Estimation of thermal decomposition temperatures of organic peroxides by means of novel local and global descriptors
��Դ�ڿ������ϴ�ѧѧ��(Ӣ�İ�)2018���7��
�������ߣ������� ţ���� ���� ������ LIU Hui(����)
����ҳ�룺1535 - 1544
Key words��organic peroxide; thermal decomposition temperature; multiple linear regression; model validation; quantitative structure-property relationship
Abstract: The thermal decomposition temperature is one of the most important parameters to evaluate fire hazard of organic peroxide. A quantitative structure-property relationship model was proposed for estimating the thermal decomposition temperatures of organic peroxides. The entire set of 38 organic peroxides was at random divided into a training set for model development and a prediction set for external model validation. The novel local molecular descriptors of AT1, AT2, AT3, AT4, AT5, AT6 and global molecular descriptor of ATC have been proposed in order to character organic peroxides�� molecular structures. An accurate quantitative structure-property relationship (QSPR) equation is developed for the thermal decomposition temperatures of organic peroxides. The statistical results showed that the QSPR model was obtained using the multiple linear regression (MLR) method with correlation coefficient (R), standard deviation (S), leave-one-out validation correlation coefficient (RCV) values of 0.9795, 6.5676 ��C and 0.9328, respectively. The average absolute relative deviation (AARD) is only 3.86% for the experimental values. Model test by internal leave-one-out cross validation and external validation and molecular descriptor interpretation were discussed. Comparison with literature results demonstrated that novel local and global descriptors were useful molecular descriptors for predicting the thermal decomposition temperatures of organic peroxides.
Cite this article as: DAI Yi-min, NIU Lan-li, ZOU Jia-qi, LIU Dan-yang, LIU Hui. Estimation of thermal decomposition temperatures of organic peroxides by means of novel local and global descriptors [J]. Journal of Central South University, 2018, 25(7): 1535�C1544. DOI: https://doi.org/10.1007/s11771-018-3846-0.

J. Cent. South Univ. (2018) 25: 1535-1544
DOI: https://doi.org/10.1007/s11771-018-3846-0
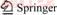
DAI Yi-min(������), NIU Lan-li(ţ����), ZOU Jia-qi(����), LIU Dan-yang(������), LIU Hui(����)
School of Chemistry and Biological Engineering, Hunan Provincial Key Laboratory of Materials Protection for Electric Power and Transportation, Changsha University of Science and Technology, Changsha 410114, China
 Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2018
Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2018
Abstract: The thermal decomposition temperature is one of the most important parameters to evaluate fire hazard of organic peroxide. A quantitative structure-property relationship model was proposed for estimating the thermal decomposition temperatures of organic peroxides. The entire set of 38 organic peroxides was at random divided into a training set for model development and a prediction set for external model validation. The novel local molecular descriptors of AT1, AT2, AT3, AT4, AT5, AT6 and global molecular descriptor of ATC have been proposed in order to character organic peroxides�� molecular structures. An accurate quantitative structure-property relationship (QSPR) equation is developed for the thermal decomposition temperatures of organic peroxides. The statistical results showed that the QSPR model was obtained using the multiple linear regression (MLR) method with correlation coefficient (R), standard deviation (S), leave-one-out validation correlation coefficient (RCV) values of 0.9795, 6.5676 ��C and 0.9328, respectively. The average absolute relative deviation (AARD) is only 3.86% for the experimental values. Model test by internal leave-one-out cross validation and external validation and molecular descriptor interpretation were discussed. Comparison with literature results demonstrated that novel local and global descriptors were useful molecular descriptors for predicting the thermal decomposition temperatures of organic peroxides.
Key words: organic peroxide; thermal decomposition temperature; multiple linear regression; model validation; quantitative structure-property relationship
Cite this article as: DAI Yi-min, NIU Lan-li, ZOU Jia-qi, LIU Dan-yang, LIU Hui. Estimation of thermal decomposition temperatures of organic peroxides by means of novel local and global descriptors [J]. Journal of Central South University, 2018, 25(7): 1535�C1544. DOI: https://doi.org/10.1007/s11771-018-3846-0.
1 Introduction
Organic peroxide (R1�CO�CO�CR2) is a derivative which one or two atoms of hydrogen in hydrogen peroxide were substituted by organic groups. Due to the free radicals provided by the peroxy group (�CO�CO�C), organic peroxides have potential applications in rubber, polymerization initiators and unsaturated polyester materials as catalysts and cross-linking agents in industry [1�C5]. Organic peroxide usually is an unstable chemical because it has a relatively low �CO�CO�C bond energy (20�C50 kcal/mol) [6]. Explosions and runaway reaction incidents of organic peroxides have happened in the past few decades. For example, more than three explosion and runaway reaction incidents of organic peroxide in a batch reaction happened in the past ten years [7]. 167 serious explosion and runaway reaction incidents happened in the United States in the past twenty years, of which 11 incidents were caused by organic peroxides [8]. In many countries, the thermal explosions or runaway reaction incidents of organic peroxides have become a serious safety topic [9�C12]. In order to decrease safety accidents, it is very important to take all available means to avoid unnecessary losses. The thermal decomposition temperature of organic peroxides plays an important role for fire and explosion prevention, design and optimization of the processes and process control [13]. As an important chemical engineering property, the thermal decomposition temperature of organic peroxide is usually inconsistent because experimental data depends on the geometry of the apparatus, the test pressure temperature and several other factors [14]. Usually the measurement of experimental data of the thermal decomposition temperature is expensive and time-consuming [15�C18]. In addition, it is difficult or even impossible for unstable organic peroxides from safety reason but also from technical reason [19]. With the increased need for reliable data for optimization of industrial process, it is an urgent demand to develop a simple, accurate, rapid and inexpensive methodology for estimating the thermal decomposition temperatures of organic peroxide.
In the recent years, a variety of methods have been used to estimate thermal decomposition temperatures of different organic compounds in the literatures [20�C25]. Of all the methods, QSPR is attractive due to its only utilizing molecular descriptors and the advantage of no needing data measurement for predictive property [26]. This method has been successfully applied in the area of drug design and predicted physical and chemical properties of desired substances [27�C32]. The molecular structure determines physicochemical property, therefore it can be used to make quick and efficient prediction of thermal decomposition temperatures of new compounds. ALSBERG et al developed a QSPR model for prediction of the thermal decomposition temperatures of 995 diverse ILs by using electronic, thermodynamic and geometrical descriptors derived from semi- empirical PM6 calculations [32]. PRANA et al [20] developed a QSPR model to predict accurately the decomposition heat and the onset temperature of 38 organic peroxides based on quantum chemical descriptors. LU et al [21] developed a simple method for prediction the detected onset temperature and heat of decomposition of organic peroxide. The predicted results of organic peroxides are in good agreement with the measured values. The method can be used to evaluate thermal stability and severity of runaway reactions. While previous researches have been focused on the thermal stability and reactivity hazards of organic compounds using any computer programs for computations.
In this study, a QSPR model for prediction the thermal decomposition temperatures of organic peroxides had been developed by using novel local and global molecular descriptors. To the best of our knowledge, this is the first time that the graph theory method had been associated with the thermal decomposition temperatures of organic peroxides.
Table 1 Comparison between calculated and experimental thermal decomposition temperatures of organic peroxides
2 Methodology
2.1 Dataset
Experimental data of thermal decomposition temperatures of 38 organic peroxides were selected as samples, 31 were taken from Arkema and 7 from Akzo Nobel [20, 33]. The concentrations were close to 97%�C99% exception given for some of them whose containing inert solvents. The prediction set of 7 organic peroxides was selected randomly from the original 38 samples. The training set of the remaining 31 compounds was used to develop a QSPR model. A complete list of organic peroxides and their corresponding experimental thermal decomposition temperatures are given in Table 1, the test samples were marked with asterisk.
2.2 Molecular descriptor generation
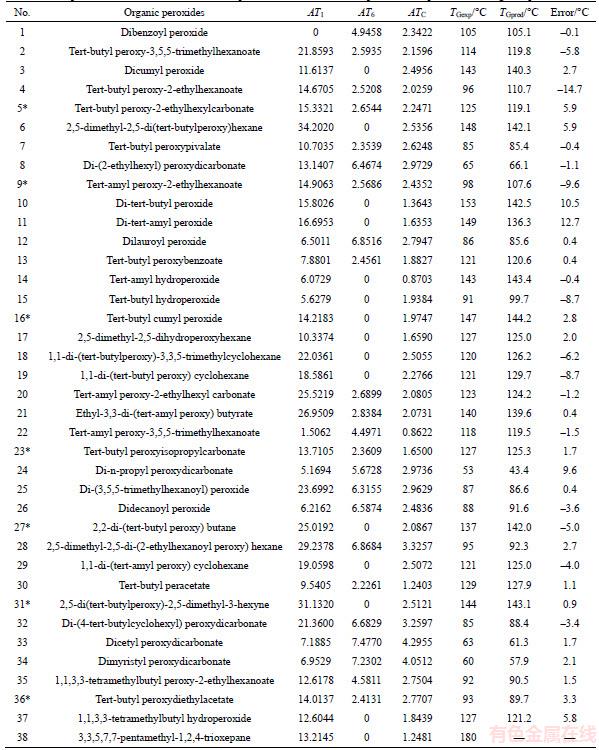
The postulation that all properties of organic molecules depend on their chemical structures and vary with it in a systematic way is a universal principle in chemistry [26]. In terms of the structure of the molecule, there are many influence factors on their physicochemical properties. To obtain more reliable results, the contributions of the component atoms or groups and their interactions must be taken into account. In this paper, we considered molecular local aspect and global aspect structures influence jointly on their physicochemical properties of compound AB (Figure 1). For monoderivatives of compounds AB, the local aspect is the contribution of groups A and B and the global aspect is their interaction between groups A and B. Accordingly, their property can be divided into three parts, and expressed by Eq. (1).
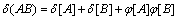 (1)
(1)
where the ��[A] and ��[B] are the local contributions of groups A and B to according property, respectively. ��[A]��[B] represents the global contribution of the interaction of groups A and B. For an organic peroxide molecule with n atoms, each pair of adjacent vertices is linked by a single bond. Based on atoms connected and chemical microenvironment, organic peroxide (R1�CO�CO�CR2) molecules include six types groups: primary (CH3�C), secondary (CH2<), ternary (�CCH<), quaternary (>C<) carbons, group (�CO�C) and (O=), respectively. Based on a previously described method, for monoderivatives of organic peroxides, their physicochemical properties are jointly the contributions of six types groups and their interaction between groups.

Figure 1 Molecular plot of A�CB
In our previous research, atom electro- negativity topological descriptors encode the molecular connectivity, charge information and individual contributions from groups in the molecules [34]. In order to elucidate the properties and interaction of vertices in a molecule, the distance matrix, D=[dij]n��n, vertex matrix, R=[rij]n��n and electro-negativity matrix X=[��ij]n��n were derived in this paper, respectively. The distance matrix D of n atoms in a molecule, a symmetric matrix, can be expressed as: D=[dij]n��n= [��Lij/LC�CC]n��n, where dij is the length of shortest path in between vertex ��i�� and vertex ��j�� in molecular graph, and is calculated by summing the bond length between two adjacent vertices in the shortest path. For example, the C�CC bond length LC�CC= 0.154 nm is taken as 1, then the relative bond length between vertices i and j is calculated: dij=��Lij/LC�CC, in organic peroxide molecule, O�CO relative bond length is 0.149/0.154=0.9675 [35]. In truth, the relative bond length of two adjacent vertices can be used to distinguish saturated, unsaturated bond and heteroatom compounds. Similarly, the distance sum Si is obtained by adding any row or column of distance matrix D, and the resultant matrix is an additive matrix S. As one of the primary physiochemical parameters of atoms, electro- negativity represents the ability of atoms to obtain or lose electrons [36]. Electro-negativity of atom can effectively reflect the chemical information at the atom and group levels. Accordingly, electro- negativity matrix X is defined to reflect every atomic chemical environmental change of a molecule. In addition, vertex matrix R is defined to distinguish the level of branching of the molecule and the atomic species. In a word, the matrix D, S, R and X are expressed as following:
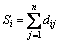 ,
, 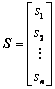 ,
, 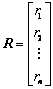 ,
, 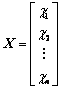
Taking into them account, novel atom electro- negativity topological descriptors is defined as:
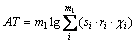 (2)
(2)
where m1 is the number of the same group in the molecule. For organic peroxides, the great majority contributions of the thermal decomposition temperatures arising from six parts, namely, the contributions of CH3�C, CH2<, �CCH<, >C<, �CO�C and O=, are expressed as: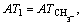
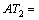
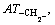
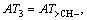
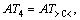
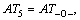
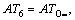 respectively. Another, their interactions between groups can seldom affect the thermal decomposition temperatures of organic peroxides. Accordingly, a new descriptor ATC is defined:
respectively. Another, their interactions between groups can seldom affect the thermal decomposition temperatures of organic peroxides. Accordingly, a new descriptor ATC is defined:
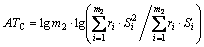 (3)
(3)
where m2 is the number of the all groups of the labeled molecular graph. And part of contributions of property is decided by their interactions between groups, that is to say, ATC plays a secondary role to contributions of the thermal decomposition temperatures of organic peroxides.
As an illustration, Figure 2 depicts the labeled molecular graph of Tert-butyl peroxy-2- ethylhexanoate.
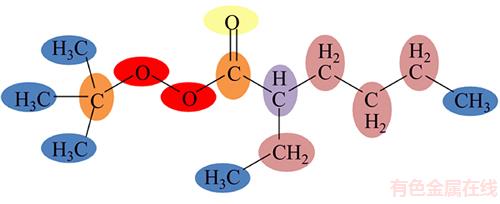
Figure 2 Labeled molecular graph of Tert-butyl peroxy-2-ethylhexanoate
According to Eqs. (2) and (3), AT1, AT2, AT3, AT4, AT5, AT6 and ATC are computed as follows:
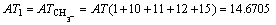
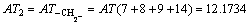
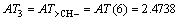
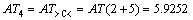
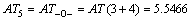
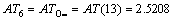
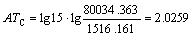
3 Result and discussion
3.1 Regression analysis
The local molecular descriptors such as AT1, AT2, AT3, AT4, AT5, AT6 and global molecular descriptor ATC were computed for organic peroxides and were listed in Table 1 and attached Table. Multiple linear regression analysis based on a combination of local and global molecular descriptors was performed to correlation thermal decomposition temperatures of organic peroxides. Generally, if the residual value is larger than ��3S, the sample can be considered as an outlier. Although the predictive performance of regression equation in this study is satisfactory, 3,3,5,7,7- pentamethyl-1,2,4-trioxepane in sample set is considered as response an outlier because the sample severely affect the prediction effect of the model. If abnormal value of MLR method was screened, the performance of the model was greatly improved. Accordingly, training set only included 30 compounds in regression model and the following regression equation was obtained.
TG=(0.9991��0.2510)AT1+(0.5506��0.0729)AT2+
(0.5591��0.1955)AT3+(2.9663��0.7124)AT4+
(�C0.2715��0.4524)AT5+(�C5.8148��0.6883)AT6+
(�C39.2220��3.3041)ATC+(166.0461��4.8950)(4)
For our general seven-descriptor model, the performance of the training set is R=0.9795,RCV=0.9328, n=30, and for the testing set TGExp=(20.8916��12.2727)+(0.8942��0.1042)TGPre,R=0.9677, n=7. The average absolute error and absolute percent error were 3.99 ��C and 3.96% for all the 30 organic peroxides in training set, respectively. The scatter plot of the calculated thermal decomposition temperature versus experimental data in the model is presented in Figure 3. From Figure 3, it can be found that the model is accurate because most of the points are located in the vicinity of bisection. From Figure 3 and squared correlation coefficient (R2=0.9595) of Eq.(4), one can say that calculated thermal decomposition temperature is in good agreement with the experimental values. In order to investigate the error distribution, we present the plot of the residuals as shown in Figure 4. Seen from Figure 4, horizontal blue lines in this figure indicate the standard deviation limits of ��2S. As the calculated residuals are distributed on both sides of the zero line, and the residuals exceed seldom the standard deviation of ��2S, the largest deviation of the calculated from the experimental value is observed for Tert-butyl peroxy-2-ethylhexanoate with the residual of �C2.24S, one may conclude that there is no systematic error in the development of the MLR model [37]. It is clear that the MLR model here can give satisfactory predictive results.
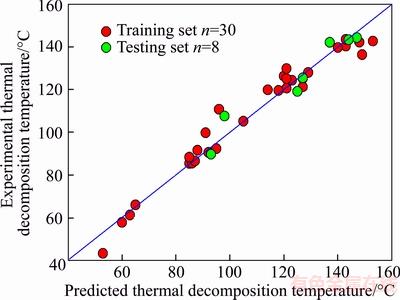
Figure 3 Correlation between predicted and experimental thermal decomposition temperature values for both training and testing sets
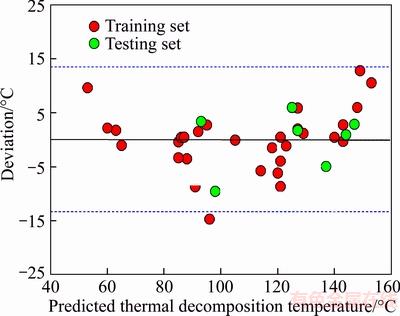
Figure 4 Residual plot of predicted versus experimental values of organic peroxide thermal decomposition temperatures
3.2 Model validation
To verify the accuracy of our proposed model, cross validation and external validation were used. Cross-validation by leave-one-out (LOO) method is the most known and used method for calculating internal validation [38, 39].
For our general seven-descriptor model, the cross validation correlation efficient R2CV=0.8701, as compared to the correlation coefficient R2= 0.9595, indicates the high stability of the linear regression model. In this study, cross validation procedure is repeated 30 times, and the frequency distribution of 30 RLOO is calculated and plotted in Figure 5 as a contour view. It can be seen from Figure 5 that RLOO and their frequency distributions are more or less randomly scattered around R derived from the general model (0.9795). The cross validated correlation coefficient RCV=0.9328, as compared to the model correlation coefficient R=0.9795, indicates the high stability of the regression equation. The cross validation result clearly demonstrates that the final model to be statistically significant. To compare the simulation results with experimental values, testing set and validation set predicted results were plotted in Figure 6. Seen from Figure 6, the results show that there are very good correlations.
In order to further test model predictive ability, the QSPR model was applied to an external set of 7 organic peroxides that are not included in the training set. By comparing the experimental values of 7 organic peroxides with predicted values obtained with Eq.(4). The accurate prediction of thermal decomposition temperatures of organic peroxides shows that this model is reliable. Therefore, we can evaluate the thermal risk of organic peroxides by using predictive thermal decomposition temperature of organic peroxide [40].

Figure 5 Frequency distribution of RLOO based on 30 organic peroxide samples
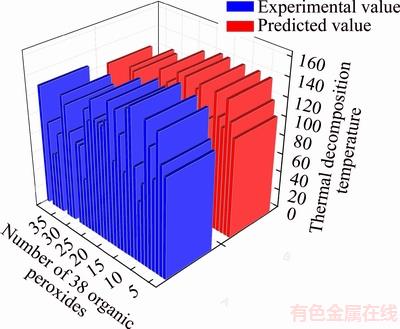
Figure 6 Plot of predicted values versus experimental values of organic peroxides thermal decomposition temperatures
3.3 Molecular descriptor interpretation
The aforementioned seven molecular descriptors have different weights that influence the increase or decrease of organic peroxide the thermal decomposition temperature according to regression equation. The six local type descriptors and one global type descriptor together determine change of the property. These molecular descriptors reflect different characters of the organic peroxide structure from local and global aspect. Each descriptor influence on the thermal decomposition temperature is judged by comparing the coefficients before descriptors [41]. The larger the absolute value of the coefficient is, the stronger the influence of the descriptor is. The significance of the descriptors involved in the MLR model decreases in the following order: ATC>AT6>AT4>AT1>AT3>AT2> AT5. The results also encode that the thermal decomposition temperatures of organic peroxide are predominantly determined by molecular size, shape and number of end peripheral group. Due to the strong influence of inter-molecular forces in crystal sublimation process, the clear presence of molecular size is strongly expected. The weigh of ATC is obviously different from other descriptors in the model. Thus, the ATC has a significant effective on thermal decomposition temperatures of organic peroxides than that of the other descriptors. The thermal decomposition temperatures of organic peroxides are predominantly decided by molecular shape. The degree of branching especially group �CCH< and >C< prevents closed contact with neighboring molecules in space and varied with degree of branching for molecules with the same number of non-hydrogen atoms. The groups of CH3�C and O= are located in the outermost layer of organic peroxide molecules. Then these groups contact area with neighboring molecules is larger than internal groups of CH2< and �CO�C. Thus, the impacts of peripheral groups on organic peroxide thermal decomposition temperature are greater than molecular internal groups. Based on a previously described procedure, it can be observed that the values of AT6 and AT1 reveal that the contributions from peripheral groups keep much greater than group CH2< and �CO�C. The regression coefficient is positive, indicating that an increase in the corresponding descriptor would result in an increase in the organic peroxide thermal decomposition temperatures, whereas negative values indicate negative contribution to thermal decomposition temperatures [42].
From the above discussion, we can conclude that the local and global molecular descriptors involved in the regression model can be interpreted reasonably. The thermal decomposition temperatures of organic peroxide mainly depend on the molecular size, peripheral atoms and molecular shape. Six local descriptors and one global descriptor have explicit physical meanings, respectively. Accordingly, the combination of these descriptors has several advantages which have high discriminating power and unique representation of the compound. Seven descriptors can account for the structural features responsible for thermal decomposition temperatures of organic peroxide.
3.4 Comparison with literature methods
The model in this paper is now compared with the different published models by other groups [20, 21]. Although it is not possible to make a perfect comparison of those previously reported models because different data sets or different algorithms were used for model building and validation [43]. Nevertheless, to our knowledge, only two references exist to predict thermal decomposition temperatures of organic peroxide using the QSPR approach. In spite of the difference of the database, the linear QSPR model firstly was proposed by LU et al [21] for predict the thermal decomposition temperatures of organic peroxide. Only a limited database of 16 organic peroxides was used to develop model with no validation set, neither explicate molecular descriptor significance. Recently, PRANA et al [20] developed a three-parameter model using 38 organic peroxides for the prediction of the thermal decomposition temperatures. The determination coefficient of PRANA was slightly lower than the model of LU in terms of fitting (0.84 vs 0.92) but the robustness was higher (0.77 vs 0.108). In this study, we established a seven-parameter MLR model using 38 organic peroxides for the prediction of the thermal decomposition temperatures. For the training set (30 data points), R2=0.9593, and the RMSE was 6.5676 ��C. The corresponding statistical parameters for the testing set (7 data points) were R2=0.9364, and RMSE=5.9079 ��C. In comparison to previous studies, our seven-parameter and simple model is provided by a dataset approximately double size than LU model.
Table 2 Comparative statistical performances of different developed models

Although the model in this study shows better results, 3, 3, 5, 7, 7-pentamethyl-1, 2, 4-trioxepane severely influences the prediction performances of the model. The results of PRANA also show that this compound is an outlier in MLR model [20]. We think the main reason is that this compound belongs to the special chemical family including three oxygen atoms in hexatomic ring. Accordingly, in this study the abnormal value is screened from the training set. It is noteworthy that the proposed method does not require any computer programs and specialized software for computations. The proposed model could be used to estimate the thermal decomposition temperatures of new organic peroxides by avoiding the costly and time consuming experiments because all the descriptors involved can be directly calculated from the molecular structure.
4 Conclusions
In this work, the QSPR method was employed to predict the thermal decomposition temperatures of organic peroxide. A seven-parameter linear model was developed by MLR, with R of 0.9795 and RMS of 6.5676 ��C for the training set. By performing the model validation, it can be concluded that the presented MLR model is a valid model and can be effectively used to predict the thermal decomposition temperatures of organic peroxides. Moreover, the contributions of molecular descriptors were discussed, in order to elucidate the structural characteristics for the thermal decomposition temperatures of organic peroxides. When comparing the results of the model to those of previously reported methods, it showed that the presented model possesses some obviously superiority.
References
[1] HERBERT K, PETER H G., RAINER S, WILFIRED M. Peroxy compounds, organic in Ullmann��s encyclopedia of industrial chemistry [M]. New York: Wiley-VCH, Weinheim, 2002.
[2] SANCHEZ J, MYERS T. Peroxides and peroxide compounds organic peroxides [M]// Kirk-Othmer Encyclopedia of Chemical Technology, 4th ed, New York: John Wiley & Sons, 1996: 230�C310.
[3] ZHANG W, XIAO J, WANG X, MIAO G, YE F Y, LI Z. Oxidative desulfurization using in-situ-generated peroxides in diesel by light irradiation [J]. Energy Fuels, 2014, 28: 5339�C5344.
[4] SAULE M, MOINE L, DEGUEIL-CASTAING M, MAILLARD B. Chemical modification of polypropylene by decomposition of unsaturated peroxides [J]. Macromolecules, 2005, 38: 77�C85.
[5] TOMMASO S D I, ROTUREAU P, CRESCENZI O, ADAMO C. Oxidation mechanism of diethyl ether: A complex process for a simple molecule [J]. Phys Chem Chem Phys, 2011, 13: 14636�C14645.
[6] BENASSI R, TADDEI F. Homolytic bond-dissociation in peroxides, peroxyacids, peroxyesters and related radicals: ab-initio MO calculations [J]. Tetrahedron, 1994, 50: 4795�C4810.
[7] SHEN S J, WU S H, CHI J H, WANG Y W, SHU C M. Thermal explosion simulation and incompatible reaction of dicumyl peroxide by calorimetric technique [J]. J Therm Anal Calorim, 2010, 102: 569�C577.
[8] CSB. Improving reactive hazard management [R]. Washington, DC, 2002.
[9] HSU J M, SU M S, HUANG C Y, DUH Y S. Calorimetric studies and lessons on fires and explosions of a chemical plant producing CHP and DCPO [J]. J Hazard Mater, 2012, 217�C218: 19�C28.
[10] TSENG J M, LIN C P. Green thermal analysis technology for evaluating the thermal hazard of di-tert-butyl peroxide [J]. Ind Eng Chem Res, 2011, 50: 9487�C9494.
[11] SWIHART M T, GIRSHICK S L. Thermochemistry and kinetics of silicon hydride cluster formation during thermal decomposition of silane [J]. J Phys Chem B, 1998, 103: 64�C76.
[12] DUH Y S, WU X H, KAO C S. Hazard ratings for organic peroxides [J]. Proc Safety Prog, 2008, 27: 89�C99.
[13] LV J Y, CHEN W H, CHEN L P, TIAN Y T, YAN J J. Thermal risk evaluation on decomposition processes for four organic peroxides [J]. Thermochim Acta, 2004, 589: 11�C18.
[14] VIDAL M, ROGERS W J, HOLSTE J C, MANNAN M S. A review of estimation methods for flash points and flammability limits [J]. Proc Safety Prog, 2004, 23: 47�C55.
[15] WANG Q, WANG L, ZHANG X, MI Z. Thermal stability and kinetic of decomposition of nitrated HTPB [J]. J Hazard Mater, 2009, 172: 1659�C1664.
[16] LV J, CHEN L, CHEN W, GAO H, PENG M. Kinetic analysis and self-accelerating decomposition temperature of dicumyl peroxide [J]. Thermochim Acta, 2013, 571: 60�C63.
[17] YEH P Y, SHU C M, DUH Y S. Thermal hazard analysis of methyl ethyl ketone peroxide [J]. Ind Eng Chem Res, 2002, 42: 1�C5.
[18] MALOW M, WEHRSTEDT K D. Prediction of the self-accelerating decomposition temperature for liquid organic peroxides from differential scanning calorimetry (DSC) measurements [J]. J Hazard Mater, 2005, 120: 21�C24.
[19] ALBAHRI T A. Flammability characteristics of pure hydrocarbons [J]. Chem Eng Sci, 2003, 58: 3629�C3641.
[20] PRANA V, ROTUREAU P, FAYET G, ANDRE D, HUB S, VICOT P, RAO L, ADAMO C. Prediction of the thermal decomposition of organic peroxides by validated QSPR models [J]. J Hazard Mater, 2014, 276: 216�C224.
[21] LU Y, NG D, MANNAN M S. Prediction of the reactivity hazards for organic peroxides using QSPR approach [J]. Ind Eng Chem Res, 2011, 50: 1515�C1522.
[22] ASTM Computer Program for Chemical Thermodynamic and Energy Release Evaluation-CHETAH [EB/OL] [2014�C01]. http: //www.astm. org.
[23] MOHAN V K, BECKER K R, HAY J E. Hazard evaluation of organic peroxides [J]. J Hazard Mater, 1982, 5: 197�C220
[24] GHARAGHEIZI F, SATTARI M, ILANI-KASHKOULI P, MOHAMMADI A H, RAMJUGERNATH D, RICHON D. Quantitative structure-property relationship for thermal decomposition temperature of ionic liquids [J]. Chem Eng Sci, 2012, 84: 557�C563.
[25] PAN Y, ZHANG Y Y, JIANG J C, DING L. Prediction of the self-accelerating decomposition temperature of organic peroxides using the quantitative structure-property relationship (QSPR) approach [J]. J Loss Prev Process Ind, 2014, 31: 41�C49.
[26] KATRITZKY A R, KUANAR M, SLAVOV S, HALL C D, KARELSON M, KAHN I, DOBCHEV D A. Quantitative correlation of physical and chemical properties with chemical structure: utility for prediction [J]. Chem Rev, 2010, 110: 5714�C5789.
[27] TROPSHA A, GOLBRAIKH A. Predictive QSAR modeling workflow, model applicability domains, and virtual screening [J]. Curr Pharm Des, 2007, 13: 3494�C3504.
[28] GHARAGHEIZI F, ESLAMIMANESH A, MOHAMMADI A H, RICHON D. QSPR approach for determination of parachor of non-electrolyte organic compounds [J]. Chem Eng Sci, 2011, 66: 2959�C2967.
[29] DAI Y M, LIU H, LI X, ZHU Z P, ZHANG Y F, CAO Z, ZHU L X, ZHOU Y. A novel group contribution-based method for estimation of flash points of ester compounds [J]. Chemom Intell Lab Syst, 2014, 136: 138�C146.
[30] JIN L J, BAI P. Prediction of the normal boiling point of oxygen containing organic compounds using quantitative structure-property relationship strategy [J]. Fluid Phase Equilibria, 2016, 427: 194�C201.
[31] XU J, WANG L, WANG L X, LIANG G J, SHEN X L, XU W L. Prediction of Setschenow constants of organic compounds based on a 3D structure representation [J]. Chemom Intell Lab Syst, 2011, 107: 178�C184.
[32] VENKATRAMAN V, ALSBERG B K. Quantitative structure-property relationship modeling of thermal decomposition temperatures of ionic liquids [J]. J Mol Liq, 2016, 223: 60�C67.
[33] ASTM E537-02. Standard Test Method for the Thermal Stability of Chemicals by Differential Scanning Calorimetry [S].
[34] DAI Y M, ZHU Z P, CAO Z, ZHANG Y F, LI X. Prediction of boiling points of organic compounds by QSPR tools [J]. J Mol Graphics Model, 2013, 44: 113�C119.
[35] DAI Y M, LIU H, NIU L L, CHEN C, CHEN X Q, LIU Y N. Estimation of half-wave potential of anabolic androgenic steroids by means of QSER approach [J]. Journal of Central South University, 2016, 23: 1906�C1914.
[36] ZHOU C, CHU X, NIE C. Predicting thermodynamic properties with a novel semi-empirical topological descriptor and path numbers [J]. J Phy Chem B, 2007, 111: 10174�C10179.
[37] DUCHOWICZ P R, CASTRO E A, FERN NDEZ F M, GONZALEZ M P. A new search algorithm for QSPR/QSAR theories: Normal boiling points of some organic molecules [J]. Chem Phys Lett, 2005, 412: 376�C380.
NDEZ F M, GONZALEZ M P. A new search algorithm for QSPR/QSAR theories: Normal boiling points of some organic molecules [J]. Chem Phys Lett, 2005, 412: 376�C380.
[38] ROY K, CHAKRABORTY P, MITRA I, OJHA P K, KAR S, DAS R N. Some case studies on application of ��r2m�� metrics for judging quality of quantitative structure activity relationship predictions: Emphasis on scaling of response data [J]. J Comput Chem, 2013, 34: 1071�C1082.
[39] DAI Yi-min, LIU You-nian, LI Xun, CAO Zhong, ZHU Zhi-ping, YANG Dao-wu. Estimation of surface tension of organic compounds using QSPR [J]. Journal of Central South University, 2012, 19(1): 93�C100.
[40] JIN L J, BAI P. QSPR study on normal boiling point of acyclic oxygen containing organic compounds by radial basis function artificial neural network [J]. Chemom Intell Lab Syst, 2016, 157: 127�C132.
[41] DAI Yi-min, HUANG Ke-long, LI Xun, CAO Zhong, ZHU Zhi-ping, YANG Dao-wu. Simulation of 13C NMR chemical shifts of carbinol carbon atoms by using quantitative structure-spectrum relationships [J]. Journal of Central South University of Technology, 2011, 18(2): 323�C340.
[42] SHI J J, CHEN L P, CHEN W H, SHI N, YANG H, XU W. Prediction of the thermal conductivity of organic compounds using heuristic and support vector machine methods [J]. Acta Phys Chem Sina, 2012, 28: 2790�C2796.
[43] PAN Y, JIANG J C, WANG R, CAO H Y, CUI Y. A novel QSPR model for prediction of lower flammability limits of organic compounds based on support vector machine [J]. J Hazard Mater, 2009, 168: 962�C969.
(Edited by HE Yun-bin)
���ĵ���
������ӱ�ľֲ���ȫ�ַ��������������л�����������ȷֽ��¶�
ժҪ���ȷֽ��¶��������л������������Σ�ճ̶�����Ҫ�IJ���֮һ���������һ�ֹ����л����������ȷֽ��¶ȵĶ����·�����38���л��������ﱻ�����Ϊѵ�����Ͳ��Լ������Ӿֲ�������AT1, AT2, AT3, AT4, AT5, AT6��ȫ��������ATC�������ӽṹ������������һ��ȷ�Ĺ����л����������ȷֽ��¶ȵĶ�����Ч��ϵģ�ͣ���Ԫ���Թ�ϵģ�͵����ϵ������ƫ�����һ����������ϵ���ֱ�Ϊ0.9795, 6.5676 ��C�� 0.9328��Ԥ������ƽ���������Ϊ3.86%��ģ���ȶ��Բ�����һ��������������֤�����ӽṹ�������л�����������ȷֽ��¶ȵ�Ӱ����к����Ľ��͡������������Ƚϱ������÷��Ӿֲ�������AT1, AT2, AT3, AT4, AT5, AT6��ȫ��������ATC����������Ч��ϵ���������л�����������ȷֽ��¶���һ����Ч�ķ�����
�ؼ��ʣ��л���������ȷֽ��¶ȣ���Ԫ���Իع飻ģ�ͼ��飻������Ч�C���ʹ�ϵ
Foundation item: Project(2015SK20823) supported by Science and Technology Project of Hunan Province, China; Project(15A001) supported by Scientific Research Fund of Hunan Provincial Education Department, China; Project(2017CL06) supported by Hunan Provincial Key Laboratory of Materials Protection for Electric Power and Transportation, China; Project(k1403029-11) supported by Science and Technology Project of Changsha City, China; Project(CX2015B372) supported by the Hunan Provincial Innovation Foundation for Postgraduate, China
Received date: 2017-07-28; Accepted date: 2017-08-20
Corresponding author: DAI Yi-min, PhD, Associate Professor; Tel: +86�C731�C85258733; E-mail: yimindai@sohu.com; ORCID: 0000- 0001-7874-5107

