Structure characteristics and adhesive property of humic substances extracted with different methods
来源期刊:中南大学学报(英文版)2011年第4期
论文作者:张元波 韩桂洪 姜涛 黄艳芳 李光辉 郭宇峰 杨永斌
文章页码:1041 - 1046
Key words:humic substances; structure characteristic; agglomeration behaviour; green ball
Abstract: The structure characteristics and adhesive property of humic substance (HS) extracted with different methods were mainly studied by terms of elementary analysis, visible spectrum, FT-IR spectroscopy, viscosity, adsorption and pelletizing experiments. The results show that HSs extracted with new method (HS-a) own higher degree of aromatization and polymerization, larger relative molecular mass and more polar functional groups than HS extracted with usual method (HS-b). The viscosity of HS-b is about 30- 40 mPa・s lower than that of HS-a. The maximum adsorption amounts of HS-a and HS-b onto iron concentrates are 9.11 mg/g and 8.08 mg/g, respectively. Meanwhile, HS-a has a better performance than HS-b in the practical application for pelletizing of iron concentrates. The difference in agglomeration behaviors with iron concentrates lies in the differences of the structure characteristics of HSs. With higher content of polar functional groups, larger relative molecular mass and viscosity of HSs, the agglomeration behavior is improved.
J. Cent. South Univ. Technol. (2011) 18: 1041-1046
DOI: 10.1007/s11771-011-0801-8![]()
ZHANG Yuan-bo(张元波), HAN Gui-hong(韩桂洪), JIANG Tao(姜涛), HUANG Yan-fang(黄艳芳),
LI Guang-hui(李光辉), GUO Yu-feng(郭宇峰), YANG Yong-bin(杨永斌)
School of Minerals Processing and Bioengineering, Central South University, Changsha 410083, China
? Central South University Press and Springer-Verlag Berlin Heidelberg 2011
Abstract: The structure characteristics and adhesive property of humic substance (HS) extracted with different methods were mainly studied by terms of elementary analysis, visible spectrum, FT-IR spectroscopy, viscosity, adsorption and pelletizing experiments. The results show that HSs extracted with new method (HS-a) own higher degree of aromatization and polymerization, larger relative molecular mass and more polar functional groups than HS extracted with usual method (HS-b). The viscosity of HS-b is about 30- 40 mPa・s lower than that of HS-a. The maximum adsorption amounts of HS-a and HS-b onto iron concentrates are 9.11 mg/g and 8.08 mg/g, respectively. Meanwhile, HS-a has a better performance than HS-b in the practical application for pelletizing of iron concentrates. The difference in agglomeration behaviors with iron concentrates lies in the differences of the structure characteristics of HSs. With higher content of polar functional groups, larger relative molecular mass and viscosity of HSs, the agglomeration behavior is improved.
Key words: humic substances; structure characteristic; agglomeration behaviour; green ball
1 Introduction
Binders are generally used to agglomerate fine iron concentrates into green pellets. Agglomeration experiments of iron ore have shown that the strength of green pellet is obviously improved with the addition of organic binders [1-2]. In recent years, a wide variety of organic binders including cellulose, starch and gum, have been tested [3]. However, these materials are found to be unsuitable due to high costs, failure to meet green pellet strength, environmental concerns, or a combination of these factors. One promising type of novel organic binders is based on humic substance (HS), namely HS-type binders, which have been proved by laboratory studies and industrial practice to be suitable for producing direct reduction iron (DRI) pellets in China [4]. The presence of HS onto surface of iron ore particles is able to improve the agglomeration and the ballability of iron ore particles. Both of the pellet strength and applied cost can meet the demand of iron-making plants [5]. Therefore, the HS-type binders appear to be a suitable replacement for the current organic binders.
Alkaline extraction is used as the usual preparation method of HSs from a wide range of sources including lignite, weathered coal and peat. Because of low extraction yields of HS from inferior lignite with traditional alkaline extraction, a new extraction method with assistant anthraquinone (AQ) has been patented and successfully used to increase the extraction yields [6]. In laboratory studies, the structure characteristic of HS from different sources have been extensively studied. Documents indicate that the structure characteristics of HS vary with original coals [7-8]. However, there have been few studies of the structure characteristics of HSs which are extracted from the same sources with different methods. Furthermore, the structure characteristics of HS have great impacts on the agglomeration behaviors with iron concentrates. But, there are fewer researches on the differences in structure characteristics and their agglomeration behaviors with iron ore concentrates of HS-type binders.
In this work, the structure characteristics of two HSs extracted from lignite with a usual method and a new method were mainly studied. Great importance was attached to the differences in elementary composition, visible spectrum, FT-IR spectra, and viscosity analysis of binders and their agglomeration behaviors with iron concentrates. These findings would be beneficial to the application of HS-type binders.
2 Experimental
2.1 Materials
The lignite sample used in this study was from Liling in Hunan Province of China [6].
The iron concentrate was obtained from Shanxi Province (Taiyuan, China). As seen from Table 1, it consists of about 94% of magnetite, and the main gangue mineral is SiO2. The content of the sample below 0.074 mm is 90.2% and the specific surface area is 1580 cm2/g.
Table 1 Chemical composition of iron concentrate (mass fraction, %)

2.2 Extraction methods of HS from lignite
Sodium hydroxide (NaOH) and chemical agent AQ were used as extractant and assistant, respectively.
The new extraction method of HS was described as follows. Under the condition of extraction temperature 80 °C, time 30 min, solid-to-liquid ratio 1:3, stirrer speed 600 r/min, alkali-to-lignite ratio 9%, assistant AQ dosage 0.75%, HS was extracted from lignite with extraction yields of 80%. Then, the HS was named as HS-a subsequently.
The usual extraction method of HS was presented as follows. Under the condition of extraction temperature 100 °C, time 60 min, solid-to-liquid ratio 1:3, stirrer speed 600 r/min, alkali-to-lignite ratio 12%, HS was extracted from lignite with extraction yields of 62%. Then, the HS was named as HS-b subsequently.
The two HSs (HS-a and HS-b) were dried at 60 °C to a constant mass and ground to less than 0.074 mm in grain size.
2.3 Analytical methods
2.3.1 Elementary composition
The elementary composition of HS was tested with a Model PE2400 CHN hydrocarbon elemental analyzer. Ash contents were obtained after roasted at 750 °C for 4 h in a muffle furnace.
2.3.2 Visible spectrum
The main testing methods were as follows. Firstly, HS of 50 mg/L was adjusted to pH 7. Visible spectrum of HS was performed from 400 nm to 760 nm on a model 722 N visible spectrophotometer with distilled water as the reference solution.
2.3.3 FT-IR spectroscopy
The infrared spectra of HSs were recorded using a Model FTS740 series Fourier transform infrared spectrometer from Digilab operating in the range of 4 000-400 cm-1. The KBr pellet technique was adopted for recording the spectra. Approximately 1 mg of the desired powder sample was thoroughly mixed with 100 mg of spectroscopic grade KBr and pressed into pellets for recording the spectroscopy.
2.3.4 Viscosity analysis
Viscosity of HS was measured with NDJ-1 viscometer with measurement error of ±5% which was used for measuring the viscous resistance and absolute viscosity of liquid. The accessory No.1-4 rotors (6 r/min, 12 r/min, 30 r/min, 60 r/min) can be chosen according to the level of liquid measured and rotating speed. In this work, HS solution was firstly adjusted to designed concentration, pH and temperature. Then, the viscosity of binder solution was tested with rotating speed of 60 r/min. Each test was repeated twice and an average value was reported.
2.3.5 Adsorption experiments
Firstly, HS of 100 mg/L was adjusted to pH 10 by minimal additions of 0.1 mol/L NaOH buffer. Secondly, adsorption amount of HS on iron ore surfaces was studied with batch adsorption experiments using HS to iron ore mass ratio of 1.0%.
The adsorbed amount of HS by iron ore was estimated as the difference in HS concentrations before and after adsorption according to Eq.(1):
![]() (1)
(1)
where Q (mg・g-1) is the amount of HS adsorbed by per unit mass of iron ore at different adsorption time; c1 (mg/L) and c2 (mg/L) are the initial and final concentrations of HS in the solution, V (mL) and m0 (g) represent the volume of the liquid phase and the mass of iron ore added into each beaker, respectively.
3 Results and discussion
The precise molecular structures of HS are not well understood so far. Most of the data on structure characteristics of HS refer to average properties with elementary composition and relative molecular mass [8]. The main index which reflects the structure characteristics of HS, as well as the comparison of their agglomeration behaviours with iron concentrates, was studied.
3.1 Elementary composition and visible spectrum analysis
The results of elementary composition of the two HSs extracted with the two methods are listed in Table 2.
As shown in Table 2, for the same lignite, the contents of main elements in two samples of HS are very similar, but there are still some differences between them. The data in Table 2 indicate that, compared with HS-b, C/O molar ratio of 2.26 in HS-a is relatively lower. This shows that HS-a possesses more polar functional groups (―C=O, ―OH, etc). And in comparison with C/H ratio of 2.24 in HS-b, C/H ratio of 2.29 is slightly higher in HS-a. Literatures indicate that C/H molar ratio means the degree of aromatization and polymerization. The higher the C/H ratio is, the higher the degree of aromatization and polymerization is [8-9]. These results show that the HS extracted with the new method owns higher degree of aromatization and polymerization than the HS extracted with the usual method.
Table 2 Elementary compositions of two kinds of HSs
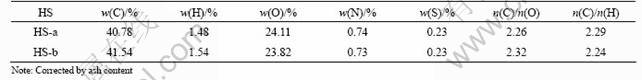
The optical properties are important to determine the structure characteristics of HS [10]. The visible spectra of HS-a and HS-b are plotted in Fig.1.
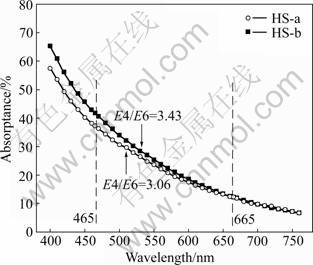
Fig.1 Visible spectra of two kinds of HSs
As seen from Fig.1, with the increase of wavelength from 400 nm to 760 nm, the absorbance of the two kinds of HSs decreases obviously. The optical density ratio (E4/E6) of solution at 465 nm and 665 nm is the molecular characteristic of HS simultaneously. The greater the E4/E6 ratio is, the smaller the relative molecular mass is. Moreover, E4/E6 ratio is also a measure of the degree of aromatization. The greater the E4/E6 ratio is, the smaller the degree of aromatization is. The optical density ratios (E4/E6) of HS-a and HS-b are 3.06 and 3.43, respectively, which indicates that HS-b owns smaller relative molecular mass and degree of aromatization than HS-a. The finding confirms the results of the elementary analysis.
3.2 FT-IR spectroscopy study
FT-IR spectroscopy has been extensively used in organic matter research and its band assignment, orientation data, and structural details have been produced [10].
The structural differences between HS-a and HS-b were also studied by means of FT-IR spectroscopy. It can be seen from Fig.2 that absorption at 3 423 cm-1 in HS-a is attributed to the stretching of ―OH groups. In addition, the very weak absorption around 2 500 cm-1 in HS-a, assigned to strongly hydrogen-bonded groups (O―H stretching of COO―H), is possible to be placed between 2 350 and 2 650 cm-1. The ―OH gives a medium-weak intensity band at 1 275 cm-1. Absorptions at 1 720 cm-1 and 1 600 cm-1 are attributed to aromaticity ―C=C― in benzene ring [11-12].
Fig.2 FT-IR spectroscopy of two kinds of HSs: (a) HS-a; (b) HS-b
As shown in Fig.2, differences in structural signatures are obvious between HS-a and HS-b simultaneously. The relative peak intensities, reflecting the relative amount of each functional group, are different for the two kinds of HSs. The spectroscopy shows bands centered around 1 700 cm-1 and 1 800 cm-1, but the bands at 1 720 cm-1 and 1 275 cm-1 for HS-a are stronger than those for HS-b. This indicates that HS-a owns higher degree of aromatization. In comparison with HS-b extracted with the usual method, HS-a extracted with the new method owns more polar functional groups such as COOR, COOH and ―OH. These results are in agreement with the elementary and visible spectrum analysis.
It is shown that the adsorption of HS onto iron ore surfaces is attributed to its polar functionality [13-14]. The difference in functional groups may lead to the different agglomeration behaviors of HS with iron ore, although this idea needs further testing.
3.3 Viscosity investigation
The viscosity of HS was mainly studied. For HS-a, the relationship curves between viscosity and pH, temperature and content of HS solution are presented in Fig.3. It can be seen from Fig.3 that, the viscosity of HS-a is significantly dependent on pH, temperature and content. The viscosity of HS-a solution obviously decreases with the increase of pH and temperature, but increases with the increase of the content of HS-a solution. The magnitude of the viscosity of HS-a solution, especially, is decreased intensely from 480 mPa・s to 6 mPa・s as pH is enhanced from 5 to 11. The main reason is that HS is easy to aggregate with each other at low pH values so that it is difficult to disperse into the solution. Therefore, comparative studies on viscosity of different HSs are necessary under the suitable and same conditions of content, pH, temperature and content.
The viscosity of the two kinds of HS is listed in Table 3. As shown in Table 3, the viscosity of the two kinds of HSs is different from each other under different conditions. Under the same condition, the viscosity of HS-b is about 30-40 mPa・s lower than that of HS-a. Under the condition of pH 10, content 20% and temperature 25 °C, the viscosities of HS-a and HS-b are 145 mPa・s and 105 mPa・s, respectively. The viscosity is a measure of the relative molecular mass at the same time [15]. And the larger the relative molecular mass is, the higher the viscosity is. The results show that the relative molecular mass of the HS exacted with the new method is larger than that of HS exacted with usual method.
3.4 Comparison of agglomeration behaviours
Adsorption property is an important factor to reflect the agglomeration behaviour of HS with iron concentrates [5, 16]. The adsorption amounts of the two kinds of HSs onto the iron concentrates against the adsorption time are plotted in Fig.4.

Fig.3 Relationship curves between viscosity and conditions of HS-a solution: (a) At different pH values; (b) At different temperatures; (c) At different contents
Table 3 Viscosities of two kinds of HSs under different conditions
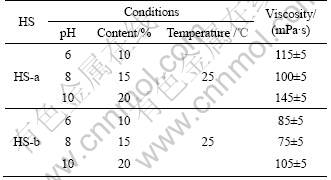
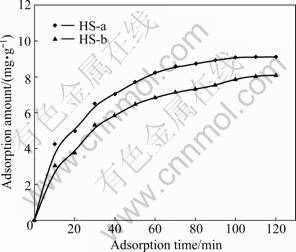
Fig.4 Adsorption amount of two kinds of HSs onto iron concentrates against adsorption time
The results in Fig.4 show that, the adsorption amount of the HS keeps unchanged as adsorption time is increased to120 min. This indicates that the adsorption of the HS onto the iron concentrates reaches the saturation. The adsorption of the two kinds of HSs differs mainly in the adsorption amount. Under the condition of the same adsorption time, in contrast with HS-b, adsorption amount of HS-a onto the iron concentrates is much higher. The maximum adsorption amounts of HS-a and HS-b onto iron concentrates are 9.11 mg/g and 8.08 mg/g, respectively. The difference in adsorption amount lies in the structure characteristics of the two kinds of HSs. The adsorption amounts of HSs are positively correlative with the functional groups. Just as described in elementary composition, visible spectrum analysis and FT-IR spectroscopy study, HS-a owns more polar functional groups such as ―COOH and ―OH. Due to the more functional groups of HS-a, its adsorption is more readily enhanced.
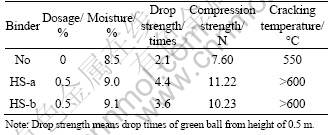
The practical balling results in Table 4 show that the pellets with HS-type binders possess much higher strengths than those with no binder, which is similar to those reported by others [16-17]. This shows that the HS-type binders are effective to iron ore pelletization. It is obvious that the pellets with HS-type binders possess cracking temperature above 600 °C. This displays that the binders possess a good heat stability. Meanwhile, it can be seen from Table 4 that, HS-a pellets have better performances than HS-b pellets. Under the condition of dosage of 0.5% binder, the drop strength and compression strength of green pellets with HS-a is 4.4 times and 11.22 N, respectively.
The results further prove that the different structure characteristics of HSs result in their agglomeration behaviors with iron concentrates. The difference in their agglomeration behaviors can be explained by two reasons. One is that the adsorption property of HS-a onto iron concentrates is better than that of HS-b, which leads to higher strength of green pellets. The other is that the viscosity of HS-type binders plays an important role in the agglomeration behaviors with iron concentrates [18]. The viscosity increases the plasticity of green pellet. If the plasticity is higher, the strength of green pellet is increased. Because of the higher viscosity of HS-a, the strength of green pellet is larger.
Table 4 Agglomeration behaviours of iron concentrates with two kinds of HSs
4 Conclusions
1) The elementary composition shows that HS extracted with a new method (HS-a) owns higher degree of aromatization and polymerization than HS extracted with usual method (HS-b). Compared with HS-b, C/O molar ratio of 2.26 in HS-a is relatively lower. The optical density ratios (E4/E6) of HS-a and HS-b are 3.06 and 3.43, respectively, which indicates that HS-b owns smaller relative molecular mass and degree of aromatization than HS-a. The FT-IR spectroscopy analyses indicate that HS-a owns more polar functional groups, such as COOR, COOH and ―OH than HS-b.
2) The viscosity results show that the viscosity of HSs is significantly dependent on pH, temperature and content. Under the same condition, the viscosity of HS-b is about 30-40 mPa・s lower than that of HS-a. Under the conditions of pH 10, content 20% and temperature 25 °C, the viscosities of HS-a and HS-b are 145 mPa・s and 105 mPa・s, respectively.
3) The adsorption of the two kinds of HSs differs mainly in the adsorption amount. Under the condition of same adsorption time, in contrast with HS-b, adsorption amount of HS-a onto iron concentrates is much higher. The maximum adsorption amounts of HS-a and HS-b onto magnetite are 9.11 and 8.08 mg/g, respectively. Through the practical application of HS-a and HS-b in balling of iron concentrates, HS-a has a better performance than HS-b.
4) The agglomeration behaviors of HSs with iron concentrates vary with their structure characteristics. Because of the better adsorption property onto iron concentrates and higher viscosity of HS-a, the strength of green pellet is larger.
References
[1] Allen A P. The role of binders in the agglomeration of minerals [C]// Proceeding of 5th International Symposium on Agglomeration, UK, 1989: 215-223.
[2] SUZUKI S, GOMI S. New organic pellet binder from bituminous materials [C]// Symposium on Pellets and Granules. Newcastle, 1994, 157-169.
[3] HEERMA R H, KORTMAN H, KATER T. Improvements of acid, olivine and dolomite fluxed iron ore pellets using an organic binder [C]// Proceeding of 5th International Symposium on Agglomeration, UK, 1998: 227-239.
[4] JIANG Tao, QIU Guan-zhou, Zhu De-qing, Fan Xiao-hui, Huang Zhu-cheng. A new process of coal-based direct reduction of iron concentrates pellets-CBP process [C]// Proceeding of 60th Iron-making Conference. Baltimore, 2001: 803.
[5] Han Gui-hong, Zhang Yuan-bo, Huang Yan-fang, Li Guang-hui, Jiang Tao. Adsorption behaviours of humic substances onto iron ore particle surface [C]// XXV Internal Mineral Processing Congress. Brisbane, 2010: 163-171.
[6] JIANG Tao, HAN Gui-hong, ZHANG Yuan-bo, HUANG Yan-fang, LI Guang-hui, GUO Yu-feng, YANG Yong-bin. Improving the extraction yield of humic substances (HS) from lignite with anthraquinone (AQ) in alkaline solution [J]. Journal of Central South University of Technology, 2011, 18(1): 68-72.
[7] LIANG Chong-shan, DANG Zhi, LIU Cong-qiang. Structure characterization of soil humic acids and adsorption equilibria on phenanthrene [J]. Chinese Journal of Analytical Chemistry, 2006, 3(24): 288-292. (in Chinese)
[8] PEURAVUORI J, Zbankova P, Pihlaj a K. Aspects of structural features in lignite and lignite humic acids [J]. Fuel Processing Technology, 2006, 87: 829-839.
[9] Be?atrice A. A comparative study on the chemical composition of humic acids from forest soil, agricultural soil and lignite deposit Bound lipid, carbohydrate and amino acid distributions [J]. Gendarme, 2006, 130: 77-96.
[10] Henrique R. Oxidation of humic substances of peat during alkaline extraction: Infrared and visible spectroscopic study [J]. Quim Nova, 1998, 47(l): 25-28.
[11] LIU Guo-gen, QIU Guan-zhou, HU Yue-hua. Study on infrared spectra of coals [J]. Journal of Central South University: Science and Technology, 1999, 30(4): 371-373. (in Chinese)
[12] LIU Guo-gen, QIU Guan-zhou, XU Jing-cang. Study on spectroscopy of HA type binder [J]. Journal of Central South University: Science and Technology, 2003, 34(3): 238-241. (in Chinese)
[13] LI Hong-xu, JIANG Tao, QIU Guan-zhou, WANG Dian-zuo. Molecular structure model and selecting criterion of organic binder for iron ore pellet [J]. Journal of Central South University: Science and Technology, 2000, 31(1): 17-20. (in Chinese)
[14] QIU Guan-zhou, JIANG Tao, LI Hong-xu, WANG Dian-zuo. Functions and molecular structure of organic binders for iron ore pelletization [J]. Colloids and Surfaces A: Physicochemistry, 2003, 224: 11-22.
[15] Peuravuori J, Pihlaja K. Molecular size distribution and spectroscopic properties of aquatic humic substances [J]. Analytica Chimica Acta, 1997, 337: 133-149.
[16] LIU Guo-gen. A study on HA-type binders for direct reduction of cold bond pellets and its raw material characteristics [D]. Changsha: Central South University, 1997. (in Chinese)
[17] Qiu Guan-zhou, Jiang Tao, XU Jing-cang. Direct reduction of cold-Bonded pellets [M]. Changsha: Central South University Press, 2001: 115-120. (in Chinese)
[18] QIU Guan-zhou, JIANG Tao, FA Ke-qing, ZHU De-qing, WANG Dian-zuo, Interfacial characterizations of iron ore concentrates affected by binders [J]. Powder Technology, 2004, 139: 1-6.
(Edited by YANG Bing)
Foundation item: Project(50725416) supported by the National Science Fund for Distinguished Young Scholars of China; Project(50804059) supported by the National Natural Science Foundation of China; Project(200805331080) supported by Specialized Research Fund for the Doctoral Program of Higher Education of China; Project supported by the Graduate Degree Thesis Innovation Foundation of Central South University, China
Received date: 2010-04-09; Accepted date: 2011-01-21
Corresponding author: JIANG Tao, Professor, PhD; Tel: +86-731-88877656; E-mail: jiangtao@csu.edu.cn

