J. Cent. South Univ. (2016) 23: 2139-2145
DOI: 10.1007/s11771-016-3270-2
TiO2 preparation by improved homogeneous precipitation and application in SCR catalyst
YAO Jie(Ҧ��)1, 2, ZHONG Zhao-ping(����ƽ)2
1. Guodian Science and Technology Research Institute, Nanjing 210046, China;
2. Key Laboratory of Energy Thermal Conversion and Control of Ministry of Education,
Southeast University, Nanjing 210096, China
 Central South University Press and Springer-Verlag Berlin Heidelberg 2016
Central South University Press and Springer-Verlag Berlin Heidelberg 2016
Abstract: Ultrasonic treatment and hydrothermal method were applied in the traditional homogeneous precipitation for nano-TiO2 preparation, which was used as carrier material for the production of honeycomb selective catalytic reduction (SCR) catalyst. The influence rules of the two improved methods on characterization of TiO2 samples, denitration activity and mechanical strength of honeycomb SCR catalyst samples were mainly focused on. The results indicate that the specific surface area, particle size and uniformity of TiO2 samples are significantly improved by both of the ultrasonic and hydrothermal treatments compared with the traditional homogeneous precipitation. Also, the denitration activities of catalyst samples are enhanced by the two improved methods (the NOx reduction ratio increases from 88.89% to 95.45% by ultrasonic homogeneous precipitation process, and to 94.12% by hydrothermal homogeneous precipitation process). On the other hand, because of good spherical shape and high particle distribution of TiO2 sample from hydrothermal homogeneous precipitation process, the corresponding honeycomb catalyst samples get the best mechanical strength, which is even higher than that of the reference sample from commercial nano-TiO2. So, it is concluded that the hydrothermal homogeneous precipitation can be a feasible and effective preparation method of TiO2 carrier for the honeycomb SCR catalyst production.
Key words: nano-TiO2; honeycomb SCR catalyst; homogeneous precipitation; ultrasonic treatment; hydrothermal method
1 Introduction
Ammonia selective catalytic reduction (SCR) is the most important industrialized flue gas denitration technology [1-5]. As the mainstream SCR catalyst, V2O5-TiO2 catalyst is widely used in the coal-fired boiler system [1-4]. TiO2 is the catalyst carrier, which can effectively support and disperse the active components to enhance the DeNOx efficiency and other properties [1-4, 6-7], so it is critical to the costs and quality of catalyst production.
Liquid method for the synthesis of nano-TiO2 generally includes direct precipitation, homogeneous precipitation, sol-gel method, hydrothermal method, etc. Among these methods, homogeneous precipitation [7-10] usually uses inorganic titanium salt and urea as raw materials. From the thermal hydrolysis process, urea decomposes and generates NH3��H2O, which makes the pH of reaction solution rise homogeneously. Then, H2TiO3 precipitates out as the reaction product. Compared to other methods, homogeneous precipitation is easy for application and industrialization. The TiO2 particles prepared from this method have uniform size, good spherical shape and high distribution, but are generally in micrometer scale, which is too large for the preparation of SCR catalyst. So, the traditional homogeneous precipitation process needs to improve.
In this work, ultrasonic treatment [11-13] and hydrothermal method [14-16] were applied in the traditional homogeneous precipitation for nano-TiO2 preparation. Moreover, honeycomb SCR catalyst samples were prepared with TiO2 as carrier material. The characterization of TiO2 samples, denitration activity and mechanical strength of catalyst samples were measured and compared with samples prepared with a commercial anatase nano-TiO2. The purpose of the work was to find a feasible and effective preparation method of TiO2 carrier for the honeycomb SCR catalyst production.
2 Experimental
2.1 Preparation of TiO2
2.1.1 Homogeneous precipitation
A certain amount of titanyl sulfate (TiOSO4, A.R.)and urea (CO(NH2)2, A.R.) were dissolved in deionized water, respectively. The two solutions were mixed and the dosages of titanyl sulfate and urea were controlled to make the initial concentration of TiOSO4 and CO(NH2)2 in the reaction mixture 0.2 mol/L and 0.8 mol/L, respectively. Then, a certain amount of DBS was added into the mixture as dispersant and its concentration was 0.01 mol/L.
The mixture was rapidly stirred under 80 ��C for 2 h in water bath. Then, the generated metatitanic (H2TiO3) was separated by vacuum filtration and the filter cake was washed by deionized water for several times to remove soluble impurities. After filtration, the metatitanic was dried at 105 ��C for 12 h and calcinated at 450 ��C for 2 h. Finally, TiO2 was obtained after natural cooling and ground as Sample 1.
2.1.2 Ultrasonic homogeneous precipitation
After the mixing of titanyl sulfate solution, urea solution and DBS, ultrasound instrument (XIANOU TECH GL-600SD, China) was used to generate ultrasound with fixed frequency of 25 kHZ. The acoustic power was fixed at 400 W and the end face of ultrasonic generator was �� 0.01 m, which was immersed under liquid surface for 10 mm. Then, the reaction was kept for 2 h with rapid stirring under 80 ��C condition. Other operations were the same as preparation of Sample 1. And TiO2 was obtained as Sample 2.
2.1.3 Hydrothermal homogeneous precipitation
After the mixing of titanyl sulfate solution, urea solution and DBS, the mixture was put in pressurized water reactor and the reaction system was heated to 140 ��C with rapid stirring for 2 h. Other operations were the same as preparation of Sample 1. And TiO2 was obtained as Sample 3.
2.1.4 Reference sample
A commercial anatase nano-TiO2 (Ishihara, ITAC- 140-7A, Japan) was selected as Sample 4.
2.2 Preparation of honeycomb SCR catalyst
Ammonium metavanadate (NH4VO3, A.R.) and ammonium tungstate ( (NH4)5H5[H2(WO4)6], A.R.) were chosen as the precursors of active component. The active component (V2O5 and WO3) was loaded by equal volume impregnation with TiO2 powders of Sample 1-4 as carriers, respectively. Considering the original certain WO3 component in commercial TiO2 powder, therefore only V2O5 was loaded for Sample 4. Then, four catalyst powders with the same ingredients of 1%V2O5-7%WO3/ TiO2 were obtained.
Afterwards, a certain amount of silica and polyacrylamide powder were added as forming agents, and the honeycomb billets were made by compression molding method [17-20]. Then, those billets were dried at 80 ��C for 24 h, and calcinated at 350 ��C, 1 h+450 ��C, 1 h+550 ��C, 3 h. After natural cooling, the honeycomb catalyst samples were obtained (Sample HC-1-HC-4 corresponded to TiO2 Sample 1-4), as shown in Fig. 1.
3 Analysis methods
3.1 Characterization analysis
X-ray diffractometer (Rigaku, D/max 2500/PC, Japan) was used to analyze the X-ray diffraction patterns. Scanning angle (2��) was from 10�� to 80��. Scanning rate was 2 (��)/min with monochromatized Cu radiation. Laser particle size analyzer (Beckman Coulter, Ls200, America) was used to analyze the particles sizes and distributions. X-ray fluorescence (TA, ARL QUANT'X, America) was used to analyze the element contents. Scanning electron microscopy (JEOL, JSM5610LV, Japan) was used to observe the TiO2 images. BET method (Micomeritics, ASAP 2020, America) was adopt to test the specific surface areas.
3.2 Mechanical strength
Electronic universal testing machine (Shenzhen SANS, CMT5105, China) was used to analyze the axial and lateral compressive strength of the honeycomb catalyst samples to evaluate the mechanical strength.

Fig. 1 Structure of honeycomb catalyst sample (a) and object picture (b) (Height of single honeycomb catalyst sample is 12 mm)
3.3 Denitration performance tests
Denitration activities of honeycomb catalyst samples were tested in self-made DeNOx activity test bench, which mainly consisted of gas distribution part, reaction part and exhaust gas analysis part. The catalyst samples were filled in reaction tube (The inner diameter of the tube was 37 mm, and single filling height of honeycomb catalyst sample was 72 mm). The simulated flue gas passed into reaction tube when it was heated to the specified temperature. Liquid water was injected into preheating section by micro-flow syringe pump, and then vaporized and mixed with other gases. NH3/N2 passed into reaction tube and mixed with the simulated flue gas after preheating. When the mixed gas flowed through the catalyst layer, SCR DeNOx reaction occurred. Then, NOx concentrations in outlet exhaust gas with and without reaction were measured by flue gas analyzer (MRU, MGA 5, Germany) to calculate the DeNOx efficiency of catalyst sample.
4 Results and analysis
4.1 Reaction mechanism and characterization of TiO2
4.1.1 Reaction mechanism
The main reaction in homogeneous precipitation for TiO2 preparation includes the hydrolysis of TiOSO4 and CO(NH2)2. For the former, total reaction can be simply described by Eq. (1), but the actual reaction is more complex than that [12, 21]:
TiO2++H2O��H2TiO3+2H+ (1)
In TiOSO4 solution, the Ti4+ exists in the form of complex Ti(OH)p(4-p)+ mainly in the beginning, which condenses with each other slowly to form the polymer as ����Ti��O��Ti�ԡ�. In this process, H+ is released with the formation of ��oxygen bridge�� in polymer. So, the titanium salt solution shows acid, and at this moment, the solution still keeps clarity.
When temperature or pH value of the reaction system rises, the equilibrium in Eq. (1) is broken and shifts to the right side. In this homogeneous precipitation process, the hydrolysis of CO(NH2)2 can be described by Eqs. (2) and (3) [10]. OH- generated constantly neutralizes H+ generated from the hydrolysis of TiOSO4, and H2TiO3 is generated as precipitate simultaneously. So, the two hydrolysis processes are promoted by each other during the whole reaction:
CO(NH2)2+H2O��2NH3+CO2 (2)
NH3+H2O��NH4++OH- (3)
In the initial reaction period of homogeneous precipitation, plenty of H2TiO3 nuclei are generated explosively. Then, the titanium polymer begins to grow and aggregate continuously centering at the nuclei with the same growth rate in all directions, because of the homogeneous rise of pH value in the reaction system. So, the TiO2 particles prepared from homogeneous precipitation have good spherical shape and uniform size.
In this work, ultrasonic treatment and hydrothermal method were applied in the traditional homogeneous precipitation as the operation improvements, respectively. Ultrasonic treatment can create micro-environment with high temperature and pressure in the liquid reaction system, so the hydrolysis of TiOSO4 and CO(NH2)2 is improved and the generation rate of H2TiO3 nuclei is increased in the initial reaction period. On the other hand, ultrasound brings the high-energy cavitation and microjet which can limit the growth of H2TiO3 particles effectively. Also, large aggregates formed will be crushed by the microjet. So, the particle size can be maintained at a low level.
The hydrothermal operation can raise the overall temperature and pressure of the reaction system. So, the generation rate of H2TiO3 nuclei can be enhanced also, and the increase of particle quantity leads to the decrease of prepared particle size. Moreover, the homogeneous characteristic of homogeneous precipitation reaction ensures the uniformity and sphericity of H2TiO3 particles. So, this method can improve the quality of TiO2 product effectively.
4.1.2 Characterization of TiO2 samples
The XRD results (Fig. 2) demonstrated that the TiO2 samples were all in anatase crystal form, which indicated that ultrasonic treatment and hydrothermal method operated in this work would not influence the crystal form of the prepared TiO2 from homogeneous precipitation. The average grain sizes of Sample 1-4 calculated by Scherrer formula [12] were 5.6 nm, 5.5 nm, 6.2 nm and 15.1 nm, respectively. The results indicated that ultrasound almost had no effect on the TiO2 grain size during the homogeneous precipitation process. It might be because the instantaneous micro-environment with high temperature and pressure generated by ultrasound could not bring a continuous impact on the growth of H2TiO3 crystal grain. However, the hydrothermal operation created a continuous and integrated environment with high temperature and pressure. It changed the overall growth condition for H2TiO3 crystal grain, and resulted in a slight increase in the average grain size. In addition, Sample 4 (commercial anatase nano-TiO2) had the highest degree of crystallinity and the largest crystal grain size. This might be due to the different operation condition during the preparation process.
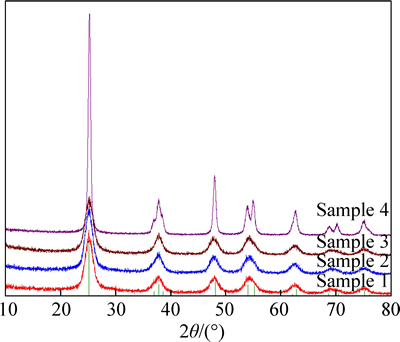
Fig. 2 XRD patterns of TiO2 samples
Particles size analysis results (Fig. 3) showed that the average particle sizes of TiO2 Sample 1-4 were 5.70, 0.65, 0.58 and 1.89 ��m, respectively, and the relative standard deviations of particle size distribution were 73.9%, 57.4%, 52.4% and 68.3%, respectively. Combined with the SEM images of the TiO2 samples (Fig. 4), it could be seen that Sample 1 prepared from the traditional homogeneous precipitation obtained good particle sphericity and larger particle size. But, the obvious crosslinking among the particles resulted in low dispersion of the TiO2 particles.
However, Sample 2 prepared from ultrasonic homogeneous precipitation presented relatively high dispersion and small particle size, because the random micro-environment with high temperature and pressure generated by ultrasound broke the homogeneous characteristic of reaction system to a certain extent. FromFig. 4(b), it could be seen that the particle sphericity of Sample 2 was low, and the particle shape was irregular. Moreover, it has been reported that ultrasound would cause the fine particles to collide and polymerize in liquid environment, so Sample 2 also showed a certain degree of agglomeration.
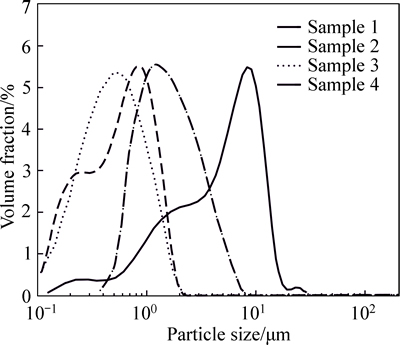
Fig. 3 Particles size distribution of TiO2 samples
TiO2 Sample 3 made by hydrothermal homogeneous precipitation, by contrast, had the lowest average particle diameter and relative standard deviation of particle size distribution, and also maintained good particle sphericity. It was just because hydrothermal process kept the homogeneous characteristic of reaction system to maintain the growth of spherical particles. In addition, almost all of the H2TiO3 nuclei were generated in the initial reaction period, because hydrothermal process enhanced the hydrolysis rate of TiOSO4. So, the H2TiO3 particles had the same growth time which brought them the similar particle size, and avoided the crosslinking and absorption among particles generated in different reaction periods.

Fig. 4 SEM images of TiO2 samples:
Compared with Sample 4 (commercial anatase nano-TiO2), Sample 2 and Sample 3 modified by ultrasonic treatment and hydrothermal method presented obvious advantages on the average particle size and distribution.
The BET surface areas of TiO2 Sample 1-4 are listed in Table 1. Sample 2 prepared from ultrasonic homogeneous precipitation obtained the highest BET surface area (121.4 m2/g), followed by Sample 3 prepared from hydrothermal homogeneous precipitation (113.6 m2/g). Sample 1 without improvement operation, by contrast, had the lower value (88.5 m2/g) than Sample 2 and 3. The results indicated that the two modifications strongly improved the specific surface area of TiO2 products. Due to the differences between the grain sizes, the specific surface area of Sample 2 was slightly higher than Sample 3.
Table 1 BET surface areas of TiO2 samples

The specific surface area of TiO2 carrier material has direct effect on the activity of the final catalyst product, so it is an important quality indicator of TiO2 materials.
4.2 Mechanical strength of honeycomb catalysts
Compressive strengths of axial and lateral direction were used for mechanical strength assessment of honeycomb catalysts (Sample HC-1-HC-4) [17-19, 22]. The results shown in Fig. 5 presented the average values of four-time measurements for each sample.
The experiment results demonstrated that the mechanical strength of Sample HC-1 was the lowest (compressive strength of axial direction was only 0.1063 MPa), which was loosely structured and friable. While with the ultrasonic treatment, the mechanical strength of sample HC-2 was improved significantly, and its compressive strength of axial direction reached 0.6818 MPa. This was because the small and uniform particles of TiO2 carrier with high dispersion brought a large contact area among these particles, and the honeycomb catalysts got higher sintering strength during the calcination process. Sample HC-3 was prepared with Sample 3, which was synthesized from hydrothermal homogeneous precipitation. Its mechanical strength wasfurther enhanced and the compressive strength of axial direction reached 0.9873 MPa. Honeycomb catalysts obtained from ultrasonic and hydrothermal homogeneous precipitation (Sample HC-2 and HC-3) had the similar average particle diameter and dispersion. The difference of mechanical strength between them might be due to different degrees of particle sphericity. Irregular particle morphology would hinder the uniformity of contact with each other during the sintering process. The created cracks weakened the connection among carrier particles just as the description in Fig. 6, therefore brought a mechanical strength reduction of honeycomb catalyst Sample HC-2.

Fig. 5 Compressive strength of honeycomb SCR catalyst samples
Compared with Sample HC-4 (compressive strength of axial direction is 0.8924 MPa), Sample HC-3 even had the better mechanical strength than the honeycomb catalyst prepared with commercial nano-TiO2. Besides, due to the structural features of honeycomb catalyst, the compressive strength of lateral direction was lower than that of axial direction, as shown in Fig. 4.
4.3 Characterization and denitration performance of honeycomb SCR catalysts
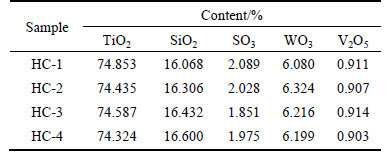
The BET surface area and the XRF analysis results of Sample HC-1-HC-4 are given in Table 2 and Table 3.
From Table 1 and Table 2, It could be seen that honeycomb catalyst samples had relatively lower specific surface area than the corresponding TiO2 samples because of the extrusion and calcination operations, while the order among each other remained unchanged. Data of Table 3 showed that honeycomb catalyst samples had the uniform component of 0.9%V2O5-6%WO3-2%SO3/TiO2-SiO2 basically. It facilitated further comparison of the denitration characteristics among different TiO2 carriers by catalyst activity tests.
SCR denitration experiments were carried out with honeycomb catalyst samples [1-4]. The testing conditions were set as follows: the catalyst height was 72 mm; volume space velocity (SV) ratio was 3000 h-1; simulated flue gas was composed of NO 0.05%, O2 5%, H2O 8%, SO2 0.3% and balance gas was N2; NH3/NO (molar ratio) was 1.05. The reaction temperature range was from 260 to 460 ��C, and the test results are shown in Fig. 7.
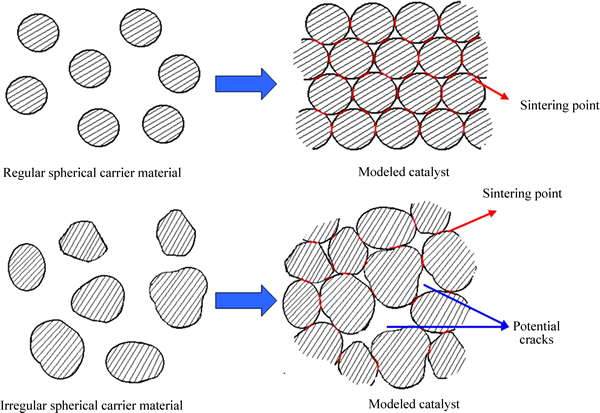
Fig. 6 Sintering properties of carrier materials with different shapes
Table 2 BET surface areas of catalysts samples

Table 3 XRF results of catalyst samples
From Fig. 7, it could be seen that, all of the four honeycomb catalyst samples presented stable NOx reduction ratio with the reaction temperature ranging from 320 to 400 ��C. It is indicated that the four samples had the same temperature range of denitration activity. The maximum NOx reduction ratio of Sample HC-1 reached 88.89% at 380 ��C nearby. It was higher than the maximum NOx reduction ratio of Sample HC-4 prepared with commercial nano-TiO2 (86.28% at 380 ��C nearby). The results presented the advantage of homogeneous precipitation process in preparation of SCR catalyst carrier TiO2. Moreover, Samples HC-2 and HC-3 presented further promotion on their NOx reduction ratio (95.45% and 94.12% at 380 ��C nearby, respectively). These catalyst samples had the uniform component, and their denitration activities showed obvious positive correlation with their specific surface area because high specific surface area would promote the contact of catalytic active sites with the reaction flue gas. It could be concluded that the ultrasonic treatment and hydrothermal method both enhanced the denitration activity of honeycomb SCR catalyst samples by the increase of specific surface area of TiO2 carrier.

Fig. 7 SCR denitration test results of honeycomb SCR catalyst samples
5 Conclusions
1) Ultrasonic treatment and hydrothermal method applied in traditional homogeneous precipitation could both decrease the product particle size of TiO2, and increase the dispersity, distribution and the specific surface area. However, ultrasound decreased the sphericity of TiO2 particles and brought some agglomeration; by contrast, the hydrothermal method made TiO2 product have the lowest relative standard deviation of particle size distribution, and also maintain good particle sphericity. In addition, both of the two improved methods would not brought obvious influence on the anatase crystal form of prepared TiO2.
2) Honeycomb SCR catalyst prepared with the TiO2 which was synthesized by ultrasonic or hydrothermal homogeneous precipitation could obtain higher denitration activity and mechanical strength than the sample prepared with traditional homogeneous precipitation TiO2 material. Besides, because of the better sphericity and dispersity of TiO2 particles, hydrothermal homogeneous precipitation brought the higher promotion on mechanical strength of honeycomb catalyst sample than ultrasonic homogeneous precipitation.
3) Compared with the commercial anatase nano- TiO2, TiO2 synthesized by hydrothermal homogeneous precipitation made the corresponding honeycomb SCR catalyst obtain the higher denitration activity and mechanical strength. The results indicated the feasibility and advantage of this method on the preparation of SCR catalyst carrier TiO2 material.
References
[1] ZHENG Yuan-jing, JENSEN A D, JOHNSSON J E. Deactivation of V2O5-WO3-TiO2 SCR catalyst at a biomass-fired combined heat and power plant [J]. Applied Catalysis B: Environmental, 2005, 60: 253-264.
[2]  O, ELSENER M. Chemical deactivation of V2O5/WO3�CTiO2 SCR catalysts by additives and impurities from fuels, lubrication oils, and urea solution I: Catalytic studies [J]. Applied Catalysis B: Environmental, 2008, 75: 215-227.
O, ELSENER M. Chemical deactivation of V2O5/WO3�CTiO2 SCR catalysts by additives and impurities from fuels, lubrication oils, and urea solution I: Catalytic studies [J]. Applied Catalysis B: Environmental, 2008, 75: 215-227.
[3] GUO Xiao-yu, BARTHOLOMEW C, HECKER W, BAXTER L L. Effects of sulfate species on V2O5/TiO2 SCR catalysts in coal and biomass-fired systems [J]. Applied Catalysis B: Environmental, 2009, 92: 30-40.
[4] CASTELLINO F, RASMUSSEN S B, JENSEN A D, JOHNSSON J E, FEHRM A N N R. Deactivation of vanadia-based commercial SCR catalysts by polyphosphoric acids [J]. Applied Catalysis B: Environmental, 2008, 83: 110-122.
[5] THEINNOI K, TSOLAKIS A, SITSHEBO S, CRACKNELL R F, CLARK R H. Fuels combustion effects on a passive mode silver/alumina HC-SCR catalyst activity in reducing NOX [J]. Chemical Engineering Journal, 2010, 158: 468-473.
[6] MA Zi-ran, WENG Duan, WU Xiao-dong, SI Zhi-chun, WANG Bin. A novel Nb-Ce/WOX-TiO2 catalyst with high NH3-SCR activity and stability [J]. Catalysis Communications, 2012, 27: 97-100.
[7] CHEN Xia, ZHANG Jun-feng, HUANG Yan, TONG Zhi-quan, HUANG Ming. Catalytic reduction of nitric oxide with carbon monoxide on copper-cobalt oxides supported on nano-titanium dioxide [J]. Journal of Environmental Sciences, 2009, 21: 1296-1301.
[8] GINSBERGA T, MODIGELLA M, WILSMANNB W. Thermochemical characterisation of the calcinations process step in the sulphate method for production of titanium dioxide [J]. Chemical Engineering Research and Design, 2011, 89: 990-994.
[9] TIAN Cong-xue, DU Jian-qiao, CHEN Xin-hong, MA Wei-ping, LUO Zhi-qiang, CHENG Xiao-zhe, HU Hong-fei, LIU Dai-jun. Influence of hydrolysis in sulfate process on titania pigment producing [J]. Transactions of Nonferrous Metals Society of China, 2009, 19: 829-833.
[10]  V, BAKARDJIEVA S, SZATMARY L. Synthesis of spherical metal oxide particles using homogeneous precipitation of aqueous solutions of metal sulfates with urea [J]. Powder Technology, 2006, 169: 33-40.
V, BAKARDJIEVA S, SZATMARY L. Synthesis of spherical metal oxide particles using homogeneous precipitation of aqueous solutions of metal sulfates with urea [J]. Powder Technology, 2006, 169: 33-40.
[11] LATT K K, KOBAYASHI T. TiO2 nanosized powders controlling by ultrasound sol�Cgel reaction [J]. Ultrasonics Sonochemistry, 2008, 15: 484-491.
[12] GHOWS N, ENTEZARI M H. Ultrasound with low intensity assisted the synthesis of nanocrystalline TiO2 without calcinations [J]. Ultrasonics Sonochemistry, 2010, 17: 878-883.
[13] TIAN Bao-zhu, FENG Che-na, ZHANG Jin-long, MASAKAZU A. Influences of acids and salts on the crystalline phase and morphology of TiO2 prepared under ultrasound irradiation [J]. Journal of Colloid and Interface Science, 2006, 303: 142-148.
[14] OH S W, PARK S, SUN Y. Hydrothermal synthesis of nano-sized anatase TiO2 powders for lithium secondary anode materials [J]. Journal of Power Sources, 2006, 161: 1314-1318.
[15] YIN Heng-bo, WADA Y, KITAMURA T, KAMBE S, MURASAWA S, MORI H, SAKATA T, YANAGIDA S. Hydrothermal synthesis of nanosized anatase and rutile TiO2 using amorphous phase TiO2 [J]. Journal of Materials Chemistry, 2001, 11: 1694-1703.
[16] PUDDU V, MOKAYA R, PUMA G L. Novel one step hydrothermal synthesis of TiO2/WO3 nanocomposites with enhanced photocatalytic activity [J]. Chemical Communications, 2007, 45: 4749-4751.
[17] LIU Qing-ya, LIU Zhen-yu, HUANG Zhang-gen, XIE Guo-yong. A honeycomb catalyst for simultaneous NO and SO2 removalfrom flue gas: Preparation and evaluation [J]. Catalysis Today, 2004, 93-95: 833-837.
[18] SUN Hong, SHU Yun, QUAN Xie, CHEN Shuo, PANG Bo, LIU Zhao-yang. Experimental and modeling study of selective catalytic reduction of NOX with NH3 over wire mesh honeycomb catalysts [J]. Chemical Engineering Journal, 2010, 165: 769-775.
[19] BERETTA A, TRONCONI E, ALEMANY L J, SVACHULA J, FORZATTI P. Effect of morphology of honeycomb SCR catalysts on the reduction of NOX and the oxidation of SO2 [J]. Studies in Surface Science and Catalysis, 1994, 82: 869-876.
[20] SHANG Xue-song, HU Gong-ren, HE Chi, ZHAO Jin-ping, ZHANG Fu-wang, XU Ya, ZHANG Yun-feng, LI Jiang-rong, CHEN Jin-sheng. Regeneration of full-scale commercial honeycomb monolith catalyst (V2O5�C WO3/TiO2) used in coal-fired power plant [J]. Journal of Industrial and Engineering Chemistry, 2012, 18: 513-519.
[21] CZEKAJ I,  O, PIAZZESI G. DFT calculations, DRIFT spectroscopy and kinetic studies on the hydrolysis of isocyanic acid on the TiO2-anatase (101) surface [J]. Journal of Molecular Catalysis A: Chemical, 2008, 280: 68-80.
O, PIAZZESI G. DFT calculations, DRIFT spectroscopy and kinetic studies on the hydrolysis of isocyanic acid on the TiO2-anatase (101) surface [J]. Journal of Molecular Catalysis A: Chemical, 2008, 280: 68-80.
[22] YASHNIK S A, ISMAGILOV Z R, KOPTYUG I V, ANDRIEV SKAYA I P, MATVEEV A A, MOULIJIN J A. Formation of textural and mechanical properties of extruded ceramic honeycomb monoliths: An 1H NMR imaging study [J]. Catalysis Today, 2005, 105: 507-515.
(Edited by YANG Bing)
Foundation item: Project(201031) supported by the Environmental Protection Scientific Research of Jiangsu Province, China; Project(BE2010184) supported by the Technology Support Program of Jiangsu Province�� Industrial Parts, China
Received date: 2015-06-08; Accepted date: 2015-12-10
Corresponding author: YAO Jie, PhD; Tel: +86-25-89620838; E-mail: tonglingyaojie@163.com