Fabrication of ferrocenyl glutathione modified electrode and its application for detection of cadmium ions
来源期刊:中南大学学报(英文版)2008年第1期
论文作者:王芳斌 范美意 刘又年 王建秀 曾冬铭 黄可龙
文章页码:44 - 44
Key words:self-assembled monolayers; synthesis; modified electrode; cadmium detection; ferrocene-glutathione
Abstract: 1′-cysteaminecarbonyl-1-glutathionecarbonyl-ferrocene (Fc-GSH) was synthesized from ferrocene dicarboxylic acid and reduced glutathione (GSH) with 4 steps. IR and 1H-NMR were used to characterize the products. Then Fc-GSH was immobilized on the surface of gold electrode. Cyclic votammetry (CV) was adopted to investigate the electrochemical properties of this Fc-GSH modified electrode in the absence and presence of Cd2+ aqueous solutions. The peak oxidation potential (Ea) and reduction potential (Ec) of Fc-GSH modified electrode were observed at Ea = 0.74 V and Ec = 0.64 V (vs Ag/AgCl) before the accumulation of Cd2+. This redox process is a monoelectron chemical reaction. The anodic shift is about 80 mV in the presence of 20 nmol/L of Cd2+ aqueous solution. Moreover, this shift is in proportion to the concentration of Cd2+ when the concentration of Cd2+ is lower than 20 nmol/L. So the modified electrode can be used as probes to detect cadmium ions with the limit of 0.1 nmol/L by cyclic voltammetry.
基金信息:the National Natural Science Foundation of China
J. Cent. South Univ. Technol. (2008) 15: 44-48
DOI: 10.1007/s11771-008-0010-2
![]()
WANG Fang-bin(王芳斌), FAN Mei-yi(范美意), LIU You-nian(刘又年),
WANG Jian-xiu(王建秀), ZENG Dong-ming(曾冬铭), HUANG Ke-long(黄可龙)
(School of Chemistry and Chemical Engineering, Central South University, Changsha 410083, China)
Abstract: 1′-cysteaminecarbonyl-1-glutathionecarbonyl-ferrocene (Fc-GSH) was synthesized from ferrocene dicarboxylic acid and reduced glutathione (GSH) with 4 steps. IR and 1H-NMR were used to characterize the products. Then Fc-GSH was immobilized on the surface of gold electrode. Cyclic votammetry (CV) was adopted to investigate the electrochemical properties of this Fc-GSH modified electrode in the absence and presence of Cd2+ aqueous solutions. The peak oxidation potential (Ea) and reduction potential (Ec) of Fc-GSH modified electrode were observed at Ea = 0.74 V and Ec = 0.64 V (vs Ag/AgCl) before the accumulation of Cd2+. This redox process is a monoelectron chemical reaction. The anodic shift is about 80 mV in the presence of 20 nmol/L of Cd2+ aqueous solution. Moreover, this shift is in proportion to the concentration of Cd2+ when the concentration of Cd2+ is lower than 20 nmol/L. So the modified electrode can be used as probes to detect cadmium ions with the limit of 0.1 nmol/L by cyclic voltammetry.
Key word:self-assembled monolayers; synthesis; modified electrode; cadmium detection; ferrocene-glutathione
1 Introduction
In recent years, self-assembled monolayers(SAMs) of alkanethiols with different functional groups on gold have been extensively studied because of their high stabilities, good orientation, and easy preparation. Now SAMs are widely used as biosensors[1-4], molecular electronic devices[5]. For example, KONDO et al[6] constructed a novel alkylthiol molecule containing ferrocene and azobenzene moieties on gold as a photochemical sensor. In fact, SAMs offered the possibility to develop biosensors among amino acids or oligopeptides by electroanalytical methods. For these applications, ferrocene and its derivatives were the most widely studied electrochemically active groups in the SAMs. They were used to conjugate with amino acids[7], peptides[8], proteins[9] and DNA[10], which provided reversible masked peptides or efficient redox systems. For instance, BARISIC et al[11] and LIU et al[12] synthesized a series of ferrocenoyl-labeled amino acid or peptide to investigate peptide electron transport properties and the interactions between intra- or inter- molecules. Besides, ferrocene- peptides were used to detect lanthanide metals and alkaline earth metals[13].
Cadmium is one of the most important industrial pollutants among the toxic heavy metals, which causes serious problem to the ecosystem. In humans, long-term exposure in an environment with excessive cadmium can cause kidney disease as a result of intake from contaminated food and water. The World Health Organization (WHO) guideline level for cadmium in drinking water is 3 ?g/L (27 nmol/L). So there are growing needs for reliable measurements of cadmium ions in a variety of aquatic media. Several methods have been developed to the determination of trace amounts of cadmium, including mass spectrometry[14], high performance liquid chromatogram[15], atom absorb/ emission spectrum[16]. Unfortunately, these methods need expensive facilities, complicated manipulate, or are of low sensitivity.
Thiol compounds such as cysteine (Cys), reduced glutathione (GSH) played an important role in heavy metal accumulation and detoxification[17]. They can coordinate with heavy metal ions, for instance, Cd2+, Cu2+, Zn2+ and Hg2+[18]. Glutathione (GSH), a biological active peptide that contains a cysteine, is a most favorable oligopeptide and has been investigated. It has eight potential binding sites to cooperate with heavy metals. The binding of Cd2+ with GSH was studied by D?AZ-CRUZ et al[18-20], and they found that Cd2+ and GSH formed 1?1 or 1?2 complexes. CHOW et al[21] used glutathione modified electrode to detect Cd2+ and got the detection limit of 5 nmol/L. But GSH itself has no redox active group, which hindered its uses in the electrochemical detection of ions binding to GSH.
In this work, 1′-cysteaminecarbonyl-1-glutathio- necarbonyl-ferrocene (Fc-GSH) was firstly synthesized.
The Fc-GSH modified electrode was fabricated, and the modified electrode was used to detect cadmium ions. And the detection limit of 0.1 nmol/L of cadmium was obtained by cyclic voltammetry (CV).
2 Experimental 2.1 MaterialsGlutathione (GSH) (reduced form, mass fraction higher than 98%), N-hydroxysuccinimide (NHS), 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide hydrochloride (EDC) were purchased from Sigma, USA. Ferrocene (Fc) and CdCl2 were purchased from Xiangzhong Chemical Co, China. Cysteamine (CSA) and trietylamine (Et3N) were from Shanghai Chemical Reagents Co, China. O-(benzotriazol-1-yl)-N,N,N′,N′- tetramethyluronium (HBTU) was obtained from Shuzhou Haofan Biochemicals Co, China.
Infrared spectra were performed using KBr pellets on a Nicolet AVATAR360 FT-IR spectrometer (USA). 1H-NMR spectra were measured on an INOVA-400 spectrometer (USA) in CDCl3 by using tetramethylsilane as an internal standard. Electrochemical measurements were carried out on a CHI660B electrochemical analytical instrument (CH Instrument Co, Shanghai, China).
2.2 Synthesis of 1′-cysteaminecarbonyl-1-glutathion- ecarbonylferrocene ( compound 4)The synthetical procedure of ferrocenoyl conjugates and fabrication of ferrocene-GSH modified electrode are shown in Fig.1.
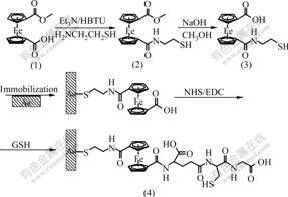
Fig.1 Synthesis of ferrocenoyl conjugates and fabrication of ferrocene-GSH modified electrode
2.2.1 Synthesis of 1′-cysteaminecarbonyl-1- methoxycar- bonylferrocene (compound 2)To the solution of compound (1) (288 mg, 1 mmol) in dry CH2Cl2, Et3N (1.0 mL) and HBTU (418 mg, 1.1 mmol) were added at room temperature. After completion, the reaction was stirred for 1 h. Then the mixture was washed successively with saturated NaHCO3 solution, 0.5 mol/L HCl, and saturated NaHCO3 solution and then distilled water. The organic phase was dried with Na2SO4, then filtered and evaporated under reduced pressure in a rotovap, the remaining solution was purified by flash chromatography on silica (volume ratio of EtOAc to hexane is 2?1, retention factor Rf=0.81) to get 150 mg crystal of active ester. Then the crystal was dissolved in dry CH2Cl2. Et3N (1.0 mmol) and cysteamine (136 mg, 1.2 mmol) was added to solution at room temperature. After completion, the reaction mixture was stirred for 2 h at room temperature, and then washed successively with saturated NaHCO3 solution, 0.5 mol/L HCl, saturated NaHCO3 and then distilled water. The organic phase was dried with Na2SO4, then filtered and evaporated under reduced pressure in a rotovap, the remaining solution was purified by flash chromatography on silica (volume ratio of EtOAc to hexane is 2?1, Rf=0.51) to get 100 mg of compound (2) with melting point of 132-133 ℃. IR (KBr, cm-1): 3 461(NH), 2 844 (S-H), 1 717 (C=O), 1 653 (N-C=O). 1H-NMR (CDCl3, δ): 7.00(s, 1H, NH), 4.91-4.38(8H, 2Cp), 3.86(t, 2H, CH2), 3.84(s, 3H, CH3), 3.31(s, 1H), 1.81(s, 1H, SH).
2.2.2 Preparation of 1′-cysteaminecarbonyl-1-ferrocene- carboxylic acid (compound 3)Compound (2) (333 mg, 1 mmol) was dissolved in MeOH (100 mL) and 1.1 mL 1 mol/L NaOH aqueous solution was added to the solution, after refluxing at 80 ℃ for 24 h, the mixture was evaporated under reduced pressure in a rotorvap to give red solid, then the solid was dissolved in water and extracted with CH2Cl2. The water layer was acidified to pH 1-2 by 1 mol/L HCl, and then filtered to give an orange solid with melting point of 135-136 ℃ and IR (KBr, cm-1): 2 923, 2 633, 2 558 and 1 674.
2.2.3 Fabrication of 1′-cysteaminecarbonyl-1-glutathione- carbonylferrocene (Fc-GSH) (compound 4) modified electrodeThe bare gold electrode(with a diameter of 2 mm)was polished to a mirror-like surface with 0.05 ?m Al2O3 slurry on microcloth pads and removed the alumina from the surface by rinsing with Milli-Q water, then brief cleaning in an ultrasonic bath, the cleaned electrode was then detected by cyclicing at the potentials between -0.2 and +0.6 V in 1 mmol/L [Fe(CN)6]4-/3-+0.1 mol/L KCl at a scan rate of 100 mV/s until reproducible scans were recorded. This freshly pretreated electrode was immersed in 10 mmol/L solution of compound (3) for 5 d to allow the Au-thiol binding forming. After that, compound (3) modified electrode was washed successively by EtOH and Milli-Q water and dried under a stream of N2, then the electrode covered with compound (3) was incubated with 10 ?L phosphate buffer solution (PBS) (pH 7.0) containing 5 mmol/L EDC and 5 mmol/L NHS as coupling reagents for 1 h, then the electrode was rinsed sequentially with PBS and Milli-Q water, after drying by N2, the activated electrode was incubated with 10 ?L PBS containing 5 mmol/L GSH for 4 h. After incubation the electrode was washed with PBS and Milli-Q and the ferrocene-GSH modified electrode was obtained.
2.3 Accumulation of cadmium ionsCdCl2 was dissolved in Milli-Q water, then the modified electrode was immersed in the solution and stirred at room temperature(23±2 ℃) for 1 h. After accumulation, the electrode was washed by Milli-Q water.
2.4 Electrochemical measurementsAll electrochemical measurements were carried out with a model CHI660B. A three-electrode system was used in the measurements, comprising a bare or modified working electrode, a platinum flag auxiliary electrode and a Ag/AgCl reference electrode, the supporting electrolyte was 0.1 mol/L KClO4 solution. All experiments were done at 25 ℃ unless otherwise stated.
3 Results and discussion 3.1 Syntheses and characterizations of compounds (2) and (3)Coupling of cysteamine to 1′-methoxycarbonyl ferrocene-1-carboxylic acid using the HBTU/Et3N procedure adapted for ferrocenoyl conjugates resulted in the formation of the desired 1′-cysteaminecarbonyl- 1-methoxycarbonylferrocene (compound (2)) as a red crystal. Because the S-H bonding of cysteamine can be readily oxidized to S-S, the yield of compound (2) in this step is rather low (<30%).
The IR spectrum of compound (2) in solid shows a typical amide band at 3 461 and 1 653 cm. In 1H-NMR spectrum, the peak of amide appears at δ 7.00.
Hydrolization of ester gave the free 1′-cysteaminecarbonyl-1-carboxylic acid (compound (3)) high yield (>90%). This compound was characterized by IR spectrum. The acid carbonyl group appears at 1 674 cm-1, indicating that compound (2) is converted to acid.
3.2 Electrochemical behavior 3.2.1 Electrochemical behavior of Cd2+ at modified electrodesThe cyclic voltammograms of modified electrodes measured in 0.1 mol/L KClO4 electrolyte is shown in Fig.2. Prior to accumulation of Cd2+, the modified electrode is active in the potential range of 0.4-1.0 V, the peaks of oxidation potential (Ea) and reduction potential (Ec) of Fc-GSH modified electrode are observed at Ea= 0.74 V and Ec= 0.64 V(vs Ag/AgCl), which is a pair of well-defined redox peaks that exhibit electrochemically and chemically reversible one electron oxidative. It is also indicated that Fc-GSH is indeed immobilized on the surface of the electrode[22]. After accumulation of Cd2+ (the modified electrode was immersed in 20 nmol/L CdCl2 aqueous solution for 1 h), it is shown that distinct redox peaks are observed at Ea=0.82 V and Ec=0.68 V(vs Ag/AgCl). The oxidation peak shifts about 80 mV to anode. These changes demonstrate that cadmium ions cooperate with Fc-GSH. Similarly, APPOH et al[13] found anode shifts for the interaction of Fc-amino acid conjugates with alkaline earth metals and lanthanide metals. The anodic shift is caused by a through space coulombic interaction of metal centers and the positively charged Fc+. This great shift can be used to detect cadmium ion at low concentrations.
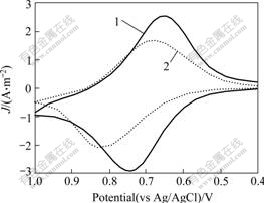
Fig.2 Cyclic voltammograms of Fc-GSH modified electrodes for detection of Cd2+: 1―Before accumulation of Cd2+; 2―After accumulation in 20 nmol/L Cd2+
3.2.2 Influence of Cd2+ concentrationThe effect of the concentration of cadmium ions on the response of Fc-GSH modified electrodes was evaluated at five different concentrations (0.1, 1.0, 5.0, 10.0, 20.0 nmol/L Cd2+, respectively). The experimental results are shown in Fig.3. Fig.3(a) shows the effects of concentration of Cd2+ on Ea, illustrating that the oxidation peak potential linearly increases with the increase of the concentration of Cd2+ when the concentration of Cd2+ is lower than 20 nmol/L. The fit line is:
Ea=0.781+0.001 96c (R=0.998) (1)
where Ea is the anodic peak potential, V; c is the concentration of Cd2+, nmol/L.
However, the peak potential changes slowly with the concentration changes when the concentration of Cd2+ is higher than 20 nmol/L. So the Fc-GSH modified electrode was used to detected cadmium ions at low concentration and got the detection limit of Cd2+ 0.1 nmol/L. The detection limit for Cd2+ with GSH-Fc modified electrode is much lower than that of GSH modified electrode (the later is 5 nmol/L[21]). This is because that the redox active group of GSH-Fc modified electrode is ferrocene, while that of GSH modified electrode is Cd2+. Ferrocene group is more sensitive than that of Cd2+ in redox reaction. So, GSH-Fc modified electrode can get much lower detection limit. ?E increases when Fc-GSH forms complex with cadmium ions. Moreover, ?E increases with the increase of Cd2+concentration (Fig.3(b)).
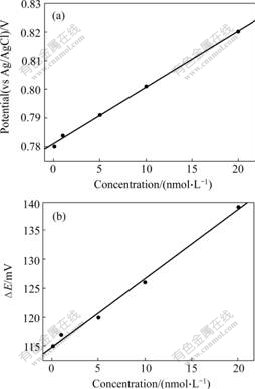
Fig.3 Cyclic voltammograms of Fc-GSH modified electrodes accumulation of different Cd2+ concentrations in 0.1 mol/L KClO4 at a scan rate of 0.1 V/s: (a) Ea vs concentration of Cd2+; (b) ΔE vs concentration of Cd2+
3.2.3 Influence of scan rateElectrode was accumulated in 5 nmol/L Cd2+ for 1 h. The effect of the scan rate on the modified electrode was studied at 5 different scan rates (0.02, 0.04, 0.06, 0.08, 0.10 V/s, respectively). Fig.4 depicts the plot of peak currents (Ip) of CV vs the scan rate. It is shown that the peak shape is independent of the scan rate, but the peak current (Ip) increases linearly with the scan rate in the range from 20 to 100 mV. According to the following equation
Ip=2.69×105n3/2ADo1/2Coυ1/2 (2)
where n is the electron number, A is the geometrical area of the gold electrode, Do is the diffusion coefficient of , Co is the concentration of cadmium ions. The peak current Ip is proportional to the scan rate υ1/2.
3.2.4 Detection for other metal ionsThe Fc-GSH modified electrode was used to detect Cu2+ and Zn2+ in aqueous solution too. These Fc-GSH modified electrode were immersed in 10 nmol/L Cu2+ and 10 nmol/L Zn2+ solution respectively for 1 h, then the electrochemical measures were carried out in the same scan ranges (from 0.4 to 1.0 V), the results are shown in Fig.5. It is observed from the experiments that the modified electrode can detect Cu2+ and Zn2+ and get the anodic peak potential about at 0.763 and 0.767 V respectively (for Cd2+ it is about 0.792 V). From Fig.5, it can be seen that the anodic peak potential and current density in Cd2+ solution are much higher than those of others because the coordination constant of Cd2+ with Fc-GSH is much larger than that of Cu2+ and Zn2+.

Fig.4 Cyclic voltammograms of Fc-GSH modified electrodes with accumulation of 5 nmol/L Cd2+ in 0.1 mol/L KClO4 at different scan rates: 1―0.02 V/s; 2―0.04 V/s; 3―0.06 V/s; 4―0.08 V/s; 5―0.10 V/s
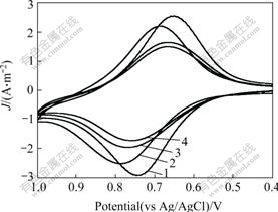
Fig.5 Cyclic voltammograms of Fc-GSH modified electrodes with accumulation of different metal ions: 1―Free of metal ions; 2―Cd2+; 3―Cu2+; 4―Zn2+
4 Conclusions1) Ferrocenoyl glutathione is synthesized and self-assemble monolayers form on gold electrode. The electrochemical properties of this modified electrode are investigated. The peak oxidation potential (Ea) and reduction potential (Ec) of Fc-GSH modified electrode are observed to be Ea=0.74 V and Ec=0.64 V(vs Ag/AgCl).
2) This modified electrode is used as probe to test Cd2+ ions because Fc-GSH and Cd2+ can form complex. And when they form coordination a large anodic shift (about 80 mV) appears. Furthermore, the shifts are in direct proportion to the concentration of ions in the solution when the concentration of Cd2+ is lower than 20 nmol/L. So the Fc-GSH modified electrode is used to detect Cd2+ at low concentration and the detection limit of Cd2+ 0.1 nmol/L is obtained.
3) GSH is a bioactive peptide, which can interact with many biomolecules. So the future research will aim at exploring these interactions and using it to recognize proteins.
References
[1] BARDEA A, KATZ E, WILLNER I. Biosensors with amperometric detection of enzymatically controlled pH-changes[J]. Electroanalysis, 2000, 12(10): 731-735.
[2] WOLOWACZ S E, YON-HIN B F Y, LOWE C R. Covalent electropolymerization of glucose oxidase in polypyrole[J]. Anal Chem, 1992, 64(14): 1541-1545.
[3] WANG Z H, VIANA A S, JIN G, ABRANTES L M. Immunosensor interface based on physical and chemical immunoglobulin G adsorption onto mixed self-assembled monolayers [J]. Bioelectrochemistry, 2006, 69(2): 180- 186.
[4] ANNE A, BOUCHARDON A, MOIROUX J. 3′-ferrocene-labeled oligonucleotide chains end-tethered to gold electrode surfaces: Novel model systems for exploring flexibility of short DNA using cyclic voltammetry[J]. J Am Chem Soc, 2003, 125(5): 1112-1113.
[5] YASUTOMI S, MORITA T, IMANISHI Y, KIMURA S. A molecular photodiode system that can switch photocurrent direction[J]. Science, 2004, 304(5679): 1944-1947.
[6] KONDO T, KANAI T, UOSAKI K. Control of the charge-transfer rate at a gold electrode modified with a self-assembled monolayer containing ferrocene and azobenzene by electro- and photochemical structural conversion of cis and trans forms of the azobenzene moiety[J].Langmuir, 2001, 17(20): 6317-6324.
[7] CHOWDHURY S, SCHATTE G, KRAATZ H B. Rational design of bioorganometallic foldamers: A potential model for parallel β-helical peptides[J]. Angew Chem Int Ed, 2006, 45(41): 6882-6884.
[8] MORITA T, KIMURA S. Long-range electron transfer over 4 nm governed by an inelastic hopping mechanism in self-assembled monolayers of helical peptides[J]. J Am Chem Soc, 2003, 125(29): 8732-8733.
[9] CHEN L Q, ZHANG X E, XIE W H, ZHOU Y F, ZHANG Z P, CASS A E G. Genetic modification of glucose oxidase for improving performance of an amperometric glucose biosensor[J].Biosens Bioelectron, 2002, 17(10): 851-857.
[10] YU C J, WAN Y, YOWANTO H, LI J, TAO C, JAMES M D, TAN C L, MEADE T J. Electronic detection of single-base mismatches in DNA with ferrocene-modified probes[J]. J Am Chem Soc, 2001, 123(45): 11155-11161.
[11] BARISIC L, CAKIC M, MAHMOUD K A, LIU Y N, KRAATZ H B, PRITZKOW H, KIRIN S I, METZLER-NOLTE N, RAPIC V. Helically chiral ferrocene peptides containing 1′-aminoferrocene-1-carboxylic acid subunits as turn inducers[J]. Chem Eur J, 2006, 12(19): 4965-4980.
[12] LIU Y N, ORLOWSKI G, SCHATTE G, KRAATZ H B. 3-Ferrocenylamido-5-methylpyrazole: Synthesis and metal coordination[J]. Inor Chim Acta, 2005, 358(4): 1151-1161.
[13] APPOH F E, SUTHERLAND T C, KRAATZ H B. Ferrocenoyl- amino acids: Redox response towards di- and trivalent metal ions[J]. J Organomet Chem, 2005, 690(5): 1209-1217.
[14] LI L, HU B, XIA L, JIANG Z. Determination of trace Cd and Pb in environmental and biological samples by ETV-ICP-MS after single-drop microextraction[J]. Talanta, 2006, 70(2): 468-473.
[15] SAITO S, DANZAKA N, HOSHI S. Direct fluorescence detection of Pb2+ and Cd2+ by high-performance liquid chromatography using 1-(4-aminobenzyl)ethylenediamine-N, N, N′, N′-tetraacetate as a pre- column derivatizing agent[J]. J Chromatogr A, 2006, 1104(1/2): 140-144.
[16] BORGES D L G, DIAS L F, DA VEIGA M A M S, CURTIUS A J. Determination of cadmium in sediments and in sewage sludge by slurry sampling electrothermal atomic absorption spectrometry using iridium as permanent modifier[J]. J Braz Chem Soc, 2003, 14(2): 291-296.
[17] STOZHKO N Y, LIPUNOVA G N, MASLAKOVA T I, ALESHINA L V, BRAJNINA K Z. A thick-film carbon-containing electrode modified with formazan for determining copper, lead, cadmium, and zinc[J]. J Anal Chem, 2004, 59(2): 179-184.
[18] DI?AZ-CRUZ M S, MENDIETA J, TAULER R, ESTEBAN M. Cadmium-binding properties of glutathione: A chemometrical analysis of voltammetric data[J]. J Inorg Biochem, 1997, 66(1): 29-36.
[19] DI?AZ-CRUZ M S, MENDIETA J, TAULER R, ESTEBAN M. Multivariate curve resolution of cyclic voltammetric data: Application to the study of the cadmium-binding properties of glutathione[J]. Anal Chem, 1999, 71(20): 4629-4636.
[20] MENDIETA J, DI?AZ-CRUZ M S, TAULER R, ESTEBAN M. Application of multivariate curve resolution to voltammetric data. II. Study of metal-binding properties of the peptides[J]. Anal Biochem, 1996, 240(1): 134-141.
[21] CHOW E, HIBBERT D B, GOODING J J. Voltammetric detection of cadmium ions at glutathione-modified gold electrodes[J]. Analyst, 2005, 130(6): 831-837.
[22] WANG Y, YAO X, WANG J, ZHOU F. Attachment of amine- and maleimide-containing ferrocene derivatives onto self-assembled alkanethiol and alkanedithiol monolayers: Voltammetric evaluation of cross-linking efficiencies and surface coverage of electroactive groups[J]. Electroanalysis, 2004, 16(21): 1775-17617.
Foundation item: Project(20676153) supported by the National Natural Science Foundation of China
Received date: 2007-04-28; Accepted date: 2007-06-12
Corresponding author: LIU You-nian, Professor, PhD; Tel: +86-731-8836964; E-mail: liuyoun@mail.csu.edu.cn

