in purity, mass fraction) and methanol (HPLC) were from Sigma-Aldrich (USA). Typical physicochemical properties of CLT are shown in Table 1 with the molecular structure. Other reagents were in analytical grade and used without purifying. All solutions used were prepared with water purified by Milli-Q Gradient water purification system.
Table 1 Typical physicochemical properties of CLT

2.2 Experimental instruments
The ultrasonic instrument was provided by Institute of Acoustics, Chinese Academy of Sciences, with a fixed frequency of 400 kHz and output power of 100 W. The reactions were carried out in an open cylindrical stainless steel reaction vessel (diameter: 100 mm, height: 200 mm), which was directly connected to the ultrasonic transducer (diameter: 80 mm) with flanges and a flexible Teflon O-ring for seal. The vessel was immersed in a water bath, which was connected to a temperature control unit. Details of the reactor for sonophotolysis were described previously [10]. The schematic diagram of the experimental apparatus is shown in Fig.1
2.3 Analysis
Chlorothalonil was analyzed by a Shimadzu LC-2010AHT HPLC equipped with a VP-ODS column (250 mm��4.6 mm) and ultraviolet detector. A mobile phase of 60/40 (volume ratio) of methanol/water at a flow rate of 1 mL/min was used for 15 min determination at 232 nm and 40 ��C column temperature.
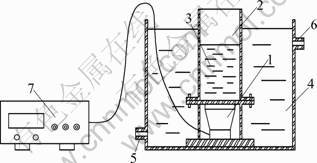
Fig.1 Schematic diagram of ultrasonic reactor: 1��Ultrasonic transducer; 2��Stainless steel vessel; 3��Solution; 4��Water bath; 5, 6��Temperature control unit; 7��Ultrasound generator
3 Results and discussion
3.1 Effect of initial concentration on chlorothalonil degradation
Fixed different initial concentrations of CLT solutions were prepared as 400, 200, 100 and 50 ��g/L, respectively. The volume of the solution was 300 mL with pH adjusted to 7.0. The solution was under the ultrasonic irradiation for 180 min and the degradation curves of CLT is shown in Fig.2. The pseudo-first-order kinetic model is used to evaluate the effect of initial concentrations on the degradation. The constant k, correlation coefficient R2 and half-life value ��1/2 are summarized in Table 2.
It is indicated in Fig.2 that initial concentrations have effect on degradation of CLT to some extent. For initial concentrations of CLT of 50, 100, 200 and 400 ��g/L, the removal rates are 92.56%, 90.13%, 85.69% and 84.84% after 180 irradiation, respectively. Theoretically, reactions of organic compounds by ultrasonic irrigation follow the pseudo-first-order kinetic model as [17]:
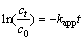 (1)
(1)
where kapp is an apparent rate constant, and c0 and ct are CLT concentrations at time 0 and t, respectively.

Fig.2 Effect of different initial concentrations on degradation of chlorothalonil by ultrasonic irradiation
Table 2 Degradation parameters of kinetics models (pseudo first-order) under different initial chlorothalonil concentrations

Based on Eq.(1), plots of ln(ct/c0) versus irradiation time (t) gives straight lines, which demonstrates the high validity of the pseudo-first-order kinetics on the degradation of CLT. Additionally, the molecular structure of CLT indicates that there is a resistant benzene ring in structure with low volatility reaching interiors of cavitation bubbles hard. Without enough extreme conditions in the cavities (thousands of degrees and hundreds of atmosphere pressure), the degradation rates of CLT were lower than those of algae toxins or BPA during previous work in laboratory. kapp decreased from 0.014 1 to 0.010 2 min-1 with the initial concentration increasing from 50 to 400 ��g/L, so the reaction does not exactly fit the first-order kinetic. According to TORRES��s work [18], CLT had little volatility and the sonolysis zone was not the bulk solution but interfacial region of cavitation bubbles. More byproducts like organic acids and polar functional groups were generated in the sonolysis which would be competitive as CLT degrading. Competed with other advanced oxidation process with a dominant of ?OH, the decline of kapp is not so tough. So it can be conferred that there are other ways for the sonolysis of CLT like mechanical and pyrolysis process acting on the whole bulk solution despite of the competition above.
With the variations of CLT initial concentration among 50-400 ��g/L, the relationship between rate constant (kapp) and initial concentration (c0) is concluded as Eq.(2):
kapp=-2��10-5c0+0.015 4 (R2=0.990 2) (2)
3.2 Effect of tertiary butyl alcohol concentration on chlorothalonil degradation
Tert-butyl alcohol (TBA) is an effective radical scavenger addition used to confirm the existence of a free radical mechanism by capturing free radical for the gaseous regions and/or interfacial regions of the cavitation bubbles [19]. TBA was added to the bulk solution (pH adjusted to 7.0, 400 ��g/L) with the concentration of 5, 10 and 20 mmol/L. Based on Eq.(1), plots of ln(ct/c0) versus time (t) are prepared, as shown in Fig.3. It is clearly indicated that the addition of TBA restrains the degradation of CLT. With the increasing of radical scavenger, the degradation rates are decreased to 0.006 0 min-1 as the initial concentration of TBA is 20 mmol/L. After 180 min, the removal rate is merely about 67%. By reacting with the ?OH, TBA inhibits the gathering of ?OH on the gaseous regions and/or interfacial regions of cavitation bubbles. Additionally, most of intermediate products of TBA are more volatile assembling the interfacial regions to prevent the formation of ?OH [20]. However, the rate constant decreases not so rapidly as the TBA concentration increases to 20 mmol/L, indicating that besides free radical reactions happening at the interfacial regions and bulk solutions, potential pyrolysis and mechanical process are also attributed to the sonolysis degradation of CLT. The pseudo-first-order kinetic model is used to evaluate the effect of different concentrations of TBA on the degradation. The rate constants kapp, correlation coefficient R2 and half-life value ��1/2 are summarized in Table 3.
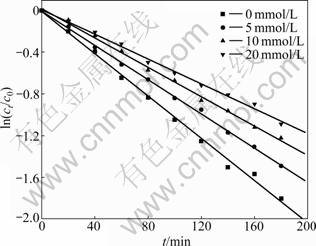
Fig.3 Effect of different concentrations of tert-butyl alcohol on degradation of chlorothalonil by ultrasonic irradiation
Table 3 Degradation parameters of kinetics models (pseudo first-order) under different tert-butyl alcohol concentrations

With the variations of concentration of TBA among 0-20 mmol/L, the relationship between rate constant (kapp) and concentration (c) is concluded as Eq.(3):
kapp=-0.000 2c+0.009 6 (R2=0.911 1) (3)
3.3 Effect of humic acid concentration on chlorothalonil degradation
Humic acids are the most representative group of humic substances, which have high molecular mass and irregular structure with many attacking regions for ��OH [21]. By adding the humic acid solution to simulate natural aquatic systems, the degradation of CLT was investigated. Different concentrations of humic acid (10, 20, 50 and 100 mg/L) were added into the bulk solution (pH adjusted to 7.0) with the initial concentration of CLT of 400 ��g/L. Plots of ln(ct/c0) versus irradiation time (t) are prepared, as shown in Fig.4.
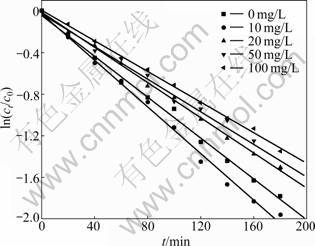
Fig.4 Effect of humic acid concentration on degradation of chlorothalonil by ultrasonic irradiation
It is indicated that lower concentration of humic acid promotes the sonolysis degradation with the kapp value increasing from 0.010 1 to 0.011 4; while as the concentration further increases, the kapp value gets descended. The rate consant decreases to 0.007 3 when the added concentration of humic acid is 100 mg/L. However, compared with other ultrasonic studies intensely restrained by humic acids accounting for the dominant free radical particle, the results also demonstrate that ?OH oxidation is not the major function in the sonolysis process [22]. The pseudo-first-order kinetic model is used to evaluate the effect of different concentrations of humic acid on the degradation. The rate constants kapp, correlation coefficient R2 and half-life value ��1/2 are summarized in Table 4.
3.4 Effect of initial pH value on chlorothalonil degradation
To investigate the effect of alkaline or acidic conditions of bulk solution on the sonolysis degradation of CLT. Solutions with different pH value (3.10-10.28) and similar initial concentration of CLT (400 ��g/L) were prepared to react for 180 min. The pH dependency on the degradation is depicted, as shown in Fig.5.
It is shown in Fig.5 that different pH value solutions make insignificant effect on sonolysis degradation of CLT. The removal rate is only changed by -0.53% to 0.44% with pH value increases from 3.10 to 10.28
Table 4 Degradation parameters of kinetics models (pseudo first-order) under different concentrations of humic acid
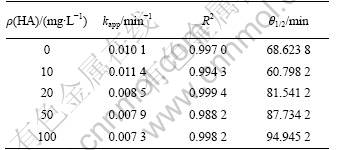

Fig.5 Effect of different pH values on degradation of chlorothalonil by ultrasonic irradiation
MA and ZHAO [23] found that neutral molecules had an easier access to the interfacial regions or the interior of cavitation bubbles than ions/polar molecules, which made the sonolysis process simply happen. OU et al [24] also found that pH value changed the structure and hydrophilic property and inhibited the degradation of MC-RR. On the degradation of chlorobenzene using ultrasonic irradiation by CHEN et al [25], the pH value could not affected the form of chlorobenzene causing little degradation rate change, which can be attributed to the same phenomenon observed on the sonolysis degradation of CLT. Meanwhile, the yield of H2O2 and ?OH can also be affected by the acidity and basicity of bulk solution. Consequently, the complex effect of pH value needs further investigation.
4 Electrical energy efficiency
Since sonolysis degradation of aqueous organic pollutants is an electric-energy-intensive process, and electric energy can represent a major part of the operating costs, simple figures-of-merit based on electric energy consumption can be very useful and informative. The international union of pure and applied chemistry (IUPAC) has proposed a figure-of-merit for advanced oxidation processes (AOPs) on the use of electrical energy for low pollutants [26-27]. It is defined as the electrical energy per order (EEo), namely the number of electrical energy required to reduce the concentration of a pollutant by one order of magnitude in 1 m3 of contaminated water. The EEo (kW?h?m-3 ) can be calculated from Eqs.(4)-(6):
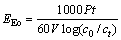 (4)
(4)
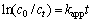 (5)
(5)
From Eq.(4) and Eq.(5), EEO can be calculated as
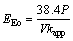 (6)
(6)
where P is the output power of ultrasonic instruments (kW), t is the irradiation time (min), V is the volume of reaction solutions (L), kapp, c0 and ct are the factors as previous stated. The effect of different impact factors above on EEo is summarized in Table 5. Results show that applying proper factors on sonolysis degradation of CLT can reduce the EEo to some extent.
Table 5 Electrical energy per order (EEo) for degradation of chlorothalonil by ultrasonic irradiation under different factors

5 Conclusions
1) Ultrasonic process is an effective advanced oxidation process for the degradation of aqueous organic pollutants especially for the recalcitrant compounds like chlorothalonil. The results follow the pseudo-first- order kinetic model with the average R2 of 0.995 0 using a batch study.
2) Apparent rate constant is sensitive to the initial concentration of chlorothalonil, tert-butyl alcohol and humic acids. Alkaline and acidic pH conditions are found to have not significantly effect on the CLT degradation. Besides the free radical reactions, mechanical and pyrolysis process might also contribute to the ultrasonic degradation of chlorothalonil.
3) The electrical energy per order (EEo) values for sonolysis degradation of CLT are introduced and calculated. Applying proper factors can reduce the EEo.
References
[1] Agency, USEP, USEPA Reregistration Eligibility Decision (RED): Chlorothalonil [S]. 1999: 320.
[2] HLADIK M L, KUIVILA K M. Analysis of chlorothalonil and three degradates in sediment and soil [J]. Journal of Agricultural and Food Chemistry, 2008, 56(7): 2310-2314.
[3] CAUX P Y, KENT R X, FAN G T, STEPHENSON G L. Environmental fate and effects of chlorothalonil: A Canadian perspective [J]. Critical Reviews in Environmental Science and Technology, 1996, 26(1): 45-93.
[4] TANG Ming-de, YI Yi-zhen, CHEN Yu-ling. The mutagenesis of pesticide chlorothalonil [J]. Journal of Environment and Health, 1989, 6(5): 37-38.
[5] PE?UELA G A, BARCELO D. Photodegradation and stability of chlorothalonil in water studied by solid-phase disk extraction, followed by gas chromatographic techniques [J]. Journal of Chromatography A, 1998, 823(1/2): 81-90.
[6] KWON J W, ARMBRUST K L. Degradation of chlorothalonil in irradiated water/sediment systems [J]. Journal of Agricultural and Food Chemistry, 2006, 54(10): 3651-3657.
[7] GUTI?RREZ R F, SANTIESTEBAN A, CRUZ-LOPEZ L, BELLO-MENDOZA R. Removal of chlorothalonil, methyl parathion and methamidophos from water by the Fenton reaction [J]. Environmental Technology, 2007, 28(3): 267-272.
[8] MOTONAGA K, TAKAGI K, MATUMOTO S. Biodegradation of chlorothalonil in soil after suppression of degradation [J]. Biology and Fertility of Soils, 1996, 23(3): 340-345.
[9] GHAUCH A, TUQAN A. Catalytic degradation of chlorothalonil in water using bimetallic iron-based systems [J]. Chemosphere, 2008, 73(5): 751-759.
[10] YAO J J, GAO N Y, LI C, LI L, XU B. Mechanism and kinetics of parathion degradation under ultrasonic irradiation [J]. Journal of Hazardous Materials, 2010, 175(1/3): 138-145.
[11] SINGLA R, GRIESER F, ASHOKKUMAR M. Sonochemical degradation of martius yellow dye in aqueous solution [J]. Ultrasonics Sonochemistry, 2009, 16(1): 28-34.
[12] MENDEZ-ARRIAGA F, TORRES-PALMA R A, PETRIER C, ESPLUGAS S, GIMENEZ J, PULGARIN C. Ultrasonic treatment of water contaminated with ibuprofen [J]. Water Research, 2008, 42(16): 4243-4248.
[13] LIN J J, ZHAO X S, LIU D, YU Z G, ZHANG Y, XU H. The decoloration and mineralization of azo dye CI Acid Red 14 by sonochemical process: Rate improvement via Fenton��s reactions [J]. Journal of Hazardous Materials, 2008, 157(2/3): 541-546.
[14] TANG Yun-lin, GAO Nai-yun, PANG Wei-hai. Progress on research of ultrasonic technology for drinking water [J]. Water & Wastewater Engineering, 2007, 33(12): 113-118.
[15] CHEN Wei, MEI Bin, FAN Jin-chu. The recent research and development of sonochemistry [J]. Shanghai Chemical Industry, 1999, 24(3/4): 24-27. (in Chinese)
[16] HUA I, HOFFMANN M R. Optimization of ultrasonic irradiation as an advanced oxidation technology [J]. Environmental Science & Technology, 1997, 31(8): 2237-2243.
[17] COLUSSI A J, HUNG H M, HOFFMANN M R. Sonochemical degradation rates of volatile solutes [J]. Journal of Physical Chemistry A, 1999, 103(15): 2696-2699.
[18] TORRES R A, PETRIER C, COMBET E, CARRIER M, PULGARIN C. Ultrasonic cavitation applied to the treatment of bisphenol A. Effect of sonochemical parameters and analysis of BPA by-products [J]. Ultrasonics Sonochemistry, 2008, 15(4): 605-611.
[19] MA Jun, LI Xue-yan, CHEN Zhong-lin, QI Fei. Removal of 2-methylisoborneol in drinking water by ozonation [J]. Environmental Science, 2006, 27(12): 2483-2487. (in Chinese)
[20] TAUBER A, MARK G, SCHUCHMANN H P, von SONNTAG C, Sonolysis of tert-butyl alcohol in aqueous solution [J]. Journal of the Chemical Society-Perkin Transactions, 1999, 2(6): 1129-1135.
[21] CHAIKOVSKAYA O N, SOKOLOVA I V, SOKOLOVA T V, YUDINA N V, MAL'TSEVA E V, IVANOV A A. Effect of humic acids on phototransformation of methylphenols in water [J]. Journal of Applied Spectroscopy, 2008, 75(4): 597-602.
[22] NADDEO V, BELGIORNO V, NAPOLI R M A. Behaviour of natural organic mater during ultrasonic irradiation [J]. Desalination, 2007, 210(1/3): 175-182.
[23] MA Jun, ZHAO Lei. Influencing factors on ultrasonic degradation of organic compound in water [J]. Journal of Natural Science of Heilongjiang University, 2005, 22(2): 142-153. (in Chinese)
[24] OU Hua-se, GAO Nai-yun, SUI Ming-hao, LI Lei. Influence factors in kinetics during degradation of MC-RR by ultrasonic irradiation process [J]. Journal of Central South University: Science and Technology, 2010, 41(2): 784-792. (in Chinese)
[25] CHEN Wei, CHEN Ling, FAN Jin-chu, QIAN Meng-lu. Study on degradation of chlorobenzene in aqueous solution by ultrasonic irradiation [J]. Water & Wastewater Engineering, 2001, 27(2): 34-37. (in Chinese)
[26] DANESHVAR N, ALEBOYEH A, KHATAEE A R. The evaluation of electrical energy per order (E-Eo) for photooxidative decolorization of four textile dye solutions by the kinetic model [J]. Chemosphere, 2005, 59(6): 761-767.
[27] SALARI D, NIAEI A, ABER S, RASOULIFARD M H. The photooxidative destruction of CI Basic Yellow 2 using UV/S2O82-process in a rectangular continuous photoreactor [J]. Journal of Hazardous Materials, 2009, 166(1): 61-66.
(Edited by HE Yun-bin)
Foundation item: Project(2008ZX07421-002) supported by the National Major Project of Science & Technology Ministry of China; Project(2008AA06A412) supported by the National High Technology Research and Development Program of China; Project(20009-K7-4) supported by the Research and Development of Ministry of Housing and Urban-Rural Development of China
Received date: 2010-12-24; Accepted date: 2011-04-12
Corresponding author: GAO Nai-yun, Professor, PhD; Tel: +86-21-65982691; E-mail: gaonaiyun@sina.com