
Effect of direct electric current on wetting behavior of
molten Bi on Cu substrate
XU Qian-gang(徐前刚)1, LIU Xi-bei(刘锡贝)1, ZHANG Hai-feng(张海峰)2
1. Department of Materials Science and Engineering,
Shenyang Institute of Aeronautic Engineering, Shenyang 110136, China;
2. Institute of Metal Research, Chinese Academy of Sciences, Shenyang 110016, China
Received 25 August 2009; accepted 10 January 2010
Abstract: The effect of direct electric current on the wetting behavior of molten Bi on Cu substrate at 370 °C was investigated by the sessile drop method. The wettability of molten Bi on Cu without an applied current is poor and the spreading time required to form the steady-state contact angle (about 102?) is approximately 30 min. With the increase of the applied electric current, the spreading of molten Bi on Cu is accelerated significantly and the steady-state contact angle decreases considerably. The cross-section SEM micrographs of the solidified Bi droplet on Cu substrate show that the electric current has a marked effect on the convection of melt. Correspondingly, the application of an electric current obviously enhances the dissolution of Cu into Bi melt, which may change the wetting triple line configuration. The improvement of wettability induced by electric current is also related to the additional driving force for wetting provided by the electromagnetic pressure gradient force.
Key words: wetting; dissolution; surface and interface; spreading
1 Introduction
Wetting of molten metal on solid is a common physicochemical phenomenon in materials fabrication and processing. Wettability and interfacial interaction between melt and solid have an important influence on the material processing and properties[1-2]. A good understanding and regulating of the wetting processes taking place at interfaces are essential in the solution of many theoretical (physics and chemistry of surface phenomena) and technological (deposition of metallic coatings, soldering, brazing, welding, infiltration, etc.) problems[3-6].
In addition to the liquid composition, wetting temperature, roughness and crystal orientation of solid material and wetting atmosphere, applied electric field is known to have a significant influence on the wettability of liquid on solid substrate. Electrowetting on dielectric is an electric means to control the wettabilty of liquid on dielectric by electrocapillary effect and has drawn much attention recently for its potential applications in microfluidic control[7-10]. When a voltage was applied
between liquid and dielectric layer, the altered stored electric charge density in the dielectric layer changed the free energy of the dielectric surface and resulted in a change in wettability[7]. However, if the substrate was a conductor, such as molten metal-solid metal wetting systems, an applied voltage between the droplet and the substrate would not induce an obvious change in the interface free energy owing to the passage of electric current through the wetting system. But an applied electric current has some effects (such as Joule heating, electromigration and electromagnetic force) on the molten metal-solid metal wetting systems[11-12], which may have a significant influence on the dissolution of metal substrate and interfacial reaction and change the wetting triple line configuration. Effect of applied electric current on the wetting of molten metal on solid metal is not clear and very few results are available on this subject. In this work, a series of wetting tests were performed to assess the influence of direct electric current on the wetting behavior of molten Bi on Cu substrate using sessile drop method. Correspondingly, the plausible mechanism was also discussed.
2 Experimental
Small spherical Bi beads (approximately 150 mg, purity >99.9%) were made by melting pieces of Bi in a rosin flux. A thin pure copper lead (with a diameter of 0.3 mm, purity is above 99.9%) was inserted into the bead during solidification and used as an electrode in wetting experiment with an applied electric current. Pure copper substrates (purity is above 99.9%) were mechanically polished with 5 μm diamond paste and then electropolished in the electrolyte (300 mL distilled H2O and 700 mL H3PO4). Cu substrates and Bi beads were ultrasonically cleaned in acetone before experiment.
Wetting examinations of Cu by molten Bi with an applied electric current were performed by the sessile drop method under the vacuum of 3×10-3 Pa at 360 °C. Contact angles were measured directly from the image of the drop section. Direct electric current was supplied by a RXN-305D DC power. By applying an electric current (1.0, 1.5 and 2.0 A) through the molten Bi bead and Cu substrate, the Joule heat induced temperature rise on the wetting sample was measured to be around 10-13 °C during wetting testing. Considering the Joule heating effect, the actual wetting temperature of molten Bi on Cu with an applied electric current was about 370 °C. To differentiate the electric current effect from the thermally induced wetting process, the wetting experiment without applied electric current was performed at 370 °C. In this work, we defined the electric current flowing from droplet to substrate as the positive current (+), otherwise as the negative current (-). After the sessile-drop experiments, the solidified samples were sectioned to examine the interface. Cross sections were polished and examined by the scanning electron microscopy (SEM) and energy dispersive spectroscopy (EDS).
3 Results
3.1 Wetting process of molten Bi on Cu substrate
Fig. 1 shows the variation of the contact angle (θ) with time for molten Bi on Cu substrate during isothermal exposure at 370 °C. The wettability of molten Bi on Cu without an applied current is poor and the spreading time required to form the steady-state contact angle (about 102?) was approximately 30 min. Both the steady-state contact angle and the isothermal exposure time required to form the steady-state contact angle decrease markedly with the increase of the applied current. In the case with an applied positive current of 2.0 A, the steady-state contact angle formed by molten Bi on Cu substrate is about 50? (only about one-half that without an applied current) and spreading time required to form the steady-state contact angle is about 10 min (only about one-third that without an applied current). Evolution of the droplet image during isothermal exposure at 370 °C for molten Bi on Cu substrate without applied current and that with an applied positive current of 2.0 A are shown in Fig. 2. The direct current direction does not have an obvious influence on the spreading rate and the steady-state contact angle of molten Bi on Cu substrate with lower applied electric current. But the steady-state contact angle of molten Bi on Cu substrate with an applied positive current of +2.0 A is smaller than that with an applied negative current of -2.0 A. Generally, an applied current may accelerate the wetting of molten Bi on Cu and decrease the steady-state contact angle considerably.
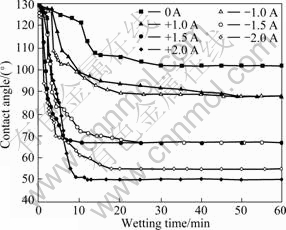
Fig.1 Variation of contact angle with time during isothermal exposure at 370 °C for molten Bi on Cu substrate with different applied currents
3.2 Interfacial interaction between molten Bi and Cu substrate
Cu-Bi system has little mutual solid solubility and there is no compound found in this system. However, there is a few solubility of Cu in molten Bi[13]. Owing to the preferential dissolution of the grain boundary, grain boundary grooves usually form at the molten Bi-Cu interface[14]. Fig.3(a) shows the SEM micrograph of cross-section perpendicular to the interface of pure Bi on Cu after wetting at 370 °C for 60 min without electric current. It can be found that dissolution of Cu into molten Bi and penetration of molten Bi into Cu occur at the interface in the wetting process. The molten Bi-Cu interface is irregular because of the preferential dissolution of the grain boundaries. The interface micrograph around the wetting triple line is shown in
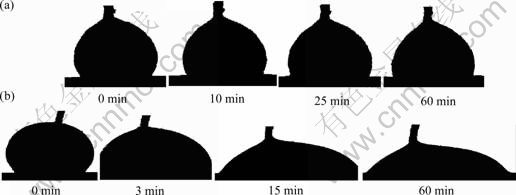
Fig.2 Evolution of droplet image during isothermal exposure at 370 °C for molten Bi on Cu substrate: (a) Without applied current; (b) With applied positive current of 2.0 A

Fig.3 SEM micrographs of cross-section perpendicular to interface of pure Bi on Cu after wetting at 370 °C for 60 min without applied current: (a) Central section interface micrograph; (b) Interface micrograph around wetting triple line (Dash line denotes initial Bi-Cu interface)
Fig.3(b). According to the surface of the substrate, the initial molten Bi-Cu interface can be shown as the dash line in Fig.3(b). During the cooling process, due to the decrease of solubility of Cu in Bi, the dissolved Cu in the molten Bi would precipitate, nucleate and grow adhered to the irregular interface. So, the solidified Bi-Cu interface goes beyond the initial molten Bi-Cu interface.
Fig.4 shows the interface micrographs around the wetting triple line of Bi droplet on Cu with applied electric current. With the increase of the applied electric current, the solidified Bi-Cu interface is gradually lower than the initial molten Bi-Cu interface, which indicates that the applied electric current enhances the dissolution of Cu in molten Bi. The solid/liquid interface is curved close to the triple line due to the dissolution of substrate in the melt, which affects the solid-liquid dihedral angle at the triple line. The true contact angle (θy) between molten Bi and Cu is larger than the apparent contact angle (θa) measured by the sessile drop method. The true contact angle θy is composed of the apparent contact angle θa and an extrapolated contact angle (θe, as shown in Fig.4(f)). The extrapolated contact angle θe enlarges with the increase of the applied electric current. It is difficult to determine the value of θe precisely due to the formation of irregular grooves. The estimated results of the extrapolated contact angle θe from Fig. 3 and Fig.4 and the true contact angle θy are listed in Table 1. Although the true contact angle θy still decreases with the increase of applied electric current, the influence of applied current on the true contact angle θy is not drastic as that on the apparent contact angle θa. The direct current direction does not have an obvious influence on the dissolution of substrate and the extrapolated contact angle θe.
Fig. 5 shows the cross-section SEM macrographs of Bi droplet on Cu substrate. No obvious precipitated Cu is found in the solidified Bi droplet after wetting experiment without applied electric current (as shown in Fig.5(a)). However, many precipitates of Cu are observed in the upper side of the solidified Bi droplet with an applied positive current of 1.5 A (as shown in Fig.5(b)). A similar phenomenon is found in other solidified Bi droplets with different applied electric currents. This indicates that the dissolved Cu at the molten metal/substrate interface is transferred to the upper side of the molten Bi under the effect of applied electric current.

Fig.4 SEM micrographs of cross-section perpendicular to interface of molten Bi on Cu after wetting at 370 °C for 60 min with applied electric current: (a) +1.0 A; (b) -1.0 A; (c) +1.5 A; (d) -1.5 A; (e) +2.0 A; (f) -2.0 A

Fig.5 SEM macrographs of cross-section perpendicular to interface of Bi droplet on Cu: (a) Without electric current; (b) With applied electric current of +1.5 A
Table 1 Measured contact angles for molten Bi-Cu wetting system with applied electric current

4 Discussion
The spreading of molten Bi on Cu substrate is a dissolutive wetting process. The experimental results above show that the applied electric current may increase the spreading rate, modify the geometry at the tripe line and decrease the steady-state contact angle of molten Bi on Cu substrate. We can combine Fig.6 to analyze the effect of the applied electric current on the wetting of Cu substrate by molten Bi. Because the Joule heat induced temperature rise on the wetting sample has been compensated in the wetting experiment without applied electric current, the effect of Joule heat on the wetting of molten Bi on Cu substrate may be neglected. Similar to the arc welding phenomena[12, 15], with the passage of current through the molten metal drop, due to the interaction of the molten conductor and the magnetic field induced by the current, inward directed electromagnetic radial pinch forces Pemr (as shown in Fig.6(a) ) are generated on the sessile drop. The radial pinch force distribution inside a conducting incompressible liquid cylinder is given by[15]
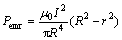 (1)
(1)
where μ0 is the permeability constant, I is the total electric current passed through a horizontal cross-section, R is the radius of a horizontal cross-section, r is the radial distance. The total radial pinch force on a horizontal cross-section may be calculated by the following equation:
 (2)
(2)
Equation (2) indicates that the total radial pinch force on a horizontal cross-section is proportional to the square of current and is independent of the radius of the horizontal cross-section, which tries to deform the molten drop in the radial direction. As the cross-section of the droplet is variable, an axial component of the electromagnetic force (Pemz, as shown in Fig.6(a)) arises, which is directed from the smaller to the larger cross-section[15]. These electromagnetic forces will set the molten metal in the droplet into convection (as shown in Fig. 6(b)). Convection provides a driving force for the transfer of the dissolved Cu within the droplet and accelerates the dissolution of the substrate. High electric current would intensify the convection in the droplet and increase the extrapolated contact angle θe.
Besides inducing convection in the droplet, electromagnetic forces also provide a driving force for the spreading of the droplet. Owing to the fact that the contact area of the droplet with the substrate (Ⅱ-Ⅱ
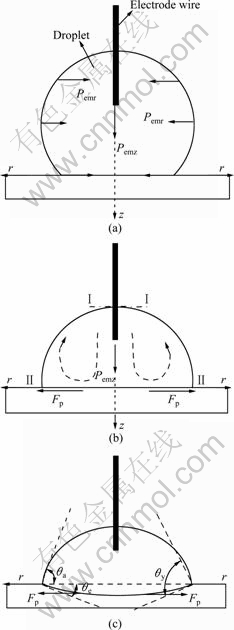
Fig.6 Schematic diagram of wetting of Bi droplet on Cu substrate with an applied electric current: (a) Initial state; (b) Convection induced by electromagnetic force in droplet; (c) Steady state
cross-section, as shown in Fig.6(b)) is larger than the area of the cross-section of the droplet contacted with the electrode (Ⅰ-Ⅰcross-section, as shown in Fig. 6(b)), the axial electromagnetic force Pemz is directed to the spreading interface and drives the molten metal to move towards the interface. Under the pinch effect of Pemz and Pemr, the melt at the spreading interface will be expanded due to the incompressibility of the molten droplet. Equation (1) suggests that there is a positive pressure gradient Fp in the radial direction at the spreading interface:
 (3)
(3)
where Rs is the spreading radius of the droplet. Therefore, in addition to the interface tension, this magnetic pressure gradient force provides an additional driving force for the wetting of molten Bi on Cu substrate with the applied electric current. The magnetic pressure gradient force is proportional to the square of current. Hence, the magnetic pressure gradient would accelerate the wetting of molten Bi on Cu with the increase of the applied electric current.
The driving force for the spreading of triple line of molten Bi on Cu substrate with the applied electric current at wetting time t may be expressed as
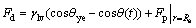
 (4)
(4)
where γlv is the surface tension of the molten metal, θye is the true equilibrium contact angle without applied current, and θ(t) is the true contact angle at wetting time t. For the dissolutive wetting, the true contact angle θ is composed of the apparent contact angle θa and the extrapolated contact angle θe (as shown in Fig.6(c)). When the wetting reaches the steady state, the driving force for the wetting will be equal to zero. Correspondingly, the true steady state contact angle θys for the wetting of molten Bi on Cu substrate with the applied electric current may be deduced from the following equation:
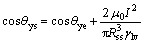 (5)
(5)
where Rss is the spreading radius of the droplet at steady state. Equation (5) shows that the true steady state contact angle θys decreases with the increase of the applied electric current.
The influence of electric current direction on the wettability of molten Bi on Cu with a higher applied electric current may be correlated to the migration direction of Bi ion under the electric field. According to the discussion of the wetting of molten Sn on Cu by WANG et al[16], applied positive current would promote the migration of Bi ion toward the wetting interface and enhance the wettability of molten Bi on Cu. In addition, the difference in the amount of the dissolved Cu in the molten Bi may have an effect on the surface tension of melt and the wetting process. The wetting process of a molten droplet on a solid substrate is made complicated by the presence of applied electric current. Further experimental and theoretical studies will help to a comprehensive understanding of the effect of applied electric current on the wetting behavior. The results of this study indicate that applied electric current is an effective method for regulating the wetting behavior of molten Bi on Cu. It would be interesting to study the effect of applied electric current on the wetting behavior in other metal-metal wetting system.
5 Conclusions
1) The wetting behavior of molten Bi on Cu substrate with applied direct current was studied using the sessile drop method. The wetting of molten Bi on Cu is a dissolutive wetting process. Melt convection induced by applied current enhances the dissolution of Cu in molten Bi.
2) With the increase of applied electric current, the wetting of molten Bi on Cu is accelerated and the steady-state contact angle decreases considerably.
References
[1] NAIDICH YU V. The wetting of solids by liquid metals [J]. Prog Surf Membr Sci, 1981, 14: 353-470.
[2] EUSTATHOPOULOS N, NICHOLAS M G, DREVERT B. Wettability at high temperature [M]. New York: Pergmon Press, 1999: 198-384.
[3] MATSUMOTO T, NOGI K. Wetting in soldering and microelectronics [J]. Annu Rev Mater Res, 2008, 38: 251-273.
[4] GERMAN R, SURI P, PARK S. Review: Liquid phase sintering [J]. Journal of Materials Science, 2009, 44(1): 1-39.
[5] FRAGE N, FROUMIN N, ALZENSHTEIN M. Reactive wetting in titanium carbide/non-reactive metal systems [J]. Current Opinion in Solid State & Materials Science, 2005, 9(4/5): 189-195.
[6] ASTHANA R, SOBCZAK N. Wettability, spreading, and interfacial phenomena in high-temperature coatings[J/OL]. http://www.tms.org/ pubs/journals/JOM/0001/Asthana/Asthana-0001.html. 2000.
[7] MUGELE F, BARET J C. Electrowetting: From basics to applications [J]. J Phys Condens Matter, 2005, 17(28): R705- R774.
[8] RAUSCHER M, DIETRICH S. Wetting phenomena in nanofluidics [J]. Annu Rev Mater Res, 2008, 38: 143-172.
[9] DARHUBER A A, TROIAN S M. Principles of microfluidic actuation by modulation of surface stresses [J]. Annu Rev Fluid Mechanics, 2005, 37: 425-455.
[10] TEH S Y, LIN R, HUNG L H. Droplet microfluidics [J]. Lab on A Chip, 2008, 8(2): 198-220.
[11] ZHAO J F, UNUVAR C, TAMBURINI U A, MUNIR Z A. Kinetics of current-enhanced dissolution of nickel in liquid aluminum [J]. Acta Materialia, 2007, 55(16): 5592-5600.
[12] DEYEV G F, DEYEV D G. Surface phenomena in fusion welding processes [M]. New York: CRC Press, 2005: 135-138.
[13] CHANG L S, STRAUMAL B B, RABKIN E, GUST W. The solidus line of the Cu-Bi phase diagram [J]. J Phase Equilibria, 1997, 18(2): 128-135.
[14] XU Q G, ZHANG H F,HU Z Q. Effect of grain boundary on the wettability and interfacial morphology in the molten bi/cu system [J]. J Mater Sci Technol, 2003, 19(3): 206-208.
[15] ANDO K, HASEGAWA M. Arc welding phenomenon [M]. SHI Y X. Beijing: China Machine Press, 1985: 249-258.
[16] WANG W M, BIAN X F, CHANG F L, QIN J Y, YE Y F. Effect of applying a direct current on wettability of liquid Sn-Pb/solid Cu [J]. Chinese Sci Bull, 1997, 42(22): 1928-1932.
(Edited LI Xiang-qun)
Foundation item: Project(50704001) supported by the National Natural Science Foundation of China
Corresponding author: XU Qian-gang; Tel: +86-24-89724144; E-mail: xu-qiangang@163.com
DOI: 10.1016/S1003-6326(09)60320-4