
Reactions between Ti and Ti3SiC2 in temperature range of 1 273-1 573 K
GU Wan-li(������)1, ZHOU Yan-chun(���Ӵ�)
1. School of Mechanical Engineering, Shandong University of Technology, Zibo 255049, China;
2. High-Performance Ceramic Division, Shenyang National Laboratory for Materials Science,
Institute of Metal Research, Chinese Academy of Sciences, Shenyang 110016, China
Received 6 March 2006; accepted 22 August 2006
Abstract: The reactions of Ti3SiC2 and Ti in the temperature range of 1 273-1 573 K under a pressure of 20 MPa were investigated. The results confirm that Ti reacts with Ti3SiC2 above 1 273 K and new phases like TiCx, Ti5Si3 and TiSi2 are identified. The reactions are closely related to temperature and content of Ti3SiC2 in Ti. During the reaction process, Ti3SiC2 decomposes in two different modes. The first is caused by the de-intercalation of Si from it and the TiCx is formed by the remained titanium and carbon; the second is that the carbon is separated from the Ti3SiC2 and reacts with titanium furthermore. The diffusing of silicon is believed to be the determinant ingredient of the reaction.
Key words: Ti3SiC2; Ti; high-temperature reaction
1 Introduction
Recently, the layered ternary carbide Ti3SiC2 has attracted attention due to its unique properties such as high toughness, high fatigue-crack growth threshold and elastic modulus, plasticity at high temperature, excellent electrical and thermal conductivity, and easy machinability[1-6]. The unique combination of these properties makes Ti3SiC2 as a promising structural material for high temperature applications. In addition, the high modulus, metallic conductivity and self-lubricated properties also make Ti3SiC2 as a potential reinforcement for soft metals such as Al, Cu, Ti and Ni. The advantage of Ti3SiC2 reinforced soft metals over other ceramics is that high strength will be achieved without loss of conductivity. During preparing Ti3SiC2 reinforced metal matrix composites, reactions between the matrix and Ti3SiC2 is one of the key factors controlling the microstructure and properties of the composites. The stability of Ti3SiC2 in metal matrix is also important in selecting the preparation method and processing parameters. The knowledge of compounds formed and the diffusion path between the ceramic and metal interface is necessary to control the interface structures, which essentially control the mechanical properties.
In practice, when used as a high-temperature structural material, Ti3SiC2 often needs to be joined with structural metals to make a complex component. While as a reinforcement of soft metals, Ti3SiC2 particulates are incorporated uniformly into metal matrix to make metal matrix composites with controlled microstructure. In both of these applications, understanding the interfacial chemical reactions and interfacial microstructure/ chemistry is of vital importance in selecting processing parameters and optimizing properties.
However, very limited information of the interaction between Ti3SiC2 and metals is available. Our previous works reported the reaction between Ti3SiC2 and Al[7], Ti3SiC2 and Cu[8], respectively. Recently, we are intrigued in investigating the interfacial reactions in Ti-Ti3SiC2 systems. The interests stem from the need in developing high temperature self-lubricated materials such as Ti/Ti3SiC2 composite, as well as joining Ti3SiC2 with Ti-based alloys for high-temperature applications. This paper reports the reaction between Ti3SiC2 and Ti powder, which is useful in both selecting processing conditions of Ti/Ti3SiC2 composite and controlling the interfacial microstructure of Ti/Ti3SiC2 joint.
2 Experimental
To investigate the possible reactions during processing of Ti/Ti3SiC2 composites, Ti and Ti3SiC2 powders were mixed and hot-pressed at 1 273-1 573 K. The average particle size of Ti powders and Ti3SiC2 powders was 40 mm (from General Research Institute For Nonferrous Metals, China) and 10 mm (prepared at author's lab), respectively. The Ti3SiC2 powder used in this work was fabricated by using the in-situ hot pressing solid-liquid reaction process, which was described in Ref.[9]. The as-prepared powders contained a small amount of TiC, which was described in the previous work[9].
According to the phase diagram of Ti-Si system [10], at low Si content (less than 1% Si, mole fraction) the solid solution of Ti(Si) forms; beyond the solubility there are five kinds of Ti-Si compounds, which are Ti3Si, Ti5Si3, Ti5Si4, TiSi and TiSi2. If Ti reacts with Si in Ti3SiC2 when Ti3SiC2 and Ti mix together at high temperatures, Ti3SiC2 will not be stable, and the reaction products depend on the content of Si, i.e. the content of Ti3SiC2. Supposing that Ti reacts with Si from the decomposed Ti3SiC2 and the Ti3Si, Ti5Si3, Ti5Si4, TiSi and TiSi2 can be formed, respectively, their mass ratio of Ti to Ti3SiC2 can be calculated as 3/4, 1.67/4, 1.25/4, 1/4 and 0.5/4, respectively. To confirm this hypothesis and
understand the high temperature reactions between Ti and Ti3SiC2, Ti/Ti3SiC2 composites with above mass ratios were prepared in the temperature range of 1 273-��? 1 573 K under a pressure of 20 MPa.
The phase compositions of the composites prepared under different conditions were identified by X-ray diffraction(XRD), wherein the data were collected by a step-scanning diffractometer (Rigaku D/max-2500PC, Japan) using ?/span>Cu K�� radiation. To understand the mechanism of the reaction and the interfacial microstructure, bulk couple of Ti/Ti3SiC2 was hot pressed at 1 473 K with a pressure of 20 MPa for 60 min. The cross-section of the hot pressed Ti/Ti3SiC2 couple was observed in an S-360 scanning electron microscope (Cambridge Instrument Ltd., UK) equipped with an energy dispersive spectroscope(EDS) system. X-ray dot maps and concentration profiles were determined to identify the interfacial chemistry.
3 Results
To study the possible reactions between Ti3SiC2 and Ti, we should know the thermal stability of Ti3SiC2. According to previous works, the dissociation temperature of Ti3SiC2 was reported from 1 573 K to ��2 073 K depending on the purity, atmosphere and sample form (bulk or powders)[11-14]. To determine the dissociation temperature of Ti3SiC2 powders prepared by the fluctuation method, the as-prepared Ti3SiC2 powders were annealed at different temperatures.
Fig.1 shows the X-ray diffraction patterns of the as-prepared Ti3SiC2 powders and those annealed at 1 573, ?/span>1 673 and 1 773 K for 30 min, respectively. In the ��?/span>as-prepared Ti3SiC2 powders there is a small amount of TiC and SiC, which is the main impurity phase in Ti3SiC2[9]. After annealing at 1 573 K, the intensity of reflections from TiCx (Here C replaced by Cx is because that the atomic ratio of Ti and C is 3 to 2 in Ti3SiC2, the subscript x in the new-formed TiCx is less than ������?/span>1) increase and a new phase TiSi2 emerges. The increase in the intensity of TiCx and the appearance of TiSi2 reveal that Ti3SiC2 powder is decomposed obviously over the temperature of 1 573 K. At 1 673 K, the intensities of diffraction peaks of TiCx are almost as high as that of Ti3SiC2, indicating that more Ti3SiC2 powders are decomposed. At 1 773 K, the reflections from Ti3SiC2 almost disappear but SiC and graphite emerge as decomposition products. The above results are similar to result of RACAULT et al[13] and different from reports of BARSOUM et al[11] and RADHAKRISHNAN et al[12] on the thermal stable of Ti3SiC2. The observed low decomposing temperature is attributed to the fact that the Ti3SiC2 powders studied in our present work have large surface area and higher activity than bulk material used in previous works[11,12]. Owing to the decomposition of Ti3SiC2 powder above 1 573 K, the temperature range for the reaction in Ti-Ti3SiC2 system is chosen below ����?/span>1 573 K.
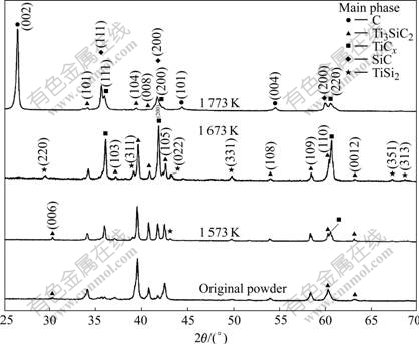
Fig.1 XRD patterns of original Ti3SiC2 powders and those annealed at 1 573, 1 673 and 1 773 K for 30 min, respectively
Fig.2 shows the X-ray diffraction patterns for samples of Ti/Ti3SiC2 composites with a mass ratio of 3/4, which were hot pressed at 1 173, 1 273, 1 373, 1 473 and 1 573 K under a pressure of 20 MPa for 30 min, respectively. For the sample hot pressed at 1 173 K, there are very weak diffraction peaks from TiCx and the main phases are Ti and Ti3SiC2, which implies that the reaction between Ti and Ti3SiC2 almost can be neglected at the temperature of 1 173 K. For the sample hot pressed at ��1 273 K, the intensities of reflections from TiCx are higher than those at 1 173 K. In addition, very weak diffraction peaks from element carbon and titanium- silicon compound Ti5Si3 can be identified, which indicates that the starting reaction should be in the range of 1 173-1 273 K. For the sample prepared at 1 373 K, the diffraction peaks of TiCx become even higher and the reflections from Ti and C disappear, indicating that the amount of TiCx increases with the reaction temperature increasing. The reflections from Ti3SiC2 are weaker than those from TiC at 1 473 K and almost disappear at 1 573 K. All the above results demonstrate that Ti reacts with Ti3SiC2 at temperatures above 1 273 K and the reaction extent is temperature dependent.
To further investigate the effect of Ti3SiC2 content on the reaction products, Ti/Ti3SiC2 powders with a mass ratio of 1.67/4 were hot pressed under a pressure of 20 MPa at 1 273, 1 373, 1 473 and 1 573 K for 30 min, respectively, and the X-ray diffraction patterns of these composites are shown in Fig.3. The difference between Figs.3 and 2 is that new reflection from TiSi2 is detected in Fig.3 besides TiCx and Ti5Si3. Meanwhile, the reflec-
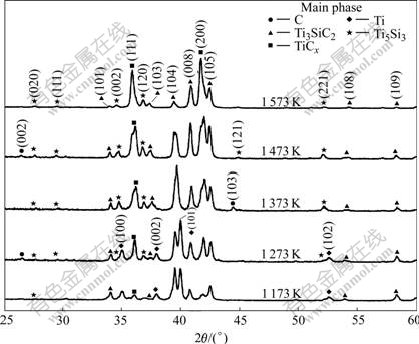
Fig.2 XRD patterns for samples of Ti/Ti3SiC2 composites with mass ratio of 3/4 hot pressed at 1 173, 1 273, 1 373, 1 473 and 1 573 K for 30 min, respectively tions from graphite emerge again, which is similar to the situation in Fig.2. The emerging of the graphite (002) peak implies that Ti3SiC2 is decomposed at 1 273 K and the element carbon is one of the products of the decomposition.
Fig.4 shows that the X-ray diffraction patterns for samples of Ti/Ti3SiC2 composites with a mass ratio of 0.5/4, which were hot pressed under a pressure of 20 MPa at 1 273, 1 373, 1 473 and 1 573 K for 30 min, respectively. No new phase can be detected besides TiSi2 and Ti5Si3. It can be found that the diffraction peak (200) from TiCx increases with the increasing temperature. In another hand, the diffraction peaks of graphite at 1 273 K and 1 373 K are higher than the same peak detected in
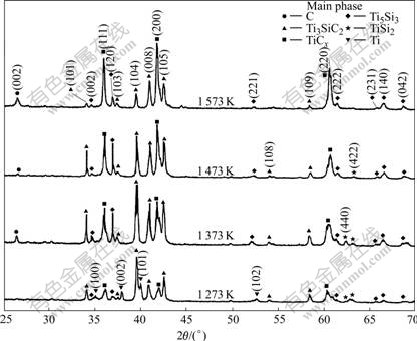
Fig.3 XRD patterns for samples of Ti/Ti3SiC2 composites with mass ratio of 1.67/4 hot pressed at 1 273, 1 373, 1 473 and 1 573 K for 30 min, respectively
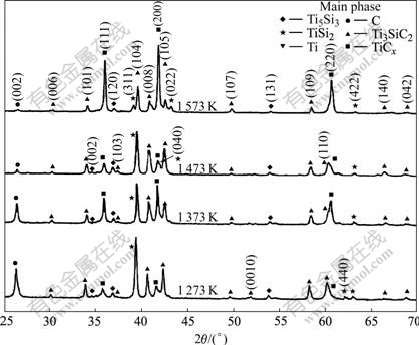
Fig.4 XRD patterns for samples of Ti/Ti3SiC2 composites with mass ratio of 0.5/4 hot pressed at 1 273, 1 373, 1 473 and 1 573 K for 30 min, respectively
Fig.3. Combined with Fig.3, it is clear that the intensity of graphite's peak has a close relationship not only with the content of Ti3SiC2 but also with the reaction temperature. Briefly, when the content of Ti is relatively low the diffraction peak of graphite is relatively high, meanwhile, for a certain mass ratio of Ti to Ti3SiC2, the diffraction peak of graphite is high at a low temperature.
As mentioned above, there are five compounds in Ti-Si system. However, the XRD results shown in Figs.2-4 indicate that only Ti5Si3 and TiSi2 can be detected during the reaction process. The important question is whether other three compounds would be detected if the mass ratio is changed. In fact, there are marked disagreements concerning Ti3Si and Ti5Si4. HANSEN et al[15] has not shown either of these compounds in their paper about the Ti-Si phase diagram. According to Ref.[10], this judgement is based on: 1) the probable purity of the alloys; 2) the fact that the phases of composition 25% and 45% (mole fraction) were observed by different authors, and 3) the present thermodynamic analysis of the system[10]. Based on these facts, it is acceptable that no Ti3Si is detected in the product of reaction. On the other hand, Ti5Si4 and TiSi will be formed above temperature of 1 843 and 1 753 K [10], respectively. It is impossible for these two compounds to be formed under 1 573 K. To conform this deduction, a sample of Ti/Ti3SiC2 composites with a mass ratio of 1/4 (with the mass ratio the compound of TiSi should be formed) is hot pressed at 1 573 K for 30 min and the XRD result is shown in Fig.5. It is clear that all the diffraction peaks of Ti-Si compound are from Ti5Si3 and TiSi2 and no TiSi can be detected. This result implies that only Ti5Si3 and TiSi2 can be formed under the condition of our investigation.
Based on the experimental results described above, the reaction between Ti3SiC2 and Ti can be described as the following equation:
Ti3SiC2+Ti��TiCx+Ti5Si3+TiSi2 (1)
The equation represents a typical reaction and maybe only one kind of Ti-Si compound can be detected for certain content as shown in Fig.2. The subscript x in TiCx represents the atomic ratio of titanium and carbon.
4 Discussion
In the above section, we have known that during the high temperature processing of Ti-Ti3SiC2 composite, Ti reacts with Ti3SiC2 and the reaction depends on both Ti3SiC2 content and the temperature. To reveal the reaction process a bulk Ti3SiC2 was polished and buried in powder of Ti, then hot pressed at 1 573 K with a pressure of 20 MPa for 60 min. The back-scattered SEM image and the chemical composition profile across the interface of the bonded Ti/Ti3SiC2 are shown in Fig.6. The composition profiles(Fig.6(b)) detected along the
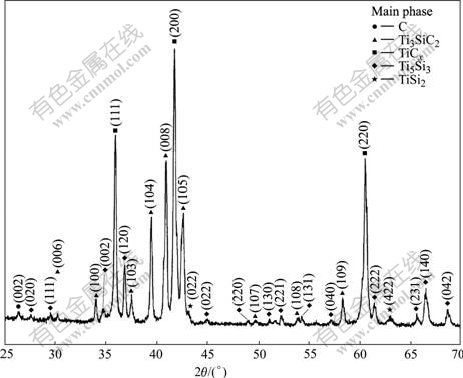
Fig.5 XRD patterns for samples of Ti/Ti3SiC2 composites with mass ratio of 1/4 hot pressed at 1 573 K for 30 min
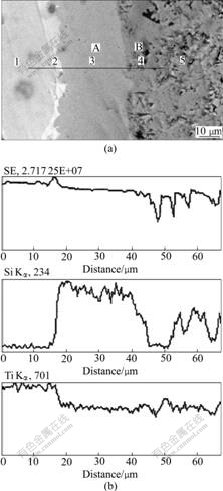
Fig.6 Back-scattered SEM image and chemical composition profile across interface of bulk Ti3SiC2 buried in powder of pure Ti hot pressed at 1 573 K with pressure of 20 MPa for 60 min
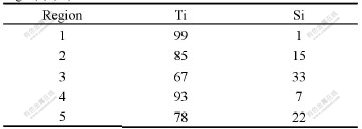
line as shown in Fig.6(a), indicate the diffusion of Si occurred on the boundary at high temperature. However, the diffusion of other elements can be neglected. On the other hand, Ti reacts with the Si in Ti3SiC2 to form Ti-Si compound (dim part of interface marked 3 in Fig.6(a)), which results in the formation of TiCx (dark part marked 4 in Fig.6(a)) near the interface. The EDS semi- quantitative analysis of Ti and Si at the five different points in Fig.6(a) is listed in Table 1, which implies that at layer A in Fig.6(a) Ti5Si3 forms and at layer B in Fig.6(a) TiCx forms. Fig.6(b) also reveals that the composition of Si declines with a steep gradient on the boundary of Ti and the layer A, which implies that the diffusion of Si from Ti3SiC2 to Ti is limited by the forming of Ti-Si compound.
Previous study on the structure characteristics of Ti3SiC2 revealed that the structure of Ti3SiC2 can be described as a layer of Si intercalated into the {111} twin
Table 1 Mole fraction of elements in different regions in Fig.6(a) (%)
boundary of TiC, and the chemical bonding between Si and Ti is relatively weak compared to the strong Ti�C bonding[16-18]. And the decomposition of this layered ternary compound is usually caused by the de-intercalation of Si from it[16-18]. In other words, if Si forms solid solution or a stable compound with other elements, Ti3SiC2 will decompose. In this work, the Si-Ti compound such as Ti5Si3 and TiSi2 forms during the reaction process, which gives a direct evidence for the de-intercalation of Si from Ti3SiC2. In addition, the presence of elemental carbon in the Ti-Ti3SiC2 reaction product implies that the decomposition mode of Ti3SiC2 in Ti may not solely follow the one described above. The experimental results shown in Figs.2-4 reveal that Ti3SiC2 can be decomposed with other ways besides the de-intercalation of Si from Ti3SiC2 and the formation of TiCx.
Analyzing the graphite's peak shown in Figs.3 and 4 we would like to hypothesize that the graphite from the decomposed Ti3SiC2 comes into existence at 1 273 K and then reacts with titanium to form TiCx at higher temperature. If the titanium is insufficient or the temperature is low, the reaction will not be sufficient and the graphite will be left. To confirm this hypothesis the reaction time is extended to 90 min for sample Ti/Ti3SiC2 composites with a ratio of 0.5/4 at temperatures of 1 273 and 1 373 K, respectively. The XRD results of the samples prepared in 90 and 30 min are compared and shown in Fig.7. It is clear that the graphite's diffraction peaks disappear for the sample with a longer reaction time. This result implies that the element carbon is an interim phase during the reaction process; the carbon from the decomposed Ti3SiC2 will react with Ti furthermore if the reaction time is long enough. Combining with the above result, Ti3SiC2 may be decomposed in two different modes. The first mode is caused by the de-intercalation of Si from it and the TiCx is formed by the remained titanium and carbon; the second mode is that the carbon is separated from the Ti3SiC2 and reacts with titanium furthermore. Thus, the reaction route for the high temperature reaction between Ti3SiC2 and Ti can be described by the following procedures.
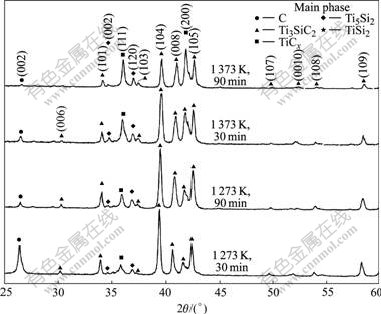
Fig.7 XRD patterns for samples of Ti/Ti3SiC2 composites with mass ratio of 0.5/4 hot pressed at 1 273 and 1 373 K for 30 and 90 min, respectively
Ti3SiC2+Ti��TiCx+Si+Ti��TiCx+TixSiy (2)
Ti3SiC2+Ti��TixSiy+C+Ti��TixSiy+TiCx (3)
The molecular formula TixSiy represents all the two sorts of titanium silicon compound detected in the reaction process. The subscript x in TiCx represents the atomic ratio of titanium and carbon.
In Fig.2, as calculated above, the mass ratio of Ti and Ti3SiC2 corresponds to the compound Ti3Si. According to this mass ratio, the element Ti should be left after the reaction is finished, because the detected compound Ti5Si3 has a relatively low Ti content compared to Ti3Si. However, the experiment result indicates that no element Ti remains. This result implies that the value of subscript x in TiCx may be less than 0.67, because more Ti will be consumed for a small value of x. On the other hand, the XRD result in Fig.4 shows that besides TiSi2, the compound should appear for this content, Ti5Si3 is detected as well. This result implies that the value of subscript x in TiCx may be larger than 0.67. All these experiment results suggest that the subscript x in TiCx will change in a large range. From Ti-C phase diagram[19] we know that the range of x will be changed from 0.5 to 1.
Considering the crystal structure of TiCx, the change of x will bring the change of lattice spacing and the remove of the diffraction peak. As described above, the results in Figs.2-4 show that the diffraction peaks of TiCx is low at 1 273 K and becomes higher and wider at 1 373 K and 1 473 K. This result implies the composite of TiCx is uneven in the different region. The peak becomes very sharp at 1 573 K, which implies a higher temperature is in favor of the harmoniousness in composition.
Our work demonstrates that Ti reacts with Ti3SiC2 at high temperatures and the reaction depends on the temperature and Ti3SiC2 content. The result of this work is instructive for selecting the preparation method and for processing parameters of Ti-Ti3SiC2 composite. Due to the reaction between Ti matrix and the Ti3SiC2 reinforcement at temperatures above 1 273 K, it is desirable to make Ti3SiC2 reinforced Ti below 1 273 K.
5 Conclusions
The chemical reactions between Ti and Ti3SiC2 have been investigated during the preparation of Ti-Ti3SiC2 composites in the temperature range of 1 273-1 573 K. The results demonstrate that Ti3SiC2 reacts with Ti to form TiCx, Ti5Si3 and TiSi2. It is shown that the hot pressing temperature is the key factor that determines the extent of reactions. The relative proportion of Ti and Ti3SiC2 has a relationship with the product of reaction. The Si diffused from Ti3SiC2 to Ti forms stable compounds like Ti5Si3 is believed to be the main mechanisms for the reaction. Because the reactions between Ti and Ti3SiC2 occur at 1 273 K, which is lower than the melting point of Ti (1 943 K), it is difficult to synthesize composite with titanium and Ti3SiC2.
References
[1] MORTENSEN A, JIN I. Solidification processing of metal matrix composites [J]. Int Mater Rev, 1992, 37(1): 101-128.
[2] MYHRA S, SUMMERS J W B, KISI E H. Ti3SiC2�A layered ceramic exhibiting ultra-low friction [J]. Mater Lett, 1999, 39(1): 6-11.
[3] LI J F, PAN W, SATO F, WATANABE R. Mechanical properties of polycrystalline Ti3SiC2 at ambient and elevated temperatures [J]. Acta Mater, 2001, 49(6): 937-945.
[4] FENG A, ORLING T, MUNIR Z A. Field-activated pressure-assisted combustion synthesis of polycrystalline Ti3SiC2 [J]. J Mater Res, 1999, 14(3): 925-939.
[5] BARSOUM M W, YOO H I, POLUSHINA I K, RUD?YU V, EL-RAGHY T. Electrical conductivity, thermopower, and Hall effect of Ti3AlC2, Ti4AlN3, and Ti3SiC2 [J]. Physical Review B, 2000, 62(15): 10194-10198.
[6] GAO N F, MIYAMOTO Y. Joining of Ti3SiC2 with Ti-6Al-4V alloy [J]. J Mater Res, 2002, 17(1): 52-59.
[7] GU W L, YAN C K, ZHOU Y C. Reactions between Al and Ti3SiC2 in temperature range of 600-650 �� [J]. Script Mater, 2003, 49(11): 1075-1780.
[8] ZHOU Y C, GU W L. Chemical reaction and stability of Ti3SiC2 in Cu during high-temperature processing of Cu/ Ti3SiC2 composite [J]. Z Metallkd, 2004, 95(1): 50-56.
[9] SUN Z M, ZHOU Y C. In situ hot pressing/solidliquid reaction synthesis and characterization of fiber-like Ti3SiC2 powders [J]. Mater Res Innov, 1999, 2(4): 227-231.
[10] MURRAY J L. Ti-Si Phase Diagram [A]. SCOTT W W Jr. Binary Alloy Phase Diagram [M]. 2nd ed. USA, 1990: 3367.
[11] BARSOUM M W, EL-RAGHY T, RAWN C J, PORTER W D, WANG H, PAYZANT E A, HUBBARD C R. Thermal properties of Ti3SiC2 [J]. Journal of Physics and Chemistry of Solids, 1999, 60(4): 429-439.
[12] RADHAKRISHNAN R, WILLIAMS J J, AKINC M. Synthesis and high-temperature stability of Ti3SiC2 [J]. J of Alloys Comp, 1999, 285(1): 85-88.
[13] RACAULT C, LANGLAIS F, NASLAIN R. Solid-state synthesis and characterization of the ternary phase Ti3SiC2 [J]. J Mater Sci, 1994, 29(10): 3384-3392.
[14] BARSOUM M W, EL-RAGHY T. Synthesis and characterization of a remarkable ceramic: Ti3SiC2 [J]. J Am Ceram Soc, 1996, 79(6): 1953-1956.
[15] HANSEN M, KESSLER H D, MCPHERSON D J. Ti-Si phase diagram [J]. Tran ASM, 1952, 44: 518.
[16] ZHOU Y C, SUN Z M, WANG X H, CHEN S Q. Ab initio geometry optimization and group state properties of layered ternary carbides Ti3MC2 (M=Al, Si and Ge) [J]. J Phys: Condens Matter, 2001, 13(10): 10001-10010.
[17] SUN Z M, ZHOU Y C.Crystallographic relations between Ti3SiC2 and TiC [J]. Mater Res Innovat, 2000, 3(5): 286-291.
[18] ZHOU Y C, SUN Z M. Electronic structure and bonding properties in layered ternary carbide Ti3SiC2 [J]. J Phys: Condensed Matter, 2000, 12(4): L457-462.
[19] MURRAY J L. Ti-C Phase Diagram [A]. SCOTT W W Jr. Binary Alloy Phase Diagram [M]. 2nd ed. USA, 1990: 888.
(Edited by YUAN Sai-qian)
Foundation item: Project(59925208) supported by the National Outstanding Young Scientist Foundation; Project(59772021) supported by the National Natural Science Foundation of China
Corresponding author: GU Wan-li; Tel: +86-533-2786499; E-mail: wlgu001@sina.com.cn