
Electroless nickel-plating on die cast magnesium alloy AZ91D
JIA Su-qiu(贾素秋)1, JIA Shu-sheng(贾树胜)2, YAO Jun(姚 军)2
1. School of Materials Science and Engineering, Changchun University of Technology, Changchun 130012, China;
2. Key Laboratory of Automobile Materials, Ministry of Education, Jilin University, Changchun 130025, China
Received 15 July 2007; accepted 10 September 2007
Abstract: Electroless nickel-plating on die cast magnesium alloy AZ91D was investigated. Growth of the electroless nickel-plating coating was characterized using scanning electron microscopy. Corrosion resistance of the coating was evaluated by open circuit potential and potentiodynamic polarization curves in 3.5% (mass fraction) NaCl solution. The results show that plating deposition is initiated on the crevices and then spread onto primary α phase. The corrosion potentials for die cast magnesium alloy AZ91D and nickel-plating coating are about -1.45 V and -0.36 V (vs. SCE), respectively. No discoloration, cracks, blisters, or peeling appear by heat-quench test. The results show that the corrosion potential of Ni-P coating is increased by 1 000 mV and corrosion resistance for die cast magnesium alloy AZ91D is improved. The adhesion between the coating and the substrate is excellent. Electroless nickel plating is a promising method to enhance magnesium alloys resistance for attacking.
Key words: electroless nickel-plating; die cast magnesium alloy; corrosion resistance; polarization
1 Introduction
Magnesium alloys have been applied in automobile industry due to their inherent low density, etc. They are promising alloys to replace aluminum alloys in order to reduce mass for saving fuel. However, magnesium is prone to atmospheric corrosion. Further, because of its high chemical affinity for aqueous solutions, magnesium is difficult for electrochemical treatment with its position in electromotive series at E0=-2.37 V. The situation is still more complex for magnesium alloys, where the alloying constituents introduce electrochemical heterogeneity [1-5]. The most widely used casting alloy, AZ91D (Mg-9%Al-1%Zn), has good castability and a sufficiently high corrosion resistance. The die cast structure of this alloy includes a supersaturated solid solution of Al in α-Mg and an intermetallic β phase Mg17Al12[6]. Die cast magnesium alloy AZ91D castings have an advantage over ingot-castings but its better corrosion resistance compared with the other Mg alloys does not satisfy the requirement for a structure application in automobile industry. This is an urgent problem for die cast AZ91D. Hence, a surface engineering technique is necessary for enhancing the corrosion resistance[7].
Electroless nickel-phosphorus coatings (Ni-P) are generally favored over electroplated deposits because of the enhanced corrosion resistance and the coating uniformity achievable with complex objects. The properties of electroless nickel and its ability to incorporate composites have enabled the process to be exploited widely[1]. General pretreatment of electroless nickel-plating on magnesium alloys includes zincating immersion and direct electroless nickel-plating[8]. Zincating immersion has been substituted by direct electroless nickel-plating because it is complex and harmful to human and environment. For example, CHEN et al[9] investigated a zinc transition layer in electroless nickel plating and this new process greatly improves the adhesion and coverage of Ni-P coating. LIU et al[10] developed a novel Ni based coating-plasma electrolytic oxidation (PEO) pre-treatment followed by electroless nickel (EN) plating, which has been developed to produce pore free Ni coatings on AZ91 magnesium alloy. The application of the PEO film between the nickel coating and the substrate acted as an effective barrier and catalytic layer for the subsequent nickel plating. ZHAO et al[11] proposed an organic coating as the interlayer between Ni-P coating and AZ31 magnesium alloysubstrate to replace the traditional chromium oxide plus HF pretreatment. ZHANG et al[12] and LIAN et al[13] researched phosphate coatings as pretreatment of electroless Ni-P coating on magnesium alloy AZ91D.
However, the direct electroless-nickel plating on cast AZ91D is still a challenge and becomes complicated process because AZ91D is highly active and heterogeneous. The available information of electroless nickel-plating on magnesium alloys is very limited. Refs.[1, 7, 14] reported electroless nickel-plating on magnesium alloys. There are many problems to be solved about electroless nickel-plating on magnesium alloys. In this work, electroless nickel-plating on cast AZ91D used nickel acetate as main salts and properties of Ni-P coating was studied.
2 Experimental
The specimens were cut from die cast magnesium AZ91D with dimension 15 mm×10 mm×3 mm. The normal compositions are 9% Al, 1% Zn and the balance Mg. The process operation details are given in Table 1. The microstructures of the deposits and the substrates were examined by scanning electron microscopy (SEM, JEOL 5600). The chemical composition of Ni-P coating was determined by energy dispersive X-ray (EDX) analysis. X-ray diffraction was used to confirm the structure of the deposits. Also the coating growth was analyzed using SEM. Polarization curves of the specimens were measured in an electrolytic cell containing about 500 mL 3.5%(mass fraction) NaCl solution at room temperature using EG & G273 electrochemical measurement system. The polarization
Table 1 Sequence of operations

started from -2.0 V and stopped at -1.2 V for the substrate and from -1.5 V to 0 V for the electroless coating with scanning rate of 1 mV/s using a saturated calomel electrode (SCE) as a reference electrode and a platinum electrode as a counter electrode. The test solution was made with analytical reagents and distilled water. All potentials were referred to the SCE [15]. The adhesion was evaluated by a heat-quench test.
3 Results and discussion
3.1 Microstructure of substrate material
Fig.1 shows the microstructure of the substrate. The samples were cut from the surface and center of the casting. It consists of primary α, eutectic α and β phases. The surface has relatively small grains and contains more β phases(Fig.1(a)) and the grain is coarse in the central area(Fig.1(b)) because the cooling rate of casting surface is larger than that of the center. This microstructural heterogeneity makes the process of electroless deposition complex[7].
3.2 Surface morphology of electroless nickel-coating
The surface morphologies of electroless nickel- plating coating on die cast AZ91D alloy at constant temperature and pH are shown in Fig.2. Most of Ni sphere -diameters are 2-4 μm and the coating is compact without pores. It can be seen that the thickness of the coating is about 17 μm after plating for 1 h (Fig.2(b)) and the phosphorus content in the deposit is 4%(mass fraction) by EDX. In Fig.2(c), the phase composition of the coating is nickel solid solution containing phosphorus by XRD.

Fig.1 Microstructure of die cast AZ91D: (a) Surface; (b) Center

Fig.2 Morphologies (a) Surface; (b) Cross section of Ni-P coating and XRD pattern (c)
The adhesion was evaluated by a heat-quench test. The specimens were heated at 250 ℃ for 1 h and quenched in water for 20 times. No discoloration, cracks, blisters, or peeling of the coating are observed. This result shows the adhesion of the plating coating to the die cast AZ91D alloy substrate is excellent [7, 14].
3.3 Coating nucleation and growth
In the present plating solution the function of nickel acetate was to provide nickel ions, and the sodium hypophosphate acted as a metal-reducing agent. The bath solution also contained citrate and bifluoride, which acted as buffers, complexing agent and accelerators. Thiourea acted as a solution stabilizer, and ammonia solution was used to adjust pH values of the plating solution. To understand the nucleation and growth of electroless nickel on AZ91D substrate, coating growth was monitored at 2 min interval using SEM in the early formation. The pretreatment process was carried out on substrates through alkaline cleaning, acid pickling and fluoride activating.
The effect of pretreatment steps on substrate surface features is shown in Fig.3. The surface is unetched during the alkaline cleaning(Fig.3(b)), and the scratches disappear after acid pickling. The acid pickling can remove the metal chippings and oxides away from the substrate surface[16]. According to Fig.3(b), the intact β phase is extruded from the substrate and the primary α is not attacked, only the eutectic α is etched. The surface is covered with MgF2 after fluoride activation and becomes dark (Fig.3(c)). Crevices on the surface after acid pickling and fluoride activation are observed.

Fig.3 Surface morphologies of pretreated surface: (a) Alkaline cleaning; (b) Acid pickling; (c) HF activation
The surface morphologies of electroless nickel nucleated on the AZ91D substrate at different intervals of time are illustrated in Fig.4. The coating preferentially nucleates on the crevices, which are the eutectic α and β, due to the galvanic coupling of β and eutectic α to primary α phase. The coating on primary α phase is formed after β and eutectic α are covered with coating. However, the coating on the β phase is discontinuous and grows slowly, which leads to convergence of defects over this region[7, 17].
3.4 Mechanism of electroless plating-nickel
It is noted that the electroless plating-nickel nucleates on the crevices. The autocatalytic reaction for nickel deposition is initiated by catalytic dehydrogenation of the reducing agent with the release of hydride ion, which then supplies electrons for the reduction of nickel ions [1]:
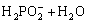 →
→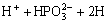 (1)
(1)
Ni2++2H→Ni0+2H+ (2)
 →
→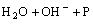 (3)
(3)
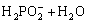 →
→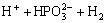 (4)
(4)
3.5 Corrosion resistance of nickel plating-coating
The open circuit potential of the Ni-P coating with time immersed in 3.5% NaCl solution is presented in Fig.5. The open circuit potential of the coated magnesium alloy fluctuates. It approaches to -0.36 V (SCE) after the immersion in 3.5% NaCl solution at room temperature for 1 h. The steady-state working potential of magnesium electrode is generally about -1.50 V, although its standard potential is -2.37 V[4]. This indicates the corrosion resistance of the plating coating prepared is enhanced, compared with the bare magnesium alloy, due to the fact that the nickel plating-coating is compact without pores and good adhesion.
Potentiodynamic polarization curves, measured in 3.5% NaCl solution, are presented in Fig.6. The curves
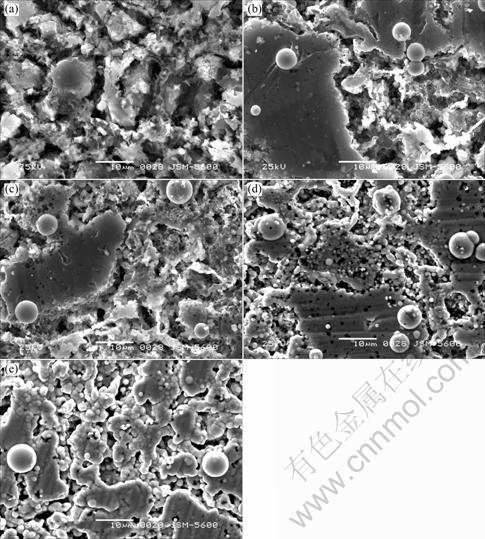 Fig.4 Nucleation and growth of Ni-P coating at different intervals of time: (a) 1 min; (b) 3 min; (c) 5 min; (d) 7 min; (e) 9 min
Fig.4 Nucleation and growth of Ni-P coating at different intervals of time: (a) 1 min; (b) 3 min; (c) 5 min; (d) 7 min; (e) 9 min
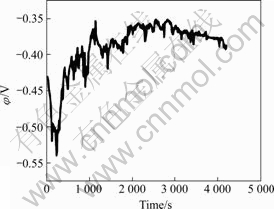
Fig.5 Open circuit potential curve of Ni-P with time
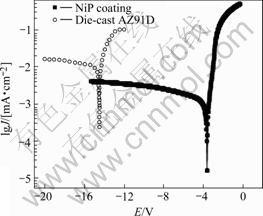
Fig.6 Polarization curves of Ni-P coating and AZ91D
indicate film breakdown at potentials immediately above the corrosion potential. The current density of AZ91D is higher than that of the Ni-P coating on the cathodic side. The corrosion potentials for die cast AZ91D and Ni-P coating are about -1.45 V and -0.36 V (SCE) respectively. These results show that Ni-P coating can increase the corrosion potential by 1 000 mV and improve corrosion resistance.
4 Conclusions
1) The surface morphology of electroless nickel-plating coating on die cast AZ91D magnesium alloy is spherical, compact without pores. The coating growth rate is about 17 μm/h and the phosphorus content in the deposit is 4%.
2) Plating deposition is initiated on the crevices, which are β phases and eutectic phases. Once the coating covers the crevices, it then spreads onto the primary α phase.
3) Potentiodynamic polarization curves, measured in 3.5% NaCl, show that the corrosion potentials for die cast AZ91D and Ni-P coating are about -1.45 V and -0.36 V (SCE), respectively. Ni-P coating can improve corrosion resistance for AZ91D and increase the corrosion potential by 1 000 mV. The adhesion of the plating coating to AZ91D alloy is excellent. Electroless nickel plating is a promising method to enhance magnesium alloys resistance for attacking.
References
[1] SHARMA A K, SURESH M R, BHOJRAJ H, NARAYANAMURTHY H, SAHU R P. Electroless nickel plating on magnesium alloy[J]. Metal Finish, 1998, 3: 10-18.
[2] ZHAO Hui, CUI Jian-zhong. Electroless plating of silver on AZ31 magnesium alloy substrate[J]. Surface & Coatings Technology, 2007, 201: 4512-4517.
[3] SONG G, STJOHN D H. Corrosion of magnesium alloys in commercial engine coolants[J]. Materials and Corrosion, 2005, 56(1): 15-23.
[4] SONG Guang-ling. Recent progress in corrosion and protection of magnesium alloys[J]. Advanced Engineering Materials, 2005, 7: 563-586.
[5] SKAR J K. Corrosion and corrosion prevention of magnesium alloys[J]. Matrials and Corrosion, 1999, 6: 2-6.
[6] UNIGOVSKI YA B, GUTMAN E M. Surface morphology of die-cast Mg alloy[J]. Applied Surface Science, 1999, 153: 47-52.
[7] RAJAN A, ZHOU W. Electroless nickel-plating on AZ91D magnesium alloy: Effect of substrate microstructure and plating parameters[J]. Surface and Coatings Technology, 2004, 179: 124-134.
[8] GRAY J E, LUAN B. Protective coatings on magnesium and its alloys―a critical review[J]. Journal of Alloys and Compounds, 2002, 336: 88-113.
[9] CHEN Jue-ling, YU Gang, HU Bo-nian, LIU Zheng, YE Li-yuan, WANG Zhen-feng. A zinc transition layer in electroless nickel plating[J]. Surface & Coatings Technology, 2006, 201: 686-690.
[10] LIU Zhen-min, GAO Wei. A novel process of electroless Ni-P plating with plasma electrolytic oxidation pretreatment[J]. Applied Surface Science, 2006, 253: 2988-2991.
[11] ZHAO Hui, HUANG Zhang-hong, CUI Jian-zhong. A new method for electroless Ni-P plating on AZ31 magnesium alloy[J]. Surface & Coatings Technology, 2007, 202: 113-119.
[12] ZHANG W X, HE J G., JIANG Z H, JIANG Q, LIAN J S. Electroless Ni-P layer with a chromium-free pretreatment on AZ91D magnesium alloy[J]. Surface & Coatings Technology, 2007, 201: 4594-4600.
[13] LIAN J S, LI G Y, NIU L Y, GU C D, JIANG Z H, JIANG Q. Electroless Ni-P deposition plus zinc phosphate coating on AZ91D magnesium alloy[J]. Surface & Coatings Technology, 2006, 200: 5956-5962.
[14] LI Jian-zhong, SHAO Zhong-cai, ZHAN Xing, TIAN Yan-wen. The electroless nickel-plating on magnesium alloy using NiSO4?6H2O as the main salt[J]. Surface & Coatings Technology, 2006, 200: 3010-3015.
[15] SONG Guang-ling, ANDREJ A, MATTHEW D. Influence of microstructure on the corrosion of diecast AZ91D[J]. Corrosion Science, 1999, 41: 249-273.
[16] SONG Ying-wei, SHAN Da-yong, HAN En-hou. A study on the pretreatment of direct electroless nickel plating on magnesium alloy AZ91D[J]. Materials Science Forum, 2005, 488/489: 835-838.
[17] LIU Zhen-min, GAO Wei. The effect of substrate on the electroless nickel plating of Mg and Mg alloys[J]. Surface & Coatings Technology, 2006, 200: 3553-3560.
(Edited by CHEN Can-hua)
Corresponding author: JIA Shu-sheng; Tel: +86-431-85095878; E-mail: jiass@jlu.edu.cn