J. Cent. South Univ. (2016) 23: 787-792
DOI: 10.1007/s11771-016-3124-y

Synthesis, characterization and antimicrobial activity of alkaline ion-exchanged ZnO/bentonite nanocomposites
Hamideh Pouraboulghasem1, Mohammad Ghorbanpour2, Razieh Shayegh2, Samaneh Lotfiman2
1. Department of Food Science, Islamic Azad University-Sarab Branch, Sarab, Iran;
2. Chemical Engineering Department, University of Mohaghegh Ardabili, Ardabil, Iran
 Central South University Press and Springer-Verlag Berlin Heidelberg 2016
Central South University Press and Springer-Verlag Berlin Heidelberg 2016
Abstract: Nanocomposites of zinc/bentonite clay were synthesized for use as an antibacterial material by a quick and simple alkaline ion exchange method. The synthesis of zinc doped bentonite nanocomposite was accomplished by placing bentonite in a melting bath of ZnSO4 for 10, 20, 40, 60 and 90 min. The complexes were characterized by XRD, SEM and DRS. XRD analyses and SEM observations confirmed the diffusion of zinc to the clay surfaces. Antibacterial activity tests against Escherichia coli showed that bentonite did not present any antibacterial properties, but after alkaline ion exchange treatment, inhibition was noted. The highest antibacterial activity was observed with ZnO/bentonite composite alkaline ion exchange for 60 and 90 min. Interestingly, the leaching test indicated that ZnO/bentonite did not present any risk for drinking water treatment.
Key words: alkaline ion exchange; ZnO/bentonite nanocomposite; antibacterial activity
1 Introduction
Overuse of antibiotics in humans and animals has caused the speedy development of pathogenic bacteria that are immune to multiple drugs. These bacteria have been known to be harmful to public health as well as to the environment. To decrease antibiotic overuse and defeat the resistance of pathogenic bacteria, safe antibacterial agents must be developed to replace today��s common antibiotics [1].
Among the known materials used for the antibacterial activities, metal and metal oxide nanoparticles have attracted extensive interest. Nanoparticles are of interest because of their high reactivity due to the large open area-to-mass ratio. It has been noted that nanomaterials display different properties at nanometer sizes compared to the properties of the same materials in bulk conditions. The considered properties of nanoparticles are size and anatomy in the range of nano scale. Some well-studied nanoparticles are Ag, ZnO, TiO2, WO3, SnO2, CuO and MgO [2-3].
Zinc oxide possesses some antibacterial effect, good thermal qualities, and color stability, with low cost and little toxicity. Rare earth ions have been used as antimicrobial agents in medicine for a long time due to their high safety and wide spectrum antibacterial action [4]. The antibacterial properties of ZnO nanoparticles have been utilized in food packaging materials for controlling food-borne pathogens and deactivating many other bacteria [5].
Although the zinc ion in small quantities is essential for various metabolic processes in most living organisms, in higher amounts, it is potentially toxic. Zinc also has a stimulatory effect on bone formation in vitro and in vivo. There are three main mechanisms for the antimicrobial activities of the zinc ion. First, metal ions bind to proteins and deactivate proteins. Second, metal ions can interact with microbial membrane, causing structural changes and permeability. Third, metal ions interact with microbial nucleic acids and prevent microbial replication [6].
It has been demonstrated that using nanoparticles alone is dangerous, because their sizes can affect human health and the environment. To overcome this problem, hybrid processes were introduced by combining nanoparticles with other environmentally friendly materials, such as inert and stable materials to utilize nanoparticles�� full properties in wide-range applications. Moreover, introducing nanoparticles with other substrates/fillers would result in composites with novel and enhanced properties that cannot be achieved by the individual components.
Clay minerals are excellent fillers for metal composites. Clay mineral has been widely attractive to manufacturers and scientists throughout the world because of their low cost, high specific surface area, high chemical and mechanical stability, variety of surfaces, high adsorption and structural properties [2]. Their adsorption capacities help to attract contaminants into the open pores and therefore raise the efficiency of the dispersed nanoparticles. Bentonite, a member of the smectite group, is mainly composed of montmorillonite clay.
Composites of metal oxide nanoparticles supported by clay can be synthesized by various techniques. However, their formulation is still mostly based on prescribed techniques such as ion exchange processes, ��-irradiation, hydrothermal synthesis, chemical reduction methods, solvothermal methods, electroless plating, hydrothermal synthesis and sol-gel [7-8]. The disadvantages of existing techniques are passing through multiple steps of ultrasonication and stirring, which take several hours or even days, followed by chemical reduction of the precursor metal salts. Thus, any new synthesis technique that helps to simplify, save time, control particle size and provide uniform dispersion of nanoparticles would be exceedingly thought provoking.
Alkaline ion exchange is an attractive alternative promoting reaction and can reduce processing time through direct heating of the reaction. Zn-alkali ion exchange is a desirable way to dope superficial layers of silicate glasses well beyond Zn solubility limits. It also provides the possibility of controlling nanocluster nucleation and growth through subsequent appropriate treatments. Metal-alkali ion exchange in glass has been widely used to dope silicate glasses to give suitable optical properties and strengthening [9-13]. The ion exchange operation is accomplished by immersing Na-bentonite in a molten salt bath containing the dopant ions, which replace alkali ions in the glass matrix. In the present work, we show some experimental results of the formation and characterization of zinc nanocomposites in bentonite by Zn ion exchange and the effect of ion exchange time on the composites�� antibacterial activity.
2 Experimental
2.1 Materials
Bentonite clay (Na-montmorillonite), used as a solid support for zinc, was obtained from the Kansas Jam Company (Rasht, Iran). All reagents were of analytical grade and used without further refinement. All aqueous solutions were prepared using distilled water. Mueller Hinton agar broth and nutrient agar were purchased from Merck. The bacterial strain used for the antibacterial activity was gram-negative Escherichia coli (E. coli) (PTCC 1270), obtained from the Iranian Research Organization for Science and Technology.
2.2 Alkaline ion exchange with zinc
Bentonite was submitted to an ion-exchange process by immersion to ZnSO4.5H2O at 550-560 ��C for 10, 20, 40, 60 and 90 min. The process was conducted using 5 g each of bentonite and ZnSO4��5H2O. After the ion exchange, the bentonite was washed carefully through sonication with distilled water. The principal aim of this step was to dissolve any compound that was not contained in the montmorillonite. After dissolution, the bentonite was dried in an oven at 25 ��C.
2.3 Characterization
Synthesized samples were analyzed by X-ray diffractometry (XRD) (Philips PW 1050, the Netherlands). The patterns were registered from 10��to 60��in the 2�� range with a scanning step size of 0.05��. Bentonite clay was used as the reference. The morphology of the molecules was investigated by scanning electron microscope (LEO 1430VP, Germany). Absorption spectra of bentonite and ZnO/bentonite nanocomposites were measured by a UV-vis diffusive reflectance spectrophotometer (Sinco S4100, Korea), in the wavelength range of 200-1000 nm.
2.4 Antibacterial activity
The antibacterial activity of the samples was studied by plate count technique on E. coli. 50 mg of powder samples (bentonite and ZnO/bentonite nanocomposite) and 100 ��L of bacterial suspension at an adjusted concentration equivalent to 0.5 McFarland standards were added to 5 mL sterile Mueller Hinton broth. The cultures were then incubated at 37 ��C on a rotary shaker for 24 h. Growth inhibition with time was followed by plating 100 ��L of the treated cultures on nutrient agar plates. Bacterial colonies were counted and compared with control after 24 h incubation at 37 ��C. The entire experiment was repeated twice to validate the data. The antibacterial effect was calculated using the following equation:
 (1)
(1)
where B is the mean number of bacteria in the control samples (CFU/sample) and C is the mean number of bacteria in the treated samples (CFU/sample).
2.5 Leaching test
In order to evaluate the stability of the nanocomposites, leaching tests were performed, with 0.2 g of each composite immersed separately in 10 mL of distilled water and vigorous shaking in a shaking water bath (30 ��C, 200 r/min) for 3 and 6 h. Supernatants from each test tube were collected by centrifugation at 4000 r/min for 10 min. Then, released zinc ions from the ZnO/bentonite nanocomposites were qualitatively determined by atomic absorption spectroscopy analysis (Varian/AAS. 20BQ, USA).
3 Results and discussion
The colour of the parent bentonite was white (Fig. 1). Alkaline ion exchange of the bentonite with zinc sulphate changes the appearance of the bentonite to a cream colour. It seems that this variation in colour may result from the amount of ion-exchanged zinc or aggregation of the zinc particles diffused into the bentonite structure. These findings will be discussed more in later characterization tests.
3.1 SEM analysis
Figure 2 presents SEM images of bentonite and Zn ion-exchanged bentonite. The scanning electron micrograph of bentonite (Fig. 2(a)) shows a typical layered structure with numerous nano-flakes of clay particles in a sheet-like morphology. After ion exchange, the overall parent structure of bentonite remains unchanged. However, the increase in zinc ions yields a more porous composite texture. This means that by increasing ion exchange time, the loading of zinc ions increases; consequently, the distance between nano- flakes increases. On the other hand, the zinc nanoparticles situated on the nano-flakes�� surface create distance among the nano-flakes.

Fig. 1 Photographes of ion-exchanged bentonite for 90 (a), 60 (b), 40 (c), 20 (d), 10 (e) and 0 (f) min

Fig. 2 Scanning electron micrographs of parent bentonite (a), ion-exchanged bentonite for 10 (b), 20 (c), 40 (d), 60 (e) and 90 (f) min
Our findings indicate that the alkaline ion exchange takes place by diffusion of zinc into the bentonite matrix and replacement with the sodium. By increasing time, the zinc loading and diffusion into bentonite increase and the structure of bentonite separates more. However, some researchers reported that composites were produced by synthesis of nanoparticles and immobilization into the bentonite. Therefore, their SEM images show typical layered structures with heterogeneous immobilized nanoparticles [2, 14-15].
3.2 XRD analysis
Figure 3 illustrates the XRD patterns of bentonite and Zn ion-exchanged bentonite. Bentonite characteristic peaks are shown at 2�� values of 19.8��, 21.8��, 36.0�� and 62.5��. The peaks appear in all patterns, consistent with other reports [11-12]. After alkaline ion exchange by ZnSO4, ZnO is deposited on the clay. Then, new peaks, discerned corresponding to the formation of ZnO in the composites, appear. These peaks, observed at 2��= 31.7�� and 36.2��, indicate the presence of ZnO and confirm that the prepared composites are composed of ZnO species. Although the XRD peaks of ZnO/bentonite contain peaks of bentonite, the intensity of the peaks is weaker than that with pure bentonite.
3.3 Diffuse reflectance spectroscopy
The diffuse reflectance absorption spectra of bentonite and Zn ion-exchanged bentonite are obtained. The UV-vis spectrum of bentonite and ZnSO4 shows an absorption peak at approximately 250 nm and 311 nm, respectively (Fig. 4). After the ion exchange process, the peak near 250 nm disappears. The peak of the ZnSO4 shifts slightly to the red wavelength by 1�C6 nm placed between these two values, i.e., 311�C318 nm. This observation is attributed to the formation of ZnO/ bentonite composites, indicating the slight increases in the size and distribution of ZnO formed in the complexes.
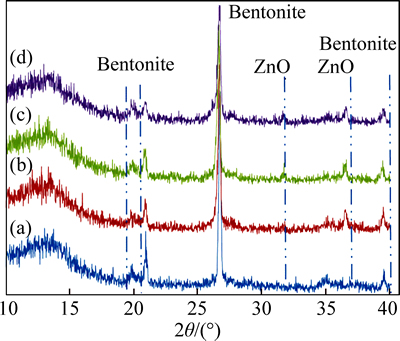
Fig. 3 XRD patterns of bentonite (a) and Zn ion-exchanged bentonite for 10 min (b), 40 min (c) and 90 min (d)
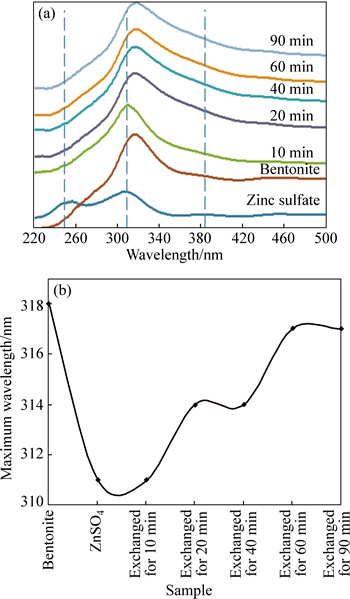
Fig. 4 Diffusive reflectance spectra and (a) wavelength of maximum absorbance (b) of prepared samples
Another interesting wavelength is around 340 nm. It can be seen at the shoulder of the bentonite spectrum. Figure 4(a) illustrates that the peak of the parent bentonite and zinc sulphate around 340 nm is almost symmetric. The absorbance of prepared nanocomposites at 340 nm has been increased by increasing the ion exchange time, through which this peak becomes asymmetric.
3.4 Antibacterial activity tests
Zinc oxide nanoparticles have been reported effective in deactivating and inhibiting both gram- negative and gram-positive bacterial growth. However, more pronounced results have been shown for the gram- negative bacteria. Generally, gram-negative organisms are more resistant to the growth-inhibiting effect of copper ions/nanoparticles than to gram-positive ones [2, 15]. The antibacterial activity of the prepared composites was tested against gram-negative E. coli bacteria as a model.
E. coli is one of the bacteria that extensively pollute drinking water. Since the ultimate goal of this research is to use the produced nanocomposites to disinfect drinking water, these bacteria were selected to evaluate nanocomposite antibacterial effects on drinking water.
Table 1 illustrates results of the antibacterial tests, which reveal that bentonite does not exhibit any antibacterial effect. The composites of ZnO/bentonite show good antibacterial properties, confirming that they can be used alone for antibacterial action. The results indicate that composites synthesised through longer ion exchange time yield greater beneficial effects in inhibiting bacterial growth.
Table 1 Mortality of ZnO/bentonite against E. coli
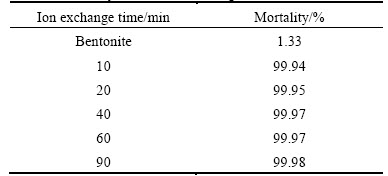
The primary objective of combining these two materials was to produce the composites for drinking water treatment. These results suggest that a composite can be an effective antibacterial material in this application. The enhanced antibacterial activity of the composites can be attributed to the chemical and physical properties of the nanoparticles with a large surface area of the clay. This implies that a great amount of ZnO is highly dispersed per unit area of the bentonite surfaces, thereby, maximizing antibacterial activity.
According to the DRS results, when the distance between base line and peak is recorded at a relatively low time setting, e.g., 10, 20 or 40 min, the distance between base line and peak increases. However, there are no discernible differences between base line and peak for the other samples generated at 40, 60 or 90 min. Interestingly, Table 1 indicates that the antibacterial activity at 40 min is higher than that at 10 and 20 min, while the tests at 40, 60 and 90 min show a similar amount of antibacterial activity. On the other hand, some reports revealed that enhanced Zn bonding resulted in increased visible absorbance [16-17]. Therefore, one could conclude that the rate of Zn bonding with bentonite increases until 40 min, thereby enhancing the antibacterial activity, while after 40 min the bonding rate decreases. This process can be explained by diffusion mass transport equations [18-19]. Given the lack of Zn in the initial bentonite, the concentration gradient and, therefore, the tendency toward Zn absorption are high. Later, Zn absorption makes concentration gradual and causes a decline in mass transport.
3.5 Leaching tests
Because the final application of these composites is to disinfect drinking water, high assurance of our results is necessary; therefore, the nanocomposites�� stability in water was measured twice. Normally, when nanoparticles are embedded into certain substrates, treatment performance stays in a fixed bed mode, in which the current conditions are nearly laminar. The vigorous agitation adopted for the leaching test therefore does not represent necessarily the most adverse condition that may be experienced, but is important to ensure that the fabric is stable under any condition. The accepted concentration of ZnO (as Zn) in drinking water as approved by the World Health Organization is 3�C5 mg/L [20]. Measurements show that the concentrations of Zn are less than 4 mg/L between 0 and 6 h (Table 2). The obtained results indicate that zinc does not pose any danger for drinking water treatment. Remarkably, the ZnO/bentonite composite exhibits minimum leaching of metals compared to findings in other studies [1-2]. This can be attributed to the desired attachment of diffused zinc to the bentonite matrix. The advantage of this stability is that the antibacterial property of ZnO/bentonite composite would last longer. On the other hand, because of the easy method of the nanocomposite��s production and using this inexpensive material, there is no reason for restoration. However, restoration can be achieved by immersing the used composite in the melted ZnSO4. Zn ions diffuse into the structure and organic materials, evaporate at 550 ��C and turn into CO2 and H2O.
Table 2 Zinc content (10-6) in water at different contact times between Zn-bentonite and aqueous media

According to the SEM analysis, increasing the porosity of the nanocomposite results in an enhanced surface bounded with Zn; therefore, antibacterial activity increases. Given the results of the leaching test, there are no differences among the realising of Zn at different times. Consequently, the important factor for antibacterial activity might be related to the Zn bounded with bentonite.
4 Conclusions
1) Nanocomposites of ZnO/bentonite are synthesized for use as antibacterial substances by speedy and simple alkaline ion exchange method.
2) XRD, DRS and SEM observations confirm the diffusion of ZnO particles to the clay surfaces.
3) The greatest antibacterial activity is detected in ZnO/bentonite composite alkaline ion exchanges lasting 60 and 90 min.
4) Leaching test results indicate that ZnO/ bentonite does not present any risk to drinking water.
References
[1] MA Y L, YANG B, BEIJING L X. Antibacterial mechanism of Cu2+ ZnO/cetylpyridinium�Cmontmorillonite in vitro [J]. Applied Clay Science, 2010, 50: 348-353.
[2] MOTSHEKGA S C, RAY S S, ONYANGO M, MOMBA M N B J. Microwave-assisted synthesis, characterization and antibacterial activity of Ag/ZnO nanoparticles supported bentonite clay [J]. Hazard Mater, 2013, 15: 439-446.
[3] APPENDINI P, HOTCHKISSJ H. Review of antimicrobial food packaging [J].Innovative Food Sci Emerg Technol, 2002, 3:113-126.
[4] TAN Shao-zao, ZHANG Kui-hua, ZHANG Li-ling, XIE Yu-shan, LIU Ying-liang. Preparation, characterization of the antibacterial Zn2�� or/and Ce3�� loaded montmorillonites [J]. Chinese Journal of Chemistry, 2008, 26: 865-869.
[5] de AZEREDO H M C. Antimicrobial nanostructures in food packaging [J]. Trends in Food Science & Technology, 2012, 30: 1-14
[6] STANI V, DIMITRIJEVI
V, DIMITRIJEVI S, ANTI
S, ANTI -STANKOVI
-STANKOVI J, MITRI
J, MITRI A M, JOKI
A M, JOKI B, PLE
B, PLE A
A I B, RAI
I B, RAI EVI
EVI S. Synthesis, characterization and antimicrobial activity of copper and zinc-doped hydroxyapatite nanopowders [J]. Applied Surface Science, 2010, 256: 6083-6089.
S. Synthesis, characterization and antimicrobial activity of copper and zinc-doped hydroxyapatite nanopowders [J]. Applied Surface Science, 2010, 256: 6083-6089.
[7] OSKAM G. Metal oxide nanoparticles: Synthesis, characterization and application [J]. Journal of Sol-Gel Science and Technology, 2006, 37: 161-164.
[8]  A, JESIONOWSKI T. Zinc oxide from synthesis to application: A Review [J]. Materials, 2014, 7: 2833-2881.
A, JESIONOWSKI T. Zinc oxide from synthesis to application: A Review [J]. Materials, 2014, 7: 2833-2881.
[9] MANIKANDAN D, MOHAN S, NAIR K G M. Optical absorption of copper nanocluster composite soda-lime glass synthesized by binary ion-exchange and ion irradiation [J]. Materials Letters, 2004, 58: 907-910.
[10] GONELLA F, QUARANTA A, CATTARUZZA E, PADOVANI S, SADA C, ACAPITO F, MAURIZIO C. Cu-alkali ion exchange in glass: A model for the copper diffusion based on XAFS experiments [J]. Computational Materials Science, 2005, 33: 31-36.
[11] TI Y, QIU F, CAO Y, JIA L, QIN W, ZHENG J, FARRELL G, Photoluminescence of copper ion exchange BK7 glass planar Waveguides [J]. J Mater Sci, 2008, 43: 7073-7078.
[12] RAMASWAMY R V, SRIVASTAVA R J. Ion exchange glass waveguide: A review [J]. Light Wave Technol, 1988, 6: 984.
[13] MA'RQUEZ H, SALAZAR D, VILLALOBOS A, PAEZ G, RINCO'N J M. Experimental study of Cu+�CNa+ exchanged glass waveguides [J]. Appl Opt, 1995, 34: 5817-5822.
[14] DRELICH J, LI B, BOWEN P, HWANG JY, MILLS O, HOFFMAN D. Vermiculite decorated with copper nanoparticles: Novel antibacterial hybrid material [J]. Applied Surface Science, 2011, 257: 9435-9443.
[15] DAS G, KALITA R D, GOGOI P, BURAGOHAIN A K, KARAK N. Antibacterial activities of copper nanoparticle-decorated organically modified montmorillonite/epoxy nanocomposites [J]. Applied Clay Science, 2014, 90: 18-26.
[16] SRIVASTAVA V, GUSAIN D, CHANDRA SHARMA Y. Synthesis, characterization and application of zinc oxide nanoparticles (n-ZnO) [J]. Ceramics International, 2013, 39: 9803-9808.
[17] RATKOVICH A, PENN R L. Zinc oxide nanoparticle growth from homogenous solution: Influence of Zn:OH, water concentration, and surfactant additives [J]. Materials Research Bulletin, 2009, 44: 993�C998.
[18] GONELLA F, CACCAVALE F. Experimental study of copper�Calkali ion exchange in glass [J]. J Appl Phys, 1998, 83: 1200-1206.
[19] KODAMA T, KOMARNENI S. Alkali metal and alkaline earth metal ion exchange with Na-4-mica prepared by a new synthetic route from kaolinite [J]. J Mater Chem, 1999, 9: 2475-2480.
[20] World Health Organization. Guidelines for drinking-water quality [S]. 4th ed. 2011.
(Edited by YANG Bing)
Received date: 2015-04-27; Accepted date: 2015-06-25
Corresponding author: Mohammad Ghorbanpour; Tel: +98-451-33512081; E-mail: Ghorbanpour@uma.ac.ir